Abstract
Background
The decision to have children can be complex, particularly for people with multiple sclerosis (MS). A key concern is the use of disease modifying drugs (DMDs) during pregnancy, and how continuing, stopping or switching them may affect the mother and child. In people with active MS, stopping medications puts the mother at risk of relapse and disease rebound.
Objectives
Review evidence on the effect of different switching strategies in people with stable relapsing remitting MS (RRMS).
Methods
We searched MEDLINE, EMBASE, EMCARE, CINAHL, SCOPUS, Cochrane Library up to March 2020. Only papers in English were included and no other limits were applied. Seven articles were included: four cohorts, two case reports and one randomized controlled trial (RCT).
Results
Two strategies were found: de-escalating, which was associated with an increased risk of relapses, and switching between first line injectables, with no change in relapse rate observed.
Conclusion
Evidence on the effect of switching strategy on disease course in stable RRMS patients planning for pregnancy is scarce, but when switching, current evidence suggests the risk of relapses mirrors known medication efficacy.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory demyelinating neurodegenerative disease estimated to affect 2.3 million people world-wide.1,2 It is more common in women than men,2,3 and most new presentations are in women of childbearing age.4–6 There is evidence of reduction in disease activity during pregnancy, but an increased chance of relapse in the first three months post-partum possibly offset by protective effect of breastfeeding (it remains uncertain if there is an effect, with some studies showing one,7–9 and others not 10 ). While pregnancy and breastfeeding both reduce the risk of relapses,8–10 they do not appear to do so to the same degree as some disease modifying drugs (DMDs). In women with more active MS who have achieved clinical stability on higher efficacy DMD there is concern that despite the protective effects of pregnancy there remains a significant risk of relapses. 11 Concerns about potential foetal effects of DMDs may lead some women to stop or delay treatment until their families are complete. 12 Emerging safety data now means women with MS planning a pregnancy are increasingly encouraged to continue some DMD treatments: Both glatiramer acetate (GA) and interferons (IFN) are increasingly continued during pregnancy and breast feeding, after the removal of their pregnancy contraindication by the EMA13–17. Natalizumab (NTZ) is considered by many to be safe for use in pregnancy, with the last dose by 34/40 week of pregnancy. 13 Other medications are either known to be teratogenic (such as Fingolimod, Cladribine, Teriflunomide) or there is limited experience in pregnancy (for example Alemtuzumab and Dimethyl fumarate). 13 Women may need to defer starting them (or in the case of Cladribine and Alemtuzumab defer pregnancy for several months after either of the treatment cycles), switch to a treatment considered safe, or stop them during conception, pregnancy and breast feeding. When stopping fingolimod or NTZ it has been suggested that switching, rather entirely stopping treatment, may be a preferable option to avoid return of disease activity and rebound.14–23
Recently a consensus paper on pregnancy planning in the UK has been published that guides with medication management when planning pregnancy and breastfeeding compatibility, 13 based on expert consensus opinion due to paucity of evidence in this area. It pragmatically did not seek to systematically review the literature on the effect switches in treatment, resulting from pregnancy planning or otherwise, have on relapse rate. Evidence on the safety of switching, and particularly de-escalating from higher potency to platform injectables in pregnancy, remains scarce. The effect of switching to another DMD type when the patient is already stable on the current one is unclear. Available studies in this area are controversial and variable. Multiple studies compare different pairs of medications switches. Some included patients who switched therapy due to treatment failure; and thus disease activity may influence the results of the switch, and make escalation therapy more appropriate and less fit for pregnancy planning. 24 Others included both stable and unstable patients 25 or included different MS types in their study when switching therapy 26 and have conflicting results even though the treatment switch was the same.
Our aim was to review the evidence for the effect on disease activity of switching from different DMDs to interferons and GA in people with stable RRMS.
Methods
This systematic review was planned according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses Guidelines (PRISMA) and registered in International Prospective Register of Systematic Reviews (PROSPERO), registration number: CRD42020172912.
