Abstract
A multicenter study involving 204 adults with relapsing-remitting multiple sclerosis (RRMS) assessed the dimensionality and item characteristics of the Mishel-Uncertainty of Illness Scale (MUIS), a generic self-assessment tool. Mokken analysis identified two dimensions in the MUIS with an appropriate item and overall scale scalability after excluding nonclassifiable items. A refined 12-item MUIS, employing a grade response model, effectively discriminated uncertainty levels among RRMS patients (likelihood ratio test p-value = .03). These findings suggest the potential value of the 12-item MUIS as a reliable measure for assessing uncertainty associated with the course of illness in RRMS.
Introduction
Illness-related uncertainty is defined as the unpredictability and ambiguity that patients experience when faced with the challenges of a health condition. 1 Multiple sclerosis (MS) is a chronic autoimmune neurological disorder that affects different functional systems, often following a trajectory that is challenging to predict. 2 Patients with relapsing-remitting multiple sclerosis (RRMS) face the uncertainty of experiencing a new relapse, the variability of everyday symptoms, the safety risks associated with disease-modifying treatments, as well as the long-term progression of disability.2–4 Uncertainty about prognosis was associated with symptom severity, anxiety, depression, and low quality of life.3–6
The Mishel-Uncertainty of Illness Scale (MUIS) is a generic self-rated questionnaire to assess the uncertainty associated with the course and the treatment of illness, providing valuable information on patients emotional well-being. 7 Giammanco et al. conducted for the first time a psychometric analysis of the MUIS in patients with RRMS, which led to the validation of only two of its four original dimensions (ambiguity and inconsistency). 1 The aim of this study was to evaluate the dimensional structure and item distributions of the Spanish version of the MUIS in a sample of patients with RRMS. Assessing the psychometric properties of the MUIS in the setting of RRMS can provide healthcare professionals with valuable information on how to effectively interpret the results yielded by this instrument. 8
Methods
We conducted a noninterventional, cross-sectional study at 19 hospital-based MS care units in Spain (FACE-MS study). Adult patients with a diagnosis of RRMS (2017 revised McDonald criteria), a disease duration of 3 to 8 years, and being on disease-modifying therapy were consecutively invited to participate in the study in the context of their follow-up visits. Patients with MS frequently experience elevated levels of anxiety and depression, a low quality of life, and an overestimation of the risk of severe disability during the initial years following diagnosis. 9 For this reason, we opted to conduct this psychometric assessment on a sample of patients in the intermediate stage of the disease who are undergoing treatment. The study was approved by the research ethics board of the Hospital de Galdakao-Usansolo (Galdakao, Spain). All participants provided written informed consent.
The MUIS scores each item on a 5-point Likert scale from 1 (“strongly disagree”) to 5 (“strongly agree”).1,7 Higher scores indicate a greater feeling of uncertainty. The validated Spanish 17-item version of the instrument was used (range: 17–85). 10
Statistical analysis
We used a nonparametric item response theory (IRT) procedure (Mokken analysis) to assess: (i) the underlying dimensional structure of the MUIS, (ii) the scalability of each item and of the overall scale, and (iii) model assumptions for unidimensional scales for the selection of a parametric IRT model, to further analyze item characteristics of discrimination and information. 11 Scalability coefficient values <0.3 are considered unacceptable to rank people on the latent trait. We employed specific IRT models to compare the fitting of the monotone homogeneity model (MHM) and the double monotonicity model (DMM) utilizing the graded response and the rating scale models, respectively. These models enabled us to derive estimates for the connection between the latent trait and item characteristics such as discrimination and information, mirroring reliability. All analyses were performed with R (v 4.0.3; https://cran.r-project.org/) using the “mokken” and “ltm” libraries.
Results
A total of 204 patients were studied. The mean age (standard deviation) was 38.7 (8.5) years and 74.3% were female. A 47.8% (n = 96) had a university degree. Median EDSS score and disease duration (interquartile range [IQR]) were 1.0 (0–2.0) and 6.0 (4.0–7.0) years, respectively. Median total number of relapses since disease onset was 2.0 (1.0–3.0). Median MUIS (IQR) score was 38.2 (30.0–45.0).
Item endorsement
Overall, 45.2% of the participants were not clear on what was going to happen to them and 36.3% stated that they couldn’t make plans for the future due to the unpredictability of the disease. Figure 1 displays the distribution of responses to the MUIS items ranked by percentage of agreement with the corresponding statement.
