Abstract
Background
There is limited data analyzing the safety and effectiveness of dimethyl fumarate (DMF) in the progressive multiple sclerosis (PMS) population.
Objective
To analyze the safety and effectiveness of DMF in patients with PMS.
Methods
We used Cox proportional hazards models to compare the time to confirmed worsening and improvement on the Expanded Disability Status Scale (EDSS) and timed 25-foot walk (T25FW) between patients treated with DMF and glatiramer acetate (GA) for at least one year.
Results
We included 46 patients treated with DMF and 42 patients treated with GA. The safety and tolerability of GA and DMF were consistent with established profiles. There was no difference in confirmed EDSS progression. A trend towards reduced T25FW was seen in the DMF compared to GA after adjustment (HR = 0.86; 95% CI:0.37, 1.98; p = 0.72 and HR = 0.60; 95% CI:0.27, 1.34; p = 0.21, respectively).
Conclusion
Dimethyl fumarate showed a trend towards reduction in T25FW but no evidence of clinically significant impact on EDSS. The small sample precluded definitive determination.
Keywords
Introduction
Dimethyl fumarate (DMF) is an FDA approved oral disease-modifying agent for relapsing forms of multiple sclerosis (MS). Two phase three trials supported an anti-inflammatory effect, with demonstrated reduction of annualized relapse rate, patient proportion with disability progression, and new MRI lesions in relapsing MS patients when compared to placebo or glatiramer acetate (GA).1,2
Currently, it is unknown whether DMF might slow worsening in a progressive multiple sclerosis (PMS) population. The INSPIRE trial, a phase three multicenter, randomized, double-blind, and placebo-controlled study of the effectiveness and safety of DMF in secondary progressive multiple sclerosis (SPMS) was terminated in 2017 after enrolling 58 participants and left unanswered questions regarding the possible effects of this drug in treating PMS. Registry study data is also limited. MSBase conducted a retrospective analysis of disease-modifying agents on short and medium-term disability outcomes in primary progressive multiple sclerosis (PPMS) and SPMS using their cohort. Unfortunately, very few DMF treated patients (n = 7) qualified for study inclusion.3,4
Mechanistically it is plausible that DMF might favorably impact progressive MS by either reducing inflammation or neurodegeneration. DMF and its hydrolyzed form, monomethyl fumarate (MMF), activates nuclear factor erythroid 2–related factor 2 (Nrf2). Nrf2 modulates cellular responses to the oxidative stress pathway.5–8 It activates hydroxycarboxylic acid receptor 2 (HCAR2), which causes downstream inhibition of NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells).5,9 It also reduces CD8+ and natural killer (NK) cells in the peripheral blood.10,11 Though this combination of DMF’s immunomodulatory and neuroprotective effects might theoretically benefit progressive MS populations, 5 data is currently limited.
Material and methods
Study design, subjects, inclusion criteria
This was an observational retrospective study in a single center. Data was collected from the Partners Oracle Database (Harvard Multiple Sclerosis Patient Database) and the Comprehensive Longitudinal Investigation of MS at Brigham and Women’s Hospital (CLIMB) database.
Patients at our institution with progressive forms of multiple sclerosis treated with DMF or GA from 2000 to January 2020 were eligible for inclusion. A diagnosis of primary progressive MS (PPMS), secondary progressive MS (SPMS), or relapsing-progressive MS by a clinician was required. Patients had to be treated for at least 12 months and were aged 18–79 years. Dimethyl fumarate and GA are approved medications for the treatment of relapse-remitting MS and the decision to start these treatments was made by the MS specialist in conjunction with the patient. In order to ensure that we were capturing a truly progressive population, we only enrolled patients with an Expanded Disability Status Scale (EDSS) > 3. We also excluded patients with an EDSS ≥7 in order to ensure that assessed patients were ambulatory. Patients were required to have a baseline visit within a 90-day-window before or after the treatment start-date. This was considered time zero for the analysis. A minimum of two recorded follow-up visits was required. Typically, visits to our clinics occurred every six months though ultimately, this was dictated by clinical need.
Patients who were reported noncompliant, received concurrent disease-modifying therapies, or had either DMF or GA preceded within 3 months by a treatment of longer duration of effect (cyclophosphamide, rituximab, alemtuzumab, cladribine, ocrelizumab, daclizumab, mitoxantrone or mycophenolate) were excluded from the study. We also performed a sensitivity analysis excluding patients who received these treatments of longer duration or natalizumab within one year prior to treatment start to assess whether this would impact our overall results.
The patients were included from treatment initiation to treatment stop date or until the last follow-up available in the medical records. There were five exceptions to this. In the DMF group, one patient was censored due to follow-up discontinuation and switch of medication at another institution. In the GA group, four patients were censored after receiving treatment for at least one year: one patient suffered a myocardial infarction which affected functional status; one patient developed GA injection allergy and interrupted the treatment for desensitizing; one patient had reduced dosage regimen; and one patient became non-adherent to medication.
We collected age, sex, diagnoses date, disease category (PPMS, SPMS, or progressive-relapsing MS), relapses during the year prior to treatment start, SPMS conversion date, treatment duration, prior treatment regimen, EDSS, timed 25-foot walk (T25FW), the assistive device used for T25FW, dalfampridine use, steroid use, discontinuation reason, new treatment initiation, and reported side effects. Absolute lymphocytes count (ALC) was collected in the DMF group to assess lymphopenia. Lymphopenia sustained for at least three months in two sequential laboratory assessments was classified according to the Common Terminology Criteria Adverse Events in grade I (800–999/μl), grade II (500–799/μl), grade III (200–499/μl), or grade IV (<200/μl). We also included MRI information for those patients with at least two MRIs during treatment to compare for new T2 lesion or gadolinium-enhancing lesion.
