Abstract
Objective
Venous thromboembolism (VTE) remains a major preventable cause of global mortality. Digital health tools may facilitate patient self-management and adherence to treatment. This study aimed to evaluate the feasibility of a mobile VTE application (mVTEA) for VTE management.
Methods
This pilot study employed a mixed-methods design, integrating a quantitative quasi-experimental evaluation with qualitative focus groups in patients using the mVTEA. From August 25, 2023 to May 10, 2024, the mVTEA was developed, incorporating patient-centered three-dimensional (3D) visualization health records, dynamic risk monitoring, and medication management support modules. The quantitative evaluation was performed 1 month after enrolment via the Chinese version of the User Version of the Mobile Application Rating Scale (C-uMARS) for design quality and the Chinese version of the mHealth App Usability Questionnaire (C-MAUQ) for usability. Qualitative data were collected via four age- and education-stratified focus groups with mVTEA users, focusing on exploring patients’ illness perceptions and in-depth adoption experiences to identify optimization priorities.
Results
Among the 43 patients who completed the 1-month follow-up, 37 (86%) provided positive feedback on the use of digital 3D health records. The mVTEA received favorable average quality (3.81±0.81) and usability (1.92±0.81) scores in the quantitative evaluation. Qualitative analysis revealed significant knowledge gaps among VTE patients and a strong desire for authoritative information. The participants expressed strong appreciation for the core functionalities of the mVTEA and desired real-time doctor–patient communication capabilities and a reliable knowledge base. Furthermore, they demonstrated a willingness to pay for comprehensive, mVTEA-enabled healthcare services.
Conclusions
In this feasibility study, the mVTEA demonstrated robust quality and usability among patients with or at high risk for VTE. Focus groups confirmed positive user feedback and identified specific optimization needs, collectively supporting the feasibility of mVTEA as a VTE management tool. This provides foundational evidence for future research to evaluate its impact on clinical outcomes.
Introduction
Venous thromboembolism (VTE), which comprises deep vein thrombosis (DVT) and pulmonary embolism (PE), affects nearly 10 million people globally each year and accounts for roughly one-quarter of all deaths.1,2 Post-thrombotic sequelae, including chronic thromboembolic pulmonary hypertension (CTEPH) and post-thrombotic syndrome (PTS), are common following VTE. Survivors of acute PE frequently experience long-term functional limitations and reduced quality of life. Post-PE impairment has been reported in up to 17% of cases, with a median of only 129 days to CTEPH diagnosis after PE. 3 Moreover, almost half of patients with VTE develop PTS, which imposes substantial demands on healthcare services and is associated with substantial costs. 4 These complications contribute significantly to morbidity, mortality, and socioeconomic burdens.
However, a substantial proportion of healthcare costs and mortality associated with VTE progression may be prevented through effective patient self-management. Patient-centered care, which prioritizes individual needs, is intended to empower patients to take an active role in managing their disease trajectory. Originating from Edith Balint's 1969 description of “understanding the patient as a unique human being”, 5 this concept has evolved into a framework that integrates collaborative care, shared decision-making, and alignment with patient values and goals. The recent Scientific Statement on Patient-Centered Adult Cardiovascular Care from the American Heart Association advocates such a collaborative approach to enhance engagement, tailor interventions, and thereby improve clinical outcomes. 6 Nevertheless, translating patient-centered principles into practice across the full spectrum of care remains challenging, particularly in facilitating meaningful communication, enhancing health literacy, and sustaining active participation in personal health management.
Patient-facing mobile health (mHealth) technologies, such as smartphone applications and wearable devices, offer significant potential to enable daily self-monitoring and self-management outside of clinical settings.7–9 Evidence has demonstrated that such interventions can facilitate behavioral changes and improve health outcomes in patients with atrial fibrillation (AF),8,10,11 heart failure,12,13 coronary heart disease, 14 diabetes,15,16 and chronic obstructive pulmonary disease. 17 In anticoagulation therapy, mHealth tools have been shown to enhance self-management for patients receiving warfarin. However, commercially available, self-administered applications remain limited, likely owing to the need for intensive monitoring and dose adjustments typically conducted within hospital settings.
Within the domain of VTE, no dedicated mobile application or digital health platform has been established to support patients throughout the course of the disease, with the goal of mitigating progression and preventing major complications. To address this gap, we have developed a smart technology-assisted, patient-centered mHealth application for managing VTE (mVTEA). 18 This pilot mixed-methods study was conducted to quantitatively evaluate the technological quality and usability of the mVTEA, and to qualitatively explore patients’ illness perceptions, attitudes, and experiences with the tool, thereby evaluating its potential for VTE home management.
Methods
Study design and setting
This pilot study employed a mixed-methods design, which integrated a quantitative quasi-experimental evaluation with qualitative focus groups in patients using the mVTEA. It was conducted at the Sixth Medical Center of the Chinese People's Liberation Army General Hospital (PLAGH) and was approved by the institutional Ethics Committee (Approval No. HZKY-PJ-2022-21). This study was registered with the Chinese Clinical Trial Registry (Approval No. ChiCTR2200063206).