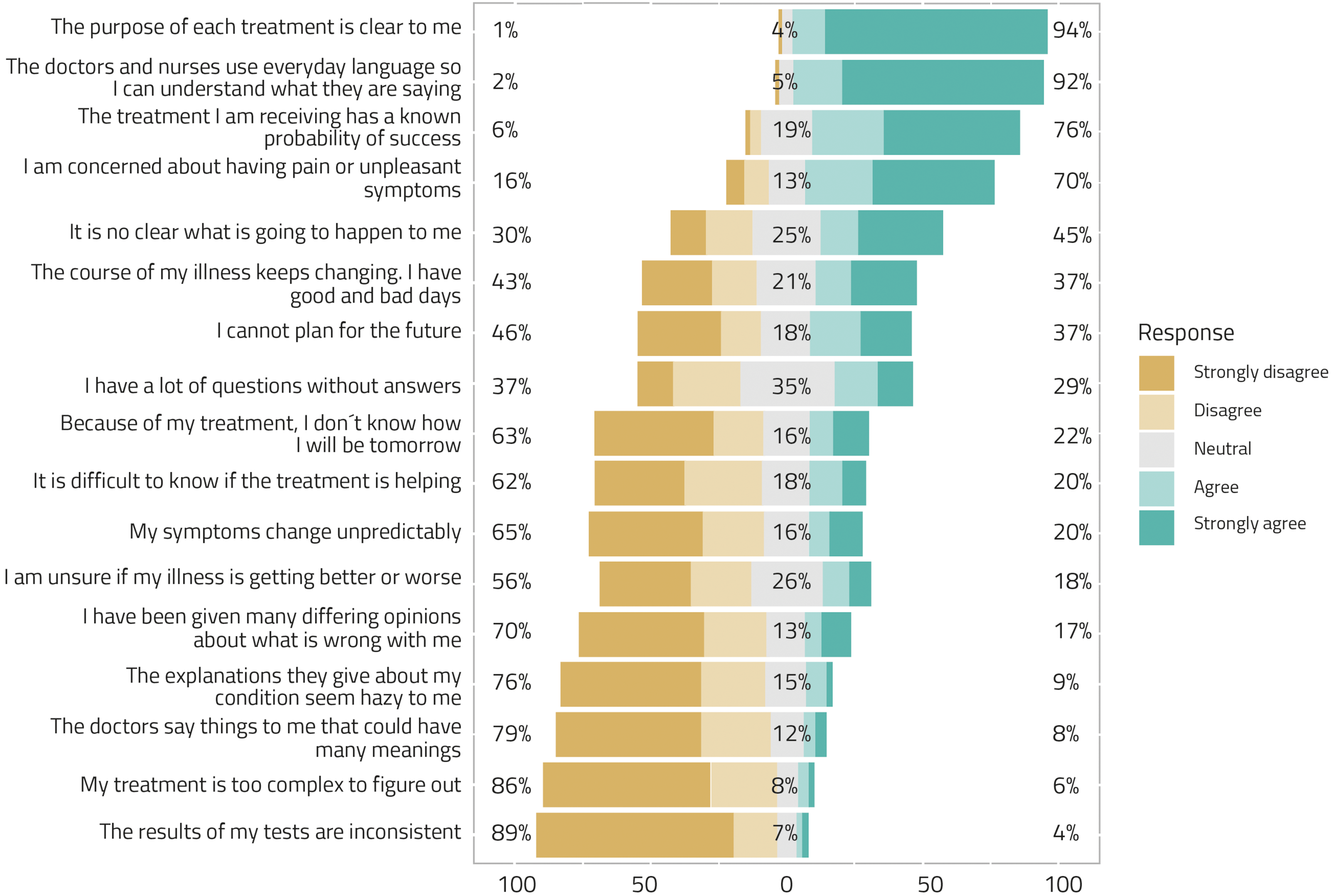
Seventeen-item MUIS (N = 204).
Dimensionality and item characteristics
The Mokken analysis suggested two dimensions for the 17-item MUIS, one general dimension indicating uncertainty and represented by 12 items, and a minor dimension composed of three items indicating confidence instead of uncertainty (item #5: “the purpose of each treatment is clear to me”; item #16: “the treatment I am receiving has a known probability of success”; item #17: “the doctors and nurses use everyday language so I can understand what they are saying”). Another two items were not classifiable into any dimension (item #1: “I have a lot of questions without answers”; item #12: “I have been given many differing opinions about what is wrong with me”).