Definition of progression
Confirmed EDSS progression was defined as an increase of 1.0 point from baseline maintained for at least 12 weeks with baseline EDSS below 5.5 or an increase of 0.5 points maintained for at least 12 weeks for baseline EDSS equal to or above 5.5. Confirmed T25FW worsening was determined by an increase of 20% from baseline assessment confirmed in a follow-up visit at least 12 weeks later. A sensitivity analysis excluding visits with a change in dalfampridine use or a change in assistive device was performed.
Definition of improvement
Confirmed disability improvement was defined by a 1.0 point or greater decline in EDSS that was confirmed on a subsequent visit at least 12 weeks later. Similarly, T25FW was considered improved if a 20% time decrease occurred and was maintained at a subsequent follow-up visit at least 12 weeks later.
Statistical analysis
To compare the difference of the time to disease progression between DMF and GA, we analyzed confirmed EDSS progression and confirmed T25FW worsening using Cox proportional hazard models. Since there were two subjects observed on both treatments, we used robust standard errors to account for the correlation within a subject. A multivariable analysis controlling for age, sex, disease duration, and baseline EDSS was performed to adjust for confounding. In addition to the multivariable Cox proportional hazards model, we also estimated the group differences adjusting for confounding using the propensity score. The propensity score model included the same confounders, and the propensity score was used in two ways: (1) adjusted for using a Cox proportional hazards model and (2) in an inverse probability of treatment weighted Cox proportional hazards model. 12 The balance between the treatment groups after inverse probability weighting was confirmed using the standardized mean difference (Supplementary Table 1). 13
We performed several sensitivity analyses to account for other treatments or devices. First, given the potential for dalfampridine to impact EDSS or T25FW, we removed all subjects treated with dalfampridine at baseline. Second, to account for the impact of assistive devices on the outcomes, we also removed subjects using an assistive device at baseline. Third, we completed all analyses excluding patients who received recurrent/concomitant steroids. Finally, we completed all analyses excluding subjects who had taken a treatment with long-term effect within 12 months.
