Abstract
Background
People living with multiple sclerosis (MS) experience a high symptom burden that interferes with daily functioning. Virtual reality (VR) is an emerging technology with a range of potential therapeutic applications that may include ameliorating the experience of some common MS symptoms.
Objective
We tested the feasibility and tolerability of a VR intervention and its preliminary effects on affect.
Methods
Participants with MS were recruited to complete a pilot study of eight sessions of VR over four weeks.
Results
A total of n = 16 participants with MS completed the study (age range: 28–63). Feasibility goals were met with 100% of the sample completing at least n = 4/8 of their intervention sessions, with a total of 119/128 (93%) completed sessions. Two of the n = 16 participants experienced brief adverse events (balance, vertigo) but these resolved with headset removal and were not otherwise treatment limiting. There was a preliminary indication of overall improved affect from baseline to intervention end, with significantly improved positive affect (t(15) = −3.19, p = 0.006) and decreased negative affect (t(15) = 2.25, p = 0.040).
Conclusion
VR interventions are feasible, safe, and tolerable for individuals living with MS and may improve affect.
Introduction
Multiple sclerosis (MS) is a chronic and progressive disorder of the central nervous system that is associated with a chronic symptom burden and results in overall reduction in quality of life.1–5 A high symptom burden is associated with depressed mood and increased irritability, which can serve to exacerbate the experience of common chronic symptoms such as pain and fatigue.6–8 As a result, affect has been identified as a moderating factor in the experience of symptoms and as a target for clinical intervention. 9
Virtual reality (VR) is an emerging technology with potential therapeutic10–13 and rehabilitative 14 applications. One established use of VR has been the reduction of pain, 15 for instance to decrease pain in burn patients. 16 These studies have indicated that psychological immersion in the VR environment can reduce the subjective awareness and experience of the physical symptom. Further, this effect has been associated with the degree of VR immersion that the subject experiences, 17 and is associated with improvements in emotional affect. 18 For example, VR immersion moderates self-reported pain in healthy controls based on the valence of affective state, with pain increasing with negative affect and decreasing with positive affect. 17
In MS samples to date, preliminary studies have demonstrated efficacy for VR as an adjuvant for physical rehabilitation (e.g., improving balance and endurance)19,20 and for assessment of cognitive and mood symptoms,21,22 but no study has yet measured the ability of VR to change affect directly.
In this pilot study, we developed and tested the feasibility and tolerability of repeated VR sessions in participants with MS. Additionally, as a secondary outcome, we tested change in positive and negative affect following VR sessions.
Materials and methods
All study procedures were approved by the NYU Langone Health Institutional Review Board. MS participants were recruited with flyers from the NYU Langone Comprehensive MS Care Center. All participants provided written informed consent.
Participants: This study was designed to have broad eligibility criteria to determine feasibility of the VR program across different ages and levels of neurologic disability. All participants had confirmed diagnoses of MS, 23 any subtype, and were at least one month from any acute relapse or steroid use. Over the course of protocol development, the initial participants (n = 10) were not required to meet any threshold for symptom severity at enrollment. We then recruited participants (n = 8) within the context of an ongoing study that is evaluating VR for chronic pain relief and included the eligibility requirement of having an interference score of at least 3 on the Brief Pain Inventory (BPI). 24
Eligibility criteria required participants to be 18 years of age or older, and have sufficient cognitive capacity to participate as measured by the Wide Range Achievement Test 4th Edition (WRAT-4) 25 reading recognition standard score ≥ 85 and a Symbol Digit Modalities Test (SDMT; verbal trial) z-score ≥ −3.0 26 (raw scores were converted to z-scores with the use of age-matched healthy control normative group scores 27 ). This SDMT z-score cutoff was selected to exclude those with severe cognitive impairment whom may have had limited ability to understand or participate in study procedures. 27
Participants were excluded if they had a history of a psychotic disorder, an uncontrolled seizure disorder, or concurrent diagnosis of vertigo, or visual, auditory, or motor deficits that would prevent the ability to operate the VR equipment.

Interactive condition content: The Karuna Virtual Embodiment Training provided virtual environments for participants to interact with and move throughout. (a) A participant is playing a modified form of Soccer in the virtual environment for enjoyment and physical exercise. (b) A participant is interacting with a virtual environment that is designed to enhance their sense of immersion. (c) A participant is completing a “target practice” exercise that requires them to physically move and stretch within their range of motion.
The second content type, referred to as “passive,” was viewing only and did not require navigation. The passive content was drawn from a published index of 360° virtual environment videos (see Li et al. 31 ) and consisted of virtual environments of both exciting experiences (e.g., such as a ride on a helicopter) and more neutral or relaxing experiences (e.g., tours through nature settings). The passive content was viewed through the Steam 360° video player application.
All sessions concluded with five minutes of a 360° guided meditation video. The meditation videos are freely available online and emphasized muscle relaxation, controlled breathing, and natural calming stimuli (e.g., ocean waves).32–34 This conclusion served as a transition from the end of the VR experience to removal of the headset.
