Abstract
Background
Alemtuzumab is an effective therapy for relapsing multiple sclerosis. Autoimmune thyroid events are a common adverse event.
Objective
Describe endocrine and multiple sclerosis outcomes over 6 years for alemtuzumab-treated relapsing multiple sclerosis patients in the phase 3 CARE-MS I, II, and extension studies who experienced adverse thyroid events.
Methods
Endocrine and multiple sclerosis outcomes were evaluated over 6 years. Thyroid event cases, excluding those pre-existing or occurring after Year 6, were adjudicated retrospectively by expert endocrinologists independently of the sponsor and investigators.
Results
Thyroid events were reported for 378/811 (46.6%) alemtuzumab-treated patients. Following adjudication, endocrinologists reached consensus on 286 cases (75.7%). Of these, 39.5% were adjudicated to Graves’ disease, 2.5% Hashimoto's disease switching to hyperthyroidism, 15.4% Hashimoto's disease, 4.9% Graves’ disease switching to hypothyroidism, 10.1% transient thyroiditis, and 27.6% with uncertain diagnosis; inclusion of anti-thyroid antibody status reduced the number of uncertain diagnoses. Multiple sclerosis outcomes of those with and without thyroid events were similar.
Conclusion
Adjudicated thyroid events occurring over 6 years for alemtuzumab-treated relapsing multiple sclerosis patients were primarily autoimmune. Thyroid events were considered manageable and did not affect disease course. Thyroid autoimmunity is a common but manageable adverse event in alemtuzumab-treated relapsing multiple sclerosis patients.
Keywords
Introduction
Alemtuzumab is an approved, highly effective disease-modifying therapy (DMT) for the treatment of relapsing forms of multiple sclerosis (RMS).1,2 In the phase 3 CARE-MS I (NCT00530348) and II (NCT00548405) trials, and the CARE-MS extension study (NCT00930553), alemtuzumab reduced relapse rate, disability progression, magnetic resonance imaging (MRI) lesion load, and brain volume loss (BVL).3–6,7 The CARE-MS studies also showed clinical and radiological superiority versus subcutaneous interferon beta-1a (IFNbeta-1a). Alemtuzumab is administered as 2 courses, given 12 months apart.3,5 Additional courses may be given for disease control ≥12 months after the previous course. 8
Alemtuzumab targets CD52-expressing B and T lymphocytes for depletion.9,10 B cells subsequently repopulate to baseline levels within ∼6 months, whereas T cells take ∼12 months to reach lower limits of normal. 11 This repopulation is accompanied by changes in immune regulatory networks including memory B cell suppression, relative increases in memory and regulatory T-cell levels, and transition from a pro- to an anti-inflammatory cytokine profile.12–15 While these mechanisms likely mediate alemtuzumab efficacy in RMS, they might also underlie non-multiple sclerosis (MS) autoimmunity that can arise. However, analysis of depletion/repopulation kinetics of the major lymphocyte subpopulations affected by alemtuzumab shows no correlation with non-MS autoimmunity, although the repopulation of specific cells that drive autoimmunity remains undetermined. 11
The most common post-alemtuzumab non-MS autoimmunity is thyroid autoimmunity, which was reported in 42% of patients over 6 years in the pooled CARE-MS I/CARE-MS II/extension studies16,17 and in 41% of patients over 7 years in the CAMMS224 and SM3 studies 18 ; incidence peaked 3 years after initial treatment and declined thereafter. Thyroid autoimmunity incidence was not related to the number of alemtuzumab courses received; at 6 years, incidence was 43%, 41%, 42%, and 38% for those receiving 2, 3, 4, and 5–6 courses, respectively. 16 Other non-MS autoimmune diseases in alemtuzumab-treated RMS patients include immune thrombocytopenia (ITP; incidence 2.2%) 19 and nephropathies (incidence 0.34%) 20 ; autoimmune cytopenias and autoimmune hepatitis are less frequently observed. A risk management plan or risk evaluation and mitigation strategy (RMP/REMS) is in place to monitor for autoimmune conditions, including thyroid dysfunction, ITP, and nephropathies.2,21
As thyroid events in the CARE-MS I/CARE-MS II/extension studies have not been evaluated systematically, a thyroid event adjudication panel (TEAP) of three expert endocrinologists (C. Dayan, B. Lecumberri, P. Senior) was convened to adjudicate all reported cases of thyroid events from the final 6-year follow-up of patients who received alemtuzumab at the start of the CARE-MS studies. Patients who received IFNbeta-1a in the 2-year CARE-MS studies were also evaluated.
Here, we report the adjudication of thyroid events, defined as adverse events (AEs) and laboratory abnormalities, that occurred in the CARE-MS I/CARE-MS II/extension studies over 6 years, as well as the therapies used to treat thyroid disease, and endocrine and MS outcomes.
Patients and methods
The design and methods of the CARE-MS I/CARE-MS II/extension studies were published previously.3–6 Thyroid events were identified under a safety monitoring program conducted by the manufacturer and reported as of March 7, 2016.