Search strategy
A comprehensive search was run using the following databases: Medline, Emcare, Embase, CINAHL, SCOPUS, Cochrane Library. English language articles only are included without any other limits applied. Manual reference searching of the eligible articles was done. The full search strategy is shown in online Appendix-1.
Inclusion and exclusion criteria
Inclusion criteria
Any type of study was eligible for inclusion if participants fulfilled the following criteria:
Adults with confirmed relapsing remitting multiple sclerosis diagnosis according to McDonald criteria.
27
Using any type of DMDs. No evidence of on treatment relapses leading to a decision to change DMD. Switching to one of the pregnancy safe DMD options (IFN or GA). Following up patients for at least six months after switching DMD.
Exclusion criteria
We excluded reviews, editorials, book chapters, commentaries, replies and conference abstracts for which we could not find full texts.
Studies looking at progressive disease, clinically isolated syndrome or that did not specify type of MS.
Studies in patients who only switched DMDs due to unclear reason or treatment failure; where those switching due to treatment failure could not be separated from those switching for other reasons (switches made for reasons other than treatment failure, such as side effects, safety concerns or patient preference, were considered for inclusion).
Data management and studies screening
Referencing was managed using Endnote X9 software. Screening Process was carried out using Rayyan, (internet-based platform). Initial article title and abstract screening, then full article screening was done by LA and NA. NA was a second reviewer for 10% of the studies in both stages. Reviewers included eligible articles independently and blindly and conflicts were resolved through a discussion meeting.
Data extraction
The following data were extracted: country, setting, study design and methods, sample size, baseline characteristics, duration of follow-up, previous DMD regimen, new DMD regimen, primary outcome (ARR), secondary outcomes (EDSS Score, new MRI lesions) and main reasons for switching.
Quality assessment
