Abstract
Background
Evidence on neurochemical mechanisms underlying response to apheresis in steroid-refractory Multiple Sclerosis (MS) attacks is limited.
Objective
To examine the effect of immunoadsorption (IA) versus plasma exchange (PLEX) on serum immunological parameters [IgG, IgA, IgM, kappa- and lambda-immunoglobulin free light chains (κ-FLC, λ-FLC), CXCL13, CXCL12] and the predictive value of these parameters on response to apheresis.
Methods
Pre- and postprocedural serum samples of 38 participants (IA: n = 19, PLEX: n = 19) from the IAPEMS trial (NCT02671682), conducted in our tertiary centre, were examined.
Results
Serum immunoglobulins were strongly reduced after both procedures (IgG: IA median −96.04%; PLEX median −85.98%). κ-FLC levels were reduced after PLEX (median −34.74%), not affected by IA. Both procedures caused a decrease in λ-FLC levels. CXCL13 slightly increased after PLEX (median +24.16%), conversely decreased after IA (median −21.92%). CXCL12 levels were reduced after IA (median −45.69%), but not significantly altered after PLEX. None of the serum parameters evaluated showed predictive value for apheresis response.
Conclusion
IA and PLEX have a differential effect on serum immunological parameters. IA appears to reduce B-cell derived inflammation more effectively. This finding requires further evaluation and comparative analysis with clinical outcomes, especially in the context of the efficacy of B-cell therapies in treating MS.
Keywords
Introduction
Relapse-associated worsening (RAW) is a key factor for long-term accumulation of disability in people with Multiple Sclerosis (pwMS).1–3 Further, a high relapse frequency or severe attacks in early disease stages are thought to be risk factors for an unfavourable disease course.4–6 Following the established attack therapy with high-dose intravenous steroids,7,8 incomplete remission of attack symptoms is often observed.9,10 The evidence regarding the optimal treatment strategy for preventing disability accumulation in this context remains limited. Treatment decisions are further complicated by the variability of attack outcomes, and the often extended period until completion of recovery. 11
Currently, apheresis procedures, i.e., therapeutic plasma exchange (PLEX) and immunoadsorption (IA), are increasingly used as escalation treatments due to their consistently high response rates,12–16 with recent work even suggesting a possible superiority of immediate IA after the initial cycle of steroids in comparison to a delayed utilisation after a second steroid course. 17
During PLEX, plasma is replaced with a human albumin solution or fresh frozen plasma. Its therapeutic effect is believed to result from the clearance of pro-inflammatory plasma components, but the non-specific mode of action also causes a loss of other plasma constituents, which may lead to complications such as thrombosis, bleeding or sepsis.18,19 IA exhibits a more selective mode of action, whereby the adsorber systems isolate immunoglobulins (Igs), allowing for the preservation of other plasma proteins. Existing evidence suggests, at least similar efficacy, if not superiority of IA, and similar tolerability between the two procedures in the treatment of MS attacks.20–22
At present, our knowledge about the mechanisms that lead to acute neuroinflammation and, consequently, to MS attacks, as well as the underlying changes in serum proteome, remains limited. The potential differential impact of attack therapies, especially the examined apheresis procedures, on serum parameters that potentially correlate or induce inflammatory activity, and its relationship with clinical outcomes, remains to be elucidated and could change clinical practice. In this context, Pfeuffer et al., 2022, 17 report a differential effect of a second steroid course compared to IA on lymphocyte subsets and chemokines, suggesting a reduction in B-cell counts after IA, but not after intravenous steroids.