Thyroid monitoring
In CARE-MS I/CARE-MS II/extension studies, thyroid function tests were performed at screening and every 3 months. Tests included thyroid-stimulating hormone (TSH) and, if TSH levels were abnormal (normal range 0.4–4.0 mU/L), free triiodothyronine (T3) and free thyroxine (T4). In such cases, TSH, free T3, and free T4 were measured at all subsequent assessments until TSH levels returned to normal. Anti-thyroid peroxidase (anti-TPO) and anti-TSH receptor (anti–TSH-R) antibodies were measured for all patients at screening; anti-TPO positivity did not exclude patients from study enrollment, although patients who screened positive for anti-TSH receptor (anti–TSH-R) antibodies were ineligible for either CARE-MS study. CARE-MS I and II sub-studies included expanded thyroid monitoring, in which TSH, free T3 and T4, and anti-TPO and anti–TSH-R antibodies were measured every 3 months for up to 2 years; the CARE-MS I sub-study was completed, but the CARE-MS II sub-study was discontinued to reduce site and patient burden, and those data are not reported here.
In the extension studies, tests for anti-TPO and anti–TSH-R antibodies were performed at the investigator's discretion. Signs and symptoms of thyroid events were reported by the investigators, and thyroid events were managed by the investigator and/or the patient's general practitioner, including consultation with an endocrinologist as appropriate.
Thyroid event adjudication
Patients with reported thyroid events were reviewed under an adjudication charter. Adjudication was conducted for all thyroid events, defined as any thyroid-related AEs and laboratory abnormalities, that occurred in the CARE-MS I/CARE-MS II/extension studies over the 6-year period. Clinical study data and thyroid case narratives were divided into three groups. Each group was reviewed by a pair of adjudicators, each blinded to the other's analysis, that is, adjudicators 1 and 2, 1 and 3, and 2 and 3 were paired such that each adjudicator reviewed two-thirds of the cases. Each adjudicator was permitted to receive assistance from other qualified endocrinologists, independent of the other adjudicators, but the final decision was the responsibility of that adjudicator. Cases were reviewed with respect to 5 domains related to thyroid dysfunction, duration, diagnosis, thyroid event onset, and outcome, with one option chosen from each domain (Table 1). Where each pair agreed on a domain, it was deemed adjudicated; where disagreement occurred, the case was referred to the third endocrinologist In these cases, the third endocrinologist was unblinded to view the decisions of the initial pair of adjudicators. This second round of adjudication focused on assessing the unmatched domains, although the third endocrinologist was able to review the entire case if needed to support their decision-making. Agreement by the third endocrinologist with either of the first two led to adjudication, but disagreement with both led to a TEAP discussion in which categories were either resolved or no consensus was reached (Figure 1). Where no consensus was reached, the case was excluded from further analyses.

Process flow for thyroid event adjudication.
Thyroid event review domains.

MS efficacy assessments
Efficacy of alemtuzumab in those with and without consensus on adjudicated thyroid events was determined by calculating annualized relapse rate (ARR), disability (Expanded Disability Status Scale (EDSS)), MRI disease activity (Gd-enhancing T1 lesions and new/enlarging T2 hyperintense lesions), and BVL, as published previously.3–6
Statistical analysis
Adjudicated thyroid events were based on the first occurrence to avoid counting an ongoing event twice; this was important for biphasic thyroid disease in which a patient could switch from hyperthyroidism to hypothyroidism or vice versa. Descriptive statistics were used to present data related to thyroid events. The statistical methods used to analyze MS outcomes are described elsewhere.3–6
Research ethics and patient consent
Registration and ethical conduct of the CARE-MS I/CARE-MS II/extension studies, and patient informed consent were reported previously.3–6
Results
Patient disposition and thyroid adjudication
As reported previously, 16 811 patients with RMS were treated with alemtuzumab 12 mg in the CARE-MS I (n = 376) and CARE-MS II (n = 435) studies, of which 742 (91.5%) entered the extension study and 659 (81.3%) remained on study for ≥6 years.
For the adjudication, 378 cases of reported thyroid events (AEs and laboratory abnormalities) in alemtuzumab-treated patients in CARE-MS I/CARE-MS II/extension studies were reviewed. Baseline characteristics were generally comparable between the groups with and without thyroid events, except for female sex, which was more common in those with thyroid events (Table 2). Consensus was achieved in all 5 adjudication domains for 316 cases (83.6%), with 62 (16.4%) lacking consensus in at least 1 category. Thirty cases were subsequently excluded from the consensus data set because the thyroid event was pre-existing (n = 23) or onset occurred after the 6-year follow-up period (n = 7) (Table 3).
Baseline demographics, disease characteristics, and treatment exposure of patients randomized to alemtuzumab in the CARE-MS and extension studies.
