Abstract
Background
Interferon beta-1a remains an important treatment option for multiple sclerosis, particularly when safety or tolerability concerns may outweigh the benefits of higher-efficacy disease-modifying therapies. The five-year phase 4 Plegridy Observational Program (POP) study (NCT02230969) collected data on real-world safety and effectiveness of Plegridy® (peginterferon beta-1a) treatment in patients with relapsing multiple sclerosis.
Objective
To explore the real-world safety and effectiveness of peginterferon beta-1a in patients with relapsing multiple sclerosis, including factors influencing treatment discontinuation.
Methods
Data were collected prospectively from patients ≥ 18 years old with relapsing multiple sclerosis for overall population analysis and for subpopulations including newly/previously diagnosed patients, age, and experience with peginterferon beta-1a. Outcome measures included annualized relapse rates, adverse events, and predictors of time to treatment discontinuation.
Results
Mean (SD) treatment duration in the overall population (N = 1172) was 896.0 (733.15) days. Incidence of adverse events was higher in new than experienced users (79.4% vs. 57.0%). New users were more likely than experienced users to discontinue (hazard ratio = 1.60; P < 0.0001). The adjusted annualized relapse rate was 0.09, and at the end of 5 years, 77.1% of patients were relapse-free.
Conclusions
Peginterferon beta-1a is an effective therapy for managing relapsing multiple sclerosis. The identification of predictors of discontinuation can help inform strategies to enhance treatment persistence.
Keywords
Introduction
Interferon beta-1a was among the first disease-modifying therapies (DMTs) approved for treating patients with relapsing forms of multiple sclerosis (MS). Despite the introduction of many new therapies for patients with MS since the introduction of interferons, interferon beta-1a therapies remain an important DMT class in the MS landscape, particularly for patients for whom safety or tolerability concerns may outweigh the benefits of higher-efficacy DMTs.1,2 Interferon treatment also demonstrates broad antiviral properties, including the possibility of affecting Epstein Barr virus infection, the primary suspected cause in MS etiology.3,4 In addition, interferon treatment may promote vaccine responses to severe acute respiratory syndrome coronavirus 2 vaccination, 5 a benefit that is particularly relevant in the post-coronavirus disease 2019 pandemic environment.
Multiple formulations and modes of administration of interferon beta-1a have been brought to market, including a subcutaneous pegylated version, peginterferon beta-1a. Pegylation extends the elimination half-life and increases systemic exposure of interferon beta-1a, which permits a reduced dosing frequency. 6 The effectiveness and safety of peginterferon beta-1a 125 μg every 2 weeks for patients with relapsing MS (RMS) were established during the phase 3 clinical trials ADVANCE and ATTAIN, which evaluated patients for up to nearly 6 years.7–9
Differences in patient populations and standards of care in real-world settings compared to controlled clinical trials mean that clinical trial data may not be generalizable to the broader patient population or long-term treatment in the postmarketing setting. For instance, differences in age demographics or racial representation are a well-known disparity between clinical trials and real-world populations. As of 31 January 2023, 86,219 patients have been prescribed Plegridy representing 187,658 patient years of exposure. The mean age of patients treated with interferon beta-1a in the postmarketing setting was estimated to be 45–50 years old in 2017, and more than half of people living with an MS diagnosis are over 55. 10 However, the mean age of patients in the ADVANCE trial was only 37, and no specific analyses have been reported for the subgroup of “older” patients included in the trial. 8 Therefore, real-world studies with more diverse patient populations are needed to assess the effectiveness, safety, and tolerability of peginterferon beta-1a in older patients, as well as in other subgroups.
The five-year, phase 4 Plegridy Observational Program (POP) collected data on the safety and effectiveness of peginterferon beta-1a treatment in patients with RMS in the real-world clinical setting. This final analysis from POP expands on clinical trial data to explore the real-world safety and effectiveness of peginterferon beta-1a in patients with RMS, including newly diagnosed and non-newly diagnosed patients, younger and older patients, and new and experienced users of peginterferon beta-1a. Injectable interferon therapies, including peginterferon beta-1a, are associated with injection site reactions (ISRs) and flu-like symptoms (FLSs), which can lead to treatment discontinuation.7,11–15 To explore factors affecting treatment persistence in the real-world setting, this analysis additionally assessed predictors of discontinuation and FLS severity over time.