Another unmet need is the identification of biomarkers for the early selection of patients at risk of RAW, allowing the timely implementation of apheresis. In a recent study, utilising serum samples from the IAPEMS trial (Dorst et al., 2019, clinicaltrials.gov: NCT02671682), 21 which investigated the use of IA versus (vs) PLEX in steroid-refractory attacks of MS or Clinically-Isolated Syndrome (CIS), we could demonstrate a predictive value of the preprocedural serum Glial Fibrillary Acidic Protein (sGFAP) levels and the serum Neurofilament Light Chain (sNfL)-to-sGFAP ratio on the clinical response to apheresis, measured by the improvement of relapse symptoms four weeks after the last apheresis session. 23
In the actual study we examined (a) the effect of IA vs PLEX on serum concentrations of immunoglobulins (IgG, IgA, IgM), κ- and λ-free immunoglobulin light chains (κ-FLC, λ-FLC), and the chemokines CXCL13, CXCL12, (b) the predictive value of these parameters on response to apheresis four weeks after the procedure.
Materials and methods
In the primary trial conducted between January 2016 and October 2018 (IAPEMS trial, clinicaltrials.gov: NCT02671682), 21 61 patients were randomized in two treatment groups (IA-group: n = 31, PLEX-group: n = 30). In both groups, apheresis was applied on five consecutive days after exclusion of an active infection. For the present study, we examined pre- and post-apheresis serum pairs available in our local biobank (stored at −80°C according to recommended biobanking protocols), 24 obtained at 0–3 days before apheresis and directly after the last apheresis session. The examined sub-cohort consisted of 38 IAPEMS participants (IA: n = 19, PLEX: n = 19), idem with the cohort examined in our previous work on the predictive value of sNfL and sGFAP. 22 No additional selection criteria were applied.
Therapeutic plasma exchange and immunoadsorption
During PLEX, 2 litres of plasma were removed per session, using the COM.TEC cell separator (Fresenius Kabi AG, Bad Homburg, Germany) and replaced with 5% human albumin solution.
IA was performed using an adsorber system (ADAsorb, medicap clinic GmbH, Ulrichstein, Germany) with regenerating protein A adsorber columns (Immunosorba, Fresenius Medical Care, Bad Homburg, Germany) after cell and plasma separation with a plasma separator (ART Universal, Fresenius Medical Care, Bad Homburg, Germany). The 2-fold total plasma volume (TPV) was processed on day 1, on the following days the 2.5-fold TPV.
Clinical outcomes
The trial participants were followed up clinically directly after the last apheresis session, two and four weeks later. In the primary trial, 21 response to apheresis was defined as an improvement of at least 10% in the Multiple Sclerosis Functional Composite (MSFC). In our sub-cohort this could be observed in 17/19 participants treated with PLEX and 16/19 participants treated with IA. For a better differentiation of study participants into “responders” and “non-responders”, we applied the model proposed by Conway et al., 2019, 25 to classify attack outcomes into “good”, “average” and “worse” by assessing the change in affected functional system scores (FSS) of the Expanded Disability Status Scale (EDSS).
Immunological serum parameters
Serum parameters potentially relevant to MS activity or treatment response were selected. Immunoglobulin removal is considered to be a key treatment mechanism for both procedures.19,20 FLC are synthesised by B-cells in the developmental stage after the pre-B-cell. They are considered to be immunologically active molecules, rather than by-products of immunoglobulin synthesis, which play an essential role in the development of autoimmunity and were proposed as markers of B-cell derived inflammation. 26 CXCL13, a B-cell attracting chemokine, has been demonstrated to be elevated in serum, cerebrospinal fluid (CSF) and active demyelinating lesions of pwMS. Elevated CXCL13 levels in CSF have been correlated with an increased relapse frequency and EDSS worsening.27,28 CXCL12 regulates cell migration. In the healthy central nervous system, it is found predominantly in endothelial cells. In MS attacks it is redistributed from the periventricular to the luminal side of the blood-brain barrier, from where it attracts mononuclear cells into the CNS. It can also be found in active MS plaques.29,30
IgG, IgA, IgM, κ-FLC and λ-FLC were measured by nephelometry on a Siemens BN ProSpec® (Siemens Healthineers, Erlangen, Germany), CXCL13 and CXCL12 using ELISA (Quantikine; R&D Systems, Minneapolis, MN, USA). The IgM assay could not differentiate values below 0.18 mg/dL. In these cases, a value of 0.18 mg/dL was assigned for statistical analysis. All measurements were performed according to the instructions provided by the respective manufacturer.
