Abstract
Background
One of the most disabling symptoms of patients with multiple sclerosis (MS) is spasticity which affects their quality of life. Nowadays, cannabinoids are used for spasticity control in patients with MS, while the efficacy and safety are not clearly understood. So, we designed this systematic review and meta-analysis to assess the efficacy of cannabinoids for controlling MS-related spasticity.
Methods
PubMed, Scopus, EMBASE, Web of Science, and Google Scholar were systematically searched by two independent researchers on 1 May 2023. They also searched gray literature (references of included studies, as well as conference abstracts).
Results
A literature search revealed 6552 records, 95 full-texts were evaluated, and finally, 31 studies remained for systematic review. Among included studies, six randomized trials were included. Nabiximols was the most commonly used medication for controlling MS-related spasticity. Mean Expanded Disability Status Scale ranged between 4.6 and 7. Most studies (17 studies) were done in Italy, followed by Germany (4 studies). The pooled standardized mean difference (SMD) of NRS (Numeric Rating Scale) (after–before) is estimated as −1.41 (95% confidence interval (CI): −1.65, −1.17) (I2 = 97%, p < 0.001). The pooled standardized mean difference (SMD) of Ashworth (after-before) is estimated as −0.39 (95% CI: −0.72, −0.06) (I2 = 69.9%, p = 0.005).
Conclusion
The results of this systematic review and meta-analysis showed that nabiximols was the most common cannabinoid which was used to control MS-related spasticity, and it was effective in controlling MS-related spasticity (significantly decreased SMD of NRS, and Ashworth after treatment).
Introduction
Multiple sclerosis (MS) is a chronic disabling disease of the central nervous system (CNS), characterized by demyelinating plaques, and significant physical complications such as walking difficulties, gait imbalance, and spasticity.1,2
One of the most disabling symptoms of patients with MS is spasticity, affecting more than half of the patients, while literature shows that near three-fourths of affected individuals suffer from spasticity 15 years after disease progression. 3 Muscle hypertonia, stiffness, weakness, and following insomnia will result in interfering with daily activities, and quality of life impairment. 4
The common treatment includes antispastic medications such as baclofen, tizanidine, or dantrolene in combination with physiotherapy, with not always fully satisfactory effects.3,5 Withdrawal is common as the side effects include falling, sedation, dizziness, and withdrawal syndrome. 6
Currently, Onabotulinumtoxin (BOTOX®, Allergan, Inc., Irvine, CA) injection has become more popular for controlling spasticity, but the duration of action is short, and administration of botox needs a high rate of specialization.7,8
These days, people with MS admit to consuming cannabinoids to control different symptoms such as pain, anxiety, spasticity, and sleep disturbances. 9
Cannabis is cultivated all over the world, containing over 483 identifiable chemicals, while only 80 cannabinoids are isolated from the plant, but the most famous ones are tetrahydrocannabinol (THC) and cannabidiol (CBD). 10
Novotna et al. 11 for the first time introduced the application of oral spray of cannabinoids for MS-related spasticity, and nabiximols has been approved for MS-related spasticity treatment.
Various studies show that cannabinoids are used for spasticity control in patients with MS, while the efficacy and safety are not clearly understood. So, we designed this systematic review and meta-analysis to assess the efficacy of cannabinoids for controlling MS-related spasticity.
Methods
We followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 for reporting our systematic review, and meta-analysis. 12
Eligibility criteria
Inclusion criteria: We included trials and observational studies that evaluated the effects of cannabinoids on spasticity in patients with MS.
Exclusion criteria: Case reports, case series, letters to editors.
We excluded studies that had no clear data for meta-analysis.
Information sources
PubMed, Scopus, EMBASE, Web of Science, and Google Scholar were systematically searched by two independent researchers on 1 May 2023. They also searched gray literature (references of included studies, as well as conference abstracts).
Search strategy