Development of the mVTEA
The mVTEA was developed on the Android platform following the Software Development Life Cycle (SDLC) framework, which consists of five phases: requirement gathering, design, implementation, testing, and maintenance. 19 A user-centered design approach was applied to ensure that the interface and functionality addressed patient needs. The user interface was informed by Nielsen's 10 usability heuristics 20 : (1) visibility of system status, (2) match between the system and the real world, (3) user control and freedom, (4) consistency and standards, (5) error prevention, (6) recognition rather than recall, (7) flexibility and efficiency of use, (8) aesthetic and minimalist design, (9) help users recognize, diagnose, and recover from errors, and (10) help and documentation. Clinical content was derived from authoritative sources and current international clinical guidelines, including those of the European Society of Cardiology, American Heart Association, American Society of Clinical Oncology, and American College of Chest Physicians,21–27 to enable the delivery of personalized, evidence-based VTE management. The core modules include patient-centered digital health records, digital VTE care pathway, and medication management support. Figure 1 illustrates the core functional interface of the mVTEA.
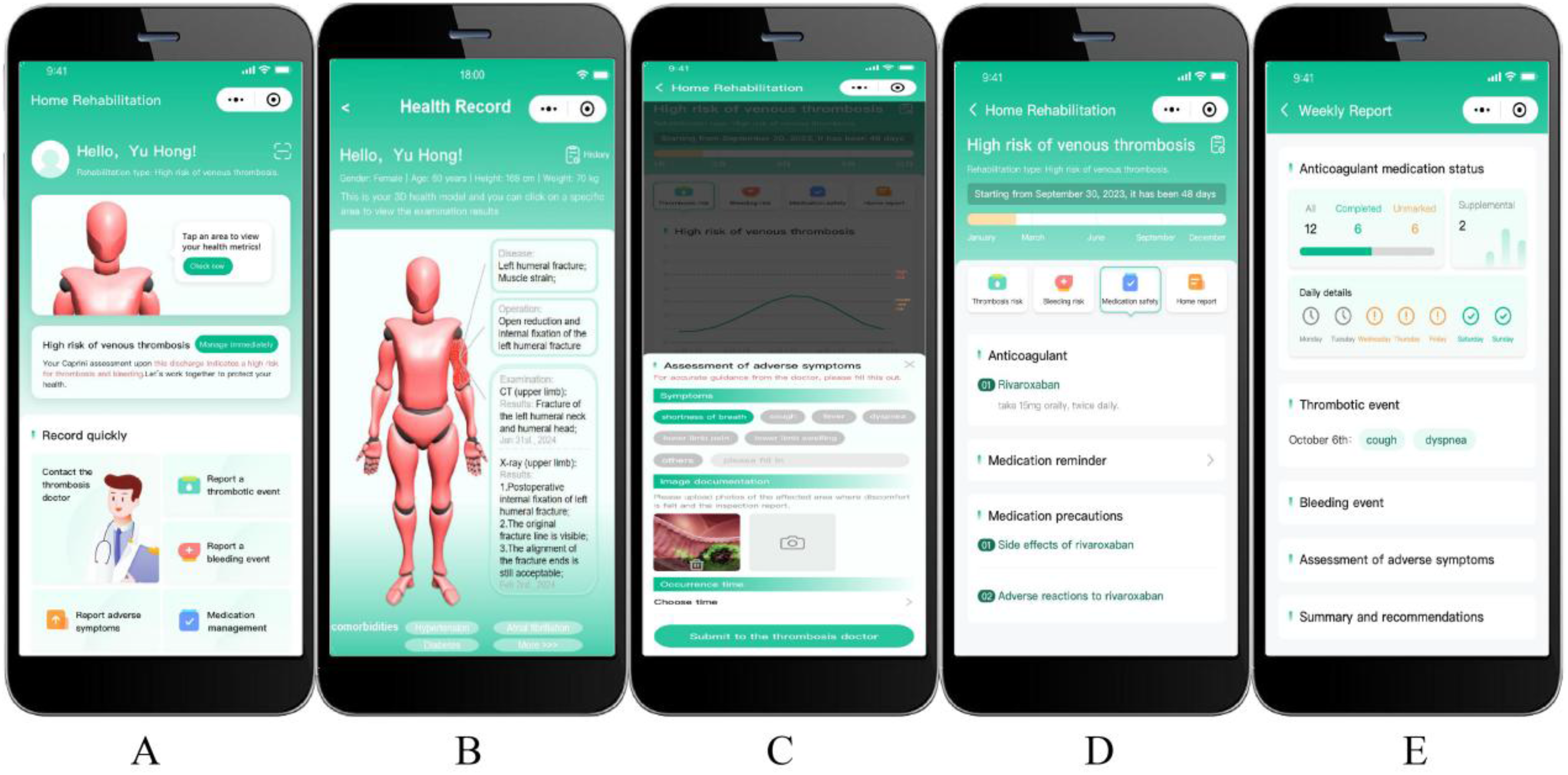
Core functional interface of the mVTEA for patient-centered VTE management. (A) Homepage with navigation to key modules, (B) interactive 3D visualized health records for intuitive review of personal medical data, (C) event reporting portal for logging thromboembolic events, bleeding incidents, and other symptoms, (D) medication management module supporting adherence tracking and reminders, and (E) weekly health summary report providing an overview of status and trends.
Patient-centered digital health records
Patient-centered digital health records were developed; incorporating three-dimensional (3D) visualization to enable patients to monitor their medical history, laboratory tests, and imaging results (Figure 1B). User satisfaction, feasibility, and acceptability of patient-centered digital health records were subsequently evaluated using a structured questionnaire (Table 1).
The questionnaire for assessing participants’ satisfaction, feasibility, and acceptability of patient-centered digital health records.