Statistical analysis
We conducted statistical analysis primarily using the software language Python (v3.1) with the Jupyter notebook software (v6.4.8) and the software packages pandas (v1.3.5) and statsmodels (v0.14.0). We employed the Mann-Whitney U-test for independent samples, the Wilcoxon signed-rank test for dependent samples, and the chi-squared test for categorical variables. We used the statistical software SPSS (version 29.0.1.0, IBM, Armonk, NY) to conduct a multivariate logistic regression analysis for evaluating the prediction of apheresis outcome. Due to the exploratory character of our study no corrections were made for multiple testing. Therefore, in result interpretation, a higher possibility of Type I error should be considered. A p-value less than 0.05 was considered significant.
Ethical approval and study registration
The IAPEMS trial received approval from the local Ethics Committee (approval number 298/15) and is listed in the National Institute of Health's registry (clinicaltrials.gov, NCT02671682). All study participants signed informed consent forms.
Results
Baseline characteristics
The baseline characteristics of the two treatment groups showed a balanced distribution (Table 1). The median age for the PLEX-group was 39 years vs 40 years for the IA-group. A high percentage of participants in both groups experienced their first demyelinating event (PLEX 53%, IA 37%). Relapsing MS was diagnosed in 74% of patients in the PLEX-group and 84% of patients in the IA-group, according to the 2017 McDonald criteria. 31 Patients with CIS did not meet the criteria for dissemination in time, but in all CIS cases, relevant differential diagnoses were excluded. A majority of study participants did not receive prior immunotherapy (PLEX-group: 84%, IA-group: 53%). One PLEX-treated and four IA-treated patients were receiving first-line immunotherapies (interferons, glatiramer acetate, dimethyl fumarate and teriflunomide), two patients in the PLEX-group and five patients in the IA group were treated with agents for active MS (S1P-modulators, Natalizumab, Ocrelizumab). Time from relapse onset to begin of apheresis and prior high-dose steroid courses were equally distributed. Optic neuritis was the most common relapse syndrome (53% in both groups), followed by myelitis and infratentorial relapse manifestations. All patients completed the planned treatment cycle of five daily apheresis sessions.
Baseline characteristics.

Baseline demographic and clinical patient data. y: years, d: days, IQR: interquartile range, RMS: Relapsing Multiple Sclerosis, CIS: Clinically Isolated Syndrome, EDSS: Expanded Disability Status Scale, IMT: Immunotherapy.
Immunoglobulins
The preprocedural IgG (p = 0.849) and IgA levels (p = 0.343) did not show significant intergroup differences. However, IgM concentrations were lower in the IA-group (p = 0.034). As reported for the overall cohort of the primary trial, 21 both procedures resulted in a marked decrease in serum immunoglobulin levels (Figure 1). This effect was most pronounced for IgG in both procedures [PRE PLEX: median 6830.00 (interquartile range (IQR) 6390.00–8020.00) mg/l, POST PLEX 994.00 (IQR 796.00–1360.00) mg/l; PRE – POST PLEX: p < 0.001; PRE IA: median 6920.00 (IQR 5335.00–7635.00) mg/l, POST IA: median 275.00 (IQR 220.50–307.00) mg/l; PRE – POST IA: p < 0.001], with a stronger reduction in IgG observed for the IA-group, resulting in lower postinterventional IgG-levels (POST PLEX – POST IA: p < 0.001) and a lower percentage difference (PLEX: median −85.98 (IQR −87.81 – −82.42) %; IA: median −96.04 (IQR −96.85 – −95.29) %; p < 0.001).