We used the revised Cochrane risk of bias tool for randomized trials, a risk of bias assessment tool for randomized controlled trials (ROB2), 28 and National Heart, Lung and Blood Institute (NIH) quality assessment tool for before-after (pre-post) with no control group cohort studies. 29 For more detailed information about the quality assessment (see online Appendix-2).
Data synthesis
Data were narratively synthesized to address the results found in the eligible articles. We used tables to present data from the articles and outcomes. A multi-colour figure was used to represent the quality of each paper in the included cohorts (see online Appendix-2). Meta-analysis was not possible due to high heterogeneity between studies.
Results
Search results
The search was run on 31 March 2020 and identified 2298 articles. After duplicate removal and title-abstract screening, 1210 records were eligible for full text screening. From this we excluded 1109; an additional two articles were identified during manual reference searching of eligible papers; we finally included seven articles that matched the inclusion criteria (Figure 1).

PRISMA chart.
The included articles are four pre-post cohort studies with no controls, two case reports and one RCT (Tables 1 to 3).
Characteristics of included observational studies.
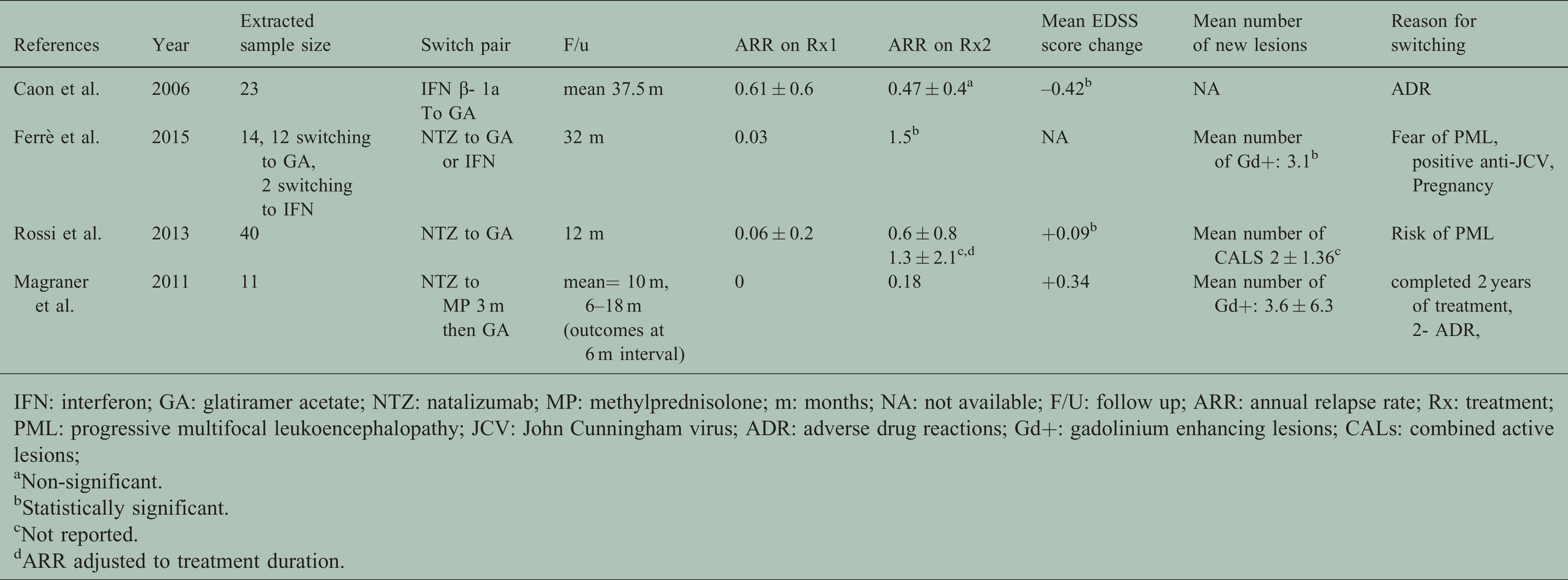
IFN: interferon; GA: glatiramer acetate; NTZ: natalizumab; MP: methylprednisolone; m: months; NA: not available; F/U: follow up; ARR: annual relapse rate; Rx: treatment; PML: progressive multifocal leukoencephalopathy; JCV: John Cunningham virus; ADR: adverse drug reactions; Gd+: gadolinium enhancing lesions; CALs: combined active lesions;
aNon-significant.
bStatistically significant.
cNot reported.
dARR adjusted to treatment duration.
Characteristics of included RCT.

IFN: interferon; GA: glatiramer acetate; NAT: natalizumab; IV: intravenous; m: months; NA: not available; F/U: follow up; ARR: annual relapse rate; Rx: treatment; PML: progressive multifocal leukoencephalopathy; nT2L: new T2 lesion; Gd + L: gadolinium enhancing lesions.
aNot provided.
bStatistically significant.
cNon-significant.
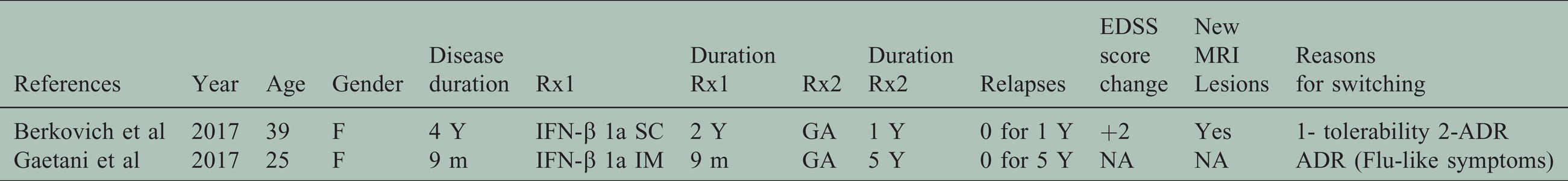
Characteristics of case reports.
