Abstract
Objective
The objective of this paper is to evaluate potential dose-dependent adverse effects of gadolinium-based contrast agents (GBCAs) on MS progression.
Methods
Outcomes from a cohort of 612 secondary progressive MS (SPMS) patients, enrolled in a two-year, placebo-controlled (negative) trial assessing the efficacy of MBP8298, were acquired. Patients received one to four (infrequent cohort; IFR) or 5–11 (frequent cohort; FR) GBCA injections between week 4 and week 104. The primary outcome was the change in Expanded Disability Status Scale (EDSS) and time to confirmed EDSS progression. Secondary outcomes included the changes in the Multiple Sclerosis Functional Composite (MSFC), Timed 25-Foot Walk (T25FW), 9-Hole-Peg Test (9HPT), and Paced Auditory Serial Addition Test (PASAT) from baseline to week 104.
Results
The 512 IFR and 100 FR participants showed no differences in baseline demographics or disease history. The mean change from baseline to week 104 in EDSS was +0.21 (IFR) and +0.13 (FR); MSFC –0.38 (IFR) and –0.14 (FR); T25FW +1.28 (IFR) and +0.55 (FR); 9HPT –0.06 (IFR) and –0.08 (FR); and PASAT +0.22 (IFR) and +0.20 (FR). The FR to IFR progression hazard ratio equaled 0.68 (p = 0.09). There were no significant differences in any of the outcomes between the two cohorts.
Conclusion
There were no differences in the disability progression measures between the two cohorts, indicating that gadolinium does not result in greater clinical worsening in SPMS after a two-year period.
Introduction
Gadolinium is a rare earth metal with paramagnetic properties. Because free gadolinium (Gd3+) is toxic (by a competition with calcium pathways or by promoting the expression of growth factors), it has to be chelated to organic ligands so that it can be used as a contrast agent.1–3 Based on the chemical structure of the ligand, gadolinium-based contrast agents (GBCAs) can be classified into four groups: linear nonionic and ionic, and macrocyclic nonionic and ionic. GBCA’s intrinsic stability is defined as the macrocyclic being the most stable, followed by linear ionic and linear nonionic being less stable. 4 It has long been considered that GBCAs were eliminated rapidly and almost completely after intravenous injection. However, recent studies have shown that gadolinium dechelates and deposits in different types of tissues. 1 Gadolinium deposition in the brain was described in 2015 by Kanda and colleagues 5 when they described an association between high signal intensity in the dentate nucleus (DN) and globus pallidus (GP) on unenhanced T1-weighted images in patients who had undergone brain magnetic resonance imaging (MRI) with linear GBCA administration. However, there is disagreement in the scientific literature regarding the presence of this signal after the administration of macrocyclic GBCA. While most studies have not demonstrated any dose-dependent increase in T1 signal with macrocyclic GBCA administration,5,6 a weak signal change has been reported in a few other analyses. 7 Asymptomatic gadolinium retention has been designated as gadolinium storage condition (GSC). 8 However, these recent observations regarding GBCA retention have given rise to questions concerning subsequent clinical effects.
In 2016, the term gadolinium deposition disease (GDD) was introduced by Semelka and colleagues and refers to a symptomatic disease process observed in individuals with normal renal function that arises within two months of GBCA administration and has no other etiologic explanation. 9 The most frequent symptoms reported of GDD include headache (>75%), bone and joint pain (>75%), skin changes (thickening and/or discoloration) (60%), and clouded mentation. Because of skin thickening, patients may complain of tightness of the hands and feet, resembling the feeling of being fitted with tight gloves and socks. They might also feel intense pain in limb extremities often described as needles or burning sensations. 8 Up to now only a small subset of patients have reported symptoms potentially related to GBCA, 10 and the causative association between these symptoms and chronically retained gadolinium has not been proven by scientific investigation. No definite GBCA dosage thought to be associated with clinical manifestations has been reported in the literature to date. However, case reports have described the emergence of symptoms after the first dose of GBCA administration 11 and have generally been manifested within the first 24 hours to one month of exposure. 10
Gadolinium-enhanced MRI is extremely valuable in the diagnosis and monitoring of multiple sclerosis (MS). Retention of gadolinium in the deep gray nuclei of the central nervous system might be thought to cause neurological symptoms, but none have yet been described. We investigated whether more frequent MRI scanning with gadolinium, such as would occur in clinical trials, could translate into detectable clinical changes such as progression of disability or changes on cognitive assessments. Therefore, we analyzed a frequent (FR) and infrequent (IFR) scanning cohort of patients taking part in a negative clinical trial in secondary progressive multiple sclerosis (SPMS) to determine whether differential GBCA accumulation affected the clinical outcomes of the trial.