Boxplots showing serum concentrations before and after therapeutic plasma exchange (PRE PLEX / POST PLEX) and immunoadsorption (PRE IA / POST IA) for IgG (a), IgA (d) and IgM (g). Scatter plots showing individual parameter change following PLEX and IA for IgG (b,c), IgA (e,f) and IgM (h,i). Indication of the results of statistical testing (nonparametric): * p < 0.05, ** p < 0.01, *** p < 0.001; n.s. indicates nonsignificant p-values (p > 0.05). For better visualization, single outliers were omitted.
In contrast, PLEX led to a more pronounced decrease in IgA [PRE PLEX: median 1630.00 (IQR 1320.00–1790.00) mg/l, POST PLEX 276.00 (IQR 200.00–356.00) mg/l; PRE – POST PLEX: p < 0.001; PRE IA: median 1480.00 (IQR 873.50–1810.00) mg/l, POST IA: median 565.00, IQR (370.50–627.00) mg/l; PRE – POST IA: p < 0.001], and IgM concentrations [PRE PLEX: median 1.03 (IQR 0.73–1.28) g/l, POST PLEX 0.18 (IQR 0.18–0.182) g/l; PRE – POST PLEX: p < 0.001; PRE IA: median 0.65 (IQR 0.49–0.99) g/l, POST IA: median 0.18 (IQR 0.18–0.23) g/l; PRE – POST IA: p < 0.001]. The comparison of post-procedure serum levels proved significant only for IgA (p < 0.001), not for IgM (p = 0.32). However, the statistical comparison of the percentage differences proved significant for both, IgA (PLEX: median −83.67 (IQR −85.59 – −81.91) %, IA: median −61.55 (IQR −65.15 – −56.43); p < 0.001) and IgM (PLEX: median −82.14 (IQR −85.88 – −73.80); IA: median −67.45 (IQR −76.69 – −63.11); p = 0.002).
Free immunoglobulin light chains (FLC, Figure 2)
The preprocedural concentrations of κ-FLC and λ-FLC were equally distributed (κ-FLC: p = 0.225, λ-FLC: p = 0.483).

Boxplots showing serum concentrations before and after therapeutic plasma exchange (PRE PLEX / POST PLEX) and immunoadsorption (PRE IA / POST IA) for kappa free immunoglobulin light chain (κ-FLC; a) and lambda free immunoglobulin light chain (λ-FLC; d). Scatter plots showing individual parameter change following PLEX and IA for κ-FLC (b,c) and λ-FLC (e,f). Indication of the results of statistical testing (nonparametric): * p < 0.05, ** p < 0.01, *** p < 0.001; n.s. indicates nonsignificant p-values (p > 0.05).
Study participants in the PLEX-group consistently exhibited a decrease in serum κ-FLC levels, for participants treated with IA we could not observe a reproducible trend (PRE PLEX: median 4.60 (IQR 3.85–6.65) mg/l, POST PE 3.30 (IQR 2.65–4.55) mg/l; PRE – POST PLEX: p = 0.011; PRE IA: median 7.10 (IQR 4.20–8.80) mg/l, POST IA: median 4.80 (IQR 4.25–8.75) mg/l; PRE – POST IA: p = 0.176). The comparison of the percentage difference between preprocedural and postprocedural serum concentrations did not reach statistical significance (PLEX: median −34.74 (IQR −45.15 – + 1.25) %; IA: median −7.95 (IQR −33.03 – + 18.56) %, p = 0.108).