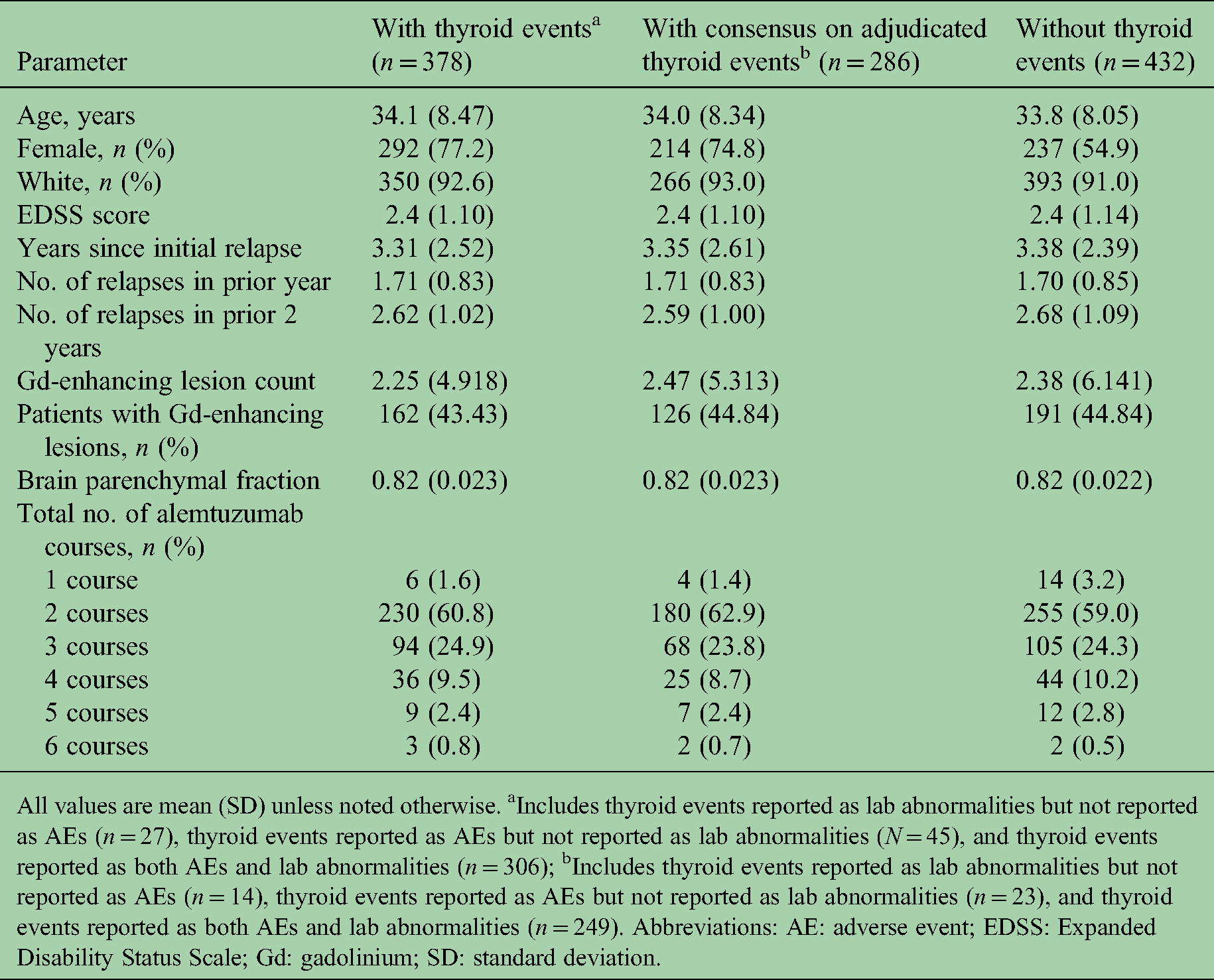
All values are mean (SD) unless noted otherwise. aIncludes thyroid events reported as lab abnormalities but not reported as AEs (n = 27), thyroid events reported as AEs but not reported as lab abnormalities (N = 45), and thyroid events reported as both AEs and lab abnormalities (n = 306); bIncludes thyroid events reported as lab abnormalities but not reported as AEs (n = 14), thyroid events reported as AEs but not reported as lab abnormalities (n = 23), and thyroid events reported as both AEs and lab abnormalities (n = 249). Abbreviations: AE: adverse event; EDSS: Expanded Disability Status Scale; Gd: gadolinium; SD: standard deviation.
Thyroid event adjudication consensus.

Cases for which consensus was reached in all 5 domains were deemed to be adjudicated. bCases for which consensus was reached in fewer than 5 domains were deemed not to be adjudicated. cIncludes 30 cases that were excluded from the consensus dataset because the thyroid event was pre-existing (n = 23) or onset was after the 6 year follow-up period (n = 7). dIncludes 5 cases that were excluded from the consensus data set because the thyroid event was pre-existing. eOf the 46 cases for which consensus was lacking for 1 domain, 1, 2, 8, 14, and 21 cases lacked consensus in domain 1, 2, 3, 4, and 5, respectively. fOf the 4 cases for which consensus was lacking for 2 domains, 1 case lacked consensus in domains 1 and 4, 1 case lacked consensus in domains 2 and 3, 1 case lacked consensus in domains 1 and 3, and 1 case lacked consensus in domains 3 and 4. gFor the single case that lacked consensus for 3 domains, consensus was not reached for domains 1, 3, and 5. hFor the single case that lacked consensus for 4 domains, consensus was not reached for domains 1, 2, 3, and 4. iOf the 8 cases for which consensus was lacking for 1 domain, 2, 0, 1, 0, and 5 cases lacked consensus in domain 1, 2, 3, 4, and 5, respectively. Each of the 5 domains are described in the Patients and methods section, Thyroid event adjudication, and in Table 1. Abbreviations: IFNbeta-1a: interferon beta-1a.
For the 389 patients treated with IFNbeta-1a in the 2-year CARE-MS studies, 30 cases of reported thyroid events were reviewed. Consensus was achieved in all 5 domains for 21 cases (70.0%), with 9 (30.0%) lacking consensus in at least 1 category. Five cases were subsequently excluded from the consensus data set because the thyroid event was pre-existing (Table 3).