Switching strategies
Switching from natalizumab to glatiramer acetate (De-escalation)
Two observational studies examined the effect of switching from NTZ to GA on disease activity. Ferrè et al. tested the effect on clinical disease activity using the Annualized Relapse Rate (ARR) measure, and the radiological activity using the mean number of new enhancing gadolinium lesions (Gd+) in MRI before and after switching. 30 Both ARR and Gd+ increased after switching to GA, reaching statistical significance (at 0.05). However, this study is judged as poor quality due to missing information (it was published as a brief communication). 30 The other study by Rossi et al. included 40 participants and measured disease activity using ARR, time adjusted ARR, Combined Active Lesions (CALs) in MRI and EDSS score change. ARR while on NTZ was (0.06 ± 0.2) and rose to (0.6 ± 0.8) and (1.3 ± 2.1) with unadjusted and treatment duration adjusted ARR, respectively. New CALs were 2 ± 1.36 at month 12 MRI assessment, without evidence of rebound. Mean EDSS score change from baseline to the end of the follow up was very minute and statistically significant. Both differences in ARR and mean CALs while on NTZ and after switching to GA increased but significance was not calculated, instead, they reported the decreased clinical and radiological activities after switching to GA compared to pre-NTZ period, and both were statistically significant. This study showed the safety and tolerability of GA and its ability to prevent disease rebound without maintaining the same efficacy of NTZ. 31
Switching from natalizumab to glatiramer acetate after “Bridging”
Switching from NTZ to GA after “bridging” with methylprednisolone for three months was tested for the effect on the disease activity as a protocol that aimed to prevent disease rebound during NTZ interruption periods by Magraner et al. Data were provided for each participant which allowed us to exclude those switching due to treatment failure. We included 11 patients who switched treatment due to risk of PML (used NTZ for two years already) but excluded the other 7 patients who experienced relapses while using NTZ (not stable). ARR changed from 0 to 0.18, while mean change in EDSS score was 0.34, and mean number of new Gd+ is 3.6 ± 6.3. However, on assessment a substantial risk of study bias was identified. All changes in measures were assessed from the point of stopping NTZ and six months after stopping it, 32 and this is very likely to be an insufficient period of time to assess the effect of a change as exposure treatment with GA alone was effectively only for three months (methyl prednisolone was given for the first three months) and the biological effects of NTZ last for approximately 12 weeks, while the changes it made on the cerebrospinal fluid distribution of immune cells may persist for up to six months from discontinuation.33–35
Switching from natalizumab to IFN-ß 1b (De-escalation)
De-escalation from NTZ to IFN-ß 1 b effect on disease activity was studied by Gobbi et al. in a one-year randomized rated blinded controlled pilot trial. We used the number of relapses reported to calculate ARR in switchers and it was higher than non-switchers (0.4 and 0 respectively). EDSS score increase ranged from 0.5–1.5 with a median of 0.5 in the switchers, while no progression was recorded in the NTZ group. Radiologically, no new lesions were reported in the control NTZ group in all MRI scans. The number of new lesions (T2 and/or Gd+) in the IFN group ranged from 0–12, but the only significant difference between groups was the number of new T2 lesions reported at month 6. The study concluded that IFN could not maintain the efficacy of NTZ but exerts some anti-inflammatory effects that made all three reported relapses mild and non-disabling. 35 Studies on a larger scale are needed to confirm these findings as this RCT was limited with its small sample size and had some quality concerns due to the fact only assessors were blinded.
Switching from IFN to GA
Changing between first line injectables - specifically from IFN to GA - was assessed in one pre-post cohort, a case series report and a case report that fulfilled our inclusion criteria. Caon et al. found that ARR was reduced by 23% after switching from IFN to GA – switched due to toxicity-, and this reduction did not reach statistical significance. 36 A case report of a 25-year-old woman with relapsing remitting MS who was initially using IFN-ß-1a for nine months and switched to GA due to the flu like side effect, showed clinical stability on GA for five years while radiological activity was not reported. 37 Lastly, a 39-year-old woman with relapsing remitting disease for four years, switched from IFN-ß-1a to GA due to side effects. One-year outcomes showed no relapses but confirmed new MRI lesions and a + 2 increase in EDSS score 38 (Figure 2).

Switching strategies.