λ-FLC were equally reduced in both treatment groups post-intervention (PRE PLEX: median 7.70 (IQR 5.20–11.85) mg/l, POST PLEX 3.60 (IQR 3.50–4.05) mg/l; PRE – POST PLEX: p < 0.001; PRE IA: median 6.60 (IQR 5.95–9.35) mg/l, POST IA: median 3.90 (IQR 3.55–4.65) mg/l; PRE – POST IA: p < 0.001). While the comparison of the postprocedural λ-FLC concentrations did not reveal a significant difference (p = 0.176), the examination of the percentage difference suggested a possibly higher effect of PLEX on λ-FLC levels (PLEX: median −54.55 (IQR −63.72 – −31.13) %; IA: median −37.25% (IQR −48.48 – −30.33) %; p = 0.042).
CXCL13 and CXCL12 (Figure 3)
Preprocedural CXCL13 (p = 0.872) and CXCL12 concentrations (p = 0.089) did not show statistically significant differences between the two treatment groups.

Boxplots showing serum concentrations before and after therapeutic plasma exchange (PRE PLEX / POST PLEX) and immunoadsorption (PRE IA / POST IA) for CXCL13 (a) and CXCL12 (d). Scatter plots showing individual parameter change following PLEX and IA for CXCL13 (b,c) and CXCL12 (e,f). Indication of the results of statistical testing (nonparametric): * p < 0.05, ** p < 0.01, *** p < 0.001; n.s. indicates nonsignificant p-values (p > 0.05).
Opposite trends in CXCL13 levels were detected after each procedure. After PLEX we observed a slight increase in CXCL13 levels (PRE PLEX: median 46.80 (IQR 34.20–59.50) pg/ml, POST PLEX 65.50 (IQR 46.75–76.95) pg/ml; PRE – POST PLEX: p = 0.003), conversely, a weak but significant decrease after IA (PRE IA: median 48.90 (IQR 36.95–65.20) pg/ml, POST IA: median 40.70 (IQR 28.05–53.90) mg/l; PRE – POST IA: p = 0.014). Thus, post-procedural concentrations (p = 0.004) and the percentage difference (PLEX: median +24.16 (IQR +4.69 – + 72.75) %; IA: median −21.92 (IQR −32.82 – + 2.84) %; p < 0.001) were lower after IA.
Similar to CXCL13, but with more profound effects, we observed a reduction of CXCL12 following IA [PRE IA: 1601.00 (IQR 1359.75–1880.50) pg/ml; POST IA: 890.50 (IQR 732.25–1043.50) pg/ml; PRE – POST IA: p < 0.001]. PLEX did not significantly affect CXCL12 levels [PRE PLEX: 1438.00 (IQR 1202.00–1574.00) pg/ml; POST PLEX: 1504.00 (IQR 1217.00–1598.00) pg/ml; PRE – POST PLEX p = 0.207]. This was confirmed by the percentage difference found in the two groups (PLEX: median +8.66 (IQR −10.85 – + 42.29) %; IA: median −45.69 (IQR −52.18 – + 33.49) %; p < 0.001).
Outcome prediction
By evaluation of the FSS improvement 25 10/19 (53%) patients treated with PLEX demonstrated a “good”, 1/19 (5%) an “average” and 8/19 (42%) a “worse” outcome. In the IA group 13/19 (68%) showed a “good” outcome, an “average” and “worse” outcome showed respectively 3/19 (16%) patients . Due to the small number of average responders, we conducted statistical analysis comparing either FSS improvement (“good” and “average”, “responders”) with “worse” response (“non-responders”).
We conducted a multivariate logistic regression analysis with the pre-procedural levels of IgG, κ- and λ-FLC, CXCL13, CXCL12, and age as dependent variables. Neither of the examined serum parameters could predict relapse outcome (Table 2). Younger age was associated with a higher likelihood of clinical improvement (p = 0.012, Odds Ratio 0.869, 95% Confidence Interval 0.779–0.969). An additional analysis of the postprocedural and the difference of the pre- and postprocedural concentrations of the studied parameters did not show any predictive value.
Evaluation of the predictive value of age and the examined serum parameters on response to apheresis.