Materials and methods
Study population
A cohort of 612 patients was acquired who were enrolled in a two-year, placebo-controlled (negative) trial assessing the efficacy of MBP8298 in SPMS patients. 12 Ethics committees at each site approved the study and all participants signed written informed consent. During the two-year follow-up, patients were scanned and received between 1 and 11 GBCA injections. A total of 512 patients received between one and four doses (infrequent/IFR group) and 100 patients received between 5 and 11 injections (frequent/FR group) (Figure 1).

Number of magnetic resonance imaging (MRI) (doses) with gadolinium-based contrast agent for the infrequent (N = 512) and frequent (N = 100) MRI cohorts.
Study design
This is a retrospective observational cohort study comparing the clinical outcomes in FR and IFR scanning groups of SPMS patients. MRI scans were performed using a standardized protocol at 1.0, 1.5 or 3T. A standard single dose of GBCA (0.1 mmol/kg gadolinium) was administered. Participants received either a linear nonionic agent (gadodiamide, Omniscan®) or linear ionic agent (gadopentetate dimeglumine, Magnevist®) or a macrocyclic nonionic agent (gadoteridol, ProHance®) or macrocyclic ionic agent (gadoterate, Dotarem®). The same GBCA was used throughout the duration of the study.
GBCA administration schedule
The population in the original study had two MRI follow-ups: in the first 100 participants (FR group), two MRIs (week –4, week 0) were performed prior to the first dose of study medication, followed by further MRIs at weeks 4, 8, and 12. Another MRI was then performed prior to the next dose (week 26), followed by three further MRIs at weeks 30, 34, and 38. Annual MRIs were then performed at weeks 52 and 104. Overall, the frequent cohort received up to 11 doses of GBCA. This close radiological follow-up was designed to capture the onset of action and anti-inflammatory effect of MBP8298. The remaining participants (IFR group) had two MRIs performed prior to first dose of medication (weeks –4 and 0) with annual MRIs at weeks 52 and 104. Therefore, the IFR cohort received up to only four doses of GBCA.
Clinical outcome
Clinical assessments available from the clinical trial data included Expanded Disability Status Scale (EDSS), 13 Cerebellar functional system (FS) score, Timed 25-Foot Walk (T25FW), 9-Hole-Peg Test (9HPT), Paced Auditory Serial Addition Test (PASAT), and the Multiple Sclerosis Functional Composite (MSFC) 14 at baseline, week 52, and week 104. Time to confirmed disease progression was defined as the date of the first EDSS increase if the EDSS increase was ≥ 1.0 and sustained for six months, and the baseline EDSS was ≤ 5.0 or if the EDSS increase was ≥ 0.5 and sustained for six months, and the baseline EDSS was ≥ 5.5. The cerebellar FS outcomes were collected at the end of this study as a post hoc analysis regarding the propensity of gadolinium to accumulate in the DN.
Statistical analysis
Of the 612 individuals in the dataset, 605 had outcomes measured at week 0 (baseline) and seven participants had no data at this time point. For the missing data at baseline of the seven remaining participants, screening results from week –4 were used. The clinical outcomes were analyzed between baseline and year 2 (week 104).
