Abstract
Background
The latitude gradient in multiple sclerosis incidence indicates that low sun exposure and therefore vitamin D deficiency is associated with multiple sclerosis risk.
Objective
Investigation of the effect of month of birth, which influences postnatal vitamin D levels, on multiple sclerosis risk and severity in Sweden.
Methods
Patients and population-based controls were included from three nationwide cohorts. Differences in month of birth between cases and controls were analyzed using logistic regression and examined for effect modification by calendar year and geographic region at birth.
Results
Males had a reduced risk of multiple sclerosis if born in the winter and increased risk if born in the early fall. Individuals born before 1960 had an increased risk if born in summer or fall. Being born in late summer and early fall was associated with more severe disease.
Conclusions
We identified a birth cohort effect on the association between the month of birth and multiple sclerosis, with a more significant effects for births before 1960. This coincides with a period of lower breastfeeding rates, recommended intake of vitamin D, and sun exposure, resulting in a lower vitamin D exposure during the fall/winter season for infants born in the summer.
Introduction
Vitamin D modulates several immune processes with deficiencies leading to impaired immune responses against infections and increased risk of certain autoimmune disorders. 1 Vitamin D deficiency is also associated with an increased risk of multiple sclerosis (MS), a chronic inflammatory disease resulting in central nervous system (CNS) demyelination. MS patients often have lower levels of circulating vitamin D, including the stabler hydroxylated form, 25(OH)D, and the active form, 1,25(OH)2D. 1 The higher frequency of genetic predisposition to low vitamin D expression among MS patients2–4 and the latitude gradient in MS prevalence with increased risk at higher latitudes and lower ultraviolet radiation levels further supports the pathological association with vitamin D, which is primarily produced photochemically. 5
The extent of vitamin D involvement in MS pathogenesis remains unclear. However, sunlight exposure, particularly during adolescence or early life, has been implicated in MS risk.6,7 This is further evidenced by the increased risk among offspring of mothers with low vitamin D intake during pregnancy, suggesting the early involvement of vitamin D during prenatal development of the immune system. 6 Previous studies have observed an association between the season or month of birth and the risk of developing MS, possibly due to differences in sun exposure during pregnancy.8–11 However, recent studies have not been able to replicate such findings.12–14 In contrast to risk, the long-term implications of vitamin D and sun exposure on MS disease severity and treatment response are inconsistent. 15 This study examines the association between the month of birth and the risk of MS development and disease severity in Sweden.
Materials and methods
Cohort description
MS patients were included from three Swedish cohorts: Genes and Environment in MS (GEMS) is a national prevalence-based study of MS patients recruited from the Swedish MS registry between November 2009 and November 2011;
3
Epidemiologic Investigation in MS (EIMS) is an incidence-based study enrolling newly diagnosed MS patients from 42 neurological clinics across Sweden;
3
and the Immunomodulation and MS Epidemiology (IMSE) study follows patients on immunomodulatory treatments for MS to examine clinical, genetic, and environmental factors that influence treatment response.
16
Population-based controls were matched to cases based on age (± 5 year intervals), sex, and residence area at the time of inclusion. Descriptive characteristics for each cohort are provided in
Description of cohort

Count with percent frequency or mean with standard deviation are provided for each multiple sclerosis (MS) cohort: GEMS, Genes and Environment in MS; EIMS, Epidemiologic investigation in MS; and IMSE, Immunomodulation and MS Epidemiology. Expanded disability status scale (EDSS), MS severity score (MSSS), and age-related MS severity (ARMSS) score were dichotomized by ≥3 and ≥6.
Data on disease characteristics were extracted from the Swedish MS registry, including age at onset, disease course, disease duration, and disability level as measured by the first available expanded disability status scale (EDSS) score. Disease severity was characterized using both the MS severity score (MSSS) and the age-related MS severity (ARMSS) score. Year, month, and region of birth were determined through their government-issued personal identification number.
Ethical approvals and patient consents
The Stockholm Regional Ethical Review Board has approved all studies under ethical permits Dnr 2017/1349-32 (EIMS, 2017-06-28), Dnr 2017/1350-32 (GEMS, 2017-06-28), and Dnr2017/1426-32 (IMSE, 2017-07-06)), and study participants have provided informed and written consent.
Data availability
Anonymized data used in this study will be shared upon request to the corresponding author by any qualified investigator pending Institutional Review Board approval.