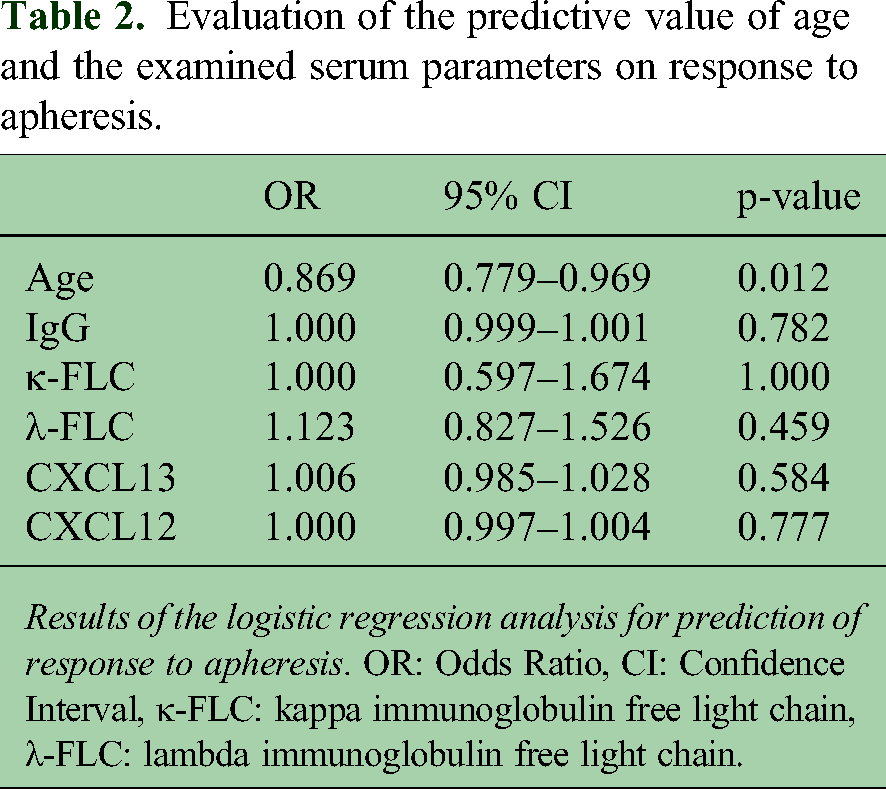
Results of the logistic regression analysis for prediction of response to apheresis. OR: Odds Ratio, CI: Confidence Interval, κ-FLC: kappa immunoglobulin free light chain, λ-FLC: lambda immunoglobulin free light chain.
Discussion
The optimal therapeutic approach for the steroid-refractory MS attack after an initial high-dose steroid cycle remains uncertain. Questioning the need for a second steroid cycle, recent evidence even suggests that apheresis is superior to a second steroid cycle in this setting.17,32
To our knowledge, IAPEMS is currently the only randomized, controlled trial that has compared the effectiveness and safety of IA and PLEX for treating steroid-refractory attacks in MS and revealed a better functional outcome after IA, measured by MSFC improvement, four weeks after the last apheresis session. 21 The actual study of serum immunological parameters reveals a differentiated effect of the two examined apheresis procedures on immunological serum parameters, suggesting a more potent effect of IA on the B-cell compartment.
Prior to interpretation of the results, it is essential to consider the underlying mechanism of action of each procedure. For PLEX, the reduction in certain serum parameters, e.g., immunoglobulins and FLC, can be attributed, at least in part, to the non-selective substitution of plasma with a human albumin solution. However, it can not explain the dynamics of the chemokines CXCL13 and CXCL12. The selective removal of immunoglobulins during IA, predominantly IgG, implies that the effects on the investigated serum parameters are most likely due to secondary immunological effects.
κ-FLC have been discussed as markers of B-cell derived inflammatory activity of autoimmune diseases. 26 No notable alterations in serum κ-FLC levels were observed after IA, while the majority of participants in the IA-group exhibited a clinical improvement. Therefore, the examination of serum κ-FLC prior to or following an apheresis cycle does not appear to be useful for the evaluation of inflammatory activity or treatment response in MS attacks.