Methods
Study design
POP (ClinicalTrials.gov no. NCT02230969) was fully enrolled and had over 150 active sites across 14 countries. The POP protocol was approved by the institutional review board at each site, and the studies were conducted in accordance with the International Council for Harmonisation Guidelines on Good Clinical Practice and the World Medical Association Declaration of Helsinki. This fifth and final prespecified analysis includes data from 12 November 2014 to 23 March 2022. Enrolled patients were required to be at least 18 years old and have a diagnosis of RMS. Patients could be either newly or currently prescribed subcutaneous peginterferon beta-1a. Patients could not be concurrently enrolled in any clinical trial of an investigational product, though participation in a non-interventional study was allowed if participation did not interfere with the POP study protocol or was unlikely to affect the ability to comply with the POP study protocol. All participants provided written informed consent prior to the evaluation of their study eligibility.
Patient subgroups
Outcomes were assessed in the overall population as well as in subgroups of patients that included newly and non-newly diagnosed patients, older and younger patients, and new and experienced users of peginterferon beta-1a. Newly diagnosed patients were defined as patients who were diagnosed with RMS ≤ 1 year prior to POP study consent and had not yet received treatment with a DMT for MS. Non-newly diagnosed patients were defined as those who were diagnosed > 1 year prior to study consent and/or had prior treatment with an MS DMT. Older patients were defined as patients aged ≥ 50 years at enrollment, and younger patients were defined as those aged < 50 years at enrollment. New peginterferon beta-1a users were defined as patients who had initiated peginterferon beta-1a ≤ 31 days prior to study consent, at study consent, or after study consent. Experienced users were defined as patients who initiated peginterferon beta-1a > 31 days prior to study consent. 16
Endpoints, assessments, and statistical analyses
Demographic and clinical characteristics were collected at enrollment and summarized using descriptive statistics. Patients were followed for a maximum of 60 months, regardless of discontinuation of peginterferon beta-1a, or until withdrawal from the study, loss of follow-up, or death, whichever occurred first. Assessments were performed approximately every 3 months (± 4 weeks) during the first year of follow-up and every 6 months (± 4 weeks) during years 2–5 of follow-up.
Safety was evaluated as the incidence of treatment-emergent adverse events (AEs) and treatment-emergent serious AEs (SAEs) in patients who met eligibility criteria and received ≥ 1 dose of peginterferon beta-1a. Symptoms were classified using preferred terms (PTs) and grouped by system organ class (SOC) according to the Medical Dictionary for Regulatory Activities. For the calculation of FLS and ISR incidence, an event presenting with multiple symptoms was counted as only a single event. FLS severity was determined through the patient-reported 3-Level European Quality-of-Life 5-Dimension (EQ-5D-3L) health questionnaire. Predictors of time to treatment discontinuation were analyzed using a Cox regression model.
Effectiveness was evaluated in eligible patients who were diagnosed with RMS and received ≥ 1 dose of peginterferon beta-1a. Because this was an observational study, the diagnosis of RMS was based on the physicians’ discretion. Annualized relapse rates (ARRs) were calculated using a Poisson regression model adjusting for baseline relapse rate and age at enrollment. Proportions of patients remaining free of relapse over 5 years were estimated using Kaplan-Meier methods. Confirmed disability worsening was defined as a ≥ 1.0-point increase from a baseline Expanded Disability Status Scale (EDSS) score ≥ 1.0 or a ≥ 1.5-point increase from a baseline EDSS score of 0.0 sustained for at least 24 weeks. Clinical no evidence of disease activity (NEDA) was defined as no relapses and no 24-week-confirmed disability worsening over the study duration. MRI activity was not considered in the definition of NEDA for this study.
Comparisons of safety and effectiveness within each stratum (newly diagnosed vs. previously diagnosed, younger vs. older, new users vs. experienced users) were descriptive.
