Abstract
Background
Eighty-five percent of people with multiple sclerosis (MS) incur gait impairments debilitating enough to significantly impact their function.
Objectives
The aim of this study was to determine if a novel combination of intermuscular electrical stimulation, followed by functional electrical stimulation combined with supported bodyweight treadmill training, would improve gait, decrease spasticity and fatigue, and improve muscle strength.
Methods
Using a pre-post experimental design, we implemented this combination six-week protocol in 16 individuals with MS. We completed summary statistics and longitudinal pre-post results using Wilcoxon sign rank tests with Bonferroni adjustment.
Results
Participants responded with median increases of 29.4 feet (p < 0.0001) during the Six Minute Walk Test, median decreases of 0.7 s (p = 0.0011) in the 25-Foot Walk Test, median increases of 3.8 toe taps to fatigue (p = 0.0306) and median increases of 5.0 heel raises (p = 0.0093). Significant changes were noted in the Modified Ashworth Scale, both after intermuscular electrical stimulation (median change = −0.5 p = 0.0039) and after treadmill walking (median change = −0.5, p < 0.0005).
Conclusions
Results of this novel protocol suggest this intervention combination has the potential to decrease spasticity, and improve gait speed and endurance in individuals with MS. Observed changes in mobility occurred without accompanying increases in fatigue.
Keywords
Introduction
Multiple sclerosis (MS) is one of the most debilitating progressive conditions affecting young adults today. 1 Of the 2.3 million people in the world who live with MS, 85% have gait deviations, making it a hallmark of the condition. 1 A confluence of factors contribute to impaired gait. One factor, fatigue, affects 80% of people with MS, 2 while another, spasticity, affects 63.1% of people with MS. Spasticity targets the plantar flexors; leading to slow cadence, short step length, and decreased endurance during gait. 3 Other factors contributing to impaired gait include muscle weakness, joint contracture, and balance instability. 3 Due to impairments impacting functional mobility, people with MS often experience difficulty living independently, maintaining employment, and retaining social interaction. 4
Previous intervention studies have focused on each of these factors individually, but have overlooked the dynamic interplay among these factors that bring about loss of function in these individuals. For example, spasticity and weakness disproportionally affect lower as compared to upper limb performance and are especially problematic in the gastrocnemius and soleus muscles, responsible for 85% of forward propulsion during normal gait. 5 Impairments in the plantar-flexors negatively impact eccentric control during stance as well as concentric propulsion during toe-off, leading to decreases in step length and stance time, as well as a lack of toe-off.5,6 In turn, these impairments are associated with slow gait speed, poor endurance, fatigue, and impaired function.3,6 Furthermore, while studies have singularly addressed both spasticity 7 and gait training,8–10 their combined potential influence on fatigue and function has largely been overlooked. 3
A central unresolved question is how to best manage spasticity, while improving walking speed and function for people with MS. Researchers have previously used trigger point dry needling (TPDN) to reduce spasticity in persons with stroke and spinal cord injury.7,11–15 These studies demonstrate that short-term decreases in muscle tone are possible, however tone modulation in connection with gait after a retention period was not assessed. 16 Functional electrical stimulation (FES) has had a positive orthotic effect on walking, but most research used stimulation to either the fibular nerve and/or the pre-tibial muscles, not focusing on the importance of the gastrocnemius/plantarflexor muscles. 17
The aim of this study was to determine if intermuscular electrical stimulation (IMES) applied to the gastrocnemius muscle, followed by treadmill training combined with FES to the gastrocnemius during stance and the pre-tibial muscles during swing, would improve gait speed, improve gait endurance, decrease spasticity in the gastrocnemius, and enhance muscle strength in the gastrocnemius and in the pretibial muscle groups in individuals with MS. The combination of IMES and FES to achieve these objectives in individuals with MS is novel, and to our knowledge has not been previously utilized.
Materials and methods
Participants
Participants in this study were individuals diagnosed with relapsing remitting, primary progressive, or secondary progressive MS. No participant experienced a relapse, change in medication status, or change in disability status for the duration of this study. Inclusion criteria included age between 18 and 64 years and the ability to walk at least 25-feet prior to enrollment. Exclusion criteria included botulinum toxin injection or deep vein thrombosis in the six months prior to enrollment. Using a non-parametric repeated measures power analysis completed on the six-minute walk test, an effect size of 15 meters (minimal clinically important difference (MCID) is 15–30 meters) an alpha of 0.05, and power of 80%, we determined the number of participants needed was 16.
