Abstract
Objective
Chronic Obstructive Pulmonary Disease (COPD) remains widely underdiagnosed in low- and middle-income countries due to reliance on costly, infrastructure-heavy diagnostic methods such as spirometry and radiographic imaging. This study aimed to design and validate a low-cost, artificial intelligence (AI)-powered Breath-Based Diagnostic (BBD) system for real-time COPD detection using exhaled volatile organic compounds (VOCs).
Methods
The BBD system integrates an array of metal oxide semiconductor sensors with a lightweight one-dimensional convolutional neural network deployed on a Raspberry Pi 5 for edge-based inference. Diagnostic performance was evaluated using a publicly available VOC dataset and a custom dataset collected under controlled conditions with the prototype device. Data augmentation strategies simulated sensor drift and environmental variability to improve model robustness. System performance was assessed in terms of accuracy, precision, latency, power efficiency, cost trade-offs, and usability. A multilingual mobile interface and Retrieval-Augmented Generation chatbot were developed to support patient engagement, while adherence to HIPAA and FHIR standards ensured regulatory compliance.
Results
The proposed system achieved 96.68% accuracy and 100% precision for COPD detection, with inference latency of 0.02 ms and power consumption below 2.5 W. A five-sensor configuration preserved 98% of diagnostic performance while reducing hardware cost by 30%. Usability testing with 31 participants yielded a System Usability Score of 86.3/100 and a chatbot trust rating of 4.4/5, confirming strong user acceptance.
Conclusion
The study demonstrates the feasibility of deploying an explainable, low-cost, and energy-efficient BBD system for early COPD detection in resource-limited settings. By combining edge AI, affordable sensor arrays, and multilingual patient engagement, the BBD system offers a scalable and ethically grounded pathway for integration into national healthcare infrastructures and global respiratory health strategies.
Introduction
Background and global relevance
Chronic Obstructive Pulmonary Disease (COPD) remains a major global health burden, accounting for approximately 3.2 million deaths annually and ranking as the third leading cause of mortality worldwide. The impact is particularly severe in low- and middle-income countries (LMICs), where healthcare systems often lack access to essential diagnostic tools such as spirometry and radiographic imaging. 1 This diagnostic gap contributes to delayed recognition, late-stage interventions, and excess mortality. In regions such as sub-Saharan Africa, exposure to risk factors—including biomass combustion, air pollution, and occupational hazards such as small-scale mining—further exacerbates the prevalence of respiratory disorders. 2 Vulnerable groups, including women and children exposed to indoor smoke, face disproportionately high risks, with studies showing a high prevalence of impaired lung function across rural communities. 3 Timely diagnosis is critical, as COPD can be effectively managed if identified early. However, conventional diagnostic modalities are constrained in LMICs by their reliance on trained specialists, high operational costs, and limited infrastructure. 4 Moreover, spirometry, though considered the clinical gold standard, has limitations in detecting early disease and requires significant patient cooperation. 5 Recent studies have also drawn attention to preserved ratio impaired spirometry (PRISm), a condition that reflects early structural and functional abnormalities before classical spirometric decline, underscoring the need for tools that can detect COPD earlier than current standards. 6
