Abstract
Background
Progressive solitary sclerosis is a unifocal demyelinating disease recently proposed as a possible multiple sclerosis variant.
Objective
To compare myelin content and brain metabolite ratio qualitatively in the normal-appearing white matter of progressive solitary sclerosis cases compared to multiple sclerosis and healthy control participants.
Methods
Case report.
Results
Progressive solitary sclerosis cases showed abnormal myelin in normal-appearing white matter tracts and global normal-appearing white matter as well as lower N-acetyl-aspartate to total creatine ratio compared to multiple sclerosis and healthy control groups.
Conclusion
Despite a single demyelinating lesion along the corticospinal tract in progressive solitary sclerosis, we showed evidence of more extensive abnormality within the normal-appearing white matter.
Keywords
Introduction
Progressive solitary sclerosis (PSS) is a unifocal demyelinating disease involving the corticospinal tract with downstream progressive weakness.1–3 This entity was recently proposed as a possible multiple sclerosis (MS) variant. 1 The current study gathers hypothesis-generating data from two PSS cases to evaluate whether pathology is restricted to a single chronically demyelinated lesion or if diffuse brain pathology beyond the focal lesion is evident. 4
Multicomponent driven equilibrium single pulse observation of T1 and T2 (mcDESPOT) is a rapid, whole-brain imaging method that can determine the myelin water fraction (MWF), a proposed marker of myelin measured by the ratio of magnetic resonance signals from the water between the myelin bilayers to the total water content. 5 Proton magnetic resonance spectroscopy (1H-MRS) can be used to quantify brain metabolite ratios, such as N-acetyl-aspartate (NAA) to total creatine (tCr) ratio (associated with axonal and neuronal integrity). 6 We compared PSS to relapsing–remitting multiple sclerosis (RRMS), primary progressive multiple sclerosis (PPMS) and healthy control (HC) participants by evaluating MWF and NAA/tCr.
Materials and methods
Cross-sectional mcDESPOT (voxel size 1.7 × 1.7 × 1.7 mm3 acquired using a series of spoiled gradient recalled echo (TR/TE = 6.5/3.6 ms, α = [2, 3, 4, 6, 9, 13, 18]°) and balanced steady state free precession (TR/TE = 5.8/2.9 ms, α = [7, 11, 15, 19, 24, 30, 47]°) acquisitions across a range of flip angles (α)) 5 and single-voxel point resolve spectroscopy 1H-MRS (TE/TR = 36/4000 ms, 6.5 × 4.5 × 1.8 cm3 primarily normal-appearing white matter (NAWM) voxel above the lateral ventricles) 7 data were acquired for two PSS cases who met the proposed inclusion criteria