To compare the difference in time to disease improvement between the groups we also analyzed confirmed EDSS improvement and confirmed T25FW improvement using a multivariable Cox proportional hazard models and propensity score methods. A linear mixed-effects model to evaluate EDSS change over time comparing both groups was also conducted. All statistical analyses were completed in the statistical package R version 3.6.3 (www.r-project.org).
Results
A search in our database identified 207 patients with progressive MS initially qualifying for inclusion: 82 patients treated with DMF and 125 patients treated with GA. After screening and validation of patients’ medical records, 88 patients were included in the study: 46 patients in the DMF group and 42 patients in the GA group (Figure 1), two patients received treatment with both GA and DMF at separate time intervals and as such were included in both groups. Population baseline characteristics at the time of either DMF or GA start are reported in Table 1. The mean age at treatment initiation was 56.4 (SD ± 10.1) in the DMF group and 52.3 (SD ± 9.1) in the GA group (p = 0.05). The baseline characteristics did not differ by sex, EDSS, or T25FW. Absence of relapses during the year before baseline was found in 44 patients in the DMF group. In the GA group, 37 patients presented with no relapses in the year prior to treatment initiation.
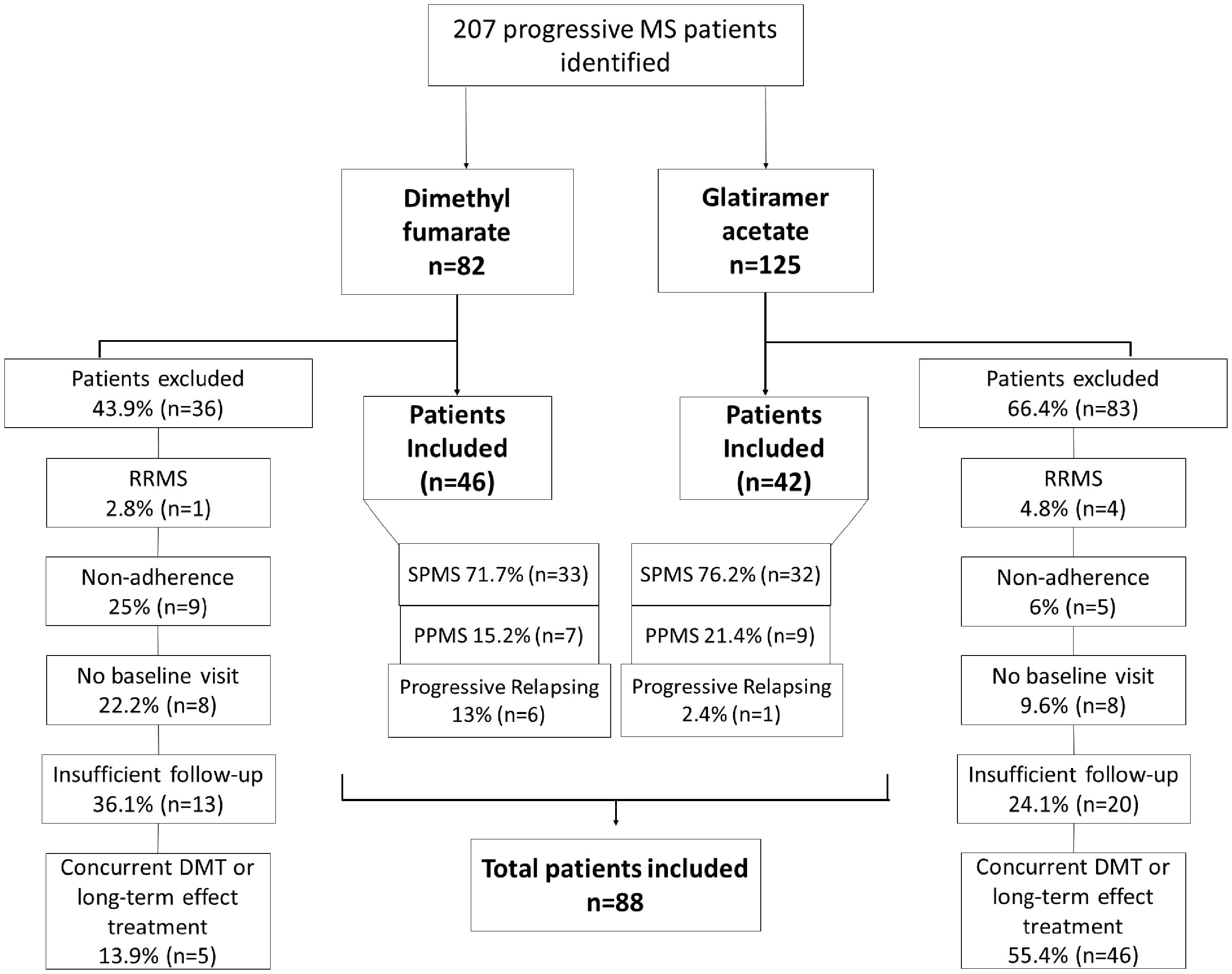
Study flowchart. RRMS: Release-remitting MS; SPMS: Secondary progressive MS; PPMS: Primary progressive MS. DMF: Disease-modifying therapies
Baseline characteristics.

Abbreviations: SPMS = Secondary Progressive Multiple Sclerosis; PPMS = Primary Progressive Multiple Sclerosis; EDSS = Expanded Disability Status Scale; T25FW = Timed 25-foot walk; SD = Standard deviation; n = number of subjects.
a Conversion from RRMS to SPMS in 8/33 patients in the DMF group and 21/33 patients in the GA group was already present since the patient’s first visit to our Institution. SPMS conversion was considered as the first visit date to the Brigham MS Center for these patients.
The most recent prior disease-modifying therapy is listed in Table 2. Fifteen patients in the DMF group were previously treated with GA and four patients in the GA group were previously treated with DMF. These patients satisfied inclusion criteria only on the occasion of DMF or GA treatment, respectively; hence, they were not included in both groups. Patients in the DMF group were treated for a mean of 3 years (SD ± 1.5), GA patients 4.5 years (SD ± 4).
Prior disease-modifying therapy.
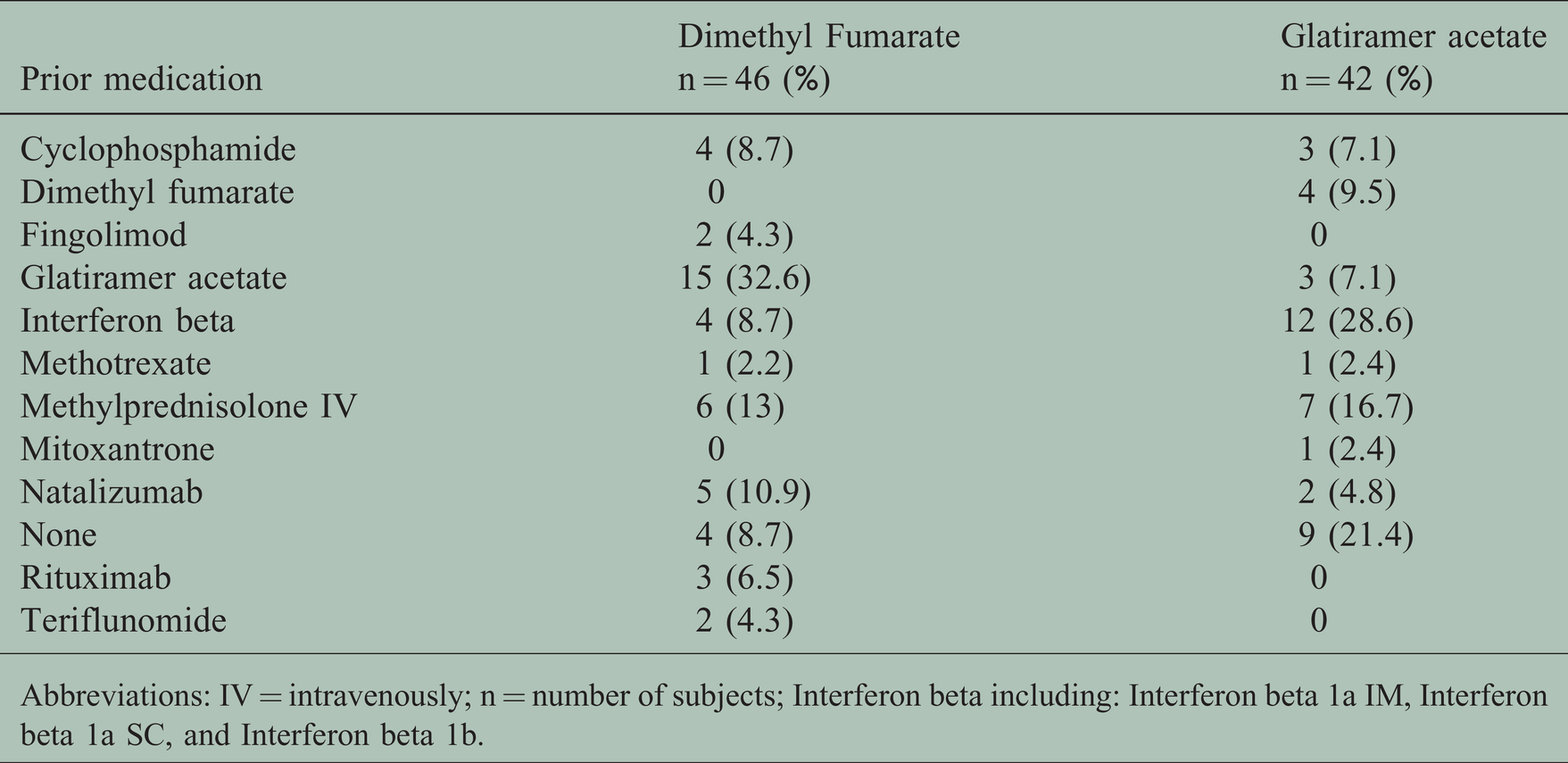
Abbreviations: IV = intravenously; n = number of subjects; Interferon beta including: Interferon beta 1a IM, Interferon beta 1a SC, and Interferon beta 1b.