Volatile organic compounds and breathomics in pulmonary diagnostics
Non-invasive breath analysis has gained growing attention as a potential solution for overcoming the diagnostic barriers associated with COPD in LMICs. Exhaled breath contains volatile organic compounds (VOCs)—metabolic byproducts linked to oxidative stress, airway inflammation, and other pathophysiological processes—that can serve as biomarkers for respiratory disease.7,8 The field of “breathomics” has emerged to systematically analyze these compounds and identify disease-specific signatures. Gas Chromatography–Mass Spectrometry (GC/MS) remains the reference standard for VOC analysis due to its high specificity and sensitivity. 9 However, its cost, technical complexity, and lack of portability make it impractical for large-scale screening or use in community health settings. As a result, there has been growing interest in sensor-based platforms such as electronic noses (E-noses), which rely on metal oxide semiconductor (MOS) sensors to detect mixtures of VOCs in real time. E-noses as shown in Figure 1 are low-cost, portable, and relatively easy to operate, making them attractive for deployment in LMICs. Nevertheless, they are hampered by limitations in selectivity, susceptibility to environmental conditions such as humidity and temperature, and lack of standardized calibration protocols. 10
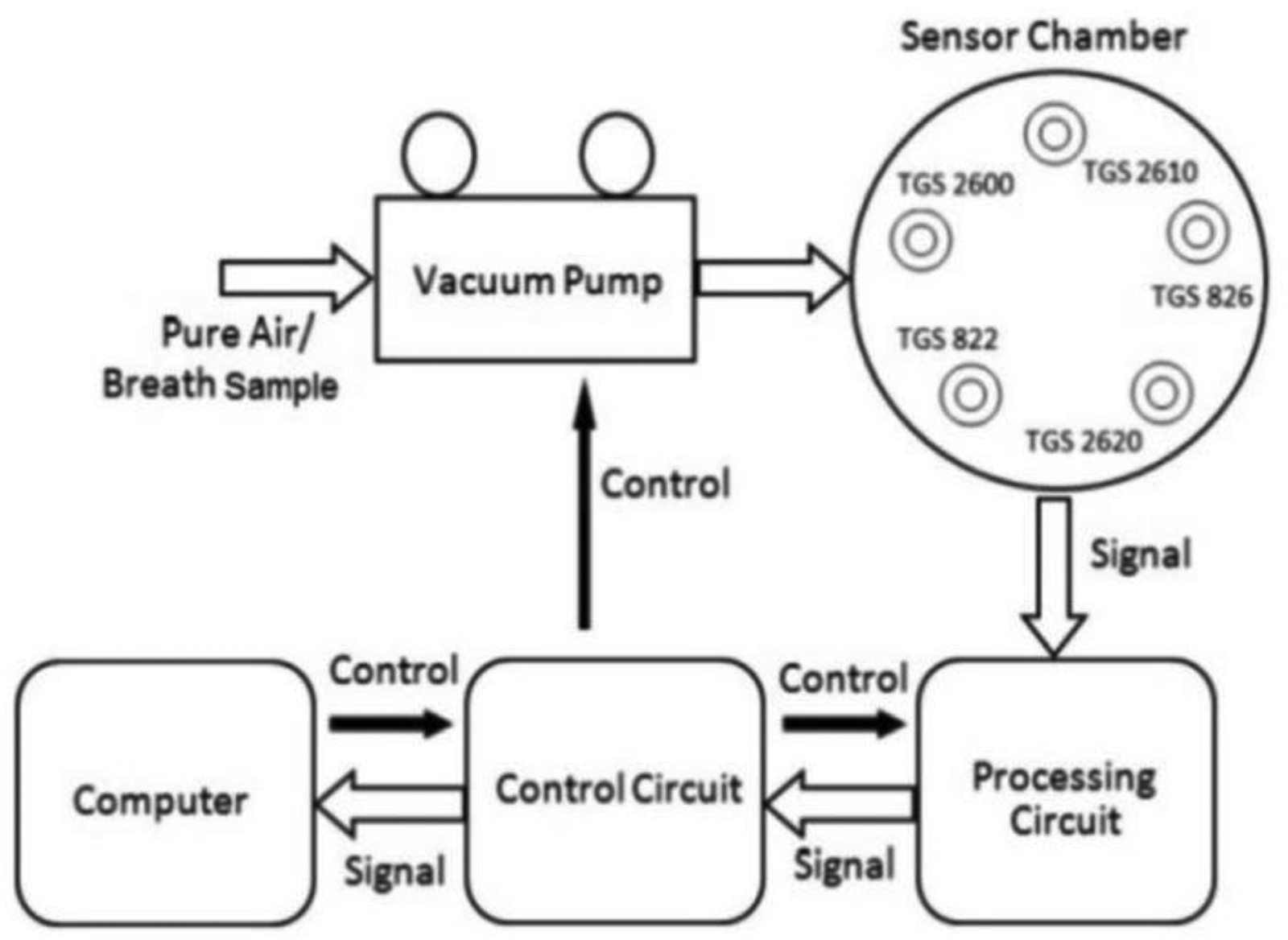
The electronic nose (E-nose) system. 10
Related studies and technological advances
Several studies have demonstrated the feasibility of combining sensor arrays with machine learning techniques for breath-based COPD detection. For instance, 11 reported an edge artificial intelligence (AI) framework capable of classifying COPD-related VOCs using synthetic datasets on a Cortex-M4 microcontroller, achieving high diagnostic accuracy in laboratory conditions. 11 However, the system relied on simulated data and lacked validation under real-world conditions. Similarly, Arduino-based E-nose prototypes employing Taguchi gas sensors achieved moderate accuracies (76.67% for COPD and 79.31% for lung cancer), 10 but were limited by poor gas selectivity, cross-sensitivity, and the absence of humidity compensation. Other breathomics studies have investigated selective detection of disease-specific VOCs, including ketones, aldehydes, and hydrocarbons. 12 While these approaches highlight the potential of VOC biomarkers, most research remains confined to clinical or laboratory environments. Furthermore, many existing studies have not accounted for confounding factors such as environmental drift or population diversity, limiting their applicability in real-world contexts. Advancements in AI, edge computing, and Tiny Machine Learning now enable lightweight models to be deployed directly on embedded hardware, eliminating dependence on continuous internet connectivity.11,13 This development is particularly relevant for LMICs, where offline-first solutions are essential. However, very few studies have validated such systems using real-world datasets collected in resource-limited settings. In addition, most prior systems overlook integration with multilingual patient interfaces, privacy-preserving data protocols, and interoperability standards, which are crucial for public health adoption. Table 1 provides a comparative analysis of some existing diagnostic systems.