Study design
This study utilized a one-group repeated measures pretest/posttest design conducted at the Oklahoma Medical Research Foundation (OMRF) Multiple Sclerosis Center of Excellence. Study review was completed by the Internal Review Boards at both the University of Oklahoma Medical Center and the OMRF (OMRF IRB #19–21).
Pretest/posttest assessments
Research personnel utilized the Modified Ashworth Scale (MAS) 17 to determine which lower extremity showed more spasticity and would receive intervention using IMES and FES. On days 1 and 14, dedicated solely to outcome measurement, we completed the following pretest/posttest assessment measures: the number of single limb heel raises to fatigue, 18 the number of toe taps completed on a force plate in ten seconds, the 25-foot walk test (T25FW),19,20 the 6-Minute Walk Test (6MWT), 21 the Berg Balance Scale, 22 and the Modified Fatigue Impact Scale (MFIS). 23 The MFIS score is a self-scored scale based upon each participant's perception of how fatigue impacts physical, cognitive, and psychosocial aspects of their daily lives (Figure 1, Figure 2).
Measures repeated before and after each intervention
Research personnel repeated the Modified Ashworth Scale (MAS), and surface electromyography (SEMG) of the medial heads of both gastrocnemius muscles before and after each dry needling session, and before and after each FES + treadmill training session. We recorded each SEMG while the participant held a bilateral heel raise for five seconds.
Intervention sessions 1–4
Intervention sessions occurred twice a week. Sessions 1–4 began with IMES to the medial and lateral heads of the gastroc-soleus muscle of the more involved leg, followed by FES with harnessed treadmill training, at each participant's self-selected speed.
IMES protocol
One physical therapist certified in dry-needling completed all IMES interventions using a 30 mm monofilament needle and a dual channel electrical stimulation unit equipped with alligator clips. The therapist used a single channel for both needles with output current and pulse rate ranging from 0–20 mA and 2–50 Hz respectively, based on a visible involuntary contraction of 1–2 twitches per second. The therapist repeated electrical stimulation 30 s on and 30 s off, three consecutive repetitions for a total of 90 s of IMES.
Functional stimulation protocol
Research personnel used two 2 × 2 inch electrodes on the tibialis anterior, and two on the gastrocnemius with an EMPI® FES unit equipped with a thumb activation switch. Personnel used a biphasic symmetrical square waveform, with a phase duration of 400 μs and stimulation frequency of 50 Hz, to participant tolerance or until there was a sustained visible contraction.
Treadmill protocol
Participants walked on a harnessed treadmill (TM), with full body weight for 20 min, at a self-selected speed. Research personnel stimulated the gastrocnemius during stance phase and the tibialis anterior during swing phase of gait using the FES unit.
Intervention sessions 5–12
Sessions 5–12 consisted of TM + FES only, as described in Figure 1.

Flow of the study.
Data analysis
EMG analysis
Research personnel collected, processed, and analyzed the EMG data using Noraxon MR3 software: a) pre-processed subject data using an infinite impulse response 10 Hz Highpass and 500 Hz Lowpass Butterworth filter; b) calculated amplitude using the root mean squared method (RMS) with a 100 ms window; and used a three second window taken from the middle of each contraction to calculate the mean and peak signal amplitude for each limb.
Statistical analysis
Summary statistics were completed for socio-demographic factors and on all outcome measures. We analyzed pre-post outcome assessment differences using Wilcoxon sign rank tests. Although conservative in exploratory analyses, we utilized a Bonferroni adjustment to avoid type I errors. 24 All statistical analyses used SAS 9.4 (Carey, NJ) with an alpha = 0.05.
Post Hoc analysis
Because we noted differences in responses, we completed a post hoc analysis in which we divided the participants into two groups based on their distance walked during the 6MWT pretest Those with Expanded Disability Status Scale (EDSS) scores greater than or equal to five, who walked 300 meters or less were classified as the low distance group (LD). Those with EDSS scores less than five, who walked more than 300 meters were classified as the high distance group (HD). We analyzed the median values on the T25FW, the 6MWT, and the MFIS for these groups separately, using Wilcoxon sign rank tests to compare within group pre/post differences, followed by the Mann Whitney U test to determine between group differences.