Results
Patient enrollment and baseline characteristics
Enrollment in POP was completed in January 2017 with a total of 1208 patients enrolled. The final safety analysis population included 1173 patients, and the effectiveness analysis population included 1172 patients. In the effectiveness population, 11.3% of patients had ≤ 12 months of follow-up, 15.1% had > 12 to ≤ 24 months, 7.4% had > 24 to ≤ 36 months, 5.7% had > 36 to ≤ 48 months, and 60.4% had > 48 months, with 617 (52.6%) patients completing the entire five-year study. The mean (SD) peginterferon beta-1a treatment duration in the effectiveness population was 896.0 (733.15) days.
In the overall effectiveness population at baseline, patients had a mean (SD) age of 43.9 (12.5) years, a mean (SD) EDSS score of 1.8 (1.5), and a mean (SD) of 0.5 (0.7) relapses in the prior year (Table 1). At baseline, although the mean age between newly diagnosed and previously diagnosed patients was similar, the newly diagnosed cohort was younger than the previously diagnosed cohort. Newly diagnosed patients had more relapses in the prior year and less disability compared to previously diagnosed patients (Table 1). In comparison with younger patients, older patients were older age at MS diagnosis, had fewer relapses in the prior year, and had a higher mean EDSS score at baseline (Supplemental Table 1). New and experienced users had similar ages at enrollment and MS diagnosis, as well as similar baseline disability and number of relapses in the prior year (Supplemental Table 1).
Baseline demographics and disease characteristics in the overall population and the newly diagnosed and non-newly diagnosed subgroups.

DMT, disease-modifying therapy; EDSS, Expanded Disability Status Scale; MS, multiple sclerosis.
Data were available for 289 newly diagnosed and 869 non-newly diagnosed patients.
Baseline EDSS data were available for 167 newly diagnosed and 388 non-newly diagnosed patients.
Safety
Treatment-emergent AEs and SAEs
In the overall population, the incidence of treatment-emergent AEs was 68.3%, and treatment-emergent SAEs were reported in 9.2% of patients (Table 2). In the overall population, nervous system disorders, infections and infestations were the most common class of SAEs, occurring in 2.2% and 2.0% of patients, respectively, (Supplemental Table 2). Treatment discontinuation due to AEs occurred in 26.3% of patients, most often due to FLS (9.5%) or ISRs (6.0%).
Treatment-emergent adverse events.

AE, adverse event; FLS, flu-like symptom; ISR, injection site reaction; MS, multiple sclerosis; SOC, system organ class; TEAE, treatment-emergent adverse event; TESAE, treatment-emergent serious adverse event.
AEs are presented by the Medical Dictionary for Regulatory Activities (MedDRA) SOC.
Deaths included respiratory failure (unknown if cause related to MS), sepsis (unrelated to MS), ST-elevation myocardial infarction (unrelated to MS), and MS complications.
The incidence of treatment-emergent AEs was similar in newly diagnosed and non-newly diagnosed patients (68.9% vs. 68.1%; Table 2). Treatment discontinuation due to AEs also occurred in similar proportions of newly diagnosed and non-newly diagnosed patients (24.7% vs. 26.8%). Treatment-emergent SAEs were reported in 8.0% of newly diagnosed patients and 9.6% of non-newly diagnosed patients.
The incidence of treatment-emergent AEs was similar between older and younger patients (68.2% vs. 68.3%; Table 2). Treatment discontinuation due to treatment-emergent AEs occurred in similar proportions of older and younger patients (26.4% vs. 26.2%). The incidence of treatment-emergent SAEs was higher in patients aged ≥ 50 years than in those aged <50 years (13.8% vs. 6.9%). The increased incidence of SAEs in the older patient group was not linked to a singular PT or SOC. The most common SAEs for both older and younger patient subgroups were nervous system disorders, occurring in 3.8% and 1.4% of patients, respectively. MS relapse and pseudo relapse were the most frequent occurring PTs within the nervous system disorder SOC.
The incidence of treatment-emergent AEs was higher in new users than in experienced users (79.4% vs. 57.0%; Table 2). A higher proportion of new users (33.0%) than experienced users (19.4%) discontinued treatment due to treatment-emergent AEs. The incidence of treatment-emergent SAEs was 7.8% in new users and 10.7% in experienced users.