A comparative analysis of existing pulmonary diagnostic systems.
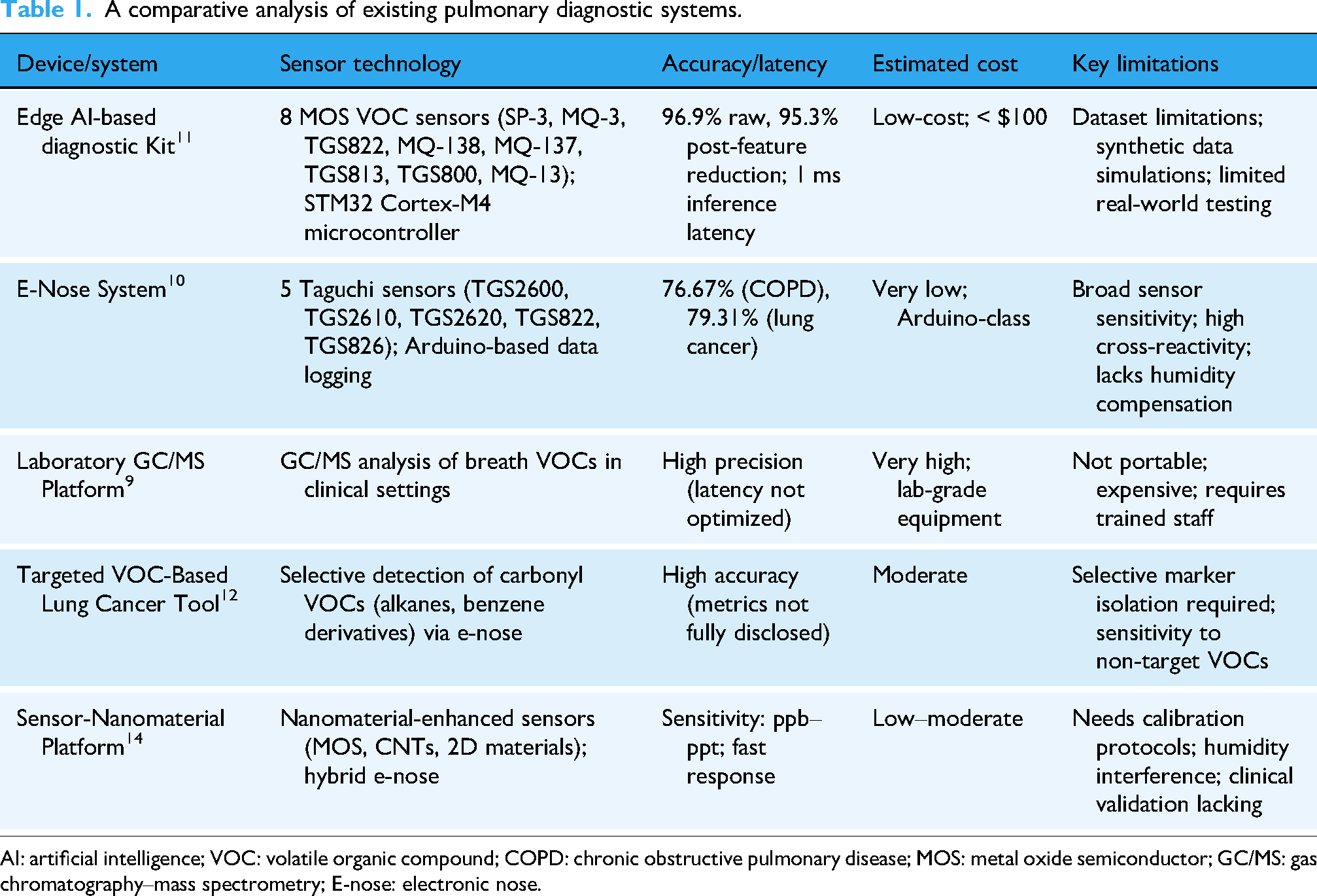
AI: artificial intelligence; VOC: volatile organic compound; COPD: chronic obstructive pulmonary disease; MOS: metal oxide semiconductor; GC/MS: gas chromatography–mass spectrometry; E-nose: electronic nose.