Adjudicated thyroid autoimmunity in CARE-MS I/CARE-MS II//extension studies
Alemtuzumab
Of the 286 confirmed cases of thyroid events in alemtuzumab-treated patients, those with a consensus diagnosis were adjudicated to Graves’ disease or Hashimoto's disease switching to hyperthyroidism (42.0%), Hashimoto's disease or Graves’ disease switching to hypothyroidism (20.3%), transient thyroiditis (10.1%), or uncertain (27.6%; Table 4). The median time of thyroid event onset from alemtuzumab initiation was 26.4 months (range, 3.0–71.5 months; Figure 2(a)) and from most recent course was 12.2 months (range, 0.03–57.1 months; Figure 2(b)). Nearly all (98%) cases were reported within 4 years of the most recent course of alemtuzumab (i.e. the RMP/REMS monitoring window).

Timing of thyroid event onset relative to alemtuzumab administration: (a) time from alemtuzumab initiation and (b) time from most recent course. The percentage of patients with serious events in years 1, 2, 3, 4, 5, and 6 from alemtuzumab initiation was 0.4%, 1.4%, 9.4%, 1.8%, 0.7%, and 0.4%, respectively, and the percentage of patients with serious events in years 1, 2, 3, 4, 5, and 6 from the most recent course was 3.2%, 8.7%, 1.1%, 0.7%, 0.4%, and 0%, respectively.
Consensus diagnoses following adjudication of thyroid AE cases reported in CARE-MS and extension alemtuzumab-treated patients.
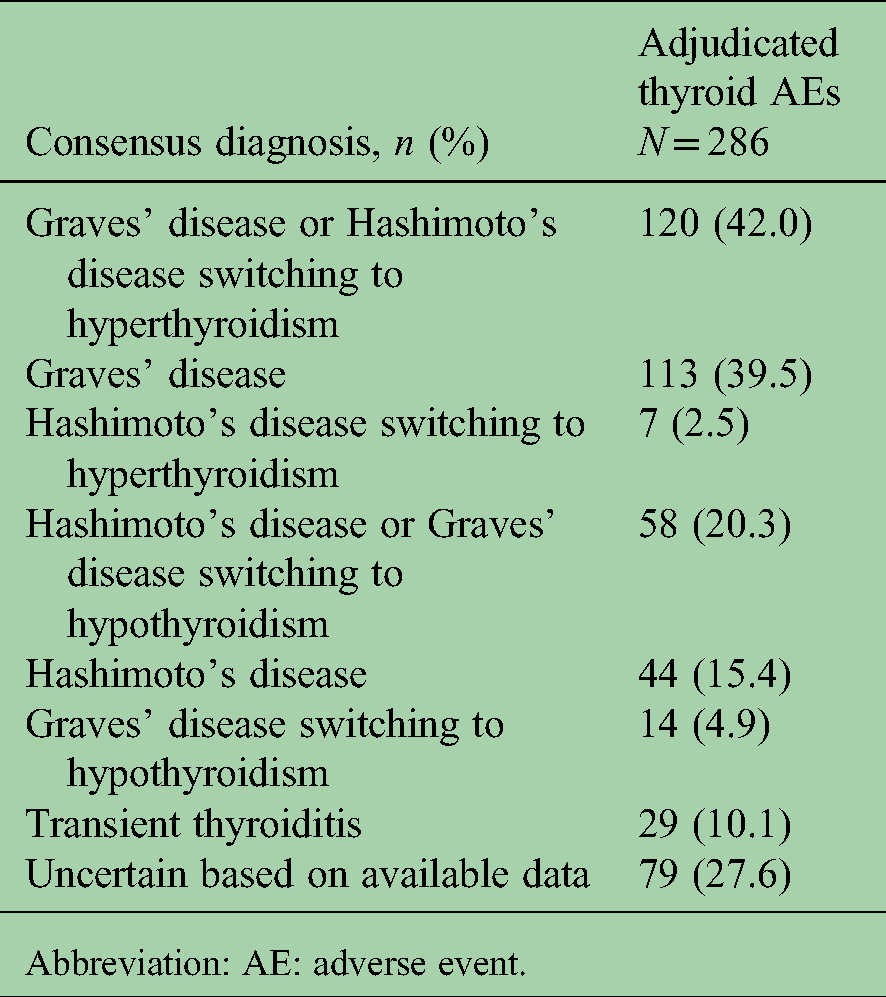
Abbreviation: AE: adverse event.
For the 286 cases with confirmed thyroid events, the proportions of patients who received 1, 2, 3, 4, 5, and 6 courses of alemtuzumab were similar to those with no reported thyroid events (Table 2), indicating that there was neither a relationship between the number of courses received and the incidence of thyroid events nor a difference in the number of courses received between those with and without thyroid events.
IFNbeta-1a
Of 16 confirmed cases of thyroid events in IFNbeta-1a–treated patients, 1 (6.3%) patient was diagnosed with hyperthyroidism (Graves’ disease), 2 (12.5%) with hypothyroidism (Hashimoto's disease), and 8 (50.0%) with transient thyroiditis. Five (31.3%) patients had insufficient data to ascertain the specific diagnosis.