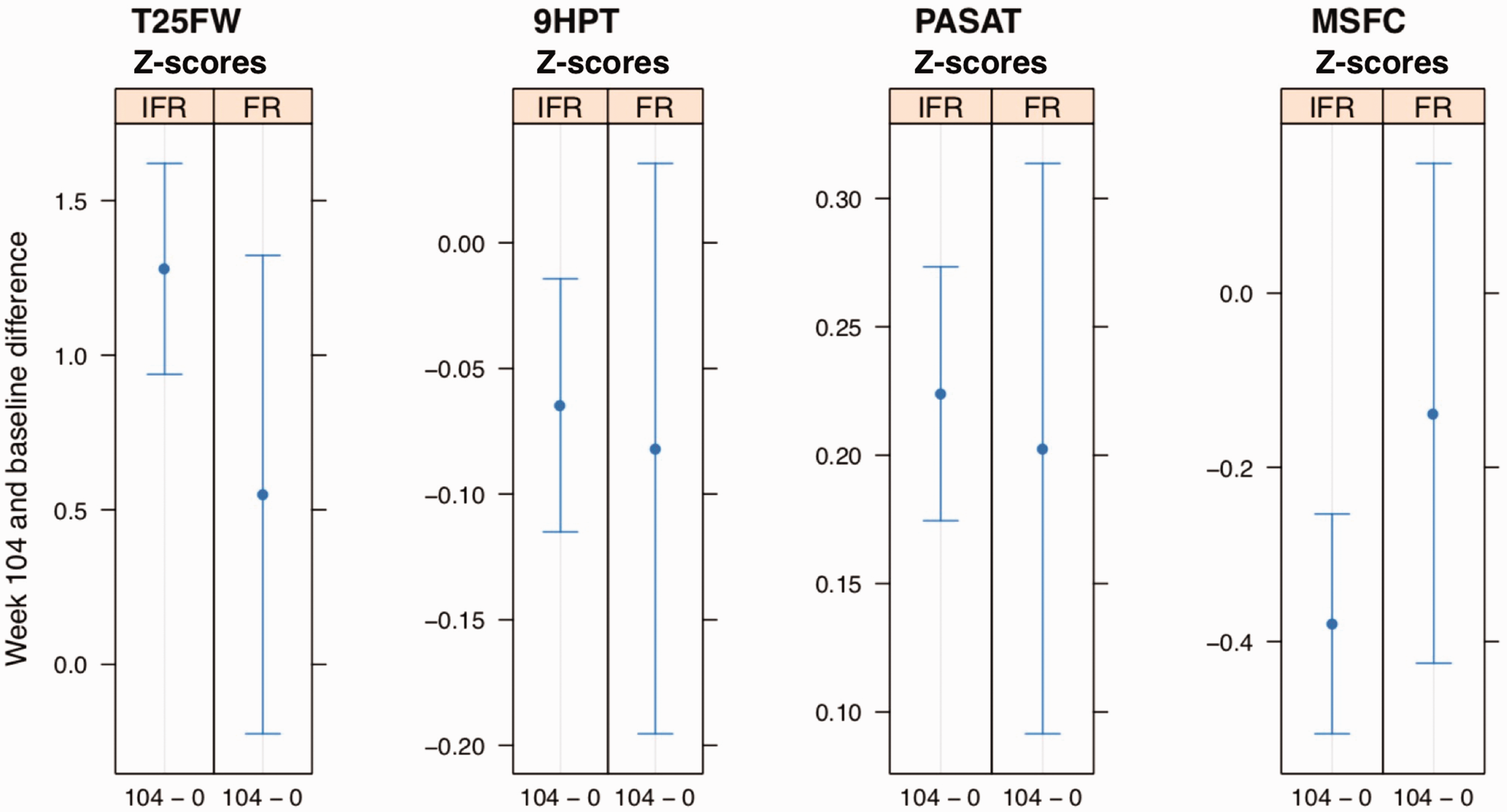
Because the components of MSFC differ in direction of change (deterioration marked by higher scores on the 9HPT and T25FW vs lower scores on the PASAT), the MSFC composite score is expressed as a Z score that is calculated from each individual component’s Z scores. Decreasing Z scores indicate neurologic deterioration. 14 The reference population used to create the Z scores was the placebo population from the initial trial. If the T25FW was not performed because of physical limitations, a score of 13.7 would be imputed for the Z score. 15 Hence, the graphs of the T25FW Z scores and MSFC Z scores pictured in Figure 2 show the difference of these two clinical outcomes between week 104 and baseline including the imputed values of 13.7.

There were no significant differences in the estimated mean change in the Timed 25-Foot Walk (T25FW), 9-Hole-Peg Test (9HPT), Paced Auditory Serial Addition Test (PASAT), and Multiple Sclerosis Functional Composite (MSFC) between weeks 0 and 104 between the infrequent (IFR) and frequent (FR) cohorts.
For the cerebellar FS and the EDSS, 9HPT Z scores and PASAT Z scores, mixed-effects models were fit with time as a categorical fixed effect with three levels (baseline, week 52, and week 104), group as a fixed effect with two levels (frequent and infrequent), and patient as a random effect. An interaction term of time with group is included in the model to allow for the changes from baseline to be estimated uniquely within each group. Mixed-effects models are not used for the outcomes T25FW Z scores and MSFC Z scores as they do not fit well because of the skewness caused by the imputed values used for incomplete assessments. Instead, paired differences between the outcomes at week 104 and baseline are calculated and then a fixed-effects model is fit to these paired differences with group included as a fixed effect. For cerebellar and all clinical outcomes, least-squares mean predictions from their fitted model are made for the comparisons of interest and the t distribution is assumed for calculating confidence intervals (CIs) and p values.
A Cox proportional hazards regression model was fit to time to disease progression that included a fixed effect for group (FR and IFR). Asymptotic normality was assumed for calculating the group hazard ratio CI.