The research challenge
In summary, COPD diagnosis remains constrained by three persistent challenges: (i) Limited access to conventional diagnostics in LMICs; (ii) the inability of existing methods to detect disease at early stages, including PRISm; and (iii) limitations of current breath-based systems, which either rely on costly laboratory methods or prototype E-noses that lack robustness in real-world contexts. There is, therefore, a critical need for an affordable, portable, and explainable diagnostic system that can reliably identify COPD at the point of care, even in offline and resource-constrained environments.
Aim and scope
The aim of this study is to develop and validate an AI-powered Breath-Based Diagnostic (BBD) system for early COPD detection. We hypothesize that breath-based VOC signatures, analyzed with lightweight convolutional neural networks optimized for edge deployment, can achieve accurate, cost-effective, and real-time COPD diagnosis under real-world conditions.
The objectives are to:
Design and optimize a portable diagnostic device that integrates low-cost MOS sensors with an embedded deep learning pipeline. Validate diagnostic performance using both a publicly available VOC dataset
15
and a real-world dataset collected with the deployed prototype. Assess trade-offs in sensor configuration, power efficiency, and cost to ensure scalability for LMIC deployment. Incorporate multilingual patient engagement and regulatory-compliant data management to enhance usability, trust, and integration into national health systems.13,16
Contribution
This study introduces a validated diagnostic system that combines edge AI, affordable sensor arrays, and multilingual support into a unified, patient-centered platform. By demonstrating accuracy of 96.68% and precision of 100% for COPD detection, the system outperforms previous E-nose prototypes while reducing cost and power consumption. Furthermore, the inclusion of explainable AI outputs, a Retrieval-Augmented Generation (RAG)-based chatbot, and compliance with HIPAA and FHIR standards distinguishes this work from prior studies. Collectively, these innovations establish a scalable and ethically grounded pathway for early COPD screening in LMICs.
Methodology
System overview
This section presents the architectural and methodological foundation guiding the development of the Low-Resource Pulmonary Diagnostic System (BBD). The system integrates sensor-based breathomics, edge-deployed deep learning, cloud-connected IoT infrastructure, and multilingual telemedicine functionality to deliver cost-effective, real-time COPD diagnostics tailored for environments with limited connectivity. Its design and implementation followed a systematic, modular approach encompassing several key phases: the selection of appropriate sensors and hardware components; calibration of sensors and preprocessing of data; development and optimization of a deep learning model suitable for deployment on edge devices; fusion of sensor inputs for improved inference reliability; integration of Bluetooth Low Energy (BLE) for secure data synchronization with cloud services; collection of user feedback through perception studies; development of mobile and web-based applications for patient and clinician engagement; and implementation of validation protocols alongside adherence to ethical and data privacy standards. The complete system architecture—illustrated in Figure 2—demonstrates the seamless interaction among hardware (breath sampling interface), edge intelligence (on-device inference), and software layers responsible for data visualization, communication, and decision support.

System architecture for low-cost, artificial intelligence (AI)-powered pulmonaiic system.