Based on early work that indicates degree of immersion may moderate overall response,12,17,18 we hypothesized that the interactive content may offer a higher degree of VR immersion and treatment response than the passive content. However, this study was underpowered to test this secondary hypothesis and the two content conditions were combined for one group analysis.
All VR sessions were completed with the participant seated for safety and to accommodate those with a range of ability levels including wheelchair dependence. Interactive condition sessions included the capability to adapt to participants’ physical ability levels (i.e., virtual objects that participants interacted with increased or decreased in velocity based on performance of the participant).
The protocol includes “stop criteria” to ensure safety and tolerability at each step. If a participant reported an adverse event (AE, e.g., nausea) during the VR session, they were given the option to pause and remove their headset. When paused, the participant was asked to rate their discomfort on a 1–10 Likert scale, with any rating of 7 or higher requiring that the session be discontinued. Otherwise, the participant was provided with up to a 5-minute rest and the option to resume when ready. If a participant re-experienced any AE, the headset was removed, and the VR session was discontinued. In addition, to ensure compliance, any participant who cancelled more than two scheduled VR sessions was discontinued from the study.
For an indicator of potential therapeutic benefit and as a secondary outcome, we recorded affect before and after each VR session using the Positive and Negative Affect Schedule–Now (PANAS-Now). 35 The PANAS-Now instructs participants to rate 20 mood-related adjectives according to their momentary experience along a scale from 1 (not at all) to 5 (extremely). The PANAS-Now has separate scores for PA and NA, each ranging from 10 to 50. Higher PA and lower NA scores indicate improvement.
All data were analyzed using SPSS version 25.
Results
Demographics and clinical characteristics.

BPI: Brief Pain Inventory; MS: multiple sclerosis; SDMT: Symbol Digit Modalities Test; WRAT: Wide Range Achievement Test.
Participant characteristics, baseline affect, and attendance.

BPI: Brief Pain Inventory.
Two of the sixteen participants experienced mild adverse events (AEs): one participant experienced four instances of a sense of loss of balance (but was seated during participation) and the second participant experienced three incidents of vertigo. All AEs resolved within seconds and participants resumed the session per protocol without any session discontinuation.
On survey, participants reported high levels of satisfaction with the VR experience and all participants (100%) reported that they would use VR again.
Secondary Outcome – Change in Affect
Affect response to VR sessions was tested by comparing the average pre vs. post session scores across the eight sessions. On average, PA significantly increased by 1.7 points, t(15) = −3.19, p = 0.006, and NA significantly decreased by 1.3 points, t(15) = 2.25, p = 0.040, indicating that the VR sessions resulted in immediate improvement in affect.
Table 3 presents the average daily change in momentary PA and NA for each intervention session, as well as the average change for each content type (interactive, passive, and pooled).
Mean change in pre-/post-session PA and NA scores.
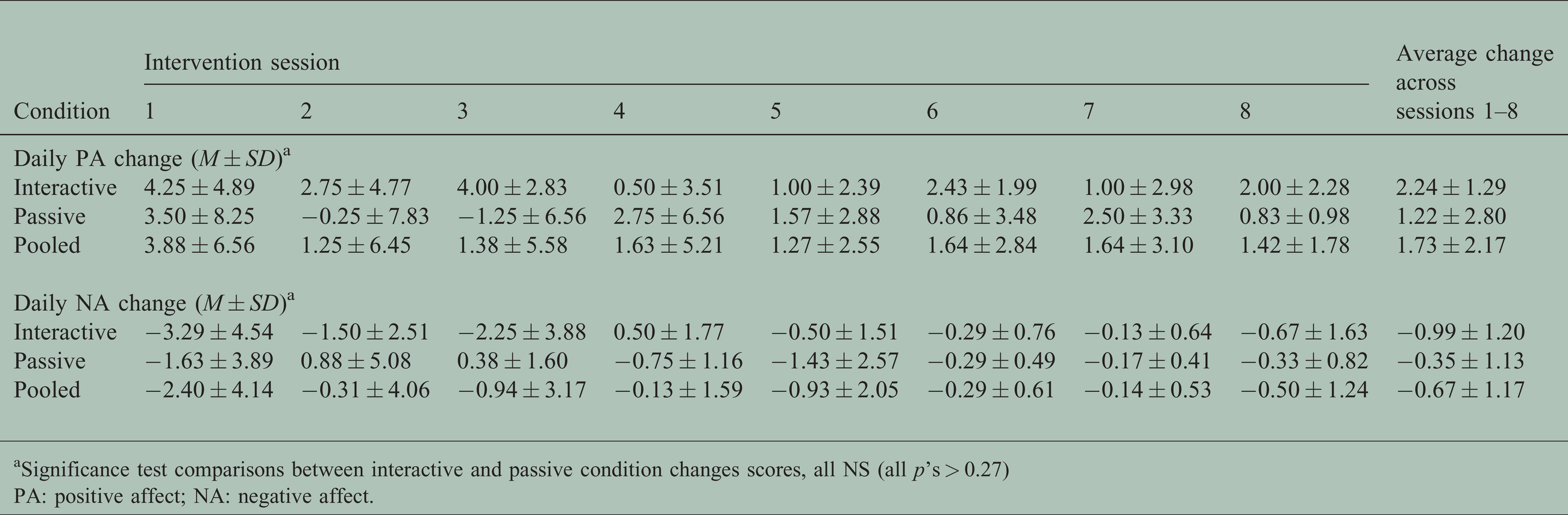
aSignificance test comparisons between interactive and passive condition changes scores, all NS (all p’s > 0.27)
PA: positive affect; NA: negative affect.