Digital VTE care pathway
The clinical care pathway for VTE integrated into the mVTEA was operationalized through the ABCDEF pathway, which was designed to achieve patient-centered care. This pathway encompassed the following components:
Appropriate Antithrombotic Management: Guided anticoagulant selection (e.g., direct oral anticoagulants, warfarin), adjusted doses based on laboratory monitoring (e.g., INR), and provided thromboprophylaxis. Bleeding Risk Management: Incorporated bleeding risk scores (e.g., HAS-BLED) and provided precautions to manage bleeding risks alongside anticoagulation. Complication Monitoring: Facilitated surveillance for recurrent VTE, PTS, and CTEPH via scheduled check-ups and patient self-reporting of symptoms. Digital Health Management: Utilized the mVTEA for telemedicine, medication tracking, symptom reporting, and wearable device integration for remote monitoring. Exercise and Rehabilitation: Provided personalized and supervised exercise programs to improve long-term physical activity and outcomes. Facilitate the Management of Vascular Risks and Comorbidities: Managed modifiable risk factors (e.g., hypertension, diabetes) and promoted lifestyle modifications through integrated care.
For patients diagnosed with VTE requiring anticoagulation, it is adapted to focus on appropriate antithrombotic management, among other components. Furthermore, the mVTEA used a hybrid data model. Authorized clinicians securely input and transmit critical data such as laboratory results and imaging reports to the patient's application via a published encrypted QRST-AB algorithm, which has been independently developed and validated in our prior study. 28 Patients can enter daily symptoms and metrics. This integrated data was visualized for patients, helping them understand their personal thrombosis and bleeding risks, track trends, and engage in informed self-management under professional guidance.
Medication management support
The Medication Management Support Module was a core component designed to enhance patients’ understanding and execution of their prescribed anticoagulation therapy. It addressed key areas of medication safety, including administration protocols, potential drug-drug interactions, and adherence to daily regimens. The system generated intelligent alerts and notifications, which were customized according to the patient's specific medication schedule and any dose adjustments made by their healthcare provider. Meanwhile, it provided a structured educational program that covered the critical importance of anticoagulant adherence, the recognition and management of potential adverse effects, and guidance on common drug-drug and food-drug interactions.
Data governance, security, and privacy measures
Given the sensitive nature of the medical data collected, a robust data governance and security framework was implemented. All data transmitted between the mobile application and the backend servers was secured using Transport Layer Security (TLS) 1.2 or higher. At rest, all personally identifiable information was encrypted using the Advanced Encryption Standard (AES-256). Access to the backend database was strictly controlled via a role-based access control system, ensuring that only authorized research personnel could access the data after de-identification for analysis purposes. Data were stored on secure, access-controlled servers within a HIPAA-compliant environment. A formal security protocol was established and adhered to throughout the study period.
Study populations
Adult patients hospitalized at the Department of Cardiology of the Sixth Medical Center of the PLAGH between August 25, 2023, and January 31, 2024, were eligible for inclusion if they had a diagnosis of VTE or were classified as being at high risk for VTE according to the Padua scale. Eligible thrombi included lower extremity or upper extremity DVT (jugular, innominate, subclavian, axillary, or brachial), PE, splanchnic vein thrombosis (hepatic, portal, splenic, mesenteric, renal, or gonadal), or cerebral vein thrombosis confirmed by appropriate imaging.
Patients were enrolled if they met all of the following criteria: (1) diagnosis of VTE or classification as high risk for VTE, (2) aged ≥ 18 years, (3) normal cognitive function without a diagnosed mental disorders, and ability to understand the study-related forms, (4) proficiency in using a smartphone, and (5) voluntary participation. Patients were excluded if they had severe hearing or language impairments that prevented effective cooperation with the researchers. Patients fulfilling these requirements and expressing interest in participation received a comprehensive, in-person explanation of the study objectives, procedures, potential benefits, and possible risks. Written informed consent was obtained from all participants prior to study initiation. All participants were followed for 1 month to allow for a quantitative evaluation of the mVTEA, including evaluation of its design quality and usability. Subsequently, 12 patients were purposively selected from this cohort for in-depth qualitative evaluation conducted through focus groups.
Quantitative research of the mVTEA evaluation
The design quality of the mVTEA was evaluated via the Chinese version of the user version of the Mobile Application Rating Scale (C-uMARS),29,30 and its usability was evaluated via the Chinese version of mHealth App Usability Questionnaire (C-MAUQ).31,32 Both questionnaires are publicly available, standardized instruments that have been validated in previous research. Their use in academic research does not require specific copyright permission according to the published terms.
The C-uMARS consists of 14 items grouped into three subscales: (A) Information, (B) Functionality, and (C) Engagement (Table 2). 30 The Information subscale evaluates the relevance, credibility, and usefulness of the application's content. The Functionality subscale assesses the application's usability, navigation, and overall performance. The Engagement subscale measures the level of user engagement with the application, including factors such as interactivity and customization options. Each subscale of the C-uMARS is rated on a 5-point scale (1-inadequate, 2-poor, 3-acceptable, 4-good, and 5-excellent).
The C-uMARS questionnaire for the quality evaluation of the mVTEA.

The C-MAUQ, which has established reliability and validity, was used for usability evaluation. 32 It contains 21 items organized into three subscales: (A) usability and satisfaction (eight items), (B) system information arrangement (six items), and (C) efficiency (seven items) (Table 3). A 7-point Likert scoring system was used, with the following coding: 1 = extremely strongly agree, 2 = strongly agree, 3 = agree, 4 = neutral, 5 = disagree, 6 = strongly disagree, and 7 = extremely strongly disagree. Therefore, lower C-MAUQ scores indicate more positive ratings of usability, which contrasts with the C-uMARS scoring direction.
The C-MAUQ questionnaire for the usability evaluation of the mVTEA.

