Abstract
Background
Elevated intrathecal immunoglobulin G (IgG; oligoclonal bands (OCBs)) or IgG in people with progressive motor impairment due to “critical” demyelinating lesions are of uncertain significance.
Objective
Compare clinical/radiological features of people with “critical” demyelinating lesion-induced progressive motor impairment with/without elevated intrathecal IgG synthesis.
Methods
A total of 133 people with progressive motor impairment attributable to “critical” demyelinating lesions (corticospinal tract location, consistent with the progressive motor deficit) were compared regarding clinical and radiological presentation with and without ≥2 unique cerebrospinal fluid (CSF) OCB and/or IgG index ≥0.85.
Results
Ninety-eight (74%) had CSF-elevated OCB and/or IgG index, higher with increased magnetic resonance imaging-lesion burden. No differences were found with/without CSF abnormalities in sex (46 of 98 female (47%) vs. 22 of 35 (63%), p = 0.11), onset-age (median 49 vs. 50 years, p = 0.5), progression from onset (62 of 98 (63%) vs. 25 of 35 (71%)), progression post-relapse (36 of 98 (37%) vs. 10 of 35 (29%), p = 0.4), and duration between demyelinating disease onset and CSF examination (30 (0–359) vs. 48 (0–323) months p = 0.7). “Critical” lesions were radiologically similar, most commonly cervical spine located (72 of 98 (74%) vs. 19 of 35 (54%), p = 0.18) both with/without CSF abnormalities.
Conclusions
People with “critical” demyelinating lesion-induced progressive motor impairment typically have elevated intrathecal IgG (OCB and/or IgG) and similar clinical and radiological presentation regardless of CSF findings, therefore representing valid presentations of progressive demyelinating disease.
Introduction
A hallmark feature of primary and secondary progressive multiple sclerosis (MS) that distinguishes it from other inflammatory myelopathies such as neuromyelitis optica and sarcoidosis is insidiously progressive, commonly asymmetric, motor impairment in the absence of active radiologically manifest inflammation. 1 The pathophysiology of progressive MS is incompletely understood, but increasing evidence supports a disproportionate role in the progressive motor impairment of individual central nervous system (CNS) demyelinating lesions in clinically eloquent locations along corticospinal tracts. We designate these as “critical” demyelinating lesions.”2,3 Progressive motor impairment due to a “critical” demyelinating lesion is exemplified by three cohorts: progressive solitary sclerosis (PSS; single CNS demyelinating lesion as the cause of progressive motor impairment) 4 ; progressive motor impairment due to one critical demyelinating lesion with 2 to 5 total CNS lesions (progressive paucisclerosis (PPS)) 5 ; and progressive, exclusively unilateral, hemiparesis or monoparesis due to a critical demyelinating lesion with unlimited (>5) CNS lesions (progressive unilateral hemiparetic MS (PUHMS)). 6
Some patients with highly restricted CNS lesion burden (PSS and PPS) do not conform specifically to the revised 2017 MS diagnostic criteria. 7 Documentation of immunoglobulin (Ig) production within the cerebrospinal fluid (CSF) characteristic of MS such as elevations in unique CSF oligoclonal bands (OCB)/IgG index replaced the need for demonstrated dissemination of lesions in time in the 2017 MS diagnostic criteria. Comparing clinical and radiological features in those with and without characteristic CSF-elevated OCB and/or IgG index may define whether all such patients with progressive motor impairment due to a critical demyelinating lesion are valid representations of progressive demyelinating disease.
Our hypothesis was that clinical and radiological features are similar in patients with progressive motor impairment due to a critical demyelinating lesion with or without characteristic CSF-elevated OCB and/or IgG index.
Patients and methods
A retrospective, comparative study on our population of Mayo Clinic patients with progressive motor impairment over 1 year due to a critical demyelinating lesion was designed by reviewing data from 1996 through 2020. The study was approved by the Mayo Clinic Institutional Review Board (IRB 09-7045).