Treatment discontinuation information is provided in Table 3. Discontinuation reason was available for 80.4% of patients in the DMF group and 52.4% of patients in the GA group (Table 3). The proportions of medication initiated after discontinuation was roughly similar with a few exceptions. A higher proportion of GA patients were transitioned to mycophenolate mofetil (20% of GA patients, 6.5% of DMF patients). More DMF patients were transitioned to ocrelizumab (35.5% of DMF patients vs. 10% of GA patients) or teriflunomide (26% of DMF patients vs. 15% GA treated patients).
Treatment discontinuation reason.

aCase previously reported. 14
Recurrent steroid therapy characterized by monthly steroid treatment for equal or more than six months was received by 13% (n = 6) of patients in the DMF group and by 40.5% (n = 17) of patients in the GA group. We identified four patients in the DMF group and two patients in the GA who qualified for sensitivity analysis based on prior long-duration treatment or natalizumab as defined in the methods above.
MRI data during treatment was available for comparison in 52.2% (n = 24) of patients in the DMF group and 69% (n = 29) of patients in the GA group. During follow-up, new T2 lesions occurred in 2 patients in the DMF group and 4 patients in the GA group.
Tolerability and Safety
One patient who initiated treatment with DMF six months after being diagnosed with PPMS developed progressive multifocal leukoencephalopathy (PML) after two years of treatment and immediately stopped DMF therapy. This case has previously been reported. 14 In the DMF group, 47.8% (n = 22) of patients developed lymphopenia [absolute lymphocyte count (ALC) <1000/μl] sustained for at least three months in two sequential laboratory assessments. Five patients had low lymphocytes (ALC 640-980/μl) at baseline. Grade I lymphopenia occurred in 8.7% of DMF treated patients, grade II occurred in 28.3% of DMF treated patients, and grade III lymphopenia occurred in 10.9% of patients. Flushing and gastrointestinal upset were reported in 43.5% (n = 20) and 15.2% (n = 7) of patients, respectively. Both side effects occurred mainly within the first month of treatment initiation. The most common side effect in the GA group was skin irritability related to injection, reported in 23.8% (n = 10) of patients. Allergic reaction and lipodystrophy were also reported in one patient each. Otherwise, GA was well tolerated.
Effectiveness – Clinical endpoints
EDSS
Confirmed EDSS progression occurred in 23.9% (n = 11) of patients in the DMF group versus 33.3% (n = 14) of patients in the GA group (Figure 2 and Table 4). There was no difference in the hazard of confirmed EDSS progression in the DMF group compared to the GA group with a wide 95% confidence interval (HR = 0.86; 95% CI: 0.37, 1.98; p = 0.72) after adjustment for age, sex, disease duration, and baseline EDSS. Results were similar when propensity score methods were used (Supplementary Table 2).