for the diagnosis as described by Keegan et al. 1 PSS data were compared qualitatively to historical data collected for RRMS, PPMS and HC participants (Table 1). All magnetic resonance imaging (MRI) was performed on a Philips Achieva 3 T scanner using the same protocol. Furthermore, a clinical history of PSS was obtained by retrospective chart review. All participants provided written informed consent prior to participation. The protocol was approved by the clinical research ethics board at the University of British Columbia.
Clinical and demographic information of the participants at the time of magnetic resonance imaging scan.
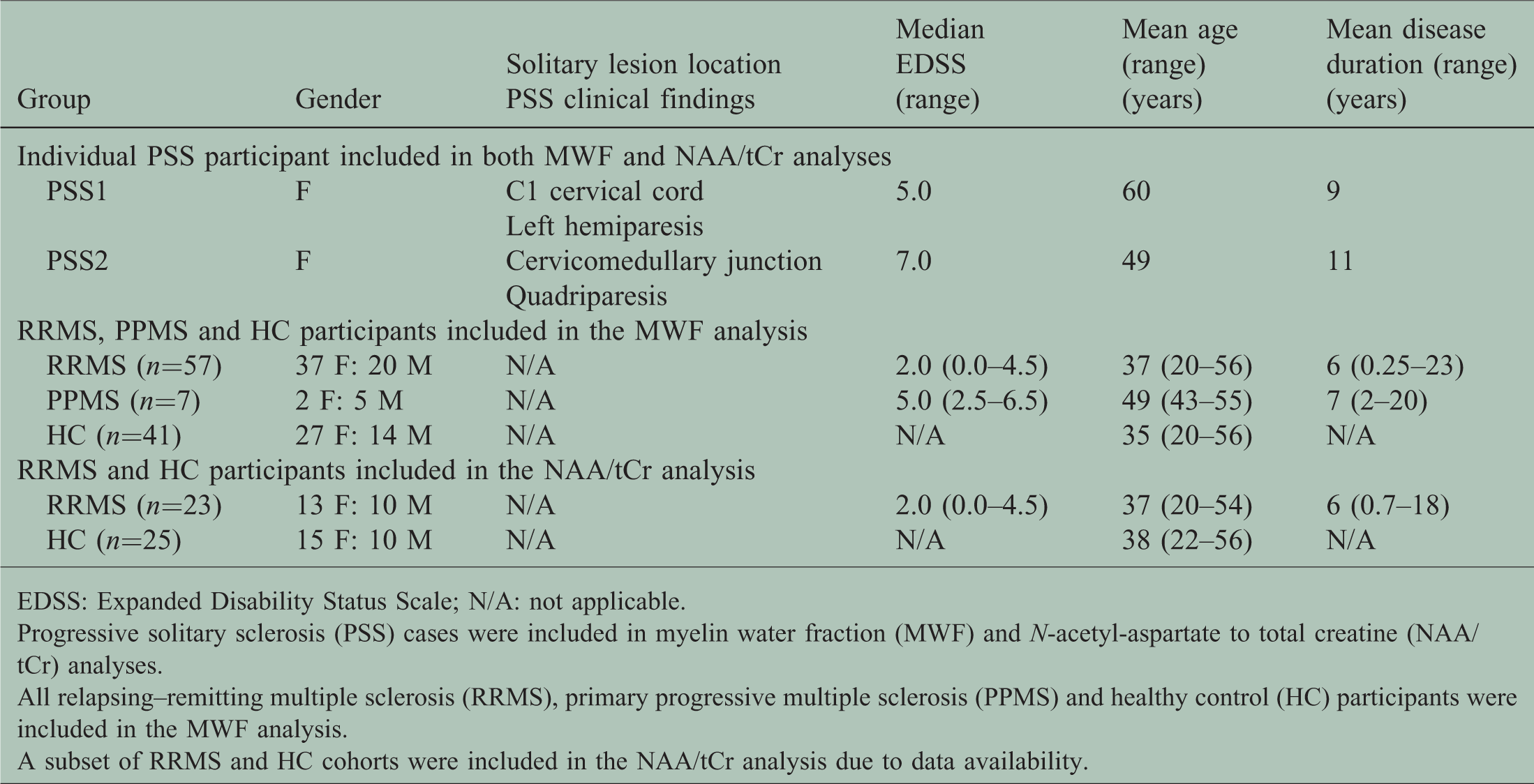
EDSS: Expanded Disability Status Scale; N/A: not applicable.
Progressive solitary sclerosis (PSS) cases were included in myelin water fraction (MWF) and N-acetyl-aspartate to total creatine (NAA/tCr) analyses.
All relapsing–remitting multiple sclerosis (RRMS), primary progressive multiple sclerosis (PPMS) and healthy control (HC) participants were included in the MWF analysis.
A subset of RRMS and HC cohorts were included in the NAA/tCr analysis due to data availability.
A whole-brain voxelwise analysis of Z-scores was computed comparing the MWF of individual PSS cases to the HC (n = 41) group distribution. The voxels with a Z-score of –2 or less were qualitatively presented as abnormal to indicate significantly lower MWF (P ≤ 0.05). Three NAWM tracts, including left and right corticospinal tracts in addition to the corpus callosum, were obtained from standard atlases and transformed to mcDESPOT space.8,9 The mean MWF was calculated within each region of interest and compared between individual PSS cases and RRMS, PPMS and HC groups. 1H-MRS spectra were fit using the LCModel version 6.3 to calculate NAA/tCr within each region of interest.
Results
The clinical and demographic information of all participants is found in Table 1. PSS1 presented at age 51 years with progressive left hemiparesis evolving over 8 years and a solitary lesion in the C1 cervical cord (Figure 1). PSS2 presented at age 38 years with rapidly progressive quadriparesis resulting in severe disability requiring a wheelchair over 1 year and a solitary lesion in the cervicomedullary junction (Figure 1). Neither of the PSS cases had further development of new neurological deficits or new demyelinating lesions on MRI. Visual evoked potentials were available for PSS1 and showed a normal response. Cerebrospinal fluid analysis was available for PSS2 and did not show oligoclonal bands or elevated IgG index. The clinical investigations did not reveal alternative diagnoses.