Focus groups qualitative research on the mVTEA use
Focus groups are a well-established qualitative research method that enables researchers to gain in-depth insights into participants’ perspectives, experiences, and understanding. 33 We employed a purposive sampling method to select participants for focus groups from the 43 patients who had completed the quantitative evaluation. Participants were stratified by age and education level, aiming to maximize the range of perspectives within the resource limitations of this initial evaluation.
The research team, comprising two male researchers, included Hao Wang (moderator) and Bin-bin Liu (co-moderator). Wang, an associate professor in the Department of Cardiology, holds an M.D. degree and has over 10 years of experience in clinical practice and qualitative research. Liu holds a Bachelor's degree in Clinical Medicine and was responsible for process documentation and assistance. In addition to two researchers, the other authors of this paper and some nursing staff also observed the online meeting. Prior to the research, the moderator had no pre-existing relationship with any participants. At the beginning of each interview, the research purpose and the academic backgrounds of the researchers were explained to the participants.
A total of four focus groups were conducted via online video conferencing. An interview guide was developed, focusing on three main themes (eTable 1 in Supplemental 1), to explore patients’ knowledge and information needs regarding VTE, their evaluations of the mVTEA and their needs for home-based VTE care. The moderator structured discussions to address predetermined themes while remaining responsive to issues that emerged during the sessions. The co-moderator documented field notes during the discussions to capture non-verbal cues and interactive contexts. All discussions were conducted and audio-recorded in Mandarin. The recordings were sent to a professional service for verbatim transcription into Chinese. Transcripts were analyzed in the original language to preserve semantic and contextual meaning. No repeat interviews were required, and transcripts were not returned to participants for comment.
Data analysis followed the thematic analysis approach. Two researchers independently performed initial coding of the entire text dataset using Nvivo 15 software and were blinded to each other's work. Inter-coder reliability was assessed prior to consensus discussions by calculating Cohen's kappa coefficient (κ) on a randomly selected subset of transcripts (30%). The initial κ value was 0.78, indicating substantial agreement according to the benchmark criteria of Landis and Koch. 34 Through multiple iterative discussions, any coding discrepancies were reviewed and resolved until full consensus was reached. The researchers subsequently collaboratively categorized and consolidated the codes to jointly develop a final coding tree (Supplemental 2). To enhance the methodological rigor, data source triangulation was employed by comparing subgroups with different ages and educational backgrounds to examine the convergence and divergence of perspectives. This section was written in accordance with the Consolidated Criteria for Reporting Qualitative Research (COREQ) (eTable 2 in Supplemental 1). 35
Statistical analysis
This study was designed as a pilot evaluation of the mVTEA. The sample size requirement was determined using the method proposed by Viechtbauer et al., 36 which estimates the number of participants needed to detect potential unforeseen problems with a given probability at a specified level of confidence. According to this method, with a minimum problem occurrence probability of 0.10 and a 95% confidence level, a minimum of 29 participants is sufficient to detect at least one occurrence. For the qualitative arm, 12 participants were included in the focus groups. This sample size is considered adequate to achieve thematic saturation in relatively homogeneous groups, as supported by established qualitative research methodology literature. 37
Continuous variables with a normal distribution are presented as mean ± standard deviation (SD). Non-normally distributed continuous variables are expressed as median (interquartile range, IQR). Categorical variables are summarized as frequency (percentage) and compared using the chi-square (χ2) test. For both the C-uMARS and C-MAUQ, the score for each question is presented as the mean of all participant responses. The score for a subscale or the total score is calculated by first computing the average of the respective items for each participant. These individual sums are then averaged across the sample to produce the reported subscale or total score. The Wilcoxon rank-sum test was performed to conduct intragroup comparisons of the subscales within the C-uMARS and the C-MAUQ, respectively. A two-sided P < .05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics version 26.0 and R version 4.4.1.
Results
A total of 43 patients (mean [SD] age, 56.9 [15.9] years; 25 men (58%) and 18 women (42%); median follow-up, 34.7 days [IQR, 29–40 days]) were included (Figure 2). Among them, 35 patients (81%) were diagnosed with VTE and eight patients (19%) were stratified as being at high risk for VTE by the Padua scale. The baseline characteristics of all patients are presented in Table 4. Hypertension was the most common comorbidity, followed by coronary heart disease and diabetes.

Patient enrolment and follow-up flowchart.
Baseline characteristics of the participants that completed the 1-month follow-up.

BMI, body mass index (calculated as weight in kilograms divided by height in meters squared). BMI ≥ 28 is defined as obesity according to the Chinese guidelines.
Patient-centered digital health records
Of the 43 patients surveyed, 41 (95%) rated the QR code scanning process for accessing digital health records as fine or excellent. Furthermore, 37 patients (86%) gave a positive rating to the digital 3D visualization of health records, and expressed satisfaction with the access to and use of patient-centered digital health records, as well as the benefits of digital 3D visualization records, such as an increased sense of control and the visualization of changes in laboratory tests (Figure 3).

The results of the investigation into satisfaction, feasibility, and acceptability of patient-centered digital health records.