The imbalance in the sample sizes between the groups is adjusted for by the models and is reflected in the uncertainty of the estimates in that the CIs for the FR group are wider compared to those for the IFR, and these uncertainties are accounted for when comparing the two groups.
Data availability
Anonymized data will be shared by request from any qualified investigator.
Results
Patient baseline demographics, including sex, average age, disease duration, clinical outcomes are presented in Table 1. In the FR cohort, 81% received more than nine doses of GBCA. The cohort distribution to the four types of GBCA is also represented in Table 1. The majority of the total study population (89.8%) received either nonionic linear or ionic linear agents.
Table of baseline demographic, clinical characteristics and cohort distribution of the four types of GBCAs.

9HPT: 9-Hole-Peg Test; EDSS: Expanded Disability Status Scale; FR: frequent cohort; GBCA: gadolinium-based contrast agents; IFR: infrequent cohort; MSFC: Multiple Sclerosis Functional Composite; PASAT: Paced Auditory Serial Addition Test; T25FW: Timed 25-Foot Walk.
aBaseline averages are based on individuals who had outcome measured both at baseline and week 104.
bFour patients had missing data for the type of GBCA used.
Change in EDSS
No significant difference (CI –0.24 to 0.09; p = 0.38) in changes in EDSS was observed from baseline to week 104 between the IFR group (+0.21 ± 0.07) and the FR group (+0.13 ± 0.15) (Figure 3).

EDSS mean estimate at week 0, week 52 and week 104 for infrequent (IFR, N = 512) and frequent (FR, N = 100) gadolinium-based contrast agent dose exposure demonstrates no difference in the change from week 104 to week 0 between the cohorts. CI: confidence interval; EDSS: Expanded Disability Status Scale score.
Time to confirmed EDSS progression
The analysis of time to confirmed disease progression in the FR and the IFR cohorts over the two-year period showed that the probability of remaining progression free after week 104 was 76% for the FR group and 63% for the IFR group. The hazard ratio was 0.68 (CI 0.44 to 1.06; p = 0.09), indicating that the IFR rate of progression was higher, but not significantly different from the FR group (Figure 4).
Kaplan-Meier plot of time to confirmed Expanded Disability Status Scale score (EDSS) progression in the infrequent (IFR, N = 512) and frequent (FR, N = 100) magnetic resonance imaging cohorts.
Cerebellar FS score (as part of EDSS)
There was a significant reduction in the estimated mean change of cerebellar FS between baseline and week 104 in the FR group compared to IFR one (–0.38 ± 0.21 vs +0.10 ± 0.09; CI –0.71 to –0.24; p = 0.0001), indicating that the cerebellar FS in the FR group improved.
T25FW, 9HPT, PASAT, and MSFC (Z scores) (Figure 2)
The estimated mean change of T25FW between baseline and week 104 showed an increase in the IFR group (+1.28 ± 0.34) and no significant increase in the FR group (+0.55 ± 0.77). The estimated mean change over time for the PASAT demonstrated a greater increase in the IFR group over the FR group (+0.22 ± 0.05 vs +0.20 ± 0.11). The change over two years in 9HPT and MSFC showed a decrease in the IFR groups (–0.06 ± 0.05) and (–0.38 ± 0.13), respectively, and no significant decrease in the FR groups (–0.08 ± 0.11) and (–0.14 ± 0.29), respectively.
The statistical analysis showed no difference between the IFR and FR groups over time for any of these clinical outcomes (T25FW: CI –1.58 to 0.11; p = 0.09; 9HPT: CI –0.14 to 0.11; p = 0.79; PASAT: CI –0.14 to 0.10; p = 0.73; MSFC: CI –0.07 to 0.55; p = 0.13)
Discussion
A previous study by Kang et al. demonstrated greater dose-dependent GBCA deposition in deep brain structures in a subgroup of 195 participants (80 FR and 115 IFR) from the cohort of this study. 16 In this larger cohort study, no dose-dependent effect of GBCA on MS progression was found although there was evidence of disability progression both in the FR and IFR cohorts. Moreover, there was no significant difference between these two groups in their probability of progression for any of the clinical disability outcome measures at week 104 except for the cerebellar FS, which appeared to have improved for the FR group rather than worsened like for the IFR group. This could potentially be explained as a false-positive result due to multiple post hoc analyses, which is consistent with the exploratory nature of this study.