Concentrations of CXCL13, a B-cell attracting chemokine, in CSF and serum are correlated with inflammatory activity in pwMS. 28 CXCL12, a chemokine regulating cell migration, is involved in attracting mononuclear cells to the site of inflammation during an MS attack.29,30 In this respect, the significant reduction in CXCL13 and CXCL12 after IA, but not PLEX, may provide initial evidence that IA has a more potent effect on B-cell activation in the context of acute neuroinflammation. In support of our findings, Pfeuffer et al., 2022, 17 report a decrease in total B-cell counts and B-cell subsets after IA but not after a second course of steroids, while Foettinger et al., 2023, 33 could not observe a change in B-cell counts after PLEX. This finding is of special interest, given the proven long-term effect of several anti-CD20 antibodies in preventing inflammatory activity and thus accumulation of disability in pwMS.34
Further studies are necessary in order to examine the possible correlation between the effect of IA on the B-cell compartment and the clinical improvement of acute symptoms in MS attacks.
Preprocedural concentrations of the examined parameters were not predictive of procedure outcome. The high rates of clinical improvement in both treatment groups, strongly support the early use of apheresis in the steroid-refractory attack, especially when treating optic neuritis, myelitis and infratentorial attacks.
Possible bias in the interpretation of our results, besides the majority of subjects being early in the disease course, is the presence of patients with a disease duration of more than 15 years, although equally distributed in both treatment groups, and the demonstration of persisting inflammatory activity, clinically and by magnetic resonance imaging prior to enrolment in the primary study. The majority of patients were either treatment-naive or on first-line therapies, however, a small number of patients in each group were on high-efficacy treatments, which could have affected the measured parameters before study entry and potentially compromised the effect of the apheresis procedures on them. Additionally, the small sample size potentially limits the generalizability of the results.
Due to the unavailability of cell containing samples, we were not able to examine cellular subsets and thus investigate a possible correlation between the reduction of CXCL13 and CXCL12 and changes in lymphocyte counts.
In summary, our study reveals a differential effect of PLEX and IA on the examined serum parameters. Of special interest is the finding of lower postprocedural CXCL13 and CXCL12 levels in serum after IA compared to PLEX, which could reflect a more pronounced effect of IA on the B-cell compartment. Given the proven efficacy of anti-CD20-therapies in treating MS, the finding could suggest the preferential use of IA in treating refractory MS attacks. A further evaluation of our finding in larger prospective studies with a focus on the link between changes in chemokine levels, lymphocyte counts and clinical improvement after apheresis is needed.
Footnotes
Consent to participate
All study participants signed informed consent forms.
Consent for publication
Not applicable
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IV has received travel support from Alexion and Novartis. JD has received consulting and speaker honoraria from Fresenius. HT has received consulting and/or speaker honoraria and/or travel support from Alexion, Bayer, Biogen, Bristol-Myers Squibb/Celgene, Diamed, Fresenius, Fujirebio, GlaxoSmithKline, Horizon, Janssen-Cilag, Merck, Novartis, Roche, Sanofi-Genzyme, Siemens, Teva, and Viatris. MS has received consulting and/or speaker honoraria from Alexion, Bayer, Biogen, Bristol-Myers-Squibb/Celgene, Janssen, Merck, Horizon, Roche, and Sanofi Genzyme. AH, BM, PE, TF and DT have no competing interests to declare that are relevant to the content of this article.
Ethical considerations
The IAPEMS trial received approval from the local Ethics Committee (approval number 298/15).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary trial 18 was supported by Fresenius Medical Care Deutschland GmbH, Bad Homburg, Germany. The sponsor had no influence on the study design, or the collection, analysis, interpretation and publication of the data. No funding was received for conducting the actual study.