Clinical evaluation
All patients were evaluated clinically by Mayo Clinic MS subspecialty neurologists. Study inclusion criteria were patients with progressive upper motor neuron impairment due to “critical” demyelinating lesion in keeping with PSS, PPS, or PUHMS with available CSF results to determine presence or absence of CSF findings characteristic of MS (i.e. unique elevations in OCB and/or IgG index). Patients were excluded if CSF analysis was not performed or if CSF analysis was recorded as performed but not confirmed within the clinical records, or if alternative etiologies for the progressive motor impairment apart from CNS demyelinating disease characteristic of MS was determined including compressive, infectious, inherited, nutritional, neoplastic or vascular disease or other immune-mediated demyelinating diseases such as neuromyelitis optica spectrum disorder (NMOSD) or myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD). Patients with the latter two diseases were excluded by the clinical presentation, neuroimaging findings, and when possible serological assessment specific to these immune demyelinating diseases. A peripheral nervous system involvement contributing to progressive motor impairment was checked by careful neurological examination by Mayo neurologists and often with nerve conduction studies and electromyography (EMG).
Clinical evaluations recorded included patient sex, age at CNS demyelinating disease onset, clinical course (relapse onset with subsequent clinical improvement prior to progressive motor impairment, “secondary progressive”; or progressive motor impairment from the onset, “primary progressive”), and the duration between demyelinating disease onset and the CSF examination. PSS was defined as progressive motor impairment for ≥1 year attributable to a “critical” demyelinating lesion with a CNS magnetic resonance imaging (MRI) burden of a single (1) lesion. PPS was defined as progressive motor impairment for ≥1 year attributable to a “critical” demyelinating lesion with a total CNS MRI burden of between two and five demyelinating lesions. PUHMS was defined as progressive, exclusively unilateral, hemi- or monoparesis motor impairment for ≥1 year attributable to a “critical” demyelinating lesion with >5 total CNS MRI demyelinating lesions.
Cerebrospinal fluid
A CSF examination was performed by lumbar puncture in all patients and interpreted either at Mayo Clinic or at local referral centers. Characteristic CSF-elevated OCB and/or IgG index were defined as either or both of ≥2 unique CSF abnormal oligoclonal bands (OCB), compared with serum OCB, or IgG index ≥0.85. CSF kappa-free light chains were not commonly recorded so were not considered in this study. Concomitant CSF values (e.g. white blood cell count, protein, glucose, autoimmune disease markers, and malignant cytology) were assessed to exclude diagnoses inconsistent with MS as the cause of the progressive motor impairment. Documentation of normal or abnormal OCB or IgG index without explicit numeric values was accepted when clearly recorded by the assessing neurologist.
Neuroimaging
Multiple MRI brain, cervical, and thoracic spinal cord performed both at Mayo Clinic or local referral centers were reviewed by an experienced neuroradiologist (SM) and MS subspecialty neurologists (EPF and BMK) to assess the location and radiological features of the “critical” demyelinating lesion assessed for anatomical correlation with the progressive motor impairment (e.g. along corresponding corticospinal tract), often associated with focal atrophy. Further review of the neuroimaging and additional investigations were performed as described previously4–6 to exclude diagnoses inconsistent with MS as the cause of the progressive motor impairment.
Statistical analysis
Statistical tests were performed with JMP 14 software. Categorical variables were described by percentages and comparisons between these analyzed using Fisher's exact test continuous variables were described by mean (standard deviation) or median and differences between these were analyzed using (unpaired) t-test. The likelihood ratio was used for variables with >20% of its categories having a value <5. A p-value of <0.05 was considered statistically significant. Due to the exploratory nature of the study, no adjustment for multiple comparisons was made.
Results
Demographics, clinical course, and CSF findings
The CSF examination results, demographics, and clinical course data for the grouped data are presented in Table 1. Characteristic CSF-elevated OCB and/or IgG index were found in 98 patients (74%). Of those with CSF-elevated OCB and/or IgG index, 53 had isolated elevations of unique CSF oligoclonal bands, 12 had isolated elevated CSF IgG index, and 29 had elevations in both CSF OCB and IgG index. Four patients with documented CSF-elevated OCB and/or IgG index but did not have numeric results available.
Demographics, clinical course, and CSF findings.

CSF: cerebrospinal fluid; PPMS: primary progressive multiple sclerosis; SPMS: secondary progressive multiple sclerosis; CMJ BSt: cervico-medullary junction, brain stem; PSS: progressive solitary sclerosis; PPS: progressive paucisclerosis; PUHMS: progressive unilateral hemiparetic multiple sclerosis.
