Abstract
Background
Between 29% and 67% of neuromyelitis optica spectrum disorder patients have cognitive alterations.
Objective
To assess the frequency of cognitive impairment in patients with neuromyelitis optica spectrum disorder in Mexico using the Brief International Cognitive Assessment for Multiple Sclerosis.
Methods
We evaluated 40 neuromyelitis optica spectrum disorder patients and 40 healthy controls from Mexico.
Results
28 (70.0%) patients with neuromyelitis optica spectrum disorder had cognitive impairment in two or more cognitive domains. Student´s T test showed statistically poor performance by neuromyelitis optica spectrum disorder patients compared to healthy controls on all three neuropsychological test scores. This significant difference was observed on the Symbols Digit Modalities Test (
Conclusions
This study showed that 70% of neuromyelitis optica spectrum disorder patients exhibited cognitive impairment in two or more cognitive domains. Determining the frequency of cognitive impairment will guide the decision of Neuropsychologists in planning cognitive rehabilitation across various domains.
Keywords
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune inflammatory disease of the central nervous system that primarily affects the optic nerve and spinal cord, usually with a severe and relapsing course.1,2 NMOSD onset is at about 39 years of age and is more prevalent among women. 3 In 2022, a prevalence of 0.71/100,000 (95% CI 0.55–0.92) was estimated in western Mexico. 4
Patients with NMOSD often experience a higher number of deficits, such as physical disability, depression, anxiety, cognitive impairment (CI), pain, and fatigue that reduces the quality of life.5–13
Investigations performed in patients with NMOSD have allowed the establishment of the frequency of cognitive alterations.6,14,15 Cognitive deficits are present in 29%–67% of NMOSD patients.16–18 Studies conducted in this population have identified the presence of alterations in attention, memory, processing speed, verbal fluency, verbal memory, and executive function.19–21 This characterization of cognitive alterations has been made possible by the existence of neuropsychological batteries designed for cognitive assessment.
In general, numerous cognitive batteries and neuropsychological tests have been developed and validated in different countries for assessing cognitive functions in patients with multiple sclerosis (MS). 22 However, there is no expert consensus on which neuropsychological batteries are better to assess cognitive function in NMOSD patients.19,23 One of the most widely used batteries to facilitate the assessment of cognitive function in MS is the Brief International Cognitive Assessment for MS (BICAMS).. 24 This clinical tool assesses processing speed, verbal memory, and visuospatial memory. 25 Several studies have examined CI in NMOSD patients. For instance. Salama et al. 26 conducted a transversal study that included 20 NMOSD patients and 18 healthy controls (HCs). This study identified a frequency of CI in 75% of patients with NMOSD, with deficits mostly seen in processing speed and verbal memory domains. 26 In 2023, Lopes et al. 27 conducted a retrospective study that included 19 NMOSD patients, 27 MS patients, and 18 HCs. The study identified that patients with NMOSD had significantly lower cognitive performance than the HCs group. 27
Despite the increasing number of investigations on NMOSD populations, there is a lack of research regarding cognitive alterations in patients with NMOSD. At the time of publication of this manuscript, there were no studies identifying the prevalence of CI in NMOSD populations from Mexico. Therefore, the objective of this study was to evaluate the frequency of CI in patients with NMOSD in Mexico using the BICAMS.
Methods
Participants
The present study included 80 Mexican participants who were divided into two groups. The first group consisted of 40 Mexican adults diagnosed with NMOSD. The Second group consisted of 40 HCs. These 40 HCs were selected to match the NMOSD sample on demographic characteristics. The groups did not differ with regard to age (NMOSD group = 39.50 ± 10.61 years; HCs group = 39.98 ± 10.01 years,
The inclusion criteria for patients diagnosed with NMOSD were as follows: (a) A diagnosis of NMOSD according to the 2015 international consensus diagnostic criteria for NMOSDs. 13 All medical records of patients with NMOSD were reviewed by 3 neurologist researchers; (b) a signed informed consent form; (c) an age between 18 and 60 years at the time of the cognitive assessment; (d) do not have visual (must be better than 20/200 in at least one eye) or auditory limitations to performed the tests; (e) dominant hand strength had to be 4+/5 or more in the modified Medical Research Council (mMRC) scale. This information was corroborated in the neurology consultations; and (f) not having an intellectual disability. We used the Wechsler Adult Intelligence Scale (WAIS-IV), and it was determined by neuropsychologist researchers. The exclusion criteria were as follows: (a) History of a neurological or psychiatric diagnosis; (b) substance and/or alcohol consumption; (c) a relapse within the last 30 days of cognitive assessment; and (d) treatment with corticosteroids within the last 30 days.