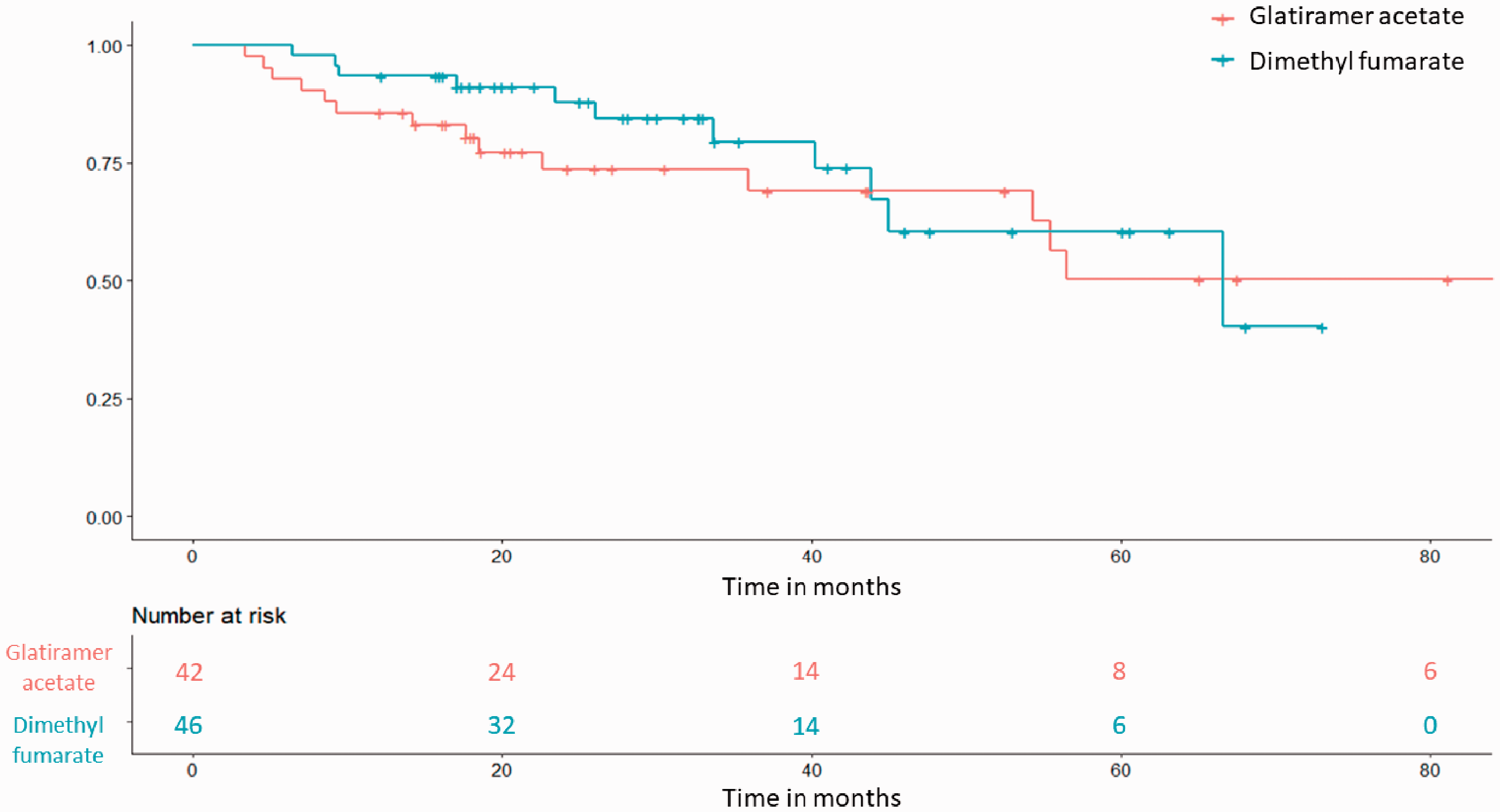
Kaplan Meier curve for the time to confirmed EDSS progression.
Confirmed EDSS progression.
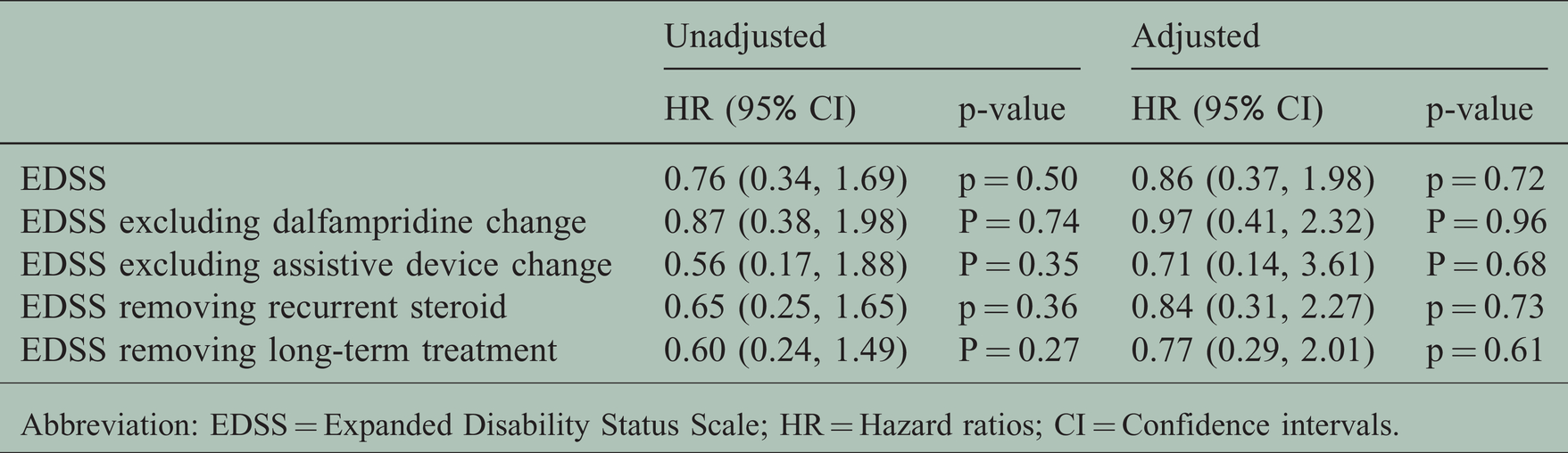
Abbreviation: EDSS = Expanded Disability Status Scale; HR = Hazard ratios; CI = Confidence intervals.
Results were maintained after sensitivity analyses were conducted removing patients 1) who experienced a change in dalfampridine use 2) who experienced a change in assistive device use 3) who received recurrent/concomitant steroid or 4) who received long-duration therapies or natalizumab one year prior to either GA/DMF start (Table 4).
Confirmed EDSS improvement occurred in 23.9% (n = 11) of patients in the DMF groups and 7.1% (n = 3) of patients in the GA group. A moderate difference in the DMF group compared to the GA group was found after adjustment for age, sex, and baseline EDSS (HR = 4.47; 95% CI: 0.97, 20.57; p = 0.05), but the confidence interval was quite wide. A linear mixed-effects model showed a limited difference in EDSS change over time between groups with EDSS change per year in GA = 0.14 (CI: 0.06, 0.21) and in DMF = 0.04 (CI: −0.21, 0.019).