Table 1 showed the endorsement frequencies and scalability coefficients of the MUIS scale once the nonclassifiable and minor dimension items were removed. This reduced 12-item scale shows appropriate scalability coefficients both at the item (all >0.30) and the overall scale (0.37) level. The number of model violations indicates that this reduced scale is compatible with an MHM but not with a DMM.
Endorsement frequencies and scalability coefficients of the 12-item version of the MUIS (N = 204).
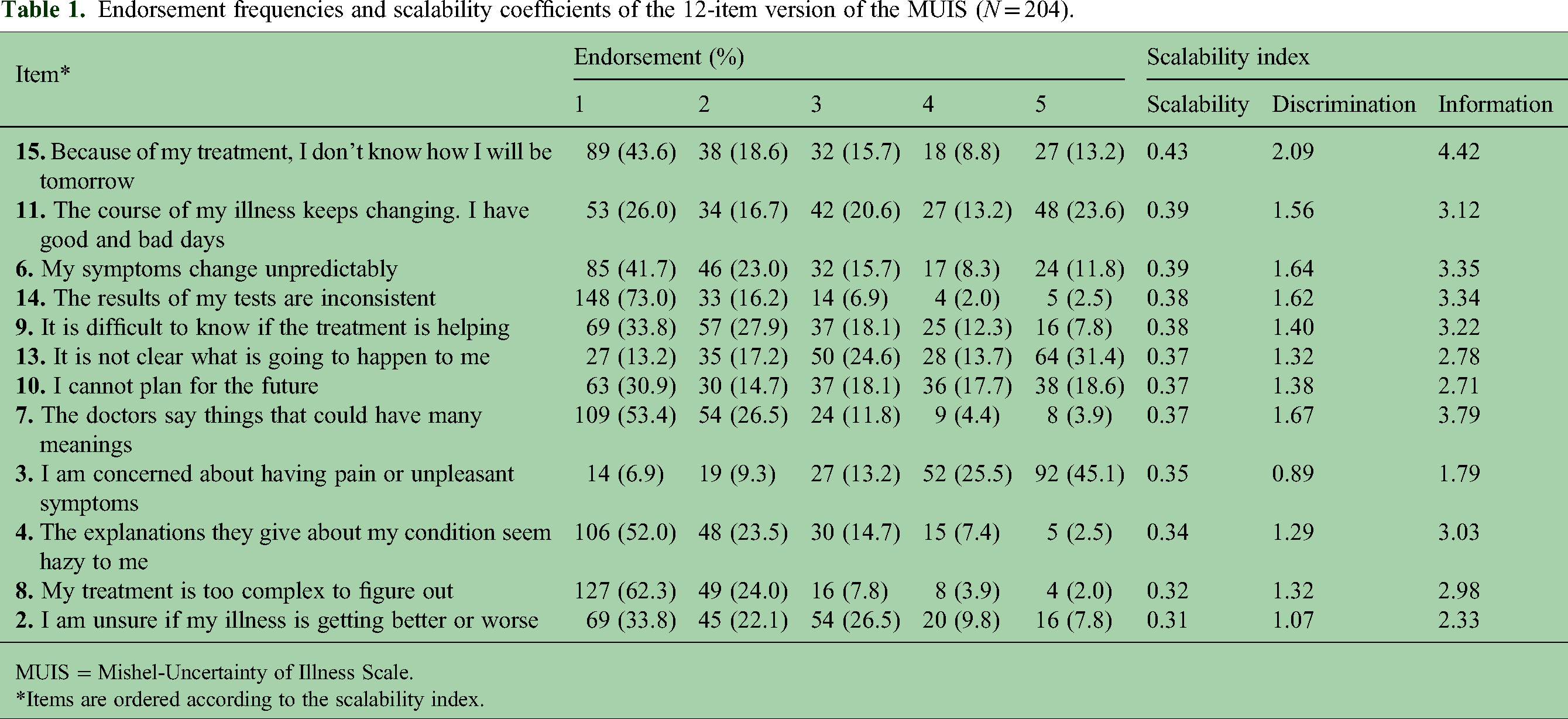
MUIS = Mishel-Uncertainty of Illness Scale.
Items are ordered according to the scalability index.