Thyroid medications, treatments, and disease outcomes in alemtuzumab-treated patients
In the consensus dataset, thyroid events were most often treated with oral medications and were captured in totality irrespective of the nature of the first or subsequent events (i.e. patients may have taken more than one medication). Eighty percent of patients received at least 1 medication. Oral medications for the treatment of thyroid AEs following alemtuzumab were mainly those typically used to treat hyperthyroidism (methimazole/thiamazole, carbimazole, and propylthiouracil) and hypothyroidism (levothyroxine); a few patients were treated for hypothyroidism with other preparations (Table 5). Levothyroxine was the most frequently administered medication, received by patients with initial hypothyroidism, those that switched from hyperthyroidism, and formerly hyperthyroid patients needing hormone replacement therapy following radioiodine thyroid ablation or thyroidectomy. Thirty-two percent of patients received at least 1 medication for each of hyperthyroidism and hypothyroidism. In addition, 7.3% and 10.8% of patients underwent radioiodine therapy and thyroidectomy, respectively. Thyroid disease outcomes as of each patient's last visit within the 6-year follow-up (n = 484 thyroid events) were 54.8% recovered, 45.0% ongoing, and 0.2% unknown. A list of medications for patients with ongoing, adjudicated thyroid events is shown in Table S1.
Oral thyroid medications used to treat adjudicated casesa.

Among patients who received alemtuzumab for up to 6 years.
Abbreviation: AE: adverse event; SD: standard deviation.
CARE-MS I expanded thyroid function sub-study
In the CARE-MS I sub-study, 143 patients (98 and 45 treated with alemtuzumab and IFNbeta-1a, respectively) had TSH, free T3 and T4, and anti-TPO and anti–TSH-R antibodies measured at screening and at quarterly visits over 2 years. Of these, 64 patients treated with alemtuzumab and 33 treated with IFNbeta-1a had assessment of anti-TPO antibodies at all sub-study time points, and 65 and 33 patients, respectively, had assessment of anti–TSH-R antibodies at all time points. Higher than normal anti-TPO antibody levels and positive anti–TSH-R antibodies were detected in post-treatment assessments among 20.4% and 21.4% of alemtuzumab-treated patients, respectively. Of IFNbeta-1a–treated patients, 8.9% displayed higher than normal anti-TPO levels and 4.4% were positive for anti–TSH-R antibodies. A thyroid AE occurred in 28/98 (28.6%) patients treated with alemtuzumab and 4/45 (8.9%) patients treated with IFNbeta-1a. The anti-TPO and anti–TSH-R antibody status of alemtuzumab- and IFNbeta-1a–treated patients appeared to be associated with the occurrence of thyroid AEs (Table 6). Forty-eight of 98 (49.0%) patients treated with alemtuzumab and 5/45 (11.1%) treated with IFNbeta-1a were eligible and included in the adjudication dataset as having a thyroid event. This confirmed a higher rate of thyroid events in alemtuzumab-treated patients compared to those treated with IFNbeta-1a, 5 and that additional cases arose after 2 years consistent with peak incidence in Year 3 (Figure 2(a)).
Anti-TPO and anti–TSH-R antibody status in alemtuzumab- and IFNbeta-1a-treated patients with and without thyroid AEs in the CARE-MS I sub-study.

The number (%) of alemtuzumab-treated patients who were ever high/ever positive for either anti-TPO or anti–TSH-R antibodies was 22 (78.6%) and 8 (11.4%) for those with and without reported thyroid AEs, respectively. The number (%) of IFNbeta-1a-treated patients who were ever high/ever positive for either anti-TPO or anti–TSH-R antibodies was 1 (25%) and 6 (14.6%) for those with and without reported thyroid AEs, respectively. Cases with thyroid AEs reported in this table were not adjudicated, as data were from the 2-year CARE-MS I sub-study rather than the CARE-MS I/CARE-MS II/extension studies for which data were collected over 6 years. Abbreviation: AE: adverse event; anti-TPO: anti-thyroid peroxidase; anti-TSH-R: anti-TSH receptor; IFNbeta-1a: interferon beta-1a.
MS outcomes
ARR, EDSS scores, MRI disease activity, and BVL over 6 years for the 286 patients with consensus on adjudicated thyroid events versus those without are shown in Figure 3. ARRs in each year ranged from 0.15 to 0.23 (0.20 over 6 years) and from 0.12 to 0.27 (0.21 over 6 years) in those with and without thyroid events, respectively (Figure 3(a)). Mean EDSS changes were −0.06 and 0.11 for those with and without thyroid events, respectively (Figure 3(b)). Moreover, 25%, 57%, and 18% of patients with thyroid events versus 20%, 59%, and 21% of patients without had improved (≥1 point decrease), stable (<1 point change in either direction), and worsened (≥1 point increase) EDSS, respectively. Annual values for freedom from MRI disease activity ranged from 61% to 78% and from 60% to 76% in those with and without thyroid events, respectively (Figure 3(c)). BVL was comparable between groups (Figure 3(d)).