Flu-like symptom
The incidence of FLS in the overall population was 47.9%, and 9.5% of patients discontinued due to FLS (Table 2). The incidence of FLS was somewhat higher in newly diagnosed than non-newly diagnosed patients (52.2% vs. 46.5%; Table 2), and treatment discontinuation due to FLS occurred in similar proportions of newly diagnosed and non-newly diagnosed patients (9.7% vs. 9.4%%). The incidence of FLS was slightly lower in older patients than in younger patients (42.3% vs. 50.7%; Table 2), and treatment discontinuation due to FLS occurred in similar proportions of patients aged ≥ 50 years and < 50 years (8.7% vs. 9.8%). The incidence of FLS was higher in new users than in experienced users (58.4% vs. 37.3%; Table 2). In addition, a higher proportion of new users (12.7%) than experienced users (6.2%) discontinued treatment due to FLS.
FLS severity
From baseline to 12 months, 77%–83% of patients reported no FLS symptoms or mild symptoms, and 17%–23% reported moderate or severe symptoms (Supplemental Figure 1A). A smaller proportion of patients taking FLS-reducing medications reported moderate or severe symptoms from baseline through 12 months in comparison with all patients reporting FLS after their most recent dose of peginterferon beta-1a (9%–13%; Supplemental Figure 1B). By 6 years, 2.6% of users reported mild FLS symptoms, whereas only 1.8% of users reported moderate to severe FLS symptoms, and only 1.1% reported moderate symptoms after taking FLS-reducing medication. Patients reported that mild FLS more frequently lasted ≤ 24 h than >24 h during the first 12 months of treatment (Figure 1A). Conversely, moderate FLS more frequently lasted > 24 h than ≤ 24 h (Figure 1B).

Percentage of peginterferon beta-1a users who experienced (A) mild or (B) moderate flu-like symptoms (FLSs) after their last injection by FLS duration.
Injection site reactions
The incidence of ISRs in the overall population was 41.3%, and 6.0% of patients discontinued due to ISRs (Table 2). The incidence of ISRs was similar in newly diagnosed and non-newly diagnosed patients (37.8% vs. 42.4%; Table 2). Treatment discontinuation due to ISRs also occurred in similar proportions of newly diagnosed and non-newly diagnosed patients (4.0% vs. 6.6%). The incidence of ISRs was similar between older and younger patients (43.6% vs. 40.1%; Table 2). Treatment discontinuation due to ISRs occurred in similar proportions of patients aged ≥ 50 years and < 50 years (5.5% vs. 6.9%). The incidence of ISRs was higher in new users than in experienced users (52.6% vs. 29.7%; Table 2). Additionally, a higher proportion of new users (9.1%) in comparison with experienced users (2.7%) discontinued treatment because of ISRs.
Predictors of discontinuation
A univariate analysis determined several covariates that were predictive of a faster time to discontinuation (Figure 2). New users of peginterferon beta-1a were significantly more likely than experienced users to discontinue peginterferon beta-1a over the course of follow-up (hazard ratio [HR] = 1.60; new users 95% CI [1.37, 1.86]). Patients presenting with ISRs were significantly more likely than those without ISRs to discontinue peginterferon beta-1a over the course of follow-up (HR = 1.50; P < 0.0001). Additional significant predictors of discontinuation included prior oral DMT use, more relapses in the year prior to study initiation, prior intravenous DMT use, fewer years since diagnosis, younger age at enrollment, and not having prior use of an injectable DMT.

Univariate analysis of predictors of time to discontinuation of peginterferon beta-1a. Cox regression model adjusting time to discontinuation/switch for the following key covariates: age, sex, CCI, comorbid conditions, MS-related symptoms, and MS severity score. aP value determined by analysis of maximum likelihood estimates. Bold text denotes a statistically significant result, P < 0.05.