Statistical analysis
Differences in the distribution of month of birth between MS cases and controls were analyzed using a logistic regression model. The season of birth was defined by three-month intervals, including winter (Dec/Jan/Feb), spring (Mar/Apr/May), summer (Jun/Jul/Aug), and fall (Sep/Oct/Nov). Analyses were stratified by calendar year and the geographic region at birth. Three regions were defined based on a latitude gradient: north (Norrland, range 60.4-69.1 N), central (Svealand, range 58.7–62.2 N), and south (Götaland, range 55.4–59.2 N). Two cut-offs, ≥3 and ≥6, were used to dichotomize disability and severity measures. Association between the month of birth and age at disease onset among MS cases were assessed using a linear regression model. All statistical analyses were performed using R v.4.0.2 (R Core Team. Vienna, Austria).
Results
In an overall analysis with 13,481 MS cases and 21,095 matched controls, month or season of birth was not significantly associated with risk of MS (
Month of birth and risk of MS among the Swedish population.
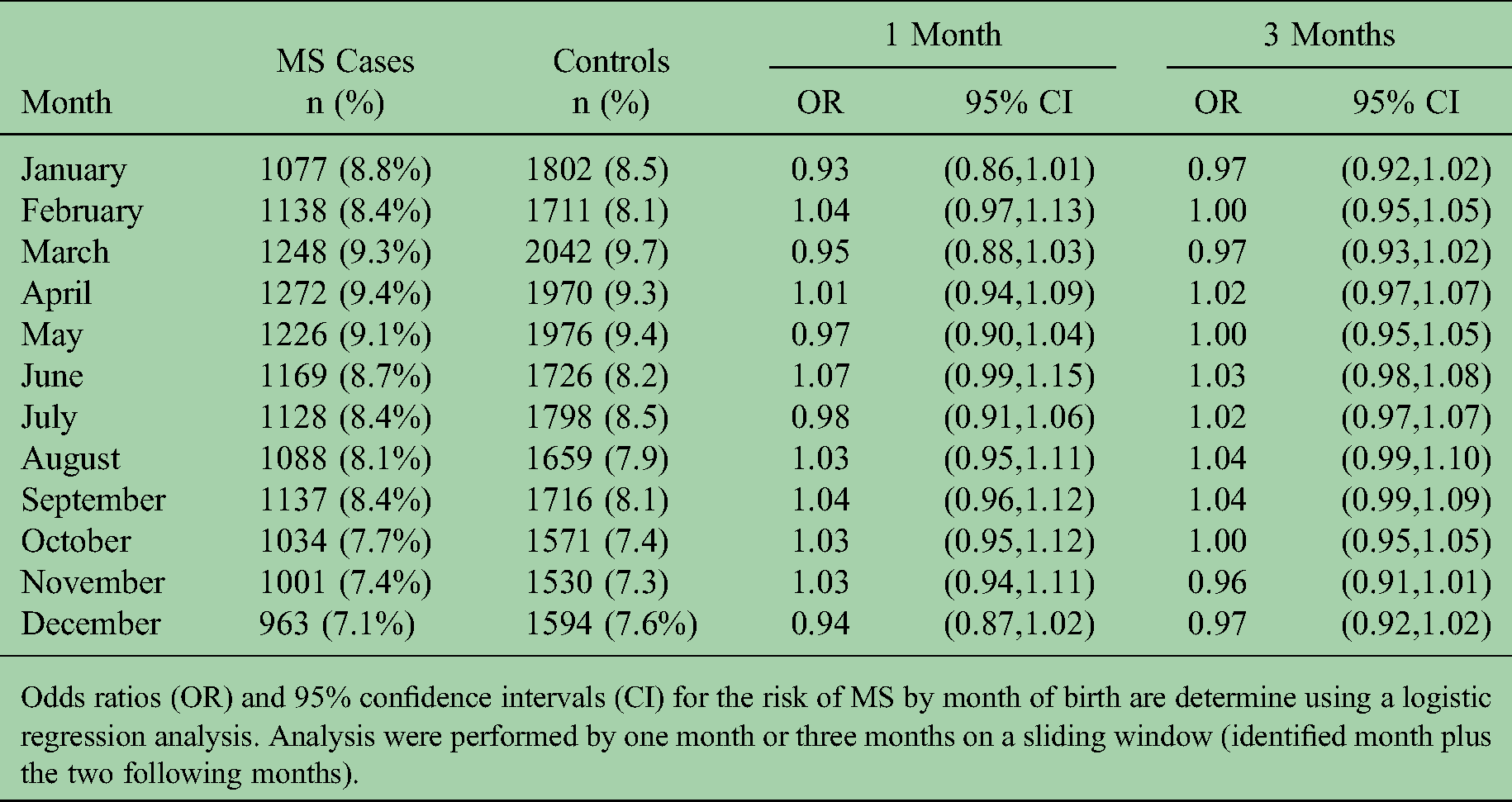
Odds ratios (OR) and 95% confidence intervals (CI) for the risk of MS by month of birth are determine using a logistic regression analysis. Analysis were performed by one month or three months on a sliding window (identified month plus the two following months).