Data acquisition, description and augmentation
The diagnostic model was trained and evaluated using a combination of two complementary datasets to ensure both generalizability and robustness. The first dataset was a publicly available VOC dataset, comprising over 1200 labeled samples across four classes: COPD, Healthy, Smoker, and Ambient Air. This dataset was collected using an E-nose array of eight MOS sensors—similar in configuration to those used in the current study—providing a valuable benchmark for model development. The second dataset, referred to as the BBD real-world dataset, was collected under controlled experimental conditions using the deployed prototype. It included 500 breath samples per second and a resolution of the ADC of 24 bits. The patients comprised of 20 COPD patients, 20 healthy patients and 4 patients exposed to polluted environments (Smokers), acquired from human participants under supervised conditions, thereby reflecting realistic variability in sensor responses and user interaction. To address the relatively limited size of the combined dataset and improve the model's ability to generalize to unseen conditions, a multi-step data augmentation strategy was implemented. This involved scaling individual sensor readings within a range of 0.9–1.1 times their original values to simulate natural sensor drift, and injecting Gaussian noise with zero mean and a standard deviation equivalent to 5% of the signal range to mimic environmental and operational variability. In addition, class-balanced data windows were randomly shuffled to reduce the risk of overfitting to temporal or sequential patterns. Collectively, these augmentation techniques expanded the dataset volume by a factor of 11 while preserving the statistical integrity of the original distributions, thereby enhancing the diagnostic model's adaptability across both controlled and real-world conditions. During the data collection the following criteria were used in Table 2 in selecting patients.
Criteria use to select patients for data collection.

Hardware design, sensor integration and calibration
The diagnostic device was designed to balance cost, sensitivity, and energy efficiency, with a focus on detecting VOCs linked to COPD, including ammonia, acetone, and benzene while detailing their importance in Table 3. Sensors were required to cost under USD 50, cover a detection range of 0.1–10,000 ppm, and consume less than 200 mA for edge deployment. Based on a comparative evaluation of commercial options (see Table 1), eight MOS gas sensors—MQ-2, MQ-3, MQ-4, MQ-6, MQ-9, MQ-135, MQ-137, and MQ-138—were selected for their biomarker sensitivity, low cost, and compatibility with microcontrollers. To address the environmental sensitivity of MOS sensors, a DHT22 sensor was added to monitor temperature and humidity, enabling real-time compensation for drift and improving measurement reliability. The system integrates an ESP32 microcontroller and a Raspberry Pi 5 microprocessor to distribute computational tasks. The ESP32, with 15 ADC channels and low-power UART support, manages analog data acquisition from the gas sensors. The Raspberry Pi 5, equipped with a quad-core CPU, 8GB RAM, and built-in BLE/Wi-Fi modules, handles machine learning inference, local storage, wireless communication, and application interfacing. This dual-platform setup allows complete on-device processing, removing reliance on cloud connectivity and ensuring functionality in rural or off-grid environments. The device enclosure, designed in SolidWorks and three-dimensional printed, was optimized for portability, thermal regulation, and reduced electromagnetic interference. It features a compact, handheld form factor with a removable top and accessible sensor ports for maintenance. For standalone use in resource-limited settings, the prototype incorporates a 16 × 2 I2C LCD display to provide immediate diagnostic feedback. Calibration was performed in clean air, with baseline resistance values (R₀) determined by averaging stable readings and stored in non-volatile memory to ensure persistence across restarts. Temperature and humidity data from the DHT22 informed compensation algorithms, maintaining robust sensor performance across variable deployment conditions.