Subgroup data based on patient cohorts are presented in Table 2. Percentages of abnormal CSF patients in the subgroups were (PSS (N = 29), 52%; PPS (N = 56), 70%; PUHMS (N = 48), 92%). No differences were found between patients with and without characteristic CSF-elevated OCB and/or IgG index in the combined group or between subgroups on demographics, clinical course, duration between symptoms onset or onset of progressive motor impairment, and timing of CSF examination. Eighty-four patients met the 2017 revised McDonald criteria for MS. Twenty-five patients not meeting the revised McDonald criteria were in the PSS group, 23 in the PPS group, and one in the PUHMS group. Serology for aquaporin-4 IgG antibody was performed in 48 patients and all were negative. Serology for myelin oligodendrocyte glycoprotein antibodies was performed in 18 patients with one having a positive serology (titer 1:100), reviewed, and concluded to be a false-positive. Nerve conduction studies and EMG were performed in 79 patients and no alternative peripheral nerve disease process was found to be a cause for the progressive impairment.
Subgroup data based on patient cohorts.

PSS: progressive solitary sclerosis; CSF: cerebrospinal fluid; PPS: progressive paucisclerosis; PUHMS: progressive unilateral hemiparetic multiple sclerosis; SPMS: secondary progressive multiple sclerosis; PPMS: primary progressive multiple sclerosis; CMJ BSt: cervico-medullary junction, brain stem.
Neuroimaging
MRI findings of both the “critical” demyelinating lesion and other CNS demyelinating lesions were similar in patients with and without characteristic CSF-elevated OCB and/or IgG index. Representative imaging findings of the “critical” demyelinating lesion in patients with and without characteristic CSF-elevated OCB and/or IgG index are presented in Figure 1. The “critical” demyelinating lesion location was similar in those with and without characteristic CSF-elevated OCB and/or IgG index both with the highest frequency location in the cervical spinal cord (Tables 1 and 2).