Quantitative research of the mVTEA evaluation
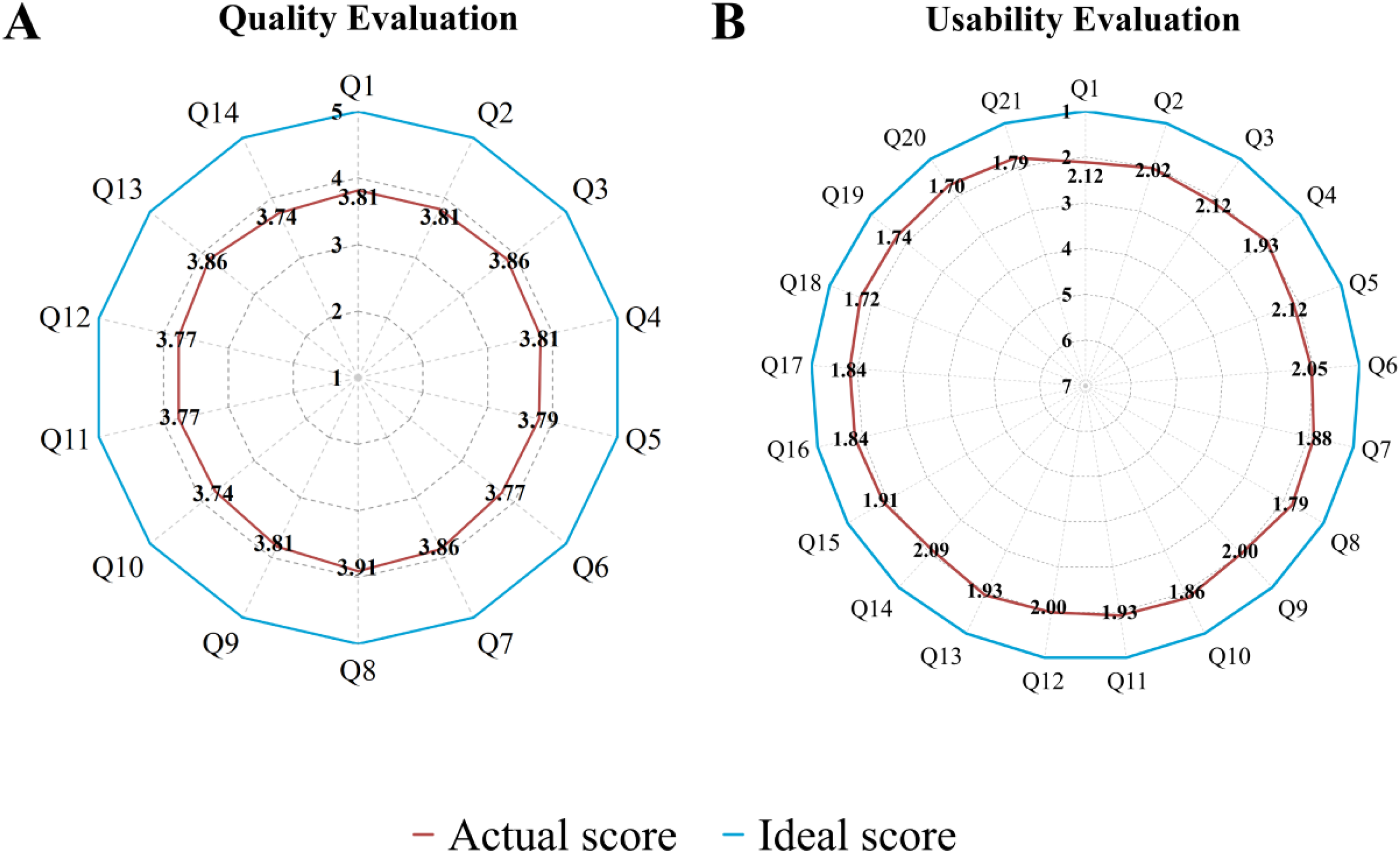
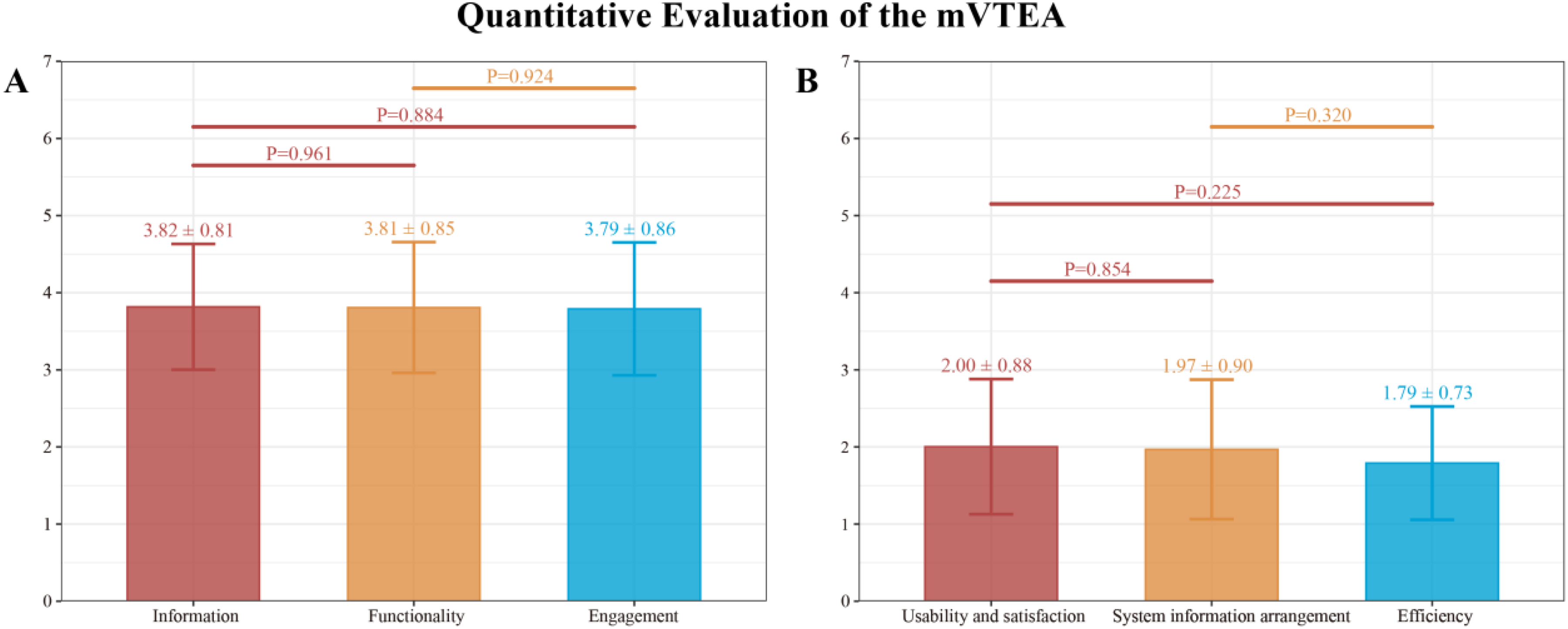
Figure 4 presents the average scores for all the items in the quantitative research including all 43 participants. The average quality score (mean ± SD) of the mVTEA was 3.81 ± 0.81. The Information subscale received the highest ratings (3.82 ± 0.81), followed by Functionality (3.81 ± 0.85), and Engagement (3.79 ± 0.86) (Figure 5A). Within the Information subscale, items 7 (information credibility) and 3 (visual esthetics) obtained the highest mean score of 3.86. Conversely, the lowest mean scores within the Functionality and Engagement subscales were both 3.74, corresponding to item 10 (navigation) and item 14 (target audience appropriateness), respectively. The overall C-MAUQ score was 1.92 ± 0.81 (range: 1.70 to 2.12), reflecting favorable usability (a lower score represents a more positive evaluation). The scores for the three subscales of Usability and Satisfaction, System Information Arrangement, and Efficiency were 2.00 ± 0.88, 1.97 ± 0.90, and 1.79 ± 0.73, respectively (Figure 5B). Specifically, within the Usability and Satisfaction subscale, item 8 (Overall, I’m satisfied with the application) was rated most favorably with a mean score of 1.79. In contrast, item 14 (This application has all the features and capabilities I expected) in the System Information Arrangement subscale (possession of expected features and capabilities) received the least favorable rating, with a mean score of 2.09.
The average score results from the quantitative evaluation of all 43 participants. (A) The average score of the C-uMARS for quality evaluation. Higher C-uMARS scores indicate more positive user evaluation (1 = inadequate; 7 = excellent) and (B) the average score of the C-MAUQ for usability evaluation. Lower C-MAUQ scores indicate more positive user evaluation (1 = extremely strongly agree; 7 = extremely strongly disagree).
Subscale scores from the C-uMARS (a 5-point scale, with higher scores indicating higher quality) and the C-MAUQ (a 7-point scale, with lower scores indicating better usability).
Focus groups on patients’ priorities regarding the mVTEA use and home management
The qualitative evaluation was conducted between March 20 and May 10, 2024, and we systematically gathered perspectives from all 12 VTE patients (eight males; aged 18–87 years; eight with tertiary education) through four focus groups, each lasting 90–120 min. Through iterative analysis of all four focus groups, the three predetermined core themes established in this study were demonstrated to be stable and comprehensive. The data analysis