T25FW
Baseline T25FW was available for 95.7% (n = 44) of patients in the DMF group and 100% (n = 42) of patients in the GA group (Figure 3 and Table 5). Of these, confirmed T25FW worsening occurred in 22.7% (n = 10) of patients in the DMF group and 33.3% (n = 14) of patients in the GA group. There was a trend towards a difference in terms of time to progression and confirmed T25FW progression between DMF and GA (HR = 0.60; 95% CI: 0.27, 1.34; p = 0.21) after adjustment for age, sex, disease duration, and baseline EDSS. Results were similar when propensity score methods were used (Supplementary Table 2).
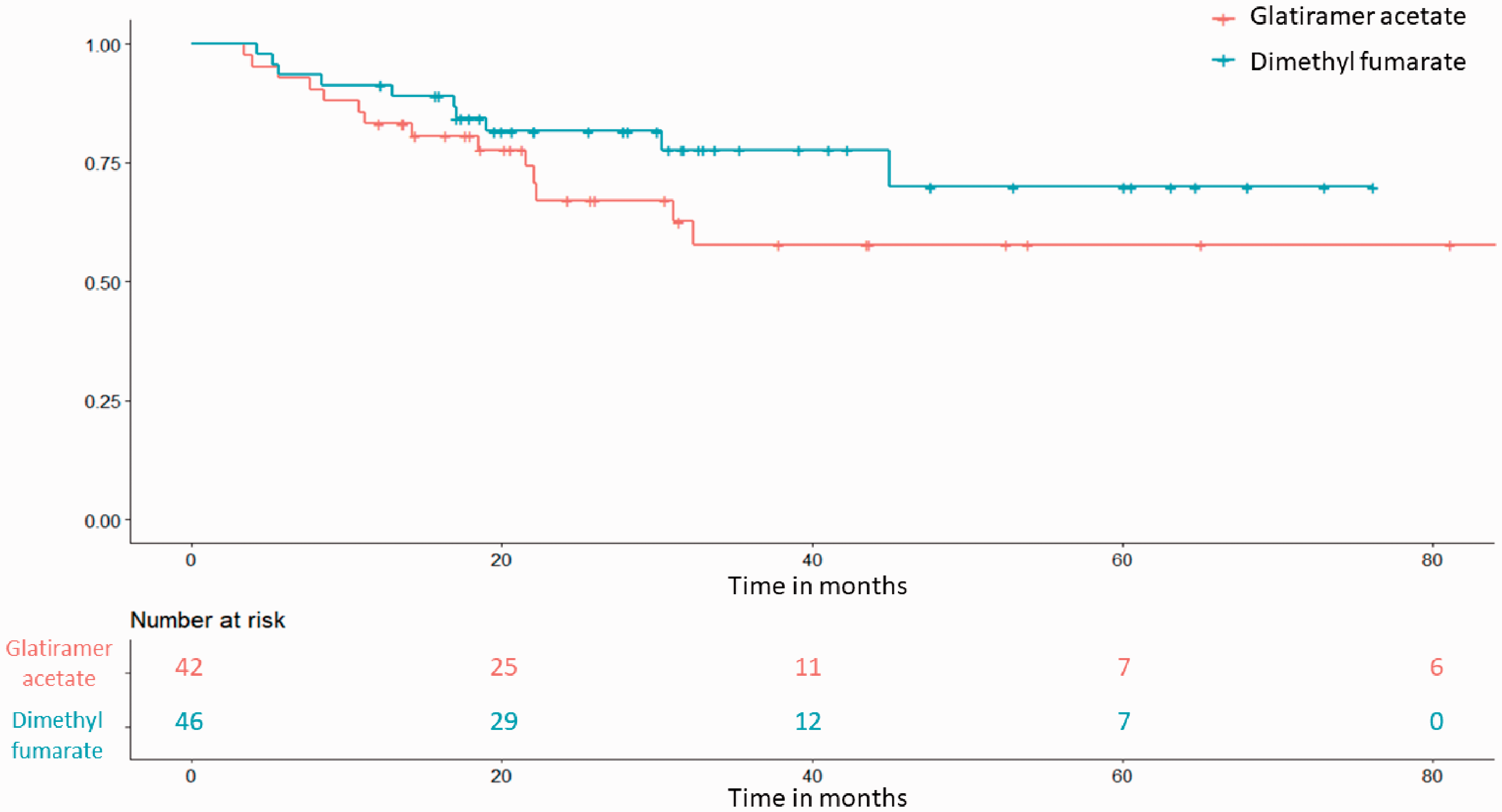
Kaplan Meir curve for the time to confirmed T25FW worsening.
Confirmed T25FW worsening.