The inclusion criteria for HCs were as follows: (a) Being born and currently living in Mexico; (b) speaking Spanish as their native language; (c) being between 18 and 60 years at the time of the cognitive evaluation; (d) having completed at least one year of formal education; and (e) being able to read and write. The exclusion criteria for HCs were (a) having a history of neurological alterations, developmental disorders, learning disabilities, and psychiatric disorders; (b) having a score ≥ 23 on the mini-mental state examination (MMSE) (MMSE = 28.65 ± 1.31) 28 ; (c) having a score ≥ 90 on the Barthel Index (Mahoney & Barthel., 1965); (d) having a score ≤ 4 on the Patients Health Questionnaire-9 (PHQ-9) (PHQ-9 = 1.42 ± 0.71) 29 ; and (e) having visual, auditory, or sensory problems.
Instruments
The Brief International Cognitive Assessment for MS (BICAMS) was developed by expert neurologists and neuropsychologists as a battery for assessing cognitive function in patients with MS.
30
This instrument consists of three different tests:
The symbols digit modalities test (SDMT). The SDMT involves a key two rows, where the upper row contains a series of nine abstract geometric symbols and the lower row contains corresponding number (1–9). Participants are instructed to verbally associate each symbol with its corresponding number as quickly as possible within a 90-s time frame. The score for this test is determined by the number of correct substitutions completed within the given time limit, with a maximum score of 110.25,31,32 The California Verbal Learning Test (CVLT-II). The CVLT-II requires the examiner to read a list of 16 words in five trials. Participants are then asked to recall as many words as they can remember (in any order) a list given by the psychometrist of the study. The score for this test is calculated based on the total number of words correctly recalled across the five trials.
33
The Brief Visuospatial Memory Test Revised (BVMT-R). In the BVMT-R, participants are shown a sheet of paper containing six geometric designs for a duration of 10 s. After the stimulus is removed, participants are given paper and pencil to draw the geometric designs from memory. Scores are determined by summing the scores from three trials (each drawing received a score of 0–2 points).
34
To measure CI we established criteria based on the clinical experience of the neuropsychologists participating in the study (EVT; AMP; TVC); and a review of the literature.14,17,35,36 The criteria for CI include
Statistical analysis
We used the statistical software R version 4.0.3 (R console Team 2020) to perform the statistical analysis. Kolmogorov-Smirnov and Levenés tests were used to evaluate normal distribution and homoscedasticity in the quantitative variables in both groups; most of the scores did meet the assumptions of normality and homoscedasticity. Qualitative variables were described as frequencies (%) and quantitative variables were described as means ± standard deviation. For univariate analysis we used chi-square test or Fisheŕs exact test (when required). For the comparison of means ± standard deviation of quantitative variables between NMOSD patients and HCs, student´s test was performed.
All composite scores were created using Stouffeŕs
Results
Clinical characteristics of NMOSD patients
We included 40 patients with an NMOSD diagnosis. Of those 40, 19 (47.5%) patients had myelitis, 14 (35.0%) patients had optic neuritis, and 7 (17.5%) had area postrema syndrome. The average time from diagnosis to cognitive assessment was 52 months, range (12–139). AQP4-IgG antibody was tested in 30 of 40 patients, with 27 of them (67.5%) having a positive result, while 10 (25%) patients had an unknown status (data not shown). At the time of the cognitive assessment a percentage of NMOSD patients were taking azathioprine (
Cognitive function
Student´s test showed statistically poor performance by NMOSD patients compared to HCs on all three neuropsychological test scores. This significant difference was observed on the SDMT (
Comparison between patients with NMOSD vs HCs on neuropsychological test scores.

Comparison between groups was made using Student
In Table 2, we show a significant difference between the groups in all cognitive domains. Processing speed (

Processing speed.

Learning and memory.