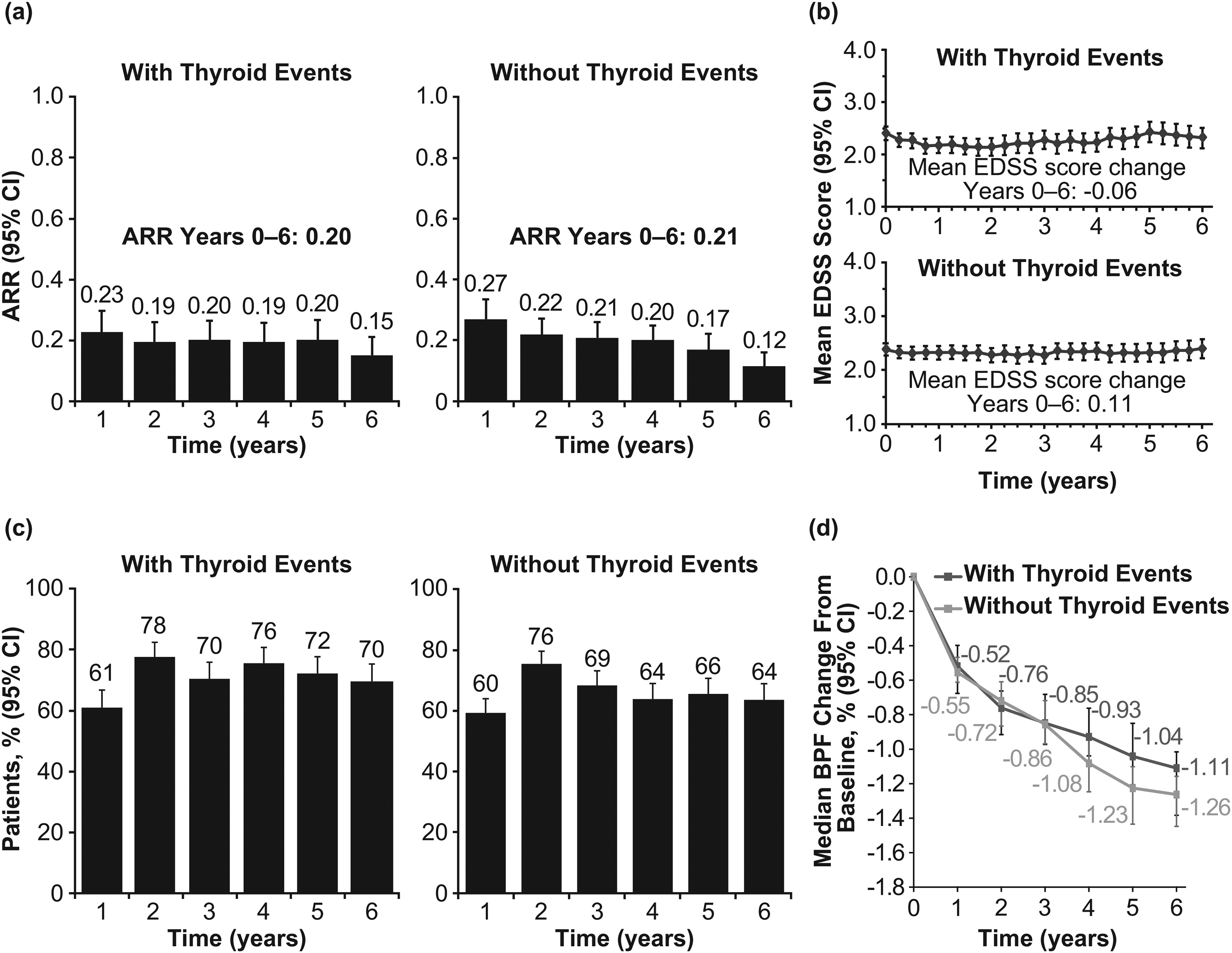
MS disease activity measures in alemtuzumab-treated patients with and without thyroid events over 6 years: (a) ARR, (b) mean EDSS score, (c) freedom from MRI disease activity, and (d) BVL. Freedom from MRI disease activity was defined as no new Gd-enhancing T1 lesions on current MRI or new/enlarging T2 hyperintense lesions since last MRI. BVL was calculated using ANCOVA, adjusted for geographic region and baseline brain parenchymal fraction. No statistically significant differences in any of the MS disease measures between patients with or without thyroid events were evident, except for freedom from MRI disease activity at Year 4 (P = 0.0045).
Discussion
While thyroid events in alemtuzumab-treated patients in CARE-MS I/CARE-MS II/extension studies have been published previously,3–6,17 those cases were not collectively reviewed by expert endocrinologists. Here, we describe the adjudication of reported thyroid events in these patients over 6 years, and their endocrine and MS outcomes.
Defining the final analysis dataset as those with at least 1 thyroid event in 6 years where agreement of all 5 domains constituted consensus and excluding those with pre-existing events or those where the event occurred after 6 years, resulted in a dataset of 286 patients representing a 35.3% incidence. While this is lower than the 40%–46% incidence over similar time periods in these and other studies,4,6,16–18,22 the incidence was 39.8% excluding the 62 patients for whom consensus was not reached, and the 30 who were excluded for pre-existing or post–6-year events. For IFNbeta-1a, the incidence rate of confirmed cases was 4.1%, and 4.3% excluding the 9 patients for whom consensus was not reached and the 5 who had pre-existing events. Regardless of the calculation used, thyroid events are common in alemtuzumab-treated patients, whereas the rate is increased, although lower, in IFNbeta-1a–treated patients. 23 This may reflect the underlying predisposition to thyroid disease in RMS patients in general, 24 which can develop with therapies that modulate the immune system, and to differing extents based on their individual mechanisms of action. IFNbeta is thought to impact the thyroid through a direct inhibitory effect on iodine organification.25,26 In contrast, the risk of secondary autoimmunity in predisposed alemtuzumab-treated patients has been linked in part to the presence of pre-existing hyperexpanded T-cell clones, which persist and expand further after treatment due to homeostatic proliferation, in combination with reduced thymopoiesis. 27
The most common adjudicated thyroid diagnosis among alemtuzumab-treated patients was hyperthyroidism (42.0%), consistent with other cohorts,18,22,28 followed by hypothyroidism (20.3%) and transient thyroiditis (10.1%), with the remaining 27.6% of patients having an uncertain diagnosis. The CARE-MS I/CARE-MS II/extension study protocols did not specify how thyroid AEs should be investigated or managed, instead leaving this to the investigators, and a paucity of clinical data for some patients made a definitive diagnosis problematic. Moreover, the etiology of hyperthyroid or hypothyroid AEs was not routinely tested. In particular, anti-TPO and anti–TSH-R antibodies were not systematically tested beyond screening or outside of the CARE-MS I sub-study. Despite these caveats, this expert adjudication represents the most detailed analysis of thyroid events occurring in the core CARE-MS and extension studies.