Reasons for selecting these biomarkers.

VOC: volatile organic compound; COPD: chronic obstructive pulmonary disease.
Model architecture, edge deployment, and human-centered IoT integration
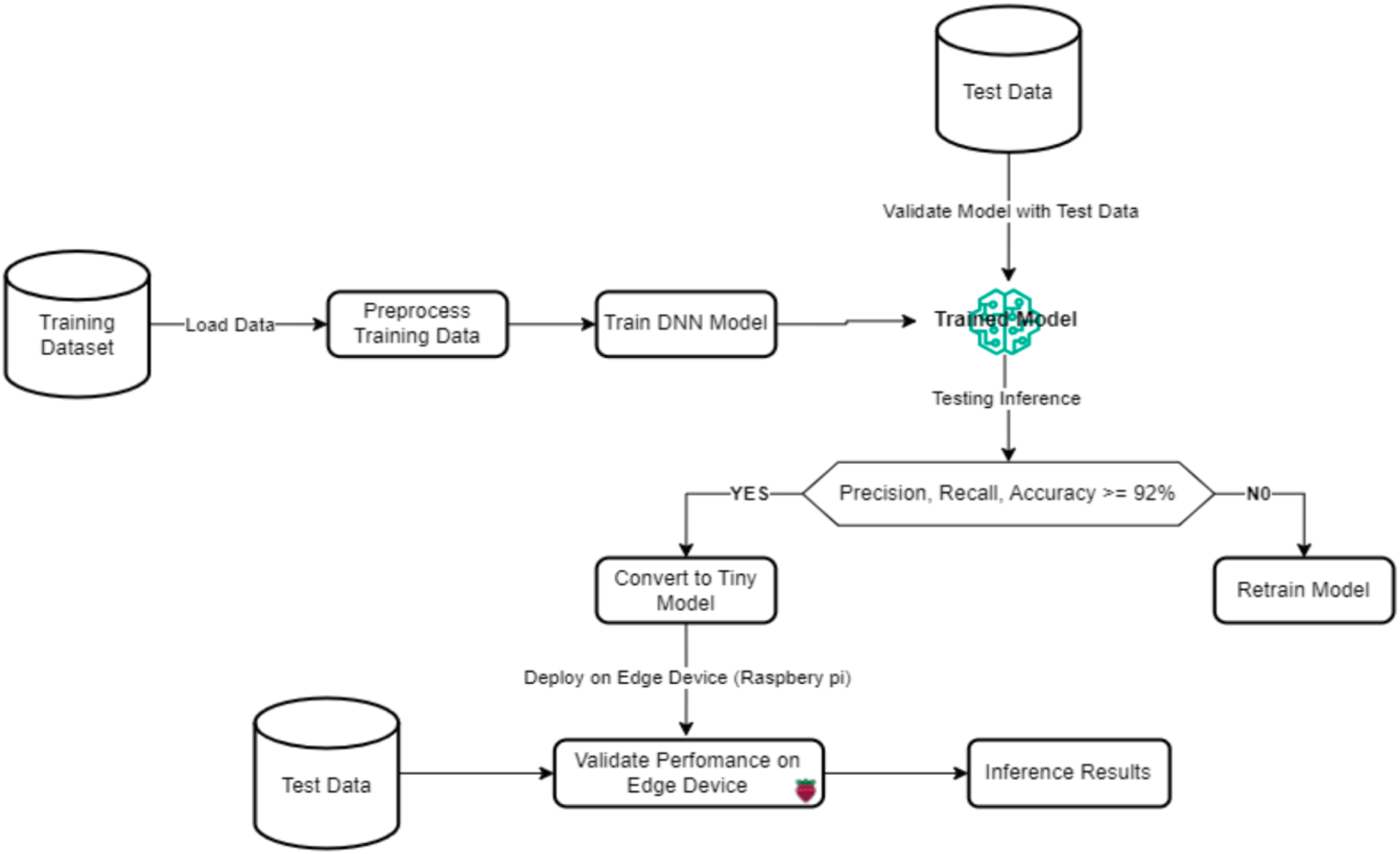
A lightweight one-dimensional Convolutional Neural Network (1D-CNN) was developed using the TensorFlow/Keras framework to classify VOC patterns captured by the sensor array. The architecture begins with an input reshaping layer for eight sensor channels, followed by two convolutional blocks—each consisting of a Conv1D layer, batch normalization, and dropout—for effective feature extraction and regularization. These are connected to fully connected dense layers with L₁ regularization to promote sparsity and reduce overfitting, and the network concludes with a softmax output layer configured for four-class prediction: COPD, Healthy, Smoker, and Ambient Air. Training employed the Adam optimizer (learning rate = 0.005), with early stopping applied to validation loss to prevent overfitting. The dataset was split into 64% training, 16% validation, and 20% testing, and class imbalance was addressed using weighted loss with class weights calculated as wi = N/Cni, ensuring balanced representation across categories. The final trained model was exported in quantized TensorFlow Lite (.tflite) format and deployed on a Raspberry Pi 5 for edge inference. Benchmarking confirmed its efficiency for real-time, low-power applications: the model achieved 96.68% classification accuracy, including 100% precision for the COPD class, with inference latency averaging 0.02 milliseconds per sample. Power consumption remained within a 2 W envelope, underscoring its suitability for resource-constrained environments. Further evaluation of sensor configurations (1, 3, 5, and 7 sensors) revealed that diagnostic accuracy improved with additional sensors but plateaued beyond five. This indicates that a five-sensor setup provides the best balance of performance, cost reduction, and energy efficiency—crucial for low-resource deployments. For data exchange, the device leverages BLE to synchronize with a companion mobile application in offline settings. Once a BLE handshake is established, diagnostic results, timestamps, and metadata are transmitted via encrypted channels to ensure privacy and integrity. When internet access is available, the system uploads diagnostic data to a Firebase-supported cloud dashboard, enabling clinician review and aggregated analytics for population-level monitoring. Security is enforced through role-based access control using OAuth tokens and device-level certificates, preventing unauthorized access during data transmission and storage. User-centered design considerations were guided by a pre-deployment perception survey of 31 participants at Ashesi University. The survey revealed that 61.3% preferred AI-driven diagnostics with human oversight, informing the integration of a transparent NLP-based chatbot that explains AI reasoning to end users. In addition, a clinician-facing dashboard was developed to support expert validation and intervention where needed. To safeguard ethical practice, informed consent protocols were embedded into the application workflow, ensuring respect for user autonomy and responsible data collection throughout the diagnostic process.