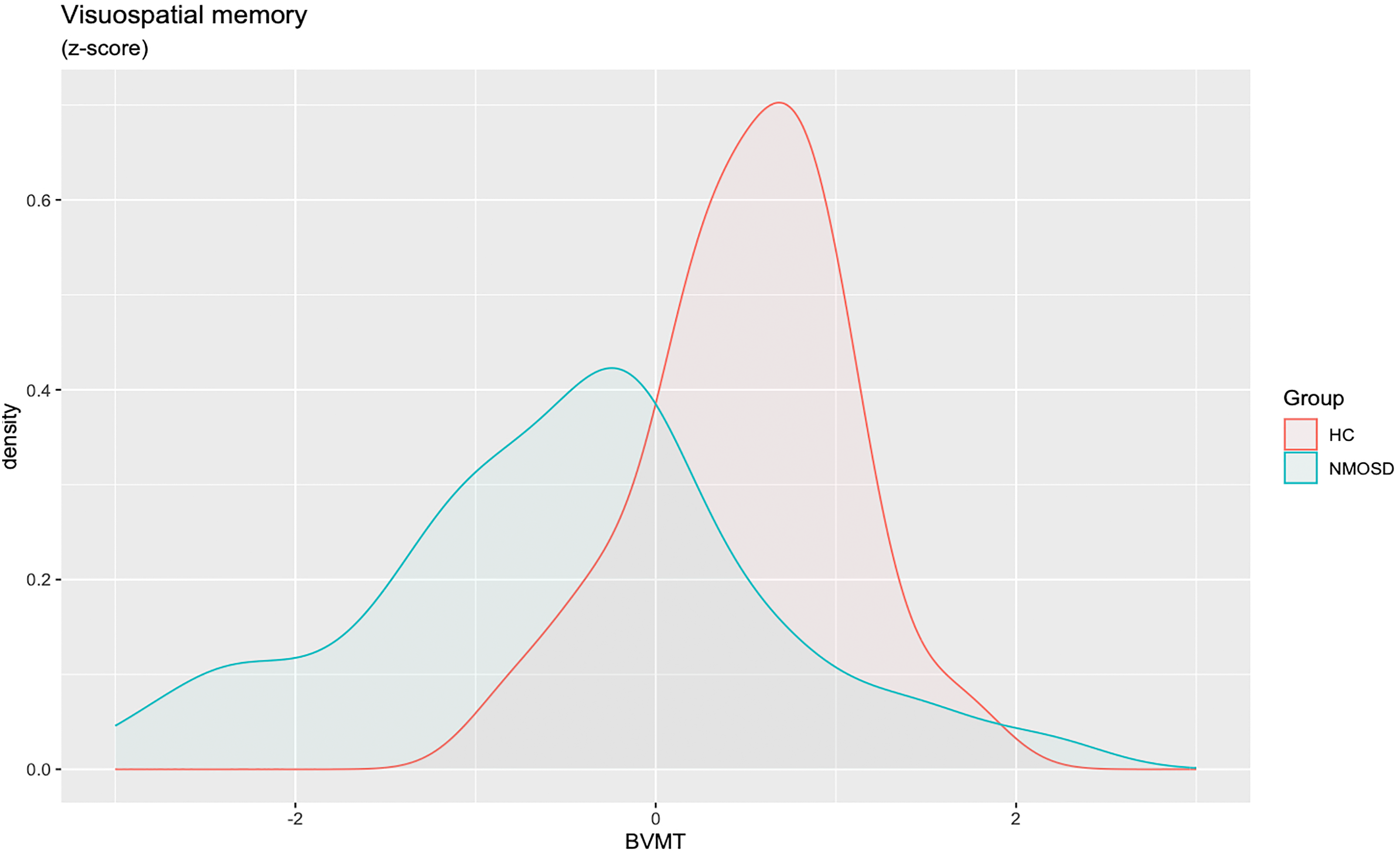
Visuospatial memory.
Average performance between patients with NMOSD vs HCs.

Comparison between means of z-scores were made using Student
We compared CI in NMOSD patients and those without CI. NMOSD patients with CI performed lower in the SDMT (
Comparison of cognitive functions between NMOSD patients with and without CI.

Comparison between groups were made using Student
Discussion
The objective of this study was to determine the prevalence of CI in patients with NMOSD in Mexico. The results revealed the following: (a) 70% of NMOSD patients exhibited CI in two or more cognitive domains. (b) 80% of the patients had impairment on verbal memory. (c) 77% of the patients had CI in processing speed (d) and 47% in visuospatial memory.