Selection process and collection
After the primary search, the obtained results were imported to ENDNOTE software. Duplicates were deleted, then titles and abstracts of eligible studies were assessed. Potential full texts were obtained, and were evaluated by two independent researchers.
Researchers extracted data and entered it in separate Excel files. If discrepancies were present, the third one solved the issue.
Data items
The first author of the publication, country of the study, publication year, duration of the study, number of study participants, total female, and male cases, type of MS, cannabinoid type, mean age at disease onset, mean Expanded Disability Status Scale (EDSS), type of the disease, numeric rating scale for spasticity, and Modified Ashworth Scale were extracted from included studies.
Study risk of bias assessment
ROBINS-I tool was used for Quality assessment of nonrandomized studies, while ROB2 was applied for quality assessment of randomized trials.13,14
Effect measures
We calculated standardized mean difference (SMD) for NRS, and Ashworth scale.
Synthesis methods
All statistical analysis was done using STATA (Version 14.0; Stata Corp LP, College Station, TX, USA). The p-values <0.05 were considered significant.
Certainty assessment
For all estimated effect sizes, we reported 95% CI. For studies that reported more than one endpoint outcome, we considered the final one.
Results
A literature search revealed 6552 records, 95 full-texts were evaluated, and finally, 31 studies remained for systematic review (Figure 1).
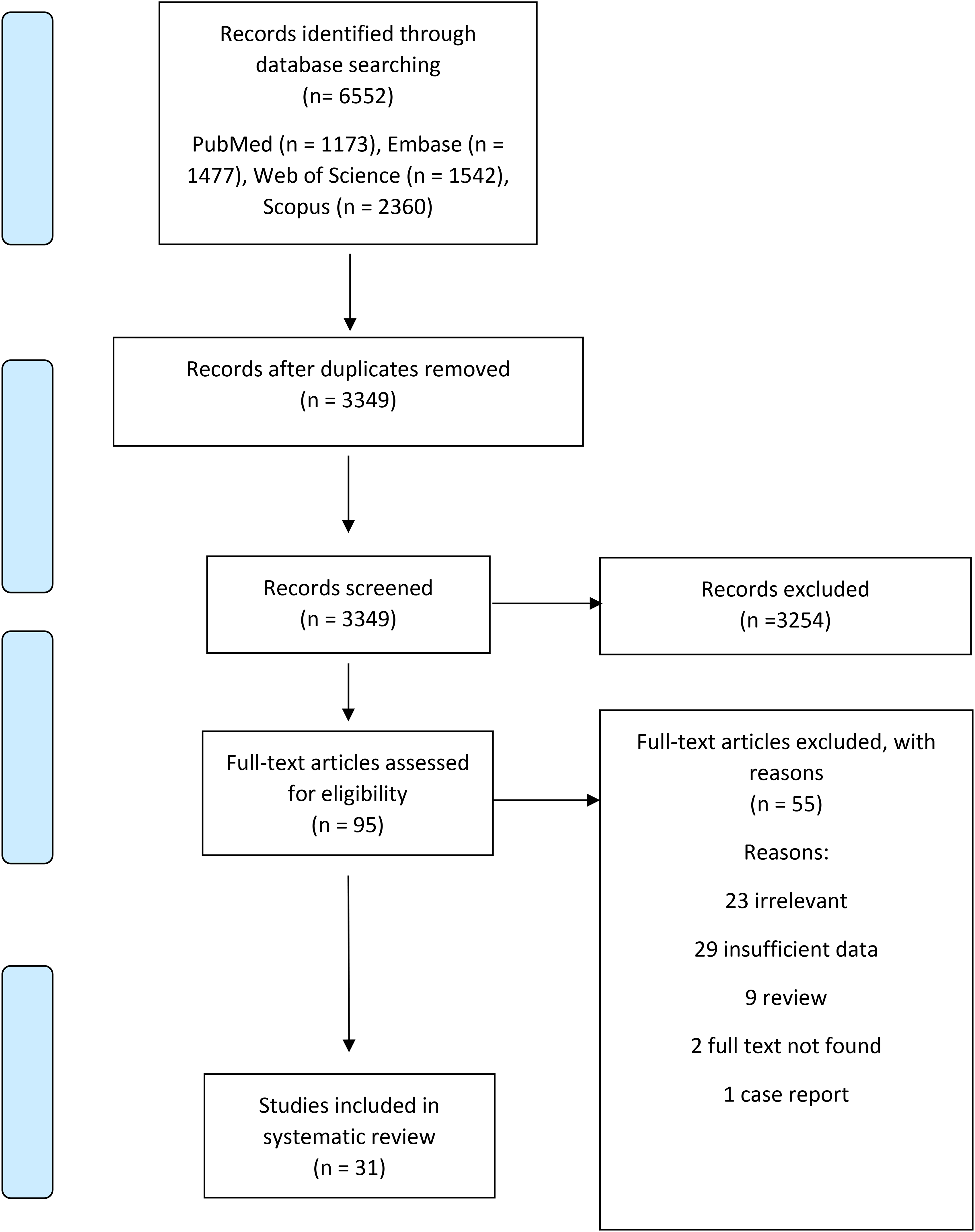
The flow chart of studies inclusion.
Included studies published between 2002 and 2023. Most studies were done in Italy, followed by Germany.
The number of patients in studies ranged between 8 and 1845, and the duration of studies ranged between 4 weeks and 1 year. Among included studies, six randomized trials were included. Except for four studies, all others used nabiximols. Mean EDSS ranged between 4.6 and 7. Most studies (17 studies) were done in Italy, followed by Germany (4 studies) (Table 1).
Included study (design and results).