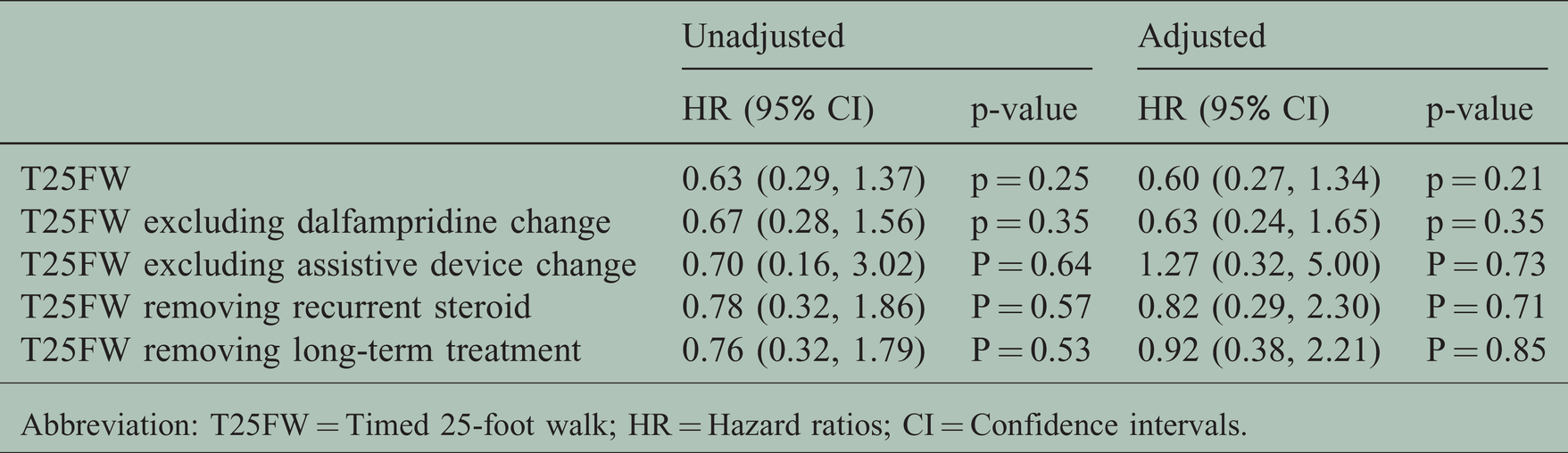
Abbreviation: T25FW = Timed 25-foot walk; HR = Hazard ratios; CI = Confidence intervals.
Similarly, we found a limited reduction in the risk of confirmed T25FW worsening after a sensitivity analysis removing patients 1) who experienced a change in dalfampridine use 2) who experienced a change in assistive device use 3) who received recurrent/concomitant steroid or 4) who received long-duration therapies or natalizumab one year prior to either GA/DMF (Table 5). Increased assistance with walking occurred in 30.4% (n = 14) of DMF and 45.2% (n = 19) of GA treated patients.
A decrease in walking assistance occurred in 6.5% (n = 3) of patients in the DMF group and 2.4% (n = 1) of patients in the GA group. Confirmed T25FW improvement did not differ between groups in the unadjusted analysis (HR = 0.91; 95% CI: 0.34, 2.42; p = 0.84) or after adjusting for age, sex, disease duration, and baseline EDSS (HR = 0.75; 95% CI: 0.26, 2.11; p = 0.58)
Discussion
The overall side effect and safety profile in this older progressive population was consistent with expectations based on phase 3 trial and post-marketing information for each medication. The most serious safety concern identified was a previously reported case of PML in the DMF group. 14 Lymphopenia (ALN <1000/μl) occurred in 47.8% (n = 22) of patients in the DMF group. Our study reported a higher number of patients with sustained lymphopenia compared to two multicenter real data studies analyzing the safety of DMF in RRMS (47.8% versus 16.5–18.7%).15,16 However, we found a similar result for lymphopenia grade III (10.7%) compared to the RRMS clinical trials, including the extension study ENDORSE (4–9%).1,2,17 We observed frequency of lymphopenia more in alignment with a recent multicenter registry study in Spain which reported sustained lymphopenia (ALN <1000/μl) in 30% of patients. An association of lymphopenia with older age (OR 1.04, p < 0.001) and higher EDSS (OR 1.10, p = 0.035) 18 was reported in this cohort. The DMF group in our study was older (mean age 56.5 versus 37.8-38.9 years)1,2,15,16 and had on average a higher EDSS (5.3 versus 2.6-2.4)1,2 than the subjects reported in the clinical trials and real-world studies. This likely contributed to the higher frequency of lymphopenia described here. The most common side effect reported in the DMF group was flushing (43.5%) followed by gastrointestinal upset (15.2%). Flushing was similarly reported in the CONFIRM study with dimethyl fumarate (35%) and gastrointestinal discomfort was less commonly reported than in clinical trial (36%). 2 In the GA group, the most common adverse event was injection reaction. Overall, both medications were well tolerated.
Our study found no difference in the risk of confirmed EDSS progression but a trend towards reduction of confirmed T25FW worsening in patients treated with DMF. The results were also supported by a difference in confirmed EDSS improvement observed in the DMF group, albeit with a small sample resulting in wide confidence intervals. Mechanistically it is plausible that DMF could delay or prevent disability in a progressive MS cohort. The pathophysiology seen in this population involves neurodegeneration in addition to inflammation. Reactive oxygen species, 19 iron deposition, 20 and glutamate toxicity,21,22 may lead to an inflammatory micro-environment contributing to neurodegeneration. The predominance of the innate immune system,23–25 mitochondrial dysfunction, and microglial activation, also contributes to neurodegenerative pathology26,27 seen in progressive MS. Several molecular mechanisms of DMF, including antioxidant effects, suggest that it could have efficacy in progressive disease. Dimethyl fumarate has demonstrated neuroprotective effects in vitro, 28 reduction of spinal cord inflammatory infiltrates, and preservation of myelin, neurons, and axons in animal models.9,28 Dimethyl fumarate and MMF also activates Nrf2 and HCAR2 and inhibit NF-κB causing upregulation of detoxifying agents,5–8 protection against oxidative stress caused by chronic demyelination,5,9 and inhibition of microglial activation.29,30 Monomethyl fumarate crosses the blood-brain barrier, modulates immune cells infiltration, reduces excitotoxicity caused by local glutamate, 6 and potentially reduces mitochondrial dysfunction. 31 Progressive MS patients reportedly have a higher percentage of CD8+perforin+ compared to RRMS patients. 32 Similarly, an increased percentage of CD56dim NK was found in SPMS and PPMS. 33 These two studies indicate that both subsets of cells potentially have a role in MS progression, and both are affected by DMF. Longbrake et al described a reduction of CD8+ perforin+ and CD56dim NK in the peripheral blood of DMF-treated patients. 10 Additionally, in vivo studies characterizing the influences of DMF in the immune system showed modulation of immune cell composition in patients’ blood samples with an increase of regulatory immune cells and a more pronounced decrease of CD8+ when compared to CD4+,11,34 which may represent another potential mechanism for DMF efficacy in PMS.