Results
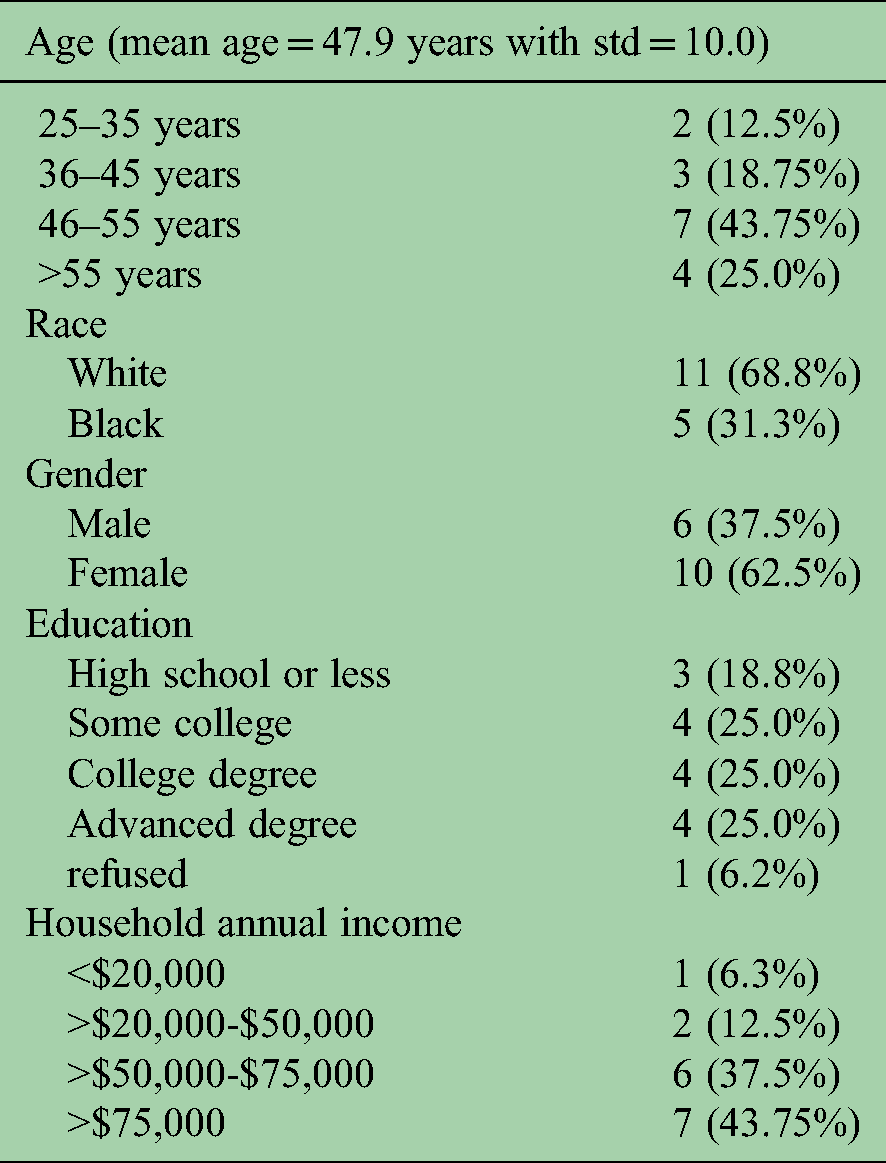
Sixteen subjects completed the study. Mean age of participants was 47.9 years with std = 10.0. Two thirds described themselves as female, two thirds reported being white, and one third reported being black. Three quarters of the participants attended at least some college, and almost half reported an annual household income of $75,000 or more (Table 1). Of the enrolled subjects, six had progressive MS and used a single point cane or walker for ambulation, and ten had relapsing remitting MS and did not use an assistive device.
Participant sociodemographic factors, number and percentage (n = 16).
Outcomes
Participants demonstrated a significant 29.4 meter median increase in distance walked during the 6MWT (p < .0001), and a significant 0.7 s median decrease in time needed to walk 25 feet during the T25FW (p = .0011). Fatigue did not change significantly; participants demonstrated a 2.0 point decrease in fatigue using the Modified Fatigue Impact Scale (p = .3549). Additionally, participants did not demonstrate significant change on the Berg balance test, with a median decrease of only 1.5 points (p = .1125) (Table 2).
Summary scores on standardized assessments at pretest, posttest and the difference. Wilcoxon signed rank test for significance of difference of the medians with S-statistic and p-value for differences between pretest and posttest (n = 16).

