Abstract
Background
Differences in pain between subtypes of multiple sclerosis are understudied.
Objective
To compare the prevalence of pain, and the association between pain and: (a) pain interference and (b) social participation in people with relapsing–remitting multiple sclerosis and progressive multiple sclerosis.
Methods
Participants completed the McGill Pain Questionnaire Short-Form-2, Pain Effects Scale and Ability to Participate in Social Roles and Activities-V2.0 questionnaires. We tested the association between multiple sclerosis subtype, pain severity, and pain interference/social participation using quantile regression.
Results
Of 231 participants (relapsing–remitting multiple sclerosis: 161, progressive multiple sclerosis: 70), 82.3% were women. The prevalence of pain was 95.2%, of more than mild pain was 38.1%, and of pain-related limitations was 87%; there were no differences between multiple sclerosis subtypes. Compared to participants with relapsing–remitting multiple sclerosis, those with progressive multiple sclerosis reported higher pain interference (mean (standard deviation) Pain Effects Scale; progressive multiple sclerosis: 15[6.0] vs relapsing–remitting multiple sclerosis: 13[5], p = 0.039) and lower social participation (Ability to Participate in Social Roles and Activities T-scores 45[9.0] vs 48.3[8.9], p = 0.011). However, on multivariable analysis accounting for age, physical disability, mood/anxiety and fatigue, multiple sclerosis subtype was not associated with differences in pain interference or social participation.
Conclusions
Pain was nearly ubiquitous. Over one-third of individuals with relapsing–remitting multiple sclerosis and progressive multiple sclerosis reported pronounced pain, although this did not differ by multiple sclerosis subtype.
Introduction
Multiple sclerosis (MS) is characterized by multiple symptoms including pain. A 2013 meta-analysis reported the prevalence of any pain type as 63% (95% CI: 55%–70%). 1 Pain is multidimensional,2,3 and its measurement often encompasses intensity, quality, location, and duration. Among people with MS, pain can often be chronic, severe, and widespread. 2 Regardless of type, pain is associated with fatigue, reduced health-related quality of life, and poor mental health.4,5 Pain may interfere with work, general activity, and social and other roles.2,6,7 In a United States veterans population with MS, 92% of 2994 participants reported bodily pain within the last 4 weeks, and 71% reported moderate or greater pain-related interference with respect to work outside the home and housework. 8
The International Progressive MS Alliance highlighted the need for studies regarding the prevalence and characteristics of pain in progressive MS populations. 9 Findings regarding associations between the prevalence of pain and subtypes of MS are limited. In the 2013 review, five studies reported a prevalence of overall pain of 50% (95%CI: 35.4–64.5%) in people with relapsing–remitting MS (RRMS), 69.8% (95%CI: 54.7–83.0%) in those with secondary progressive MS and 70.3% (95%CI: 59.9–79.8%) in those with primary progressive MS. 1 However, a more recent single-center Italian study found no difference in the prevalence of chronic pain or neuropathic pain, by MS subtype. 10 Similarly, another recent study found no difference in pain prevalence or pain interference between participants with RRMS and progressive MS. 11 Thus uncertainty remains as to whether pain characteristics or interference differ by MS subtype. As greater accrual of spinal cord lesions and more advanced neurodegeneration could lead to differences in the experience and impacts of pain, a better understanding of potential differences in pain characteristics by MS subtype would aid clinical management and health care resource planning.
We compared the pain prevalence, intensity, and characteristics in relapsing and progressive MS subtypes, and assessed the associations between pain and participation in social activities. We hypothesized that, regardless of MS subtype, those with the highest severity and frequency of pain would report the highest effects on social participation.
Methods
Study population
From November 2014 through July 2016, participants were enrolled in a single-center prospective study in Manitoba, Canada to evaluate psychiatric comorbidity in immune-mediated inflammatory diseases, including MS. 12 All participants were ≥18 years or older, provided informed consent, and agreed to study participation for 3 years. Herein, we included participants with definite MS (confirmed by records review) who completed the final (fourth) study visit, the only visit when pain characteristics were measured. Ethics approval was obtained from the University of Manitoba Health Research Ethics Board.