Certain limitations need to be considered. First, the two-year study duration and the maximum of 11 dose exposures might not be sufficient to capture enough GBCA accumulation to be clinically significant. Prior gadolinium exposure was not available and therefore not incorporated into the number of dose accumulation and analyses. Moreover, a possible delayed clinical effect cannot be excluded. However, there is no definite GBCA dose thought to be associated with clinical manifestations and no delayed clinical effect has been reported in the literature to date. Case reports have described the emergence of symptoms after the first dose of GBCA11 and have indicated that clinical manifestations after GBCA administration arise mostly within the month of exposure. 10 Further studies with a longer follow-up duration should be conducted to capture the presumed long-term effect of GBCA. Second, the type of GBCA used varied across sites. However, a standard dose of GBCA was used at each participating site for all participants and the same GBCA was consistently used throughout the study. In addition, 89.8% of the entire cohort received linear GBCAs. As linear agents have the highest association with deep brain structure accumulation, 5 they may be the most suitable to identify clinical signs in a high exposure cohort. Finally, the clinical outcomes may have been insensitive to detect the impact of GBCA accumulation, particularly with respect to the preferential deposition in the deep gray matter structures. The PASAT cognitive test may partially reflect these structures. 17 However, in this study the change over two years marginally improved in both cohorts, although the difference between the two cohorts was not significant. Fine motor skills could also be affected by injury to deep gray matter structures, and this study observed significant worsening in the 9HPT only for the IFR cohort. No statistical difference between the two groups was observed. Given the propensity of gadolinium to accumulate in the DN, 18 an impact on the cerebellar FS might be suspected. Post hoc analysis of the cerebellar FS showed a significant difference between the two cohorts, with the FR group showing an improvement over time contrary to what would be expected for an adverse effect on the cerebellar system. However, it is important to highlight that the changes in all the clinical outcomes over time and between the two cohorts were consistently small.
In summary, this large, retrospective, longitudinal study did not find any evidence of GDD manifesting as greater disability progression, cognitive impairment, or worsening cerebellar function in SPMS patients receiving frequent dosing (up to 11 doses) of GBCA over a two-year follow-up period.
Footnotes
Acknowledgments
The authors thank Andrew Riddehough (MS/MRI research group – University of British Columbia) for his assistance in gathering the data and information used in this analysis.
Statistical analysis performed by Carolyn Taylor, MSc.
Conflict of Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Nathalie Ackermans has received a grant funding from Sanofi-Genzyme and Biogen for her one-year research fellowship in MS. Carolyn Taylor has nothing to declare. Roger Tam has received research support as part of sponsored clinical studies from Novartis, Roche, and Sanofi Genzyme. Robert Carruthers has received grants/research funding from MedImmune, Teva, and Guthy Jackson; has received speaking fees for unbranded lectures from Biogen, Genzyme, and Teva; and has received consulting fees from Novartis, EMD Serono, and Genzyme. Shannon Kolind has received research/educational grant funding from Genzyme; has received honoraria or consultation fees from Acorda and Genzyme; and is a member of a company advisory board, board of directors or other similar group of Acorda and Genzyme. Heejun Kang has nothing to declare. Mark S. Freedman has received a research/educational grant from Genzyme; received honoraria or consultation fees from Actelion, BayerHealthcare, BiogenIdec, Chugai, Clene Nanomedicine, EMD Canada, Genzyme, Merck Serono, Novartis, Hoffman La-Roche, Sanofi-Aventis, and Teva Canada Innovation; is a member of a company advisory board, board of directors or other similar group of Actelion, Bayer HealthCare, Biogen Idec, Hoffman La-Roche, Merck Serono, MedDay, Novartis, and Sanofi-Aventis; and is on the speakers bureau for Genzyme. David K.B. Li has received research funding from the Canadian Institute of Health Research and Multiple Sclerosis Society of Canada. He is the emeritus director of the UBC MS/MRI Research Group, which has been contracted to perform central analysis of MRI scans for therapeutic trials with Novartis, Perceptives, Roche, and Sanofi-Aventis. The UBC MS/MRI Research Group has also received grant support for investigator-initiated independent studies from Genzyme, Merck Serono, Novartis, and Roche. He has acted as a consultant to Vertex Pharmaceuticals and served on the Data and Safety Advisory Board for Opexa Therapeutics and Scientific Advisory Boards for Adelphi Group, Celgene, Novartis, and Roche. He has also given lectures that have been supported by nonrestricted education grants from Biogen Idec, Novartis, Sanofi-Genzyme, and Teva. Anthony L. Traboulsee has received grant funding from the MS Society of Canada, Canadian Institute for Health Research, Roche, and Genzyme; and has received honoraria or travel grants from Teva Canada Innovation, Roche, Genzyme, and Chugai Pharmaceuticals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