To examine effect modification by calendar year, overlapping strata based on year of birth were used to investigate the association between season of birth and MS (

Effects modification of MS risk and season of birth by calendar year and geographic region at time of birth
Being born in the summer was associated with a younger age at onset (Jun/Jul/Aug; β = -0.57, P = 0.008) with the peak association in July (β = -0.87, P = 0.01,

Effects of the month of birth on the age at onset, disease disability, and severity among MS cases
Discussion
Our findings indicate that month of birth was associated with MS development primarily among men with a higher risk if born in the early fall and lower risk in the early winter. This association was modified by the calendar year of birth, with both males and females having an increased risk of developing MS if born in the summer or fall before 1960. Differences in the risk by sex may indicate a synergistic interaction between the early environmental exposures associated with the month of birth and sex-based differences in immune development and tolerance, 17 although the factors and mechanisms involved require further investigation.
The prenatal period is particularly sensitive to vitamin D exposure, since both low dietary intake of vitamin D during pregnancy and low vitamin D levels as a newborn are risk factors for the later development of MS.6,7,18 However, it is difficult to distinguish whether the effects of sun exposure, as predicted by the month of birth, occur during pregnancy or after birth as levels are inversely correlated. Low neonatal vitamin D levels measured in a subset of the same cohort as this study were not associated with an increased risk of developing MS later in life, although concerns regarding sample quality have been raised. 19 This suggests that the effects may not be related to exposure during pregnancy but during the first months after birth.
Individuals born in the fall have less sunlight exposure during their first six months due to fewer daylight hours and more frequent indoor activity. In our study, these individuals had an increased risk of MS that declined during the 1960s, coinciding with a period of low breastfeeding rate in Sweden (

Changes in factors that affect vitamin D levels during the first 6 months of life and immunity to infections, based on data and general recommendations from Sweden years 1950–2019.
Active sunbathing and travelling to sunnier climates have also steadily increased since the 1960s. Incidence of cutaneous melanoma, a population-level indicator of high ultraviolet radiation (UVR) exposure, has gradually increased by approximately 5% annually.22,23 The rate of subsequent melanomas among patients previously diagnosed with a primary cutaneous melanoma has also steadily increased since the 1960s,(
24
Breastfeeding may also have a direct role in the risk of MS. 25 In a recent study from the same cohort, prolonged breastfeeding was associated with reduced MS risk only among men. 26 Possible explanations include effects on microbiota known to influence autoimmunity, 27 Th1 shift in formula-fed children, 28 and potential molecular mimicry between myelin oligodendrocyte glycoprotein, a MS autoantigen, and bovine butyrophilin in formula. 29 Differences in the risk association to breastfeeding by sex may indicate developmental differences in the immune system in utero or during childhood 30 or influence from other potential environmental confounders. 26 However, the seasonal timing of breastfeeding was not considered in the reported study, 26 and it is unknown if this effect changes over time.
Associations between MS and month of birth may also be independent of vitamin D. Exposure to UV radiation can also have direct immune-suppressive effects by altering inflammatory cytokine profiles and modulating regulatory T-cell activity independently from vitamin D production.
31
However, it is unclear whether the relationship stems from direct exposure in the infant or maternally either during pregnancy or through breastfeeding. A higher risk of infection during the winter could also contribute to the increased MS risk in those born in the fall. This may also explain the trends in the MS risk association as vaccination rates increased after the 1960s resulting in fewer active infections (
Differences in response to infection-related exposures may also explain the stronger risk association among males observed in this study. Passive immunity from breastfeeding is less beneficial for males resulting in a higher risk for neonatal respiratory infections. 33 Differences in immune response between sexes are also evidenced by the stronger antibody response from early vaccinations for females. 30 Infections less protected by passive immunity such as the respiratory syncytial virus, rotavirus, and influenza may affect the balance between immune tolerance and resistance during a susceptible period of development, potentially leading to an increased risk of MS. Vitamin D levels may also modulate the severity of the immune responses, which further motivates the possible relationship between the season of birth and MS risk.
In conclusion, our findings suggest that time of birth may influence the risk of MS when factoring in sex, year of birth, and geographical residence; however, the findings will require follow-up and additional validation.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PS, JHu, AKH, LA, JHi, AM, and IK declare(s) that there is no conflict of interest. Outside of this work, TO has received unrestricted MS research grants from Biogen, Novartis and Merck, including personal honoraria for advisory boards
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Magretha af Ugglas foundation, Swedish research council (2017-00777, 2020-01638), Horizon 2020 EU grant (MultipleMS, 733161), endMS Doctoral Studentship from the Multiple Sclerosis Society of Canada (EGID:304), the Swedish brain foundation, and the Knut and Alice Wallenberg foundation.