Measures
All measures included herein were collected concurrently during the final visit. 12
Sociodemographic and clinical characteristics
Self-administered questionnaires captured birth date, gender, race, marital status, education, annual household income, and smoking status. Gender was categorized as man versus woman. Race was categorized into White or other because few individuals identified as non-White. Marital status was dichotomized as single (including single/never married/divorced/separated/widowed) or partnered (including married/common law). Education was categorized as ≤ high school or > high school. Annual household income was reported in Canadian dollars and categorized as <$50,000, ≥$50,000, or “I do not wish to answer.” Smoking was reported as current smoker or not. Participants reported age at MS symptom onset and diagnosis, and comorbidities using a validated questionnaire, 13 verified by medical records review. Physical comorbidities were summarized as a count: 0, 1, 2, ≥ 3. Types of MS was determined by chart review. Consistent with other studies, we grouped participants with primary progressive and secondary progressive MS into one progressive MS group (PMS). 11
Measured characteristics included height and weight from which we calculated body mass index (BMI), the Timed 25-foot Walk (T25FW) test, 14 Nine-hole Peg test(9HPT), 14 and the Expanded Disability Status Scale (EDSS). 15 The T25FW and the 9HPT were completed by certified research assistants and provided validated measures of lower limb and upper limb function, respectively. These were averaged into one z-score, similar to the approach used in the MS Functional Composite, 14 providing a summary measure of physical function. The EDSS is an ordinal measure of disability derived from the neurological examination. 15 Scores range from 0 (no disability) to 10 (death due to MS). 15 Due to its poor inter-rater reliability and poor responsiveness to change in individuals with severe disability, the EDSS was used to describe the study sample, not as a covariate. The T25FW has a better signal-to-noise ratio in RRMS, 16 and is more sensitive to change in PMS than the EDSS, 17 while the 9HPT can detect physical changes in individuals who are non-ambulatory better than the EDSS.
Psychological distress
Participants completed the Hospital Anxiety and Depression Scale (HADS) to assess symptoms of depression (HADS-D) and anxiety (HADS-A) within the past week. The HADS includes 7 items each for depression and for anxiety, with total scores for each subscale ranging from 0 to 21. Higher scores indicate more severe symptoms; scores of ≥11 indicate clinically meaningful symptoms. The HADS is valid and reliable.18,19
Fatigue
Participants completed the Daily Fatigue Impact Scale, which includes eight items, each scored from 0 (no problem) to 4 (extreme problem). 20 Total scores range from 0 to 32 with higher scores indicating more fatigue.
Pain and participation
In the International Classification of Functioning, Disability and Health, sensory functions and pain fall into the domain of body function. We also assessed activities and participation.
Pain characteristics were assessed with the McGill Pain Questionnaire Short-Form-2 (MPQ-SF2). 21 The MPQ-SF2 includes 22 items that ask about pain intensity in the past week, with responses ranging from 0 (none) to 10 (worst possible); responses are summed across all items. Subscale scores can be generated for pain qualities such as continuous (six items: Throbbing pain, cramping pain, gnawing pain, aching pain, heavy pain, tender), intermittent (six items: Shooting pain, stabbing pain, sharp pain, splitting pain, electric-shock pain, piercing), neuropathic (six items: Hot-burning pain, cold-freezing pain, pain caused by light touch, itching, tingling or “pins and needles”, numbness) and affective (four items: Tiring-exhausting, sickening, fearful, punishing-cruel).
Pain interference was assessed using the Medical Outcomes Study Pain Effects Scale (PES), a valid and reliable measure derived from the PES. 22 The PES includes 6 items related to mood, ability to walk or move around, sleep, work (outside the home and at home), recreational activities, and enjoyment of life in the past 4 weeks. Responses indicate the degree of pain interference, with responses ranging from 1 (not at all) to 5 (to an extreme degree). Total scores range from 6 to 30, where higher scores indicate greater pain interference; scores >6 indicated “any” pain interference.
Social participation was assessed using the Ability to Participate in Social Roles and Activities (APSRA) V2.0 from the Patient-Reported Outcome Measures Information System (PROMIS) assessment center. 23 This measure was collected based on the recommendation of our study's Patient Advisory Group, who indicated that the effect of pain and fatigue on this outcome were particularly important. This 8-item measure assesses participation restrictions, including related to social interaction, employment, transportation, community, social, and civic life. Responses for each item are never (5), rarely (4), sometimes (3), usually (2), and always (1), and are summed across all items. Raw scores range from 8 to 40 and are transformed into T-scores with a mean of 50 and standard deviation (SD) of 10. A score of 50 is considered average for the US general population. Lower T-scores represent greater restrictions in social participation.