In the sub-study, we observed a higher proportion of patients with high anti-TPO/positive anti–TSH-R antibodies who experienced thyroid AEs compared with those who did not (Table 6). Taken together with prior reports of alemtuzumab-treated cohorts in which autoantibody-mediated thyroid disease has been documented,22,28–30 our findings support ongoing monitoring of these biomarkers in patients who receive alemtuzumab. Consensus diagnosis data suggest that testing for anti-TPO and anti–TSH-R antibodies is important for a more complete adjudication. Indeed, the proportion of alemtuzumab-treated patients with uncertain diagnosis decreased by 5.7% when anti-TPO and anti–TSH-R antibody data were included, although it is important to emphasize that this finding is based on a sub-study over 2 years with a relatively small sample size, and in some cases, anti-TPO and anti–TSH-R antibodies were not tested at the time of thyroid dysfunction occurrence (2.34 + /- 1.04 years). Nevertheless, for most cases that had both consensus and enough evidence for a confirmed diagnosis, the hallmark findings of autoimmune thyroid disease were evident. These conclusions were based largely on the evidence of sustained, well-documented thyroid dysfunction, combined with concomitant medications and supplemented by autoantibody testing in a minority of cases.
The published guidelines 31 recommend autoantibodies be tested in all cases of thyroid disturbance following alemtuzumab treatment to clarify its etiopathogenesis and guide the most appropriate treatment. This adjudication indicates that, consistent with Daniels et al., 28 the majority of thyroid AEs are due to humoral autoimmunity rather than cell-mediated autoimmune destruction or inflammatory thyroiditis. Indeed, routine anti–TSH-R antibody testing may indicate that some cases considered here as thyroiditis were due to transient autoantibody induction. However, such an association would be most clearly answered by thyroid scintigraphy. The CARE-MS I sub-study data support this notion, since 64.3% and 53.6% of alemtuzumab-treated patients with a reported thyroid AE were ever high for anti-TPO antibodies and ever positive for anti–TSH-R antibodies compared with 4.3% and 8.6%, respectively, without an AE (Table 6). This is clinically important, as hypothyroid cases that are anti–TSH-R positive may be due to blocking anti–TSH-R antibodies rather than destruction of the thyroid gland and may be reversible. Long-term follow-up is therefore justified to allow for subsequent supervised withdrawal of thyroxine. 31
We found a high rate of switching between hyperthyroidism and hypothyroidism and vice versa (7.3%; i.e. biphasic disease) in our alemtuzumab cohort. Grave's disease with fluctuating thyroid status has been described previously in alemtuzumab-treated patients.22,28 To our knowledge, this is the first report of switching to hyperthyroidism from Hashimoto's disease in alemtuzumab-treated patients. The transition to hyperthyroidism occurred in 13.7% of Hashimoto's disease cases in our cohort, which is greater than the 1.2% incidence reported in the general Hashimoto's disease population. 32 Switching between hyperthyroidism and hypothyroidism in our cohort speaks to the complexity of alemtuzumab thyroid effects and is likely due to the coexistence of thyroid stimulatory and blocking autoantibodies that change in proportion over time.22,30,33
MS outcomes in patients with and without thyroid events were similar, indicating no additional impact on MS disease course. Similarly, health-related quality of life outcomes in patients from CARE-MS I/CARE-MS II/extension studies over 6 years in those with and without thyroid events were similar for those with non-serious AEs, although quality of life worsened toward the end of the study in those with more serious thyroid AEs who were treated with radioiodine or thyroidectomy. 16 By contrast, Sovetkina et al. reported significantly fewer relapses, longer relapse-free survival, and reduced disability progression over 2 years in alemtuzumab-treated MS patients with thyroid autoimmunity versus those without. 34 There are several important differences between Sovetkina et al. and our study that could explain these disparate findings, including a much smaller sample size (n = 33 patients with thyroid AEs versus n = 286 in our study), higher baseline EDSS score (mean 4.5 for patients with thyroid AEs versus 2.4 in our study), and higher percentage of patients treated with carbimazole (66.7% versus 8.4% in our study), a drug with considerable anti-inflammatory properties. 35 Future research should ascertain whether pre-treatment disability status or carbimazole use influences MS outcomes for alemtuzumab-treated patients.
The adjudication of thyroid events by a panel of expert endocrinologists is a key strength of this analysis. Previous work has shown that the accuracy of outcome adjudication is proportional to the expertise of assessors, but also the number of assessors for outcomes with significant inter-assessor disagreement. 36 In our study, there was an overall low level of disagreement between assessors (approximately one in four thyroid events), showing the value of panel adjudication for thyroid outcomes. Moreover, expert endocrinologists classified 7% of adjudicated thyroid events as pre-existing, which contributed to the slightly lower frequency of thyroid events than in the CARE-MS trials. Considering the limitations of our study, it is pertinent to note that although the CARE-MS I and CARE-MS II trials included robust designs and statistical comparisons, thyroid outcomes were not the focus of these trials; consequently, comparisons of MS outcomes for patients with and without thyroid AEs are post hoc and should be interpreted with this caveat in mind. Additional data that may have informed adjudication decisions, such as post-procedure complications or treatment for patients with ongoing events, were not available for this study. Furthermore, outcomes are largely reported using descriptive measures, and no adjustments were made for possible confounding variables.