*Applying the Bonferroni adjustment for multiple comparisons makes the significant p-value = 0.01.
With regard to tone, the MAS decreased by a median score of 0.5 points after the completion of IMES (p = .0039) and a median of 0.5 points after the completion treadmill training (p = .0005). Median Peak EMG amplitude activity in the gastrocnemius muscle did not change significantly during heel raises after the completion of IMES portion of the intervention (median peak amplitude change of 1.9 with p = .7354), or at the end of all intervention (median peak amplitude change of 16.4 with p = 0.0574). Participants demonstrated a significant median increase of 5.0 heel raises to fatigue on the involved limb (p = .0093), compared to an insignificant median increase of 2 heel raises to fatigue on the uninvolved limb (p = .1099). The median number of toe taps a participant could complete in 10 s on the more involved leg increased by 3.8 taps (p = .0306) (Table 2).
Post Hoc analysis
The six-minute walk test (6MWT)
The LD group increased their walking distance by a median 49.2 meters (p = .0078), compared to a median 15.2 meters in the HD group (p = .0078), with a significant between group difference (p = .0136). Additionally, four of the eight LD participants exceeded the minimal clinically important difference of 15–30 meters,25–27 compared to one of the eight in the HD group (Table 3, Figure 3).

Standardized assessments utilized in this report.
Wilcoxon signed rank test for significance of difference of the medians with S-statistic and p-value for differences between pretest and posttest on the Six Minute walk test, 25-foot walk test, and the modified fatigue impact scale analyzed in the group of people scoring higher (n = 8) and a second group scoring lower (n = 8) on the Six-Minute walk pretest .

*Applying the Bonferroni adjustment for multiple comparisons makes the significant p-value = 0.01.
Timed 25 foot walk test (T25FW)
The LD group decreased their T25FW time by a median 1.8 s (p = .0156) compared to a median 0.2 s (p = .01953) for the HD group, with the difference between the groups not significant after the Bonferroni adjustment (p = .0239) (Table 3, Figure 4).

Difference between pretest and posttest in the six minute walk test in both the lower distance (LD) and higher distance (HD) groups (median and 95% CI).
Modified fatigue impact scale (MFIS)
Although the between group comparison of the MFIS was not statistically different (p-value = .4613), 80% of participants that exceeded the MCID of two points of were in the LD group (Table 3, Figure 5).

Absolute difference between pretest and posttest in the 25-foot walk test in both the lower distance (LD) and higher distance (HD) groups (median and 95% CI).

Absolute difference between pretest and posttest in the modified fatigue impact scale in both the lower distance (LD) and higher distance (HD) groups (median and 95% CI).
Discussion
In this study involving people with different types of MS, we completed a novel combination intervention using intermuscular electrical stimulation (IMES), functional electrical stimulation, and treadmill training. The primary focus of this study was to investigate whether this intervention was associated with meaningful changes in both gait speed and endurance without a concomitant increase in fatigue. We combined IMES to the gastrocnemius muscle with functional electrical stimulation (FES) to the pretibial muscle group during swing and the gastrocnemius during stance, combined with supported body weight treadmill training. Our intervention was associated with increases in distance walked during the six-minute walk test (6MWT), improvement in walking speed during the timed 25-foot walk test (T25FW), and improvements in spasticity as evidenced by increases in repetitions of heel raises and changes in the Modified Ashworth Scale (MAS). We did not, however, observe significant changes in Berg Balance Scale (BBS) scores, Modified Fatigue Impact Scores (MFIS), or number of toe taps (after Bonferroni adjustment).