Discussion
In this review, we found two main DMD switching strategies in people who were stable on treatment, switching from a higher potency drug to GA or IFNs and switching between interferons and GA. Our findings confirm that risk of relapse when switching mirrors, the medication’s known efficacy (NTZ provides higher protection from relapses than the injectable therapies). Nothing in our findings leads us to question the conclusions of the UK consensus on pregnancy in multiple sclerosis. 13 However, our most striking finding is how little high-quality information there is about the relapse risk associated with changing treatment, and that none of the studies identified specifically considered this in the context of pregnancy (and the reduction in relapse risk associated with pregnancy and breast feeding).
When balancing risks, it is worth recalling that people on higher potency treatments will tend to have had more active disease before starting these medications. Alroughani et al., who followed up 99 pregnancies, measured ARR before and during pregnancy, 21 of the participants were using fingolimod, 24 used NTZ, 42 used IFN, 2 teriflunomide users,1 DMF and 9 were taking no treatment. Seventeen relapses occurred during pregnancy, 70% of them happened in patients who used either NTZ or FTY before conception. 11 This reminds us that for patients with previously more active disease, controlled with more potent agents, switching (and in the case of NTZ now continuing), rather than stopping, may be preferable. In addition, we now have available two treatments that can induce a sustained response after two courses of treatment (cladribine and alemtuzumab), and while they cannot be used during pregnancy due teratogenic risk, conception is advised to be attempted from six (cladribine) or four (alemtuzumab) months after a cycle of treatment, and this may be preferable to using a lower potency agent through pregnancy.
GA and IFNs are considered of a comparable efficacy39–41 and while evidence of the effects of switching between them appears consistent with this, this is of less interest than it was previously as both treatments can now be continued during pregnancy.42–48
We identified only one high potency treatment (NTZ) being stepped down to GA or IFN, yet there are now many more with higher potency (than GA or IFN) commonly used in clinical practice, and people with MS now also have a choice between treatments that can induce and sustain remission after two courses (alemtuzumab and cladribine) and others that maintain remission when taken regularly such as (interferons, fingolimod and natalizumab).
While the recent UK consensus guidance paper on pregnancy in MS adopts a pragmatic approach, supported by the limited data we have identified, the optimal approach to DMD management in women planning pregnancy has yet to be established. To help women take informed shared decisions about pregnancy and DMD management, we need a more robust evidence base that includes managing the full range of commonly used treatments, and in particular we also lack direct evidence of DMD efficacy during pregnancy, compared with pregnancy alone.38,44,45,49,50
We included studies where a switch in DMD was prompted by side-effects, but only where patients were not also having relapses. While this is not the situation in pregnancy planning, treatment side effects per se are unlikely to substantially bias comparison of the risk of relapse before and after switching, except where this affects treatment compliance. However, if we had excluded such studies, we would have been left with one out of the seven we did identify, further highlighting the paucity of data directly applicable to pregnancy planning.
In addition to noting how few studies there are on treatment switching, it should also be noted that those we did identify were small, meaning that we should be cautious about generalising the results. Further, a formal meta-analysis was not possible due to limited treatment overlap between the studies, and only one study was an RCT.
In conclusion, the results of the few studies that there have been on switching treatments in clinically stable relapsing-remitting patients appear to be predictable based on a medication’s known efficacy and highlight the risk of de-escalation.
Supplemental Material
sj-pdf-2-mso-10.1177_20552173211001571 - Supplemental material for Switching treatments in clinically stable relapsing remitting multiple sclerosis patients planning for pregnancy
Supplemental material, sj-pdf-2-mso-10.1177_20552173211001571 for Switching treatments in clinically stable relapsing remitting multiple sclerosis patients planning for pregnancy by Lubna Almouzain, Fiona Stevenson, Declan Chard, Nur Abdul Rahman and Fiona Hamilton in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Within the last three years (to November 2020), Declan Chard has been a consultant for Biogen and Hoffmann-La Roche. He has received research funding from the International Progressive MS Alliance, the MS Society, and the National Institute for Health Research (NIHR) University College London Hospitals (UCLH) Biomedical Research Centre.
This review was published as conference abstract in ACTRIMS/ECTRIMS MS 8th Joint Meeting 2020.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review is funded by King Saud University, Riyadh, Saudi Arabia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