Effectiveness
Relapses
In the overall population, the ARR was 0.09 (95% confidence interval [CI] 0.08–0.10). At the end of Years 1–5 years of the study, 86.6%, 82.8%, 79.7%, 78.3%, and 77.1% of patients in the overall population were relapse-free (respectively). ARRs in newly diagnosed and non–newly diagnosed patients were 0.09 (95% CI 0.07–0.11) and 0.09 (95% CI 0.08–0.10), respectively (Figure 3A). At 5 years, 70.9% of newly diagnosed patients and 79.3% of non-newly diagnosed patients were relapse-free (Figure 4A). ARRs in older and younger patients were 0.06 (95% CI 0.05–0.08) and 0.11 (95% CI 0.10–0.13), respectively (Figure 3B). At 5 years, 85.1% of older patients and 73.1% of younger patients were relapse-free (Figure 4B). ARRs in new and experienced users were 0.10 (95% CI 0.08–0.11) and 0.08 (95% CI 0.06–0.09), respectively (Figure 3C). At 5 years, 73.9% of new users and 80.4% of experienced users were relapse-free (Figure 4C).

Adjusted annualized relapse rates in (A) newly diagnosed and non-newly diagnosed patients, (B) patients < 50 and ≥ 50 years of age, and (C) new users and experienced users. Adjusted ARRs based on a negative binomial regression model adjusting for baseline relapse rate and age at enrollment.
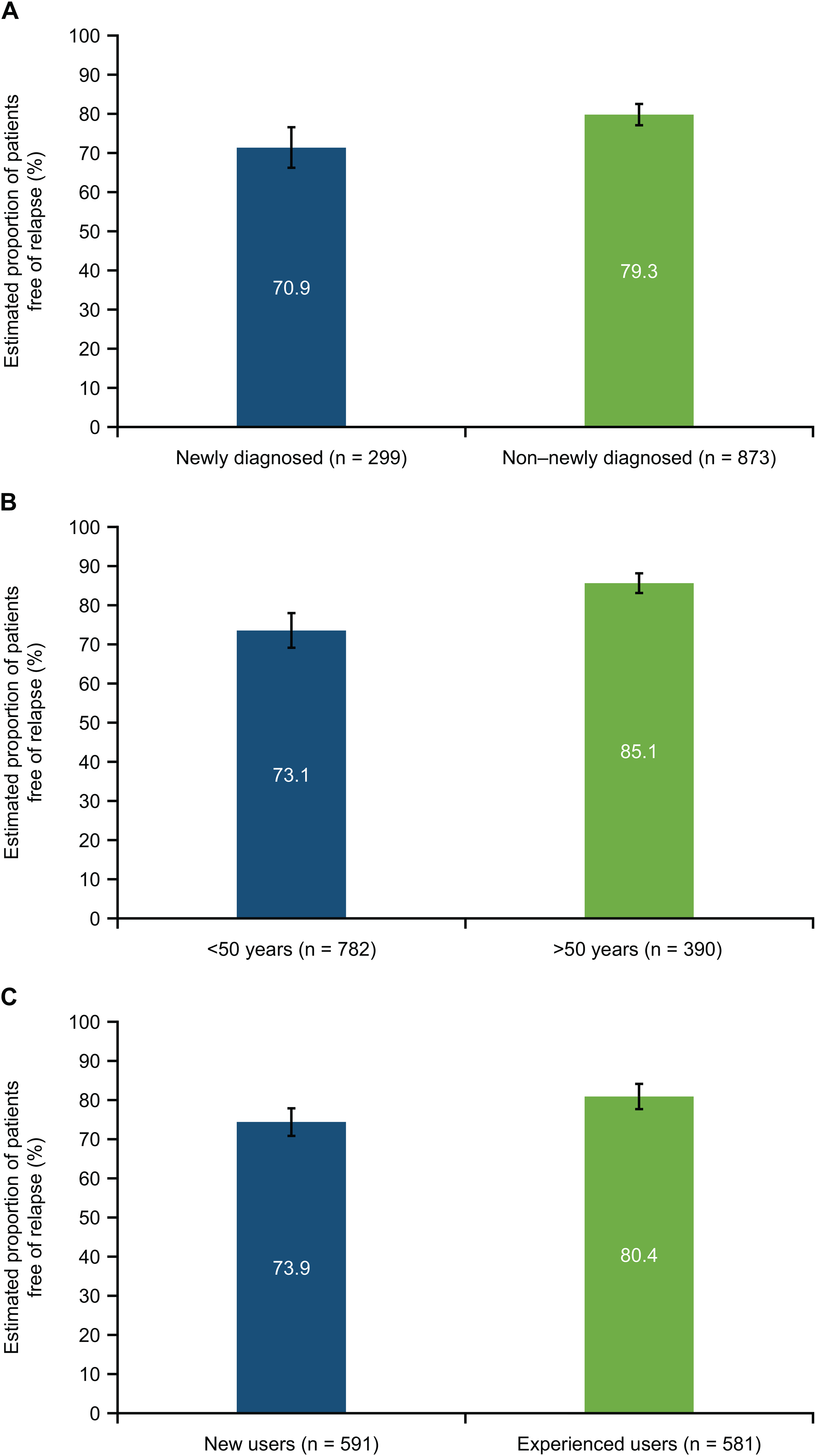
Estimated proportions of patients free of relapse at 5 years for (A) newly diagnosed and non-newly diagnosed patients, (B) patients < 50 years and ≥ 50 of age, and (C) new users and experienced users.