Several previous studies have assessed and compared CI in MS and NMOSD patients.14,17,18,20,39–41 In Latin America (LATAM) there is a lack of research and information regarding the prevalence of CI in NMOSD patients. To date, only one study has been conducted in LATAM (Argentina; 39) that included patients diagnosed NMOSD (
On the other hand, it is important to highlight some methodological differences between the present study and previous research. For instance: (a) the sample size of the groups is different, especially in the NMOSD group, as we included 40 NMOSD patients, while a study conducted in Egypt by Salama et al., 26 included 20 NMOSD patients and found that 75% of them had CI in two or more cognitive domains (Information processing speed and memory). In Turkey, a retrospective study conducted by Yabalak et al., 41 included 22 patients and reported that 45.5% of NMOSD patients had a CI (Memory, attention, and processing speed). In Japan, a study conducted by Fujimori et al. 35 included 12 patients with NMOSD and showed that 41.7% had CI (Perceptual organization, working memory, and processing speed). In Spain, Lopez et al. 42 included 41 NMOSD patients and showed that 34% had a CI (visual memory and attention). A recent paper by Portugal, Lopes et al. 27 included 19 NMOSD patients and reported significant CI (compared to HCs) in the areas of processing speed, concentration, language processing, and executive function. (b) Additionally, the number of cognitive domains evaluated by the neuropsychological test batteries varied from study to study. For Example, some studies assessed CI using the Brief Repeatable Battery of Neuropsychological Test,18,35,41–43 while others used the BICAMS to assess.26,27
Currently, the majority of instruments used in NMOSD patients have not been standardized and validated for use in these populations (Table 4). In the present study, the BICAMS was used to assess the primary cognitive domains affected in patients with NMOSD. Our results indicated that patients with NMOSD had significantly lower scores compared to HCs. In summary, the findings of the present study validate the utility of the BICAMS battery in differentiating between HCs and those with NMOSD diagnoses. However, the significance of this study lies in the fact that cognitive assessment was conducted on Mexican patients diagnosed with NMOSD to determine the prevalence of CI.
Cognitive performance in previous studies that include NMOSD patients.
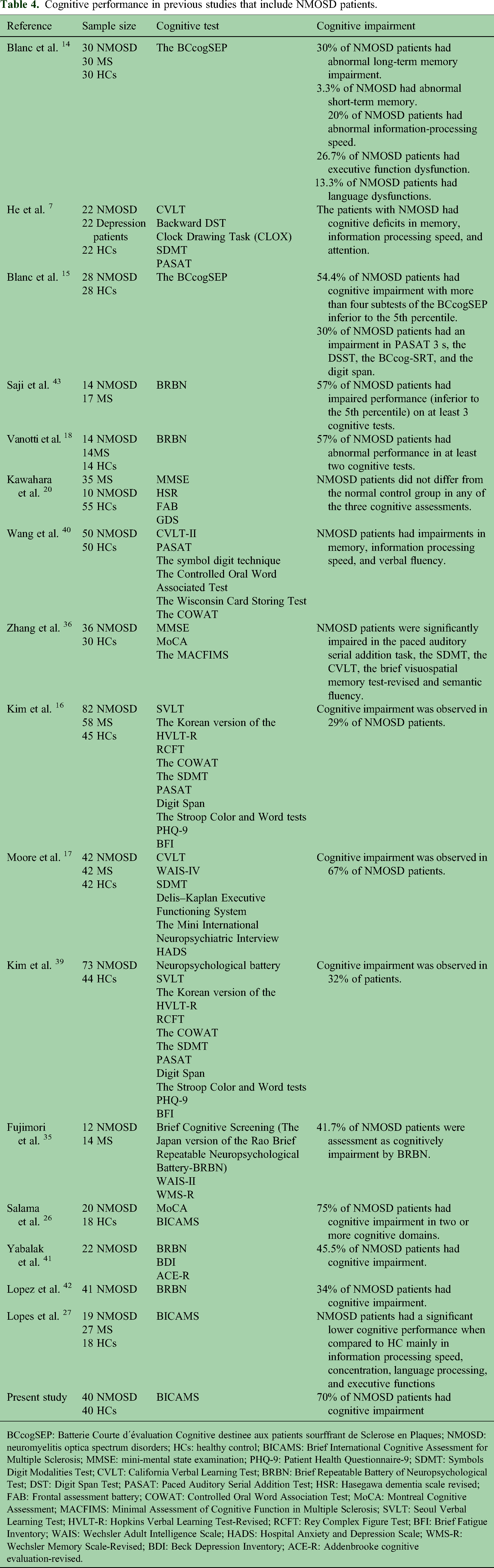
BCcogSEP: Batterie Courte d´évaluation Cognitive destinee aux patients sourffrant de Sclerose en Plaques; NMOSD: neuromyelitis optica spectrum disorders; HCs: healthy control; BICAMS: Brief International Cognitive Assessment for Multiple Sclerosis; MMSE: mini-mental state examination; PHQ-9: Patient Health Questionnaire-9; SDMT: Symbols Digit Modalities Test; CVLT: California Verbal Learning Test; BRBN: Brief Repeatable Battery of Neuropsychological Test; DST: Digit Span Test; PASAT: Paced Auditory Serial Addition Test; HSR: Hasegawa dementia scale revised; FAB: Frontal assessment battery; COWAT: Controlled Oral Word Association Test; MoCA: Montreal Cognitive Assessment; MACFIMS: Minimal Assessment of Cognitive Function in Multiple Sclerosis; SVLT: Seoul Verbal Learning Test; HVLT-R: Hopkins Verbal Learning Test-Revised; RCFT: Rey Complex Figure Test; BFI: Brief Fatigue Inventory; WAIS: Wechsler Adult Intelligence Scale; HADS: Hospital Anxiety and Depression Scale; WMS-R: Wechsler Memory Scale-Revised; BDI: Beck Depression Inventory; ACE-R: Addenbrooke cognitive evaluation-revised.