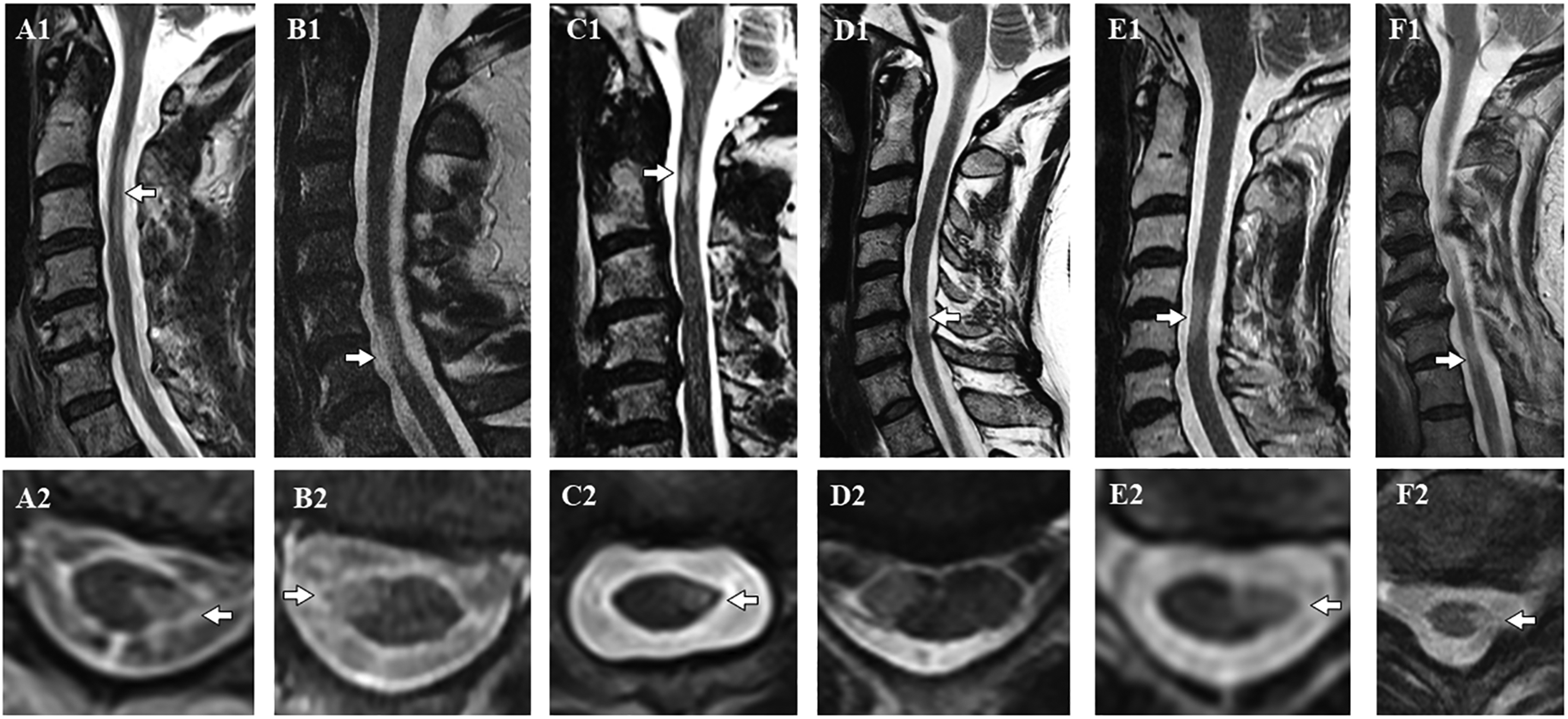
(A1) Sagittal T2 and (A2) axial MRI cervical cord in a man with PPS and abnormal CSF with a progressive from onset clinical course, (B1, B2) sagittal T2 and axial MRI cervical cord in a man with PSS, and abnormal CSF (elevated OCB/IgG index) with a progressive from onset clinical course, (C1, C2) sagittal T2 and axial MRI cervical cord in a woman with PUHMS and normal CSF with a relapse onset progressive hemiparetic clinical course, (D1, D2) sagittal T2 and axial MRI cervical cord in a woman with PSS, and normal CSF with a progressive from onset clinical course, (E1, E2) sagittal T2 and axial MRI cervical cord in a woman with PPS, and normal CSF with progressive from onset clinical course, (F1, F2) sagittal T2 and axial MRI cervical cord in a man with PUHMS and normal CSF with progressive hemiparetic clinical course.
Discussion
This study showed no demographic, clinical, or radiological differences in the “critical” demyelinating lesion between those with and without characteristic CSF-elevated OCB and/or IgG index of MS (elevated unique OCB and/or IgG index) in patients with progressive motor impairment due to a critical CNS demyelinating lesion. This finding supports the consideration of both those with and without CSF-elevated OCB and/or IgG index as valid clinical presentations of progressive demyelinating disease. This finding was for the whole study population of those with progressive motor impairment due to critical CNS demyelinating lesions and was maintained when considering subgroups with highly restricted lesion load (PSS and PPS), some of whom may not satisfy the 2017 revised MS diagnostic criteria. This finding remained regardless of progressive from onset (primary progressive), relapse onset with subsequent progressive motor impairment (secondary progressive) clinical course, duration between disease onset, and CSF examination.