While other studies have investigated the short-term effects of dry needling/IMES for the treatment of spasticity in individuals with stroke and spinal cord injury, our study is novel in that it used a two-pronged approach to address spasticity and gait impairments in individuals with MS.7,12–15 Slowed gait speed has been associated with fall risk in people with MS, 28 and the T25FW is often used to measure gait speed in this population as it is highly correlated with falls. 29 Coleman and associates suggest a 20% decrease in time in the T25FW constitutes a minimal clinically important difference (MCID). 29 In our study, 80% of participants decreased their T25FW time and those participants who entered with the slowest 6MWT times demonstrated almost two seconds median improvement in their T25FW walk time, meeting the MCID. Our results compare favorably to decreases in time between 0.4–1.2 s reported in the literature, achieved with a variety of interventions of similar or increased duration (8–12 weeks) and frequencies (3–5 times per week).25–27,30,31
Participants in this study also demonstrated meaningful changes in walking endurance as evidenced by their scores on the 6MWT. While the MCID on the 6MWT for people with MS has not been reported, across multiple ages and different pathologies studied, changes of 15–35 meters are considered minimally clinically important. 21 In our study, 75% of participants exceeded a change of 14 meters and the entire lower functioning group exceeded 32 meters. While these changes could be associated singularly with either the TM + FES portion of the intervention, or the IMES portion of the intervention, they could also be associated with their combination. The IMES portion of this intervention was associated with decreases in spasticity, as measured by the MAS, the number of toe taps and the number of heel raises, and may have partially or fully facilitated the observed changes. Improvements in the 6MWT compare favorably to those reported in the literature of between 14–56 meters achieved with a variety of interventions of longer duration (12 weeks); and higher frequency (3–5 times per week).25–27,30–35
The Modified Ashworth Scale (MAS), one of the most common clinical assessments of spasticity, has correlated well with self-reported spasticity. 17 In the current study, we measured waning spasticity in three ways: A decrease in participants’ MAS scores, an increase in the number of toe taps our participants could complete in ten seconds, and an increase in the number of heel raises participants could complete before fatigue. The findings from this study suggest that the number of toe taps and heel raises increases as spasticity in the plantar flexors wanes, notably in participants with greater spasticity at baseline.34,35
We used SEMG of the gastrocnemius as one measure of motor recruitment, which could potentially contribute to changes in muscle force. Sixty percent of the participants in this study demonstrated a positive change in the number of heel raises in the involved lower extremity. This improvement may indicate that as spasticity waned, participants were more successful in gastrocnemius muscle recruitment. Gastrocnemius recruitment was enhanced through intervention provided by the FES plus supported bodyweight treadmill training. In this study, only the more involved lower extremity demonstrated a statistically significant change in the number of heel raises to fatigue. The same extremity received IMES and TM + FES (Table 2). This finding lends support to the combined approach. Although the median changes in SEMG were not statistically significant (p = 0.0574), 66% of participants in this study increased their mean peak amplitude, prompting future examination with a comparative study.
We evaluated change in fatigue for people with MS using the MFIS, as it relates to the critical interaction between spasticity and gait. Nine of the sixteen participants (56%) reported improvements in their fatigue levels as measured by the MFIS, and five exceeded the MCID of 3.86–8.11 reported by Coleman and associates in 2012.
29
Of the eight lower functioning participants, six (75%) showed decreased scores on the MFIS, with four (50%) exceeding the MCID. This change suggests that the increased activity associated with this intervention did
Limitations
Because we did not have literature to support our choice for an efficacious dose of IMES in people with MS, we used parameters based on clinical judgement and previous studies in other neurological populations.7,12–15 Another limitation of the study was that we could not attribute changes we observed in spasticity to IMES, TM + FES, or a combination of these interventions. Additionally, this study might be subject to selection bias, as many of the participants were able to walk longer distances. Establishing more specific exclusion and inclusion criteria based on a functional test would have eliminated some of the ceiling effects we observed. Finally, we utilized the Berg Balance Scale as a measure of balance and fall prediction in this population of outpatients with MS. The lack of a statistically significant change in participants’ scores may be partially related to their high-test scores at baseline resulting in a ceiling effect. 33 Seven of our participants with lower baseline scores on the BBS did meet or exceed the MCID of two points for the Berg Balance Test in outpatients with MS. 33
Our study design did not include a control group, making the findings preliminary and proof of concept. However, we purposefully used a repeated measure design to determine the incremental effects of this combination of intervention techniques, and the dose related to change. These preliminary results will inform the next larger intervention study that will include a control group. For example, we will compare an IMES group with a combination IMES and treadmill group.
In conclusion, our findings suggest people with MS who have impaired gait speed and endurance may respond favorably to a combination of IMES combined with TM + FES. Our results suggest individuals with higher levels of gait impairment may have a more dramatic response, without related changes in fatigue, than individuals whose functional levels are already high. Our findings also suggest that the BBS and the MFIS might not be optimal measures of balance and fatigue in higher functioning individuals with MS, as they may lead to a ceiling effect. A longer duration of intervention may be needed for significant decreases in fatigue and improvements in balance to occur.
Footnotes
Acknowledgements
This study was funded by a grant from the Presbyterian Health Foundation, Oklahoma City, Oklahoma.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Presbyterian Health Foundation (grant number 21765-08).