Data analysis
We described the study population using means (SD) for continuous variables, median (interquartile range), and frequency (percent). Prevalence of pain was reported as the number (%) of participants that reported a total MPQ-SF2 score >0. “No more than mild pain” has been identified as a more relevant pain outcome, and has been classified as a score <3/10 on an 11-point rating scale. 24 Therefore, we also report the proportion of participants with an averaged MPQ-SF2 score ≥3 across all 22 items. Bivariate analyses were used including student's t-tests, ANOVA, Kruskal–Wallis, chi-squared or Fisher's exact tests as appropriate.
We used two sets of multivariable quantile regression models to examine the association between the independent variables of interest, MS subtype and pain severity (total MPQ-SF2 score), and the outcomes: (a) pain interference (as measured by the PES), and (b) social participation (as measured by the APSRA). We used quantile regression as it does not require distributional assumptions and permits assessment of the relationship of an independent variable across the entire range of a dependent variable instead of its conditional mean. 25 Our primary outcome was the median quantile, or 50th percentile. The 20th and 80th percentiles were examined as complementary outcomes. To determine whether the associations between pain severity and the outcomes (pain interference, social participation) differed between MS subtypes we included an interaction term between pain and MS subtype. We report regression coefficients and bootstrapped 95% confidence intervals.
Covariates for all models included age (continuous), gender (woman as reference), race (white as reference), marital status (partnered as reference), education (≤high school as reference), income (≥$50,000 as reference), smoking status (smoker as reference), BMI (continuous), physical function (continuous z-score), count of comorbidities (none as reference), and fatigue (continuous).26,27 Depression (HADS-D < 11 as reference), and anxiety (HADS-A < 11 as reference) symptoms were included only in the social participation model because the PES captures the effects of pain on mood.
Statistical analyses used IBM SPSS Statistics Version 29 (IBM Corp, Armonk, NY).
Results
Participants
Of the 255 participants enrolled initially, 234 provided data at the planned final study visit; 8 participants died before the last visit. Participants who completed all visits were more likely to have post-secondary education but did not differ on other demographic or MS-specific characteristics (Supplemental Table e1). Two of the 234 participants were excluded as their course of MS was unknown, and one was excluded because of non-completion of the MPQ-SF2. Thus, 231 participants were included in the final analysis (Supplemental Figure e1), of whom 161 had RRMS.
Overall, participants were predominantly women, self-identified as White, with a mean (SD) age of 53.9 (12.7) years. Compared to participants with RRMS, those with PMS were 12 years older on average, had higher disability scores as measured by the EDSS, and had worse performance on the T25FW test and 9HPT whether examined individually or combined (Table 1).
Baseline characteristics of participants.

MS: multiple sclerosis; RRMS: relapsing remitting multiple sclerosis; PMS: progressive multiple sclerosis (secondary progressive multiple sclerosis and primary progressive multiple sclerosis); DFIS: Daily Fatigue Impact Scale; EDSS: Expanded Disability Status Scale; HADS: Hospital Anxiety (A) and Depression (D) Scale; SD: standard deviation; IQR: interquartile range. a-Missing data: Race n = 1 with RRMS, Education = 1, Timed 25foot walk n = 12, 9-hole peg test n = 11.
Bold indicates statistical significance.
Pain intensity and characteristics
Overall, 220 participants had a total MPQ-SF2 score >0, indicating a prevalence of any pain of 95.2% in the past week, while 88 (38.1%) reported more than mild pain. The prevalence of any pain did not differ between those with RRMS and PMS, nor did the prevalence of more than mild pain (Table 2). Unadjusted total MPQ-SF2 scores did not differ between the groups nor did the subscale scores, including those related to neuropathic and affective aspects of pain. The distribution of the total scores was similar in the two groups (Figure 1).

Distribution of pain severity according to subtype of multiple sclerosis.
Descriptors of pain characteristics and interference according to subtype of multiple sclerosis.

MPQ-SF2: McGill Pain Questionnaire Short Form 2; PES: Pain Effects Scale; APSRA: ability to participate in social roles and activities; SD: standard deviation.
Bold indicates statistical significance.