Disability
At 6 years, the estimated cumulative proportion of patients with confirmed disability worsening was 3.8% in the overall population. Disability worsening was 3.7% in newly diagnosed patients and 3.8% in non-newly diagnosed patients; 5.1% in older patients and 3.1% in younger patients; and 2.7% in new users and 4.8% in experienced users.
Clinical NEDA
Over 6 years, the percentage of patients with clinical NEDA was 66.7% (370 of 555) in the overall population. Within each subgroup, the percentage of patients with clinical NEDA over 6 years was 62.9% (105 of 167) in newly diagnosed patients and 68.3% (265 of 388) in non–newly diagnosed patients, 65.8% (269 of 409) in younger patients and 69.2% (101 of 146) in older patients, and 64.9% (192 of 296) in new users and 68.7% (178 of 259) in experienced users (Figure 5).

Percentage of patients in each subgroup with clinical no evidence of disease activity (NEDA) over 6 years.
Discussion
The safety profile in POP is consistent with that observed in the phase 3 ADVANCE/ATTAIN studies.2–4 No new or unexpected safety signals were reported. Consistent with the phase 3 ADVANCE and ATTAIN studies,2–4 the most common AEs were ISRs and FLS. The incidence of AEs was similar in newly diagnosed and non-newly diagnosed patients, and although the incidence of AEs was similar in younger and older patients, older patients had a higher incidence of SAEs than younger patients. This higher incidence of SAEs may be due to increased comorbidities among older patients. 17
The incidence of AEs, especially FLS and ISRs, was higher in new patients than in experienced patients. New patients may have less experience with FLS and ISRs, and a lack of effective mitigation and management strategies may have contributed to the higher incidence of AEs reported in this population. However, because experienced patients may be less likely to report minor AEs, self-selection cannot be ruled out as a factor contributing to the lower incidence of AEs among experienced patients.
New users of peginterferon beta-1a, younger patients, and those experiencing ISRs were more likely to discontinue peginterferon beta-1a. These results indicate that new users of peginterferon beta-1a treatment as well as younger patients may benefit from additional information on ISR mitigation and management through discussions with their healthcare professionals and support the results communicated by several expert panels.12–15 Alternative modes of administration, including intramuscular peginterferon beta-1a, may also be an option. 18
Peginterferon beta-1a is an effective therapy for patients in the real-world clinical setting. In the overall population and in all subgroups, the adjusted ARR was ≤ 0.11, and the estimated proportion of patients remaining relapse-free over 5 years was >70%. At 6 years, the estimated cumulative probability of disability worsening was 3.8% in the overall population and <6% across all subgroups. Over 6 years, more than 62% of newly diagnosed and non–newly diagnosed patients experienced clinical NEDA. Similar to what has been reported for intramuscular interferon beta-1a, 19 no significant differences were found between older and younger patient subgroups in terms of effectiveness. In addition to first-line therapy for RRMS, peginterferon beta-1a may also serve as an effective alternative treatment for those patients who need de-escalation strategies after immunosuppression with monoclonal antibodies, cladribine, or S1P-modulators. The robust safety profile makes peginterferon beta-1a a good candidate for patients who need ongoing immunomodulation at a higher age.