yielded seven sub-themes (Figure 6) that further enriched the thematic structure, while no new concepts emerged that necessitated revision of the core framework. This finding confirms that thematic saturation at the core thematic level was achieved with the data collected via purposive sampling. Detailed patient perspectives and partial representative quotations derived from the sub-themes analysis are presented below. Full representative quotations are provided in eTable 3 in Supplemental 1.

Theme and sub-themes of focus groups on patients with the mVTEA use.
Theme 1: VTE awareness and information needs
Patients demonstrated significant gaps in their knowledge regarding VTE. This cognitive state directly contributed to a significant psychological burden and motivated an active search for reliable information sources.
Gaps in VTE foundational knowledge
Patients consistently demonstrated inaccurate comprehension of fundamental concepts such as the definition and etiology of VTE. Their understanding of VTE treatments was predominantly limited to anticoagulation therapy, while failing to extend to a clear understanding of potential side effects. Only a minority of patients were aware of the increased risk of bleeding associated with anticoagulation. ‘I’m afraid that using anticoagulants might cause bleeding’ (P5, female, 57 years old, primary education).
Psychosocial impact of VTE
Patients generally perceived VTE, particularly PE, as highly dangerous. This perception leads to significant psychological distress and lifestyle impacts, primarily manifesting as behavioral changes such as a fear of engaging in physical activity, which substantially diminished their quality of life. ‘When I was first diagnosed with a thrombosis, I was utterly terrified… Now I still live in constant fear, never knowing when it might recur’ (P11, male, 43 years old, tertiary education).
Information-seeking behavior and channel evaluation
Patients primarily acquired VTE knowledge from healthcare professionals and online sources. Triangulation revealed that younger, highly-educated patients tended to cross-verify information through multiple channels, while older or less-educated counterparts relied more heavily on direct guidance from healthcare professionals. Despite these behavioral differences, all participants expressed a strong desire for access to authoritative VTE knowledge. ‘I always feel online information isn't sufficiently accurate. While I trust doctors as the most authoritative source, it's impossible to consult them at any time. Is there a more convenient way to obtain reliable knowledge about thrombosis’ (P10, male, 50 years old, tertiary education).