CBD: cannabidiol; EDSS: Expanded Disability Status Scale; F: female; M, male; NRS: Numeric Rating Scale; PPMS: primary progressive multiple sclerosis; PRMS: progressive-relapsing multiple sclerosis; RRMS: relapsing-remitting mustiple sclerosis; SE: standard error; SEM: standard error of the mean; SPMS: slowly progressive multiple sclerosis; THC: tetrahydrocannabinol.
The quality assessment of trials and observational studies are summarized in Tables 2 and 3.
Quality assessment of nan-randomized studies (ROBINS-I).

Quality assessment of randomized trials (ROB2).

The pooled SMD of NRS (after–before) is estimated as −1.41 (95% CI: −1.65, −1.17) (I2 = 97%, p < 0.001) (Figure 2), indicating that cannabis use is effective in decreasing numeric rating scale of spasticity in patients with MS.

The pooled SMD of NRS (after–before). NRS: Numeric Rating Scale; SMD: standardized mean difference.
The pooled SMD of Ashworth (after–before) is estimated as −0.39 (95% CI: −0.72, −0.06) (I2 = 69.9%, p = 0.005) (Figure 3), indicating that cannabis use helps reducing Ashworth spasticity scale in patients with MS.

The pooled SMD of Ashworth (after–before). SMD: standardized mean difference.
Discussion
To our knowledge, this is the first systematic review and meta-analysis in this field. According to our results, the administration of cannabinoids for controlling spasticity in patients with MS is helpful as the pooled SMD of NRS and Ashworth were significantly improved after administration of these medications. As our results show, the SMD of Ashworth scale was −1.78, which showed a great impact of cannabinoids on MS-related spasticity.
In a multicentric observational study, Guger et al. enrolled patients with MS who suffered from spasticity, and evaluated spasticity-treatment using nabiximols oromucosal spray. Their results showed near 40% reduction in NRS for spasticity after administration of the medication. 43
In a single-center study, Sartori et al., evaluated the effects of botulinum toxin injections (BTI), and nabiximols on MS-related spasticity in patients with MS. Their results showed that BTI was more effective than nabiximols in treating MS-related spasticity. 27
Vecchio et al., evaluated the effects of cannabinoids (cannabinoid spray) on spasticity in patients with MS. Participants were allowed to use the maximum dose of 12 puffs per day. They found that after 6 weeks, pain, and spasticity were improved significantly. 15
Among included studies, some found that cannabinoids are not effective in controlling spasticity, while others did find. The difference among the findings is due to different inclusion and exclusion criteria, diverse definitions of spasticity, and follow-up duration variation.
The first large-scale clinical trial to evaluate the effects of cannabinoids on MS-related spasticity was developed by Zajicek et al. in 2005, and 630 patients were recruited.
They were assigned to oral cannabis extract, Δ9-tetrahydrocannabinol, or placebo.
They reported improvement in spasticity as 61% in the first group, 60% in the second group, and 46% in the placebo group. We did not include this study as they reported a mean change of Modified Ashworth Scale for spasticity not crude numbers. 46
Patients with MS suffer from spasticity based on demyelinating plaques of CNS, and damage to descending spinal pathways (corticospinal, reticulospinal, and vestibulospinal). 47 Factors such as male sex, duration of MS disease, higher level of disability, and relapses play roles in developing MS-related spasticity.
Urinary tract infections, distension of the urinary bladder and rectum, pain, and pressure sores could lead to development, and aggravation of spasticity in MS. 48
The most common medication that is used for MS-related spasticity is baclofen followed by benzodiazepines, while their efficacy is not very satisfactory. Botulinum toxin type A is another medication that could reduce muscle tone, but it is partially effective. 48
In animal models of MS, both endogenous and exogenous cannabinoids improve spasticity and tremors. 49
Cannabinoids activate G protein-coupled receptors (GPCRs), leading to increased synthesis of cyclic adenosine monophosphate (cAMP), and activation of cAMP-dependent protein kinase (PKA) that helps phosphorylation of channel protein. All these effects result in ionic permeability modification. 50
The administration of cannabinoids to control MS-related spasticity may help patients, but larger multicentric studies are needed.
This study had some limitations. First, the inclusion and exclusion criteria differed between included studies. Second, some studies applied NRS while others used Ashworth scale for spasticity evaluation. Third, the duration of disease differed between studies.
Conclusion
The results of this systematic review and meta-analysis showed that nabiximols was the most common cannabinoid which was used to control MS-related spasticity, and it was effective in controlling MS-related spasticity (significantly decreased SMD of NRS, and Ashworth after treatment).
Footnotes
Authors’ contribution
MA was involved in conceptualization, investigation, writing—original draft, and writing—review & editing; MP in conceptualization, formal analysis, andwriting—review & editing; FG in investigation, data curation, and writing—review & editing; SZE-R in project administration, supervision, visualization, and writing—review & editing; ETB in visualization, data curation, and writing—review & editing; MG in methodology, software, formal analysis, and writing—original draft; and MR in methodology, software, formal analysis, and writing—original draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