Pain interference
Overall, 201 (87%) participants had PES scores >6, indicating some pain-related interference. This did not differ between those with RRMS and PMS (Table 2). However, participants with PMS had higher average PES scores than those with RRMS (Tables 2 and 3, Supplemental Figure e2). After adjustment for the two covariates that differed between MS subtypes (age and physical function), pain-related interference did not differ between PMS and RRMS participants (Table 3, model 2). This did not change after adjustment for additional covariates including fatigue (Table 3, models 3 and 4). Higher pain intensity as measured by total MPQ-SF2 scores was associated with greater pain interference even after adjustment for all covariates (Table 3).
Regression coefficients (95% confidence intervals) for the association between subtype of multiple sclerosis, MPQ-SF2 scores and PES a .
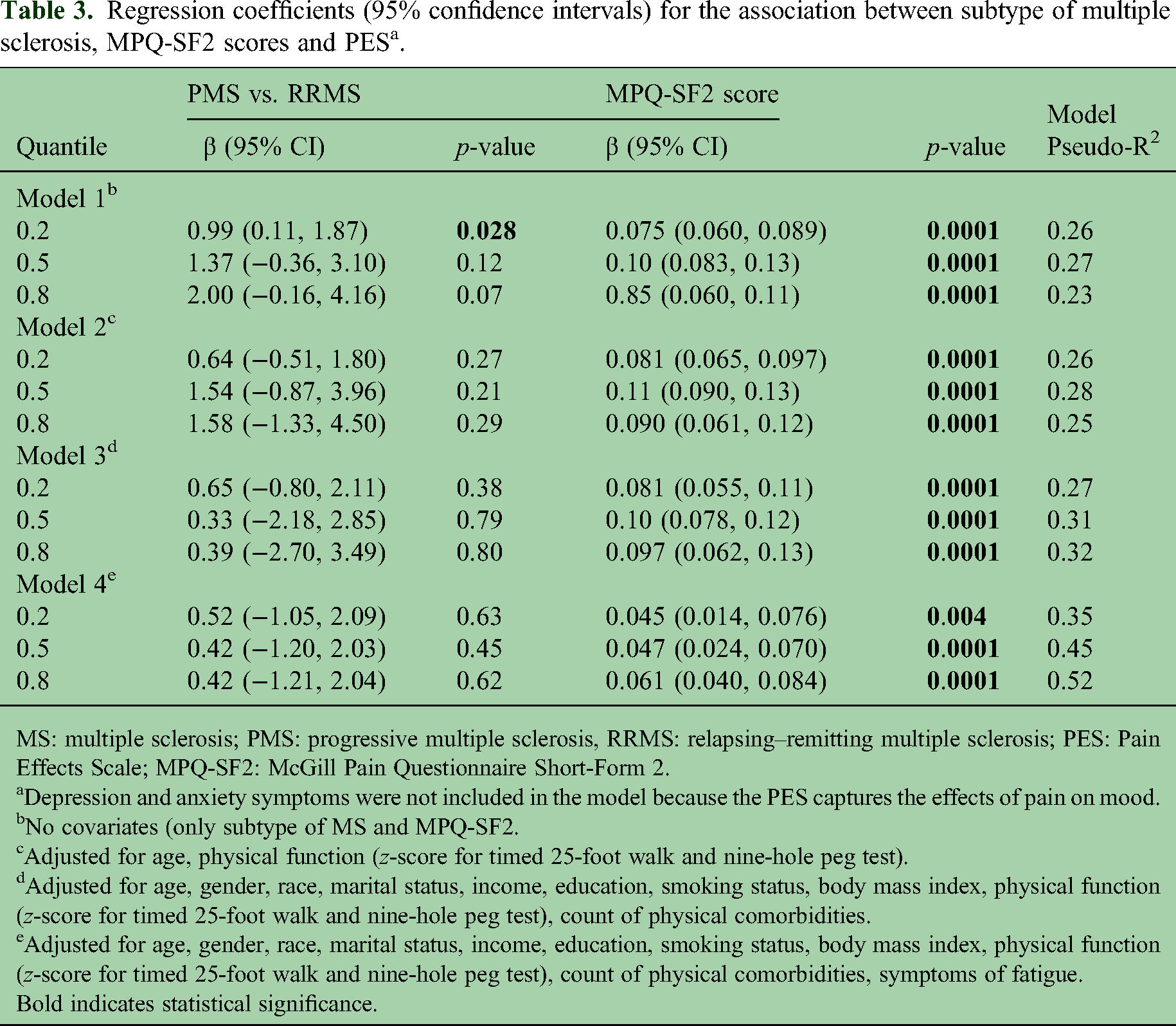
MS: multiple sclerosis; PMS: progressive multiple sclerosis, RRMS: relapsing–remitting multiple sclerosis; PES: Pain Effects Scale; MPQ-SF2: McGill Pain Questionnaire Short-Form 2.