Application development, IoT architecture and NLP pipeline
The BBD platform was designed as a modular, scalable ecosystem that integrates hardware, mobile and web interfaces, cloud services, and a secure NLP pipeline. The system combines offline-first capabilities with real-time connectivity, ensuring usability in diverse healthcare environments, including rural and low-resource settings. The mobile application (Figure 3), developed in Flutter, and the clinician-facing web dashboard, built with ReactJS, are linked through Firebase Firestore and Firebase Storage. These services support secure patient record management, real-time visualization of diagnostic outputs, and the encrypted uploading of multimedia data such as breath recordings and symptom-related images. Firestore provides real-time synchronization and offline caching, enabling continuous functionality in areas with intermittent connectivity, while its horizontally scalable design ensures support for a growing user base. Some limitations were observed in handling complex relational queries, suggesting opportunities for future optimization or hybrid back-end integration.
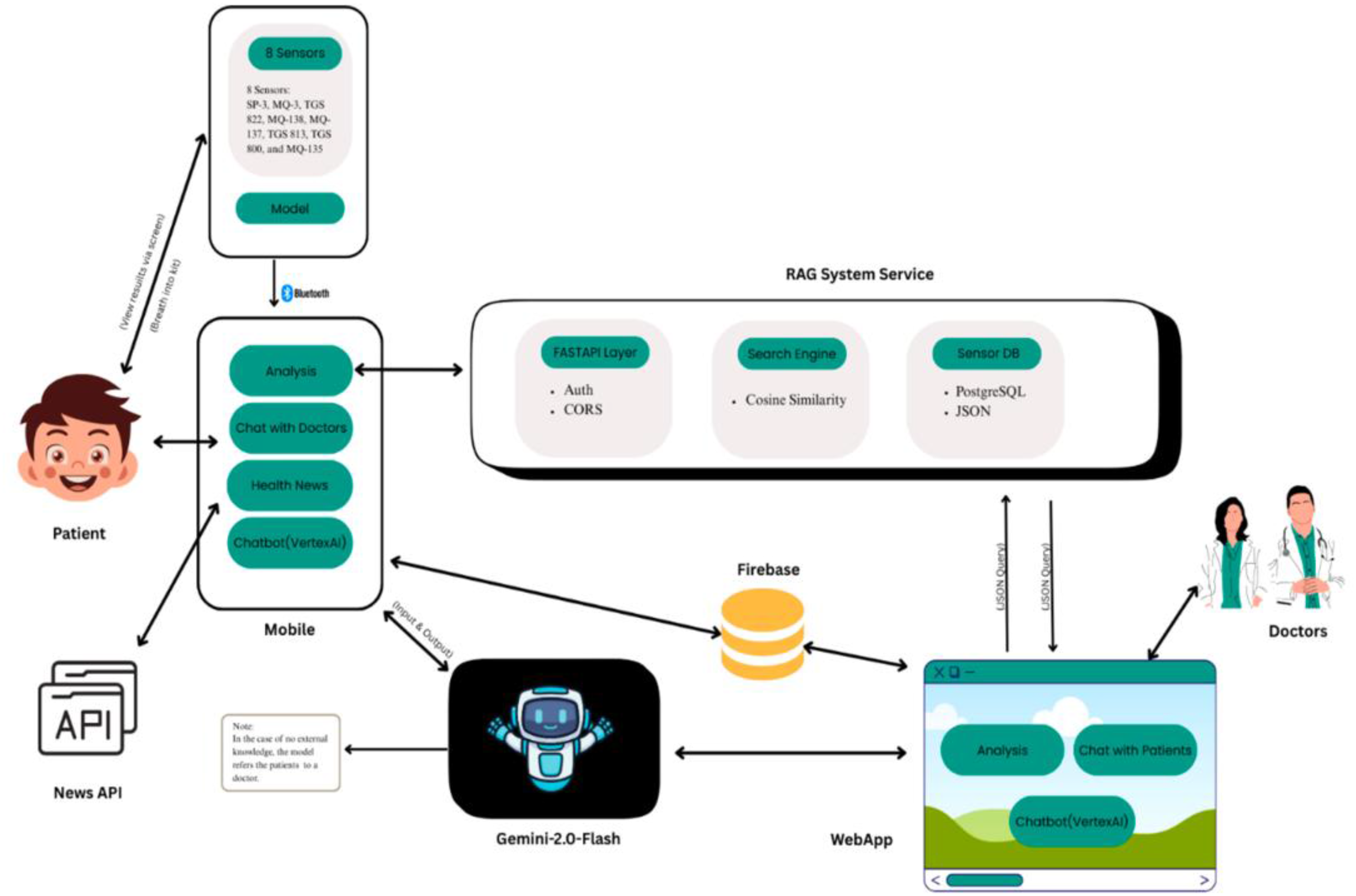
High-level architecture of mobile application system.