Global and regional myelin abnormality in normal-appearing white matter of progressive solitary sclerosis (PSS). (a) Example slices through the myelin water fraction (MWF) Z-score maps of two PSS cases show areas marked in blue, which correspond to regions with significantly lower MWF (P ≤ 0.05), where Z is –2 or less, when compared to an MWF atlas constructed from 41 healthy control (HC) participants. The Z-score map of the oldest HC (56 years) is illustrated to show that the abnormalities seen in PSS cases are likely to be due to the disease rather than age-dependent myelin changes. The Z-score maps were overlaid on a standard MNI-152 2 mm template displayed axially (left) and sagitally (middle). A solitary lesion in each PSS case is marked by a red arrow on conventional images (right). (b) MWF (a marker for myelin) in left and right corticospinal tracts and corpus callosum was compared between an individual PSS case and relapsing–remitting multiple sclerosis (RRMS), primary progressive multiple sclerosis and HC cohorts. N-acetyl-aspartate to total creatine (NAA/tCr; associated with axonal and neuronal integrity) in the voxel above the ventricles was compared between an individual PSS case and HC and RRMS cohorts.
The Z-score map of individual PSS cases showed global diffuse reduction of myelin in the brain indicated by the number of voxels that were –2 or fewer standard deviations away from the mean MWF constructed from 41 HC participants (P ≤ 0.05) (Figure 1(a)). Both PSS cases had lower mean MWF in all NAWM tracts than the average across each of the RRMS, PPMS and HC groups, as well as lower NAA/tCr than the median NAA/tCr of the RRMS and HC groups (Figure 1(b)).
Discussion
The MWF and NAA/tCr data show abnormal myelin and metabolic dysfunction in the non-lesional tissue of two PSS cases. This suggests that PSS pathology may be similar to that seen in MS, demonstrating diffuse NAWM abnormality rather than damage restricted to a specific lesion in the central nervous system. PSS cases appear to have greater diffuse myelin damage in NAWM as measured by MWF than RRMS, PPMS and HC groups. Moreover, PSS cases were found to have low NAA/tCr, which may indicate axonal or neuronal loss (low NAA) and/or inflammation (high tCr). 6 Wallerian degeneration along the pathway of the focal lesion or transsynaptic degeneration may contribute to diffuse pathology in PSS.
Limitations of the study include a small sample size of PSS cases, which reflects the low prevalence of this variant and prevents group-level statistical analyses. There was a large variation in MWF and NAA/tCr for all groups, which is expected when analysing these measures in heterogenous groups. The age of one PSS case was outside the range of our comparison groups by 4–6 years due to the availability of historical data. A retrospective chart review of the clinical history did not provide cerebrospinal fluid or visual evoked potential findings for PSS1 and PSS2, respectively. Further study involving longitudinal advanced MRI metrics in PSS cases is warranted.
In conclusion, advanced MRI findings demonstrate more extensive abnormalities in the normal-appearing brain of two PSS cases despite a solitary demyelinating lesion along the corticospinal tract, which may in part contribute to the gradual progression of disability. Central nervous system injury in PSS may not be isolated to a solitary lesion.
Supplemental Material
Supplemental material for Advanced imaging findings in progressive solitary sclerosis: a single lesion or a global disease?
Supplemental Material for Advanced imaging findings in progressive solitary sclerosis: a single lesion or a global disease? by Lisa Eunyoung Lee, Jillian K Chan, Emilie Nevill, Adam Soares, Irene M Vavasour, Erin L MacMillan, Hideki Garren, David Clayton, B Mark Keegan, Roger Tam, Anthony L Traboulsee, Shannon H Kolind and Robert L Carruthers in Multiple Sclerosis Journal—Experimental, Translational and Clinical
Footnotes
Acknowledgements
The authors would like to thank sincerely the participants and MRI technologists at the UBC MRI Research Centre. They also thank the UBC MS/MRI Research Group for help with generating lesion masks and Ms Carina Graf and Dr Brian Weinshenker for helpful discussions.
Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LEL, EN, AS and IMV declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. JKC has received fellowship funding from Biogen and consulted for Roche. ELM has received salary support from Philips Healthcare Canada. HG and DC are employees of Genentech. BMK has research funded by Biogen and has received publishing royalties for Common Pitfalls in Multiple Sclerosis and CNS Demyelinating Diseases. RT has received research support as part of sponsored clinical studies from Novartis, Roche and Sanofi Genzyme. ALT has received research funding from Chugai, Roche, Novartis, Genzyme and Biogen, and consultancy honoraria from Genzyme, Roche, Teva, Biogen and Serono. SHK has received research funding from Sanofi Genzyme and F Hoffmann La Roche. RLC is a site investigator for studies funded by Novartis, MedImmune and Roche, and has received research support from Teva Innovation Canada, Roche Canada and Vancouver Coastal Health Research Institute. He also has done consulting work and has received honoraria from Roche, EMD Serono, Sanofi, Biogen, Novartis and Teva.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Milan & Maureen Ilich Foundation, Genentech and F Hoffmann-La Roche.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