Next, as a potential indicator of affect response to the intervention, change from baseline to treatment end was assessed by comparing the average pre/post session scores of session one and session eight for the n = 12 participants that completed all eight treatment sessions. There was a significant decrease in NA (−2.1 point mean difference, t(11) = 2.50, p = 0.029) and a trend towards increased PA (3.1 point mean difference, t(11) = −2.05, p = 0.065) from baseline to treatment end. As shown in Table 3, the average pre/post VR session change scores for PA and NA did not significantly differ between VR content conditions (all p’s > 0.27).
Discussion
This study demonstrates the potential feasibility and tolerability of this VR protocol for individuals with MS and signals the potential for affective regulation. This study confirms that VR is tolerable for individuals with MS, with only two participants experiencing mild and transient AEs during the course of the VR sessions. Further, our study protocol includes designated stop criteria that may be useful for future investigations of repeated VR sessions targeting symptom management. Encouragingly, all of our participants reported that they would like to use VR again.
Both types of VR session content (interactive and passive) were feasible and acceptable to our participants. In general, a theory of VR use for pain relief contends that both the emotional valence and the psychological immersiveness of virtual content influence symptom relief. 18 We hypothesize that immersion may be a key feature for symptom relief, as it has previously been shown to moderate emotional affect response. While the literature to date broadly supports VR as an immersive experience independent of content, the role of specific types of content remains an ongoing question, and the difference in immersion between interactive and passive content has yet to be determined. Here, we did not find any signal of difference between the content conditions and opted to combine our intervention groups for analyses in efforts to report on the overall effects of VR. We believe an important future direction is to measure VR immersion and determine the role of specific content types for optimal therapeutic use. While both types of content were feasible in this study, a consideration for future directions should include that those with more advanced disability may have a preference for passive viewing due to the lower physical requirements.
While we did not select for those with lower PA and/or higher NA at baseline, we found that, on average, momentary PA and NA both improved following the daily VR sessions. Further, there was indication that both PA (trend) and NA (significant) may improve with repeated VR sessions as an intervention. However, due to the use of momentary assessments with the PANAS-Now, interpretation is limited and individual fluctuations at each session’s assessments may have contributed to statistical variance in our results. Nonetheless, this finding contributes to accumulating evidence that VR can influence cognitive-affective processes in MS.21,22
Future work should include selecting those with baseline affective disturbance to determine the overall influence of repeated VR sessions on affect over time, with the further goal of linking change in affect to symptom experience. Beyond symptomatic management, emotional affect influences coping strategies and overall quality of life36,37 and interventions that assist patients in managing the emotional stressors of chronic disease may have far-reaching impact. An appealing non-drug intervention with the potential to improve emotional affect, such as VR, warrants further clinical research.
As the home use of VR is rapidly becoming available and widespread in use, VR may become an important tool for telerehabilitation applications as well. However, it would be especially important to establish safety protocols for home use for people living with MS. For instance, participants may be encouraged to remain seated for the duration of the VR session, with content that does not encourage interactive movements that may affect balance. In addition, ongoing remote supervision, for instance via live teleconference, would be recommended to monitor patients for safe and tolerable use in their home settings.
Limitations of this study include the small sample size and exploratory pilot design. The study was overall underpowered to adequately address the relevance of VR content (i.e., interactive condition vs. passive condition). We are not able to address whether disease status or symptom severity, even while including the subset we recruited with pain symptoms, influences VR response. Future studies should include more specific designs and outcomes to evaluate the potential therapeutic effects of VR for specific symptoms (e.g., pain, fatigue) and rehabilitation applications.
Conclusion
Our findings support the growing evidence of the feasibility, safety, and potential for benefit of VR use for individuals with MS.
Conflict of Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
sj-pdf-1-mso-10.1177_20552173211006139 - Supplemental material for Virtual reality is a feasible intervention platform in multiple sclerosis: A pilot protocol and acute improvements in affect
Supplemental material, sj-pdf-1-mso-10.1177_20552173211006139 for Virtual reality is a feasible intervention platform in multiple sclerosis: A pilot protocol and acute improvements in affect by Michael T. Shaw Maria J.Palmeri MartinMalik Dobbs Bryan Leigh E. Charvet in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgements
We would like to acknowledge Sebastien Scioscia, Jon Links, Amy Ro, Allan George, and Danielle Ladensack for their contributions and assistance with distribution of intervention, monitoring of safety, and data entry efforts.
Funding
The Lourie Foundation, Inc., National MS Society PP-1808-32457.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