Depression and anxiety symptoms were not included in the model because the PES captures the effects of pain on mood.
No covariates (only subtype of MS and MPQ-SF2.
Adjusted for age, physical function (z-score for timed 25-foot walk and nine-hole peg test).
Adjusted for age, gender, race, marital status, income, education, smoking status, body mass index, physical function (z-score for timed 25-foot walk and nine-hole peg test), count of physical comorbidities.
Adjusted for age, gender, race, marital status, income, education, smoking status, body mass index, physical function (z-score for timed 25-foot walk and nine-hole peg test), count of physical comorbidities, symptoms of fatigue.
Bold indicates statistical significance.
Social participation
Overall, the average (SD) APSRA T-scores were 47.3 (9.0). Participants with PMS had lower APSRA T-scores than those with RRMS (Table 2, Supplemental Figure e2). This was true across all quantiles of APSRA scores, Table 4, Model 1). After adjustment for age and physical function, there was no significant difference in limitations in social participation between PMS and RRMS participants (Table 4, Models 1–3). In models that adjusted only for MS subtype, higher pain intensity as measured by total MPQ-SF2 scores was associated with lower social participation (Table 4, Model 1). The magnitude of the effect of total MPQ-SF2 scores was greater for participants with lower APSRA T-scores. After adjustment for demographic characteristics and physical function, higher pain intensity remained associated with lower social participation. However, the association between pain intensity and social participation was fully attenuated after additional adjustment for symptoms of depression, anxiety, and fatigue (Table 4). We did not observe any interaction between MS subtype and MPQ-SF2 pain score on APSRA T-scores.
Regression coefficients (95% confidence intervals) for the association between MPQ-SF2 scores, subtype of MS and ability to participate in social roles and activities scores.

MS: multiple sclerosis; PMS: progressive multiple sclerosis, RRMS: relapsing remitting multiple sclerosis; MPQ-SF2: McGill Pain Questionnaire Short-Form-2.
No covariates (only subtype of MS and MPQ-SF2.
Adjusted for age, physical function (z-score for timed 25-foot walk and nine-hole peg test).
Adjusted for age, gender, race, marital status, income, education, smoking status, body mass index, physical function (z-score for timed 25-foot walk and nine-hole peg test), count of physical comorbidities.
Adjusted for age, gender, race, marital status, income, education, smoking status, body mass index, physical function (z-score for timed 25-foot walk and nine-hole peg test), count of physical comorbidities, symptoms of fatigue, and symptoms of depression and anxiety.
Bold indicates statistical significance.
Discussion
While pain is recognized as a highly prevalent and often debilitating symptom of MS, differences in pain experience amongst persons with different MS subtypes have had limited study. We compared the prevalence of pain and its impact on participation in social activities according to MS subtype. The overall prevalence of any pain in our cohort was 95.2%, 31.8% reported more than mild pain, and 87% experienced pain-related interference. We found no difference between subtypes in the prevalence of any pain, more than mild pain, or in the average intensity of pain. Greater pain intensity was associated with greater pain interference as measured by the PES, which focuses on how pain interferes with mood, mobility, sleep, work, recreational activities and enjoyment of life. Findings differed somewhat when we examined social participation using a questionnaire that captured limitations in work and leisure activities as well as activities with friends and family and for fun, but that did not specify pain as the reason for those limitations. MS subtype was not associated with pain interference or social participation after adjustment age and physical function. Greater pain intensity was associated with lower social participation even after adjustment for age and physical function, but not after we also adjusted for symptoms of fatigue, depression and anxiety.
Previous studies reported widely varying estimates of the prevalence of pain among persons with MS which could reflect differences in the MS population, definitions of pain, and measures used.5,6 The prevalence of any pain in our sample was high. A prior meta-analysis reported that the prevalence of overall (any) pain was 50% in individuals with RRMS, and 70% in individuals with PMS. 8 Some studies in the meta-analysis which reported a higher prevalence of pain in people with PMS than RRMS were largely descriptive and accounted for few covariates (e.g., age, sex, disability), if any.28–30 The lack of difference between RRMS and PMS participants in our study may be because we accounted for a broad set of covariates. Consistent with our findings, a recent study found that average pain intensity, evaluated using an 11-point numeric rating scale, did not differ between people with RRMS and PMS, nor did the prevalence of greater than mild pain (RRMS: 51% vs. PMS: 50%). 11 Like our study, this study controlled for multiple demographic and disease-related factors including age, disability status, education, employment status, sex, race, income, marital status, alcohol use, and smoking status.