Theme 2: evaluation and expectations for the mVTEA
Patients widely acknowledged the existing functional value of the mVTEA and expressed clear expectations for its future development, while their specific foci varied across demographic groups.
Willingness to use the mVTEA and evaluation of its functionality and usability
All patients expressed strong endorsement for core functionalities, including the digital 3D visualization records, medication management support, adverse event reporting, and weekly health summaries. Concurrently, they emphasized the critical importance of data privacy. Specific suggestions for improvement were also provided, primarily concerning the intelligent medication reminder and the user interface. These needs were particularly evident among elderly users. 'These functions are really useful as they enable doctors to adjust my treatment plan, but patient privacy must be carefully protected’(P1, male, 30 years old, tertiary education).
Expectations for a future version of the mVTEA
Nearly all patients anticipated that the mVTEA would deliver two core functions: real-time doctor–patient communication and a specialized VTE knowledge base. Several participants also recommended incorporating lifestyle intervention (such as exercise reminders and hydration prompts). ‘It would be ideal to get quick answers from doctors through online consultation when I feel unwell. My only concern is that doctors might sometimes be too busy to reply promptly’(P6, female, 58 years old, secondary education). ‘I believe reminders for daily activity and drinking water would be quite useful’(P2, male, 32 years old, tertiary education).
Theme 3: home-based VTE care
Patients generally expressed a desire to access healthcare services based on mVTEA and demonstrated willingness to purchase mechanical thromboprophylaxis devices.
Willingness to pay for the mVTEA and desired self-care functions
Patients demonstrated a strong demand for home-based VTE management services through the mVTEA platform, with their ideal service model being a multifunctional integrated service. This comprehensive package would include annual/monthly online consultations, cross-institutional examination report interpretation, and monthly health reports. The acceptable price range was concentrated between RMB 1000 and 2000 annually. A notable age-related divergence emerged: younger patients showed not only greater willingness to pay for perceived beneficial services but also stronger preference for long-term service models. In contrast, older patients prioritized cost-effectiveness, preferring the lowest price for equivalent services. ‘If my condition requires me to purchase this service, I would only purchase a long-term subscription(such as an annual plan). Otherwise, I would not purchase it at all’(P9, male, 18 years old, tertiary education).
Willingness and acceptability of mechanical prevention
Under mVTEA platform-based end-to-end management, patients explicitly prefer direct purchase of mechanical thromboprophylaxis devices to secure ownership. The device rental model, however, has not gained favor. ‘If I genuinely need it, I would rather purchase it than rent it’(P3, male, 49 years old, tertiary education).
Mixed methods integration
A joint display (Table 5) was developed to integrate quantitative results from the C-uMARS and C-MAUQ assessments with qualitative themes from focus groups. This display highlights convergence between data streams, explanatory complementarity for lower-scoring dimensions, and expansion where qualitative evidence uncovered new user priorities.
Integration of quantitative and qualitative findings.

Discussions
This study employed a combined quantitative and qualitative research to evaluate the feasibility of the mVTEA. The quantitative results indicated that the design quality and usability of the application were robust evaluated by patients with VTE and those at high risk for VTE. Qualitatively, patient feedback from focus groups provided depth to these metrics, revealing strong endorsement of core functionalities, significant unmet information and communication needs, and a clear willingness to engage with and pay for integrated home-based care services delivered through the mVTEA. These findings collectively demonstrate mVTEA's preliminary feasibility and provide directions for optimization.
In the quality evaluation, the Information subscale received the highest evaluation, with Information Credibility scoring particularly well. This result was consistent with the qualitative finding of knowledge gaps among VTE patients and their clear need for authoritative information. This consistency indicates that the credibility and professionalism of information are the foundation for users’ acceptance of mVTEA, while also highlighting the importance of subsequently integrating a professional VTE knowledge base. Additionally, the analysis reveals a critical tension between standardized digital design and the different abilities within the users. Quantitative scores for Navigation and Target Audience Appropriateness were comparatively lower. Qualitative research corroborated this finding: elderly users generally perceived the interface as somewhat complex and insufficiently intuitive. This highlights the limitations of standardized design in addressing user diversity and underscores the need for the mVTEA to adopt more personalized, inclusive design principles (such as providing a simplified interface with larger fonts for elderly users) to accommodate varying levels of digital literacy. Furthermore, when users perceive high core utility in an application, they may exhibit greater tolerance for certain usability limitations. This was reflected in the usability assessment, where overall satisfaction was relatively high while ratings for interface esthetics were lower. This can be attributed to the considerable enthusiasm users express for core features such as interactive 3D digital health records. The visual and exploratory nature of such features appears to enhance both comprehension and perceived value, 38 indicating that optimizing highly practical core functionalities is equally crucial to refining interface details during early development stages. Qualitative findings revealed a clear willingness among patients to pay for integrated VTE management services delivered through the mVTEA platform, although preferred service models and price sensitivity varied across demographic groups. This not only supports the economic viability of implementing such a digital tool but also underscores the importance of tailoring service programs to meet diverse user expectations.
The findings of this pilot study offer concrete solutions to address key challenges in the current management of VTE. VTE, as a preventable and manageable disease, has led to severe economic and social burdens.39,40 The conventional VTE management model has limitations such as high treatment costs, a shortage of high-quality medical resources, suboptimal adherence to guidelines, insufficient prevention and treatment of complications, and a lack of post-diagnosis rehabilitation management. Thrombotic and bleeding risks are not static, determined by a “one-time” assessment of baseline factors, but rather dynamic and continually influenced by factors such as aging, incident comorbidities, and changes in medications. 41 Many of these factors are transient and potentially modifiable, underscoring the need to explore new, adaptive pathways for VTE management.