Discussion
Uncertainty about the risk of physical disability and cognitive impairment can lead to psychological distress and anxiety, as RRMS patients often face the unknown and try to make sense of their situation.2–5 In this context, there is a lack of validated tools to identify and quantify the pervasive uncertainty experienced by individuals facing the unpredictable progression of the disease. 1
Our results agree with those reported in the Italian validation of the MUIS in patients with MS but further reduced the scale from 17 items representing two dimensions of ambiguity (10 items) and inconsistency (seven items) to 12 items representing a single dimension of uncertainty or unpredictability. 1 This reduced scale includes items with different discrimination in measuring the construct of interest (illness-related uncertainty) and allows an aggregate score to be used to reflect the uncertainty level of a patient across the natural history of illness. When applied to nonhospitalized patients, the reduction of the number of scale items from the 33 original items to 17 did not decrease the number of dimensions tapped with it.1,7 In contrast, our proposal for RRMS patients already beyond the early stage of the disease fits a single dimension of disease uncertainty.
The study population included a sample of patients with RRMS with at least three years of disease history and low physical disability. Therefore, the results might not be generalizable to individuals in the early or advanced stages of the disease, where either the assimilation of the diagnosis's impact is still recent or significant physical and cognitive disabilities have already manifested.
Conclusions
This study assessed the dimensionality and item characteristics of the MUIS scale within a cross-sectional study of patients with RRMS. The suggested 12-item reduced version showed appropriate psychometric properties at the item level for scalability, discrimination, and information, as well as including a single dimension that allows computation of a total uncertainty score. It may constitute a valuable and easy-to-implement instrument to measure illness-related uncertainty in patients with RRMS.
Footnotes
Acknowledgments
The authors are most grateful to all patients, neurologists, and nurses participating in the study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ES and JM are employees of Roche Farma Spain. JS received honoraria for lecturing, consulting, or travel expenses from Biogen, Merck, Teva, Novartis, BMS, Janssen, Almirall, Roche, and Sanofi. JMM served on scientific advisory boards and/or received speaking honoraria, research funding and support to attend scientific meetings from Biogen, Merck, Novartis, Roche, and Teva. FJB received compensation for consulting services and speaking honoraria from Almirall, Biogen, Genzyme, Merck, Novartis, Roche, Sanofi, and Teva. AA received compensation for consulting services from Biogen, BMS, Sanofi, Roche, Janssen, and Novartis; and speaking honoraria from Biogen, BMS, Sanofi, Roche, Janssen, Merck, Almirall, and Novartis. JLSM received support to attend scientific meetings from Novartis, Merck, and Biogen; speaking honoraria from Biogen, Novartis, Sanofi, Merck, Almirall, Bayer, and Teva; and participated in clinical trials from Biogen, Merck, and Roche. LB received compensation for consulting services, speaking honoraria, and support to attend scientific meetings from Bayer, Celgene, Biogen, Sanofi, Merck, Novartis, Roche, Almirall, and Teva. MGG received speaking honoraria from Biogen, BMS, Janssen, Merck, Mylan, Novartis, Roche, Sanofi, and Bial. SE received consulting fees and lecture honoraria from Biogen, Novartis, Sanofi, Merck, Roche, and Almirall. CC received compensation for consulting services, speaking honoraria and support to attend scientific meetings and courses from Merck, Teva, Sanofi, Novartis, Biogen, Roche, and BMS. EFD received honoraria and travel expenses for participation in scientific meetings and advisory boards from Almirall, Biogen, Merck, Roche, and Sanofi. OC received honoraria for speaker services and advisory boards from Sanofi, Roche, Janssen, and Merck. AO received research grants, travel support, advisory activities, and honoraria for speaking from Almirall, Biogen, BMS, Merck, Mylan, Novartis, Roche, Sanofi, and Teva. ALR received speaker and consultation fees, and congress travel support from Biogen, Janssen, Merck, Novartis, Roche, and Sanofi. AM received research grants, travel support or honoraria for speaking engagements from Biogen, Merck, Novartis, Roche, Sanofi, and Teva. EA received speaking honoraria from Roche, Novartis, Merck, Sanofi, and Biogen. The rest of the authors declared no potential conflicts of interest. The abstract of this article was presented at the European Congress of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) as a poster presentation with interim findings (PCR178; Copenhagen, Denmark; November 14, 2023).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by Roche Farma Medical Department, Spain (ML43469).