Limitations of this analysis include that POP is an observational study with an open-label design. This study is not sufficiently powered for statistical comparisons between groups. Data on disability, as measured by EDSS scores, were limited and only recorded for relatively few patients at later timepoints. However, many more patients had complete data for this final analysis compared with previous analyses, increasing the robustness of the NEDA analysis.
These results confirm and extend previous studies of peginterferon beta-1a in patients enrolled in POP and indicate that peginterferon beta-1a has the potential to provide treatment benefits to patients with RMS, including newly diagnosed patients, patients ≥ 50 years of age, and new users of peginterferon beta-1a. The safety data provide important information on how AEs such as FLS and ISRs affect adherence and persistence and can therefore inform mitigation strategies.
Supplemental Material
sj-docx-1-mso-10.1177_20552173241238632 - Supplemental material for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program
Supplemental material, sj-docx-1-mso-10.1177_20552173241238632 for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program by Marco Salvetti, Sibyl Wray, Gereon Nelles, Nicholas Belviso, Achint Kumar, Thijs Koster, Wanda Castro-Borrero, Megan Vignos and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-2-mso-10.1177_20552173241238632 - Supplemental material for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program
Supplemental material, sj-docx-2-mso-10.1177_20552173241238632 for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program by Marco Salvetti, Sibyl Wray, Gereon Nelles, Nicholas Belviso, Achint Kumar, Thijs Koster, Wanda Castro-Borrero, Megan Vignos and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-3-mso-10.1177_20552173241238632 - Supplemental material for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program
Supplemental material, sj-docx-3-mso-10.1177_20552173241238632 for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program by Marco Salvetti, Sibyl Wray, Gereon Nelles, Nicholas Belviso, Achint Kumar, Thijs Koster, Wanda Castro-Borrero, Megan Vignos and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-ai-4-mso-10.1177_20552173241238632 - Supplemental material for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program
Supplemental material, sj-ai-4-mso-10.1177_20552173241238632 for Safety and clinical effectiveness of peginterferon beta-1a for relapsing multiple sclerosis in a real-world setting: Final results from the Plegridy Observational Program by Marco Salvetti, Sibyl Wray, Gereon Nelles, Nicholas Belviso, Achint Kumar, Thijs Koster, Wanda Castro-Borrero, Megan Vignos and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
Luke Ward, PhD, of Ashfield MedComms, an Inizio Company, wrote the first draft of the manuscript on the basis of input from the authors. Celia Nelson of Ashfield MedComms copyedited and styled the manuscript per journal requirements.
Credit authorship contribution statement
Marco Salvetti: conceptualization, methodology, investigation, writing–review and editing, and supervision. Sibyl Wray: conceptualization, methodology, investigation, writing–review and editing, and supervision. Gereon Nelles: conceptualization, methodology, investigation, writing–review and editing, and supervision. Nicholas Belviso: formal analysis, data curation, writing–review, and editing. Achint Kumar: writing–review and editing. Thijs Koster: writing–review and editing, and supervision. Wanda Castro-Borrero: writing–review and editing, and supervision. Megan Vignos: writing–review and editing, and supervision.
Data sharing statement
Data requests can be submitted to the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MS has received grant support and/or speaker honoraria from Biogen, Merck, Novartis, Roche, Sanofi, and Teva Neuroscience. SW is a paid consultant, speaker, and/or contract researcher for Biogen, Celgene/Bristol Myers Squibb, EMD Serono, Genentech/Roche, Genzyme/Sanofi, Novartis, Receptos, and TG Therapeutics. GN has received speaker honoraria from Bayer, Biogen, Celgene, Merck, Novartis, and Roche. NB, AK, WC-B, and MV are employees of and may hold stock and/or stock options in Biogen. TK was an employee of Biogen at the time of these analyses and may hold stock and/or stock options in Biogen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The POP study was funded by Biogen. Biogen also provided funding for medical writing support in the development of this manuscript. Biogen reviewed the manuscript and provided feedback to the authors. The authors provided their final approval of all content.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