There is limited information in the literature about the efficacy of DMF in progressive MS patients. The number of patients required to have adequate power to ascertain whether DMF might have efficacy in PMS is difficult to obtain in a uniform registry cohort. A pilot observational single-center study investigating the DMF efficacy and safety in 26 patients with PMS by assessing the EDSS increase showed no disease progression in 75% of their sample in a mean follow-up of 13.2 months. 35
A randomized phase II clinical trial with 54 participants is currently investigating the safety and efficacy of DMF in PPMS (FUMAPMS) [NCT02959658]. In our study, we reported a well-characterized PMS population observed in a real-life clinical setting, with each treatment group being followed on average over three years and a similar sample size compared to published data. We believe our findings are intriguing with some clinical outcome measures hinted at a better disease trajectory for DMF treated patients when compared to GA treated patients. However, we acknowledge a lack of sufficient power to offer a definitive determination. We did not include SPMS duration at treatment start in the analysis because 63.6% of patients in the GA group and 24.2% of patients in the DMF group were categorized as SPMS at their first visit to the Brigham MS Center. Because we were unable to validate information regarding their conversion, we opted to not adjust for this in the analysis. Other than presenting basic MRI information regarding acquisition of new lesions in follow-up imaging, we elected to forego further analysis because MRI’s were not obtained in a homogenous or scheduled way in these patient groups. If standardized MRI acquisition and timing had been employed an informative dataset would have been available. We did not adjust for multiple comparisons and though we controlled for a number of factors like age, sex, disease duration, and EDSS, patients may have been enrolled at times when the treatment landscapes differed; for example, only four patients were started on GA after DMF FDA approval in 2013. The relatively increased availability of new therapies (teriflunomide and ocrelizumab, for example) may have led to fewer progressive patients starting GA and a lower threshold to switch patients in the DMF group who were perceived to be progressing.
Glatiramer acetate was chosen as a comparator to avoid potential confounding by indication bias that might have occurred if we had instead elected to study untreated patients. We believe GA serves as a reasonable reference point because it was previously demonstrated to be ineffective in patients with PPMS in a large-scale trial (n = 943) comparing GA to placebo. 36
It is worth noting that in the DMF group, more patients were categorized as progressive relapsing which was a potential factor contributing to better treatment effectiveness results in this group. However, the DMF group was older and with longer disease duration at baseline when compared with GA. We understand that these differences likely benefitted the GA group effectiveness outcomes. Additionally, more patients in the GA group were likely to be excluded because they had received a longer acting (potent) prior medication, again biasing the GA group to be more benign on the whole. Another characteristic that potentially favored the GA group over the DMF patients was the larger number of patients who received recurrent and concomitant steroids during GA treatment, yet a sensitivity analysis excluding these patients was in line with overall findings. Lastly, a large proportion of patients in the DMF group had received prior treatment GA (32.6%). Since these patients previously discontinued GA for reasons that may have been related to diminished efficacy related to refractory disease, including these patients in the DMF group and excluding from the GA may have favored the GA group in this analysis. Given some suggestion that clinical outcomes may have been better for the DMF treated group compared to the GA treated group, further studies should be undertaken with larger cohorts to facilitate an understanding of whether DMF holds promise for the treatment of progressive MS.
Supplemental Material
sj-pdf-1-mso-10.1177_20552173211010832 - Supplemental material for Effectiveness and safety of dimethyl fumarate in progressive multiple sclerosis
Supplemental material, sj-pdf-1-mso-10.1177_20552173211010832 for Effectiveness and safety of dimethyl fumarate in progressive multiple sclerosis by Vanessa F Moreira Ferreira, Yanqing Liu, Brian C Healy and James M Stankiewicz in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Author contributions
VFMF: Conceptualization, Methodology, Investigation, Writing – Original Draft. YL: Formal analysis, Writing – Original Draft. BCH: Formal analysis, Writing – Original Draft. JMS: Conceptualization, Methodology, Supervision, Funding acquisition, Writing – Original Draft, Review & Editing.
Data statement
The data that support the findings of this study are available from the corresponding author, JMS, upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VFMF reports no disclosures. YL reports no disclosures. BCH has received research support from Analysis Group, Verily Life Sciences, Novartis, Merck Serono, and Genzyme. JMS is currently affiliated with Novartis and he has received consulting fees from Roche-Genentech, Biogen, Genzyme, Novartis, EMD Serono, and Celgene in the past two years.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an Investigator-Initiated Trial grant from Biogen. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Standard protocol approvals,registrations,and patient consents
Brigham and Women’s Hospital institutional review board approved this research- related secondary use of data. The IRB determined that this project meets the criteria for exemption which informed consent is not required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