CSF-elevated OCB and/or IgG index were more common in subgroups with higher total MRI lesion burden (PUHMS>PPS>PSS). The reported frequency of characteristic CSF-elevated OCB and/or IgG index varies with MS clinical course, often the overall MRI disease burden and occasionally disease duration with highest abnormalities seen in relapsing–remitting MS (RRMS), patients with higher MRI lesion burden and longer disease duration. CSF-elevated OCB and/or IgG index was found in 65% in the largest international cohort of a radiologically isolated syndrome 8 CSF-elevated OCB and/or IgG index in clinically isolated syndrome (CIS) overall is reported to be 61% with associations found on: baseline MRI lesion burden (OCB-positive: 27% with 0 lesions, 64% with 1–9 lesions; 83% with 10 or more lesions) and degree of fulfillment of Barkhof MRI criteria (BC) 9 (OCB-positive: 31% with 0 BC; 69% with 1–2 BC; 85% with 3–4 BC). 10 Frequency of CSF-elevated OCB and/or IgG index in RRMS range from 82% to 95%.11–13 One small RRMS comparison study found no difference in OCB-negative (n = 11) versus matched OCB-positive (n = 22) MS patients regarding BC, but OCB-negative patients were found to have more juxtacortical, and fewer infratentorial MRI lesions. 14 Reports of CSF-elevated OCB and/or IgG index in progressive forms of MS range as well from 85% in secondary progressive MS, 12 and 78% to 91.1% in primary progressive MS.12,15,16
A large Canadian study showed most patients with MS had detectable OCBs (72.5%) with a slightly higher prevalence in those with primary progressive MS (PPMS) than those with relapse onset MS without difference in disease progression over time. 17 A large multinational MSBASE study reported CSF-elevated OCB and/or IgG index in clinical courses of MS to be: CIS (63%), primary progressive (83%), progressive–relapsing (90%), relapsing–remitting (82%), and secondary progressive (85%); however, most of the cohort did not have CSF analysis performed or reported. 12 Among 170 patients with serial CSF examination, 22% changed from “normal” to “abnormal” demonstrating that repeat CSF later in the clinical course can alter the testing result.
Patient demographic and latitudinal location have been assessed in CSF-elevated OCB and/or IgG index in demyelinating disease. A Swedish study found no difference in sex predominance, age at disease onset, the proportion with a primary progressive course, or rate of MRI positivity between OCB- positive and -negative MS patients. 18 Other factors such as latitudinal gradient have been found to alter CSF OCB prevalence in large multinational studies,12,19 but this latitudinal gradient was not confirmed in Argentina. 20
The use of CSF-elevated OCB and/or IgG index in the diagnostic criteria for MS has changed over subsequent revisions regaining diagnostic importance on the 2017 revised McDonald criteria as a substitute for dissemination in time that has assisted in facilitating an earlier diagnosis of MS. 21 While it is possible that there are separate processes leading to two distinct forms of MS (i.e. those with and without abnormal CSF findings), there is sparse evidence to support that. One study showed differences in human leukocyte antigen -DR beta 1 (HLA-DRB1) genotypes between OCB-positive and -negative MS patients. 18 Increasing inflammatory MRI lesion load may contribute to more active B cell follicles that perhaps are intrathecal producers of CSF OCB and IgG index, 22 and this is a plausible explanation for the higher frequency of CSF-elevated OCB and/or IgG index in the PUHMS group in our study.
Limitations to the current study include that this study was a convenience sample and was not powered to detect differences between the groups with and without CSF-elevated OCB and/or IgG index. While CSF-elevated OCB and/or IgG index evaluated in this study are highly characteristic of MS, they occur in other non-demyelinating CNS diseases such as neurosarcoidosis, CNS lymphoma, and some CNS infections. These were carefully excluded prior to inclusion in this study. Neither elevations in unique CSF OCB and IgG index, nor insidiously progressive motor weakness, nor the accompanying radiologic characteristics of spinal cord lesions evaluated in this study are common in alternative demyelinating diseases such as aquaporin-4 IgG seropositive NMOSD and MOGAD. Many other clinical and radiological features discriminate those conditions from progressive demyelinating diseases including MS as well, but serological testing for these alternative diagnoses was not available for all patients.23,24 Assessment of CSF oligoclonal bands continuously evolved over the time period of the study and was performed in laboratories apart from Mayo Clinic. While some institutions assess alternative patterns of OCB, we exclusively regarded those OCBs unique to the CSF and not those additionally present in the serum.
While CSF examination findings vary substantially in MS and the presence of markers of intrathecal IgG synthesis are not entirely specific for MS, their detection provides strong support in an appropriate clinical context for diagnosing MS and assessing for alternative diagnoses. There remains a need for great vigilance when assessing patients with progressive MS to rule out alternative causes of the impairment with confidence. We found that CSF-elevated OCB and/or IgG index did not distinguish patients with progressive motor impairment due to critical demyelinating lesions, and as such, both those with and without such abnormalities are valid presentations of progressive demyelinating disease.
Footnotes
Acknowledgement
Dr. Keegan acknowledges the Center for MS and Autoimmune Neurology Research and Applebaum Family Philanthropic support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