Mobile health technology has emerged as the key to addressing these challenges. The mAFA, by implementing an integrated management pathway specific to AF, has increased the anticoagulation rate among high-risk patients to over 80% and significantly reduced clinical adverse events. 8 This demonstrates the substantial efficacy of a structured digital pathway in chronic disease management. However, its entire design logic is closely centered on AF and cannot be directly applied to the management of VTE. Alfalfa focuses on optimizing the warfarin anticoagulation therapy through remote INR monitoring and dose adjustment, increasing the time in therapeutic range to 79.35% and effectively improving anticoagulation quality. 42 Nevertheless, its medication-oriented model shows clear limitations within the current paradigm of patient-centered disease management.
The mVTEA developed in this study aims to address this gap. It is the first to apply integrated, structured digital clinical pathways to VTE, aiming to establish a comprehensive platform covering the entire VTE care and to address the core requirements of VTE care. Our study demonstrates that the mVTEA exhibits robust quality and usability. Patients’ willingness to adopt this tool, coupled with their strong endorsement of its core features, indicates its feasibility and promising potential for VTE management. Future studies should consider evaluating long-term engagement, behavioral mechanisms of change, applicability in broader patient populations, integration with clinical workflows, and cost-effectiveness to better understand the application's effectiveness in enhancing patient self-management. 18
Limitations
This study has limitations regarding generalizability. The single-center design inherently limits the diversity of the patient population and clinical practices reflected in our sample. A notable selection bias was introduced by the requirement for smartphone ownership and digital literacy, as illustrated in the recruitment flow (Figure 2). The participants therefore likely represent a subgroup of patients who are more comfortable and engaged with digital technology. Consequently, the positive quality and usability findings may not be generalizable to the broader VTE population, particularly older adults or those with limited digital access or skills, which should be considered when interpreting the results. The qualitative evaluation included 12 participants. This sample size is appropriate for a feasibility study aiming to deeply explore user perceptions and gather detailed feedback for tool optimization, and we observed signs of information saturation. However, it also implies that the identified themes and insights, while valuable, may not fully represent the perspectives of a broader cohort of VTE patients with varying digital skills or disease experiences. Consequently, these qualitative findings should be regarded as exploratory discoveries providing key directions and hypotheses for subsequent research. The lack of a control group in this pilot means that positive user feedback could be influenced, in part, by the increased attention inherent to participation in a study (e.g., Hawthorne effect). 43 This is an inherent limitation of a single-arm feasibility study.
Another limitation was that user satisfaction was measured as a feasibility metric. The scores thus represented initial perceptions to inform design refinements and were not definitive. The development and full validation of a satisfaction instrument, including establishing its content validity and internal consistency, were reserved for a subsequent, larger-scale study following this iterative development phase.
Lastly, the current study did not include objective behavioral or clinical endpoints necessary to draw definitive conclusions on effectiveness. Based on our previous research on secure data transmission technology and the current feasibility study of smart technology-enabled VTE management, and as we now commence the randomized controlled trial (SmaVTE study), addressing the effectiveness and the impact on clinical and safety outcomes.18,28
Conclusion
In this pilot study, the mVTEA demonstrated robust quality and usability among patients with or at high risk for VTE. Focus groups further clarified specific optimization needs and clinical deployment requirements. These findings indicate the application's feasibility and potential as a VTE management tool and provide a foundation for future real-world evaluations of its clinical effects.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261430465 - Supplemental material for Smart technology facilitated patient-centered venous thromboembolism management: A mixed-methods study of the digital feasibility
Supplemental material, sj-docx-1-dhj-10.1177_20552076261430465 for Smart technology facilitated patient-centered venous thromboembolism management: A mixed-methods study of the digital feasibility by Binbin Liu, Zhigeng Jin, Hao Wang, Hui Zhang, Zheqi Zhang, Hong Wang, Ying Yang, Zhenguo Zhai and Yutao Guo in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076261430465 - Supplemental material for Smart technology facilitated patient-centered venous thromboembolism management: A mixed-methods study of the digital feasibility
Supplemental material, sj-docx-2-dhj-10.1177_20552076261430465 for Smart technology facilitated patient-centered venous thromboembolism management: A mixed-methods study of the digital feasibility by Binbin Liu, Zhigeng Jin, Hao Wang, Hui Zhang, Zheqi Zhang, Hong Wang, Ying Yang, Zhenguo Zhai and Yutao Guo in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors thank all the staff and participants at the Sixth Medical Center of the PLAGH and all the patients who voluntarily took part in the study. We appreciate Liu Chen, from the DrBreath Company (DrBreath Co., Ltd., Beijing, China) for her technical assistance with the mVTEA.
ORCID iDs
Ethical approval
This study was approved by the Ethics Committees of the Sixth Medical Center of the PLAGH (Approval No. HZKY-PJ-2022-21). The trial was registered in the Chinese Clinical Trial Registry (Approval No. ChiCTR2200063206).
Author contributions
Study planning, project conception, and coordination were conducted by Zhigeng Jin, Zhenguo Zhai and Yutao Guo; all authors contributed to the design of the intervention; recruitment and data extraction were conducted by Binbin Liu and Hao Wang. All authors reviewed and approved the final version of the manuscript. Technical development and maintenance of the mVTEA were provided by DrBreath Co., Ltd, under the scientific direction of the authors.
Funding
This study was funded by National Key Research and Development Program of China (2023YFC2507201).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
Yutao Guo.
Supplemental material
Supplemental material for this article is available online.