In summary, adjudication of thyroid events that occurred over 6 years following alemtuzumab treatment in patients with RMS was primarily autoimmune and was managed with oral medications. Furthermore, the substantial frequency of biphasic disease highlights the importance of appropriate endocrine investigation and close monitoring of treatment response in patients identified with thyroid abnormalities after alemtuzumab treatment. The non-serious nature of thyroid autoimmunity in most patients, coupled with available guidance on how to monitor, detect, and treat thyroid autoimmunity,2,21,37–39 helps maintain a favorable risk profile where patients can benefit from the efficacy of alemtuzumab.
Supplemental Material
sj-docx-1-mso-10.1177_20552173221142741 - Supplemental material for Endocrine and multiple sclerosis outcomes in patients with autoimmune thyroid events in the alemtuzumab CARE-MS studies
Supplemental material, sj-docx-1-mso-10.1177_20552173221142741 for Endocrine and multiple sclerosis outcomes in patients with autoimmune thyroid events in the alemtuzumab CARE-MS studies by Colin M. Dayan, Beatriz Lecumberri, Ilaria Muller, Sashiananthan Ganesananthan, Samuel F. Hunter, Krzysztof W. Selmaj, Hans-Peter Hartung, Eva K. Havrdova, Christopher C. LaGanke, Tjalf Ziemssen, Bart Van Wijmeersch, Sven G. Meuth, David H. Margolin, Elizabeth M. Poole, Darren P. Baker and Peter A. Senior in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
The authors and Sanofi thank the patients for their participation in the CARE-MS I, CARE-MS II, and extension studies, as well as the steering committees and the investigators. The authors wish to acknowledge Salman Afsar for his contribution to the manuscript.
Author Contributions
Conceptualization: DPB, EMP, DHM; Methodology: CMD, BL, PAS, DPB, EMP, DHM; Validation: CMD, BL, PAS, DPB; Formal Analysis: DPB; Investigation: CMD, BL, PAS, Resources: DPB, EMP, DHM; Data Curation: DPB; Writing – Original Draft, DPB; Writing, Review and Editing: all authors; Visualization: DPB; Project Administration: DPB, EMP, DHM.
Data sharing
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient-level data will be anonymized, and study documents will be redacted to protect the privacy of trial participants. Further details on Sanofi's data sharing criteria, eligible studies, and process for requesting access can be found at: https://![]() .
.
Declaration of conflicting interests
CMD has received compensation for serving on an advisory board and/or speaking fees (Sanofi). BL has nothing to disclose. IM has received honoraria and travel expenses for lecturing (Sanofi). SG has nothing to disclose. SFH has received consulting agreements, speaker honoraria, and grant/research financial support (AbbVie, Acorda, Actelion, Adamas, Avanir, Bayer, Biogen, Novartis, Osmotica, Questcor, Roche, Sanofi, Synthon, and Teva). KWS has received consulting and/or speaking fees (Biogen, Merck, Novartis, Roche, Sanofi, and Synthon). H-PH has received consulting and/or speaking fees (Bayer, Biogen, BMS Celgene, CSL Behring, GeNeuro, Horizon Therapeutics, Merck Serono, Novartis, Octapharma, Roche, Sanofi, and TG Therapeutics). EKH has received honoraria and grant support (Actelion, Biogen, Merck Serono, Novartis, Receptos, Roche, Sanofi, and Teva), and support (Ministry of Education of Czech Republic [PROGRES Q27/LF1]). CCL has received compensation for consulting (Acorda Therapeutics, Bayer, Biogen, Cephalon, EMD Serono, Novartis, Pfizer, Questcor, Sanofi, Strativa, Teva, and UCB). TZ has received consulting and/or speaking fees (Almirall, Bayer, Biogen, Merck, Novartis, Roche, Sanofi, and Teva) and grant/research support (Biogen, Novartis, Sanofi, and Teva). BVW has received advisory board and/or speaking fees (Almirall, Biogen, Merck, Novartis, Roche, and Sanofi); research support (Biogen, Merck, Novartis, and Sanofi), and contracted research (PI) (Biogen, Merck, Sanofi, Novartis, and Roche). SGM has received honoraria for lecturing, travel expenses for attending meetings, and financial research support (Almirall, Amicus Therapeutics, Bayer HealthCare, Biogen, Celgene, Diamed, HERZ Burgdorf, Merck Serono, Novartis, Novo Nordisk, ONO Pharma, Roche, Sanofi, and Teva). DHM and EMP were employees of Sanofi during study conduct and analysis, and may hold shares and/or stock options in the company. DPB is an employee of Sanofi and may hold shares and/or stock options in the company. PAS has received grant research support (Novo Nordisk). The adjudicators (CMD, BL, and PAS), and IM and SG who assisted CMD), received no compensation for review of the cases reported in this study.
Funding
Editorial support for the development of this paper was provided by Elevate Scientific Solutions (Panos Xenopoulos PhD, Richard Hogan PhD, and Renee E. Granger PhD) and was funded by Sanofi. The CARE-MS I, CARE-MS II, and extension studies were funded by Sanofi and Bayer Healthcare Pharmaceuticals.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