Study limitations
Results from the current study should be interpreted in the context of limitations.
Procedure
All participants gave their informed consent before being included in the study. This study was designed following the principles of the 64th Declaration of Helsinki (last revision Fortaleza, Brazil 2013). The Research and Ethics Committee of the Centro Medico Nacional de Occidente at the Instituto Mexicano del Seguro Social, in Guadalajara, Mexico, approved this study (code of approval: R-2019-1301-023). The medical records of patients admitted to the neurology unit with an NMOSD diagnosis were reviewed to select patients who met the inclusion criteria. Patients meeting the inclusion criteria were contacted and invited to participate, and those who accepted were contacted to schedule the evaluation. After the informed consent was given and signed, all participants completed the BICAMS battery to measure cognitive function.
Conclusion
This study observed that 70% of NMOSD patients had CI. In our sample, the cognitive domains most affected were verbal memory and processing speed. Whereas visuospatial memory impairment was observed in less than 50% of NMOSD patients. The utilization of BICAMS can offer valuable insights into CI in NMOSD patients, providing Neuropsychologist working with NMOSD patients from Mexico with information that can be utilized for planning cognitive rehabilitation across various domains.
Highlights
70% of NMOSD patients had CI.
The most affected CI was verbal memory.
NMOSD patients obtained lower scores in all cognitive functions.
Prompt recognition of CI is paramount to cognitive rehabilitation.
Footnotes
Author contribution
Conceptualization was done by Edgar R Valdivia-Tangarife, Fernando Cortés-Enríquez, and Miguel A Macías-Islas. Data curation was done by Alejandra Morlett-Paredes, Teresita Villaseñor-Cabrera, Mario A. Mireles-Ramírez, and Laura González-López. Formal analysis was done by Edgar R. Valdivia-Tangarife and Jorge I. Gámez-Nava. Investigation was done by Alejandra Morlett-Paredes, Teresita Villaseñor-Cabrera, and Laura González-López. Methodology was designed by Edgar R Valdivia-Tangarife, Jorge I. Gámez-Nava, and Laura González-López. Supervision was done by Miguel A Macías-Islas, Fernando Cortés-Enríquez, and Jorge I Gámez-Nava. Writing—original draft was done by Edgar R Valdivia-Tangarife, Fernando Cortés-Enríquez, Alejandra Morlett-Paredes, Gámez-Nava, Mario Alberto, Mireles-Ramírez, and Miguel Ángel Macías-Islas. Writing—review & editing was done by Edgar R Valdivia-Tangarife, Fernando Cortés-Enríquez, Alejandra Morlett-Paredes, Teresita Villaseñor-Cabrera, Jorge I. Gámez-Nava, Mario A Mireles-Ramírez, Laura González-López, and Miguel Ángel Macías-Islas. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The Research and Ethics Committee of the Centro Medico Nacional de Occidente at the Instituto Mexicano del Seguro Social, in Guadalajara, Mexico, approved this study (code of approval: R-2019-1301-023).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
All participants gave their informed consent before being included in the study.