On unadjusted analyses, participants with PMS had higher pain interference scores and lower social participation. The minimally important difference for between-groups comparisons on the APSRA scale is 2–6 T-score points, 31 and the difference between participants with RRMS and PMS fell in this range. However, these differences were no longer significant after adjustment suggesting the findings were not directly due to subtype of MS but due to differences in covariates. These findings from our adjusted analyses are similar to those of another study that compared pain interference, as measured by the PROMIS Pain Interference Short Form, between participants with RRMS and PMS. 11 That study did not find any differences in pain interference between the groups whether in analyses that were unadjusted, or adjusted for disability status, education, employment status, sex, race, age, income, marital status, alcohol use, and smoking status. 32 Notably, pain intensity was not associated with social participation after we accounted for symptoms of fatigue, depression, and anxiety. These three symptoms are strongly associated with each other among people with MS, even at diagnosis, 33 and bi-directional, dynamic relationships exist among them. 34 Collectively, this symptom cluster is also associated with work impairment. 7 One prior study examined the effect of pain intensity on participation in social roles using 4 items from the APSRA. 32 In that study, individuals with higher pain intensity had lower same-day social participation on unadjusted analyses, but not after adjustment for fatigue and depressed mood. However, within-person changes in pain remained associated with lower same-day social participation even post-adjustment.
This study had several strengths. We examined pain intensity overall, and with respect to neuropathic, affective, constant and intermittent features, which have received relatively limited attention previously. 10 We also considered any, and more than mild pain. The MPQ-SF2 has demonstrated validity and reliability in populations with chronic pain. 35 We accounted for multiple covariates including demographic characteristics, physical function, and health behaviors. Limitations should also be noted. Our sample was predominantly women with at least some post-secondary education so our findings may not generalize as well to other demographic subgroups. Due to the high data collection burden on participants, pain characteristics were captured at only one study visit and we did not capture information regarding pain management.
We found a very high prevalence of pain that did not differ between RRMS and PMS subtypes; older age and greater physical impairment appeared to account for differences in pain-interference between subtypes. Pain severity was associated with greater pain interference. In contrast, pain severity was not associated with limitations in participation of social roles and responsibilities in people with MS after accounting for elevated symptoms of fatigue, depression and anxiety, suggesting that the latter factors may be more significant drivers of social activity limitations than pain per se. This highlights the importance of conceptualizing, evaluating and effectively treating pain from a biopsychosocial perspective, which accounts for the complex interrelationships between the highly prevalent and disruptive symptoms of pain, mood, and fatigue in people with MS. Further studies are needed to evaluate the effectiveness of pain management on improving participation in important social domains.
Supplemental Material
sj-docx-1-mso-10.1177_20552173231188469 - Supplemental material for Pain and participation in social activities in people with relapsing remitting and progressive multiple sclerosis
Supplemental material, sj-docx-1-mso-10.1177_20552173231188469 for Pain and participation in social activities in people with relapsing remitting and progressive multiple sclerosis by Dhruv Jain, Charles N Bernstein, Lesley A Graff, Scott B Patten, James M Bolton, John D Fisk, Carol Hitchon, James J Marriott, Ruth Ann Marrie and in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors thank the members of the CIHR Team for defining the burden and managing the effects of psychiatric comorbidity in chronic immunoinflammatory disease. The CIHR Team members included Ruth Ann Marrie, James M Bolton, Jitender Sareen, Scott B Patten, Alexander Singer, Lisa M Lix, Carol A Hitchon, Renée El-Gabalawy, Alan Katz, John D Fisk, Charles N Bernstein, Lesley Graff, Lindsay Berrigan, Christine Peschken, and James Marriott. The authors acknowledge Shared Health/Health Sciences Centre Winnipeg. The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Crohn's and Colitis Canada, Institute of Musculoskeletal Health and Arthritis (grant number n/a, THC-135234) and the Waugh Family Chair in Multiple Sclerosis (to RAM). CNB is supported in part by the Bingham Chair in Gastroenterology.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