At the system's core is embedded middleware on the Raspberry Pi 5, which manages local data preprocessing, machine learning inference (Figure 4), BLE data transfer, and system status reporting. The Flutter-based mobile app serves as the primary patient interface, offering diagnostic results, BLE device connectivity, a symptom-tracking module, multilingual support (English, Twi, and Ewe), and a conversational chatbot for health education. For clinicians, the ReactJS dashboard provides access to diagnostic records, searchable patient registries, trend analysis, and downloadable PDF reports for documentation. Both applications are anchored by Firebase, which provides secure authentication, real-time database capabilities, and synchronized file storage.
Machine learning process flow.
The BBD device employs BLE for energy-efficient, secure wireless communication with the mobile application. Diagnostic data—structured in JSON format—are transmitted via custom BLE services, with the mobile app functioning as the central node that polls data at predefined intervals or in response to user input. For environments without reliable internet access, the app adopts an offline-first architecture, storing records locally in an SQLite database. Once connectivity is restored, the data synchronize asynchronously with Firebase Cloud Firestore, ensuring seamless updates without disrupting the user experience. Google Firebase was selected as the backend-as-a-service for its integration with mobile and web platforms, real-time database functionality, and strong security features. Firebase Firestore manages structured data such as patient profiles and diagnostic outputs, while Firebase Storage secures multimedia files including audio samples, clinical images, and signed consent forms. User authentication is implemented via Firebase Authentication, which supports email/password logins, two-factor authentication (2FA), and OAuth token exchange. Backend logic is extended through Firebase Cloud Functions, which automate processes such as role verification, alert generation, and query routing within the chatbot pipeline. Offline persistence and automatic synchronization enhance reliability for users in bandwidth-limited environments. Access is governed through role-based control with four user categories: Patients, clinicians, administrators, and the NLP-powered chatbot. Patients access their diagnostic results, chatbot, and symptom tracker; clinicians manage patient data and monitor progress; administrators oversee devices, permissions, and policy updates; and the chatbot is restricted to health education queries. Permissions are enforced through Firebase Authentication and Firestore's document-level security rules. To comply with HIPAA standards, all data are encrypted using AES-256 at rest and TLS 1.3 in transit, with audit trails maintained for all user interactions. Data collection follows explicit informed consent and ethical data minimization principles, with the platform formally approved under institutional ethics protocols. The chatbot system is built on a RAG architecture (Figure 5) to support patient education in low-literacy and multilingual contexts. User queries are first embedded into high-dimensional vectors using a BERT-based model, then matched against a knowledge base through cosine similarity. The most relevant documents are forwarded to the Gemini 2.0 Flash large language model, which generates context-aware, natural language responses. Middleware APIs provide token-based authentication, query logging, and caching to ensure secure, scalable, and traceable operation. In pilot evaluations, the chatbot demonstrated strong reliability, achieving an estimated factual accuracy rate of 83.6%, and was well-received as a trustworthy medium for patient engagement.

Pipeline of the custom retrieval-augmented generation (RAG) system.
Results
This section outlines the evaluation methodology from the development and validation of the BBD system for early detection of COPD. The experimental procedures were structured to assess multiple dimensions of system performance, including diagnostic accuracy, sensor effectiveness, computational efficiency during edge deployment, and overall user experience. Evaluation metrics were selected based on established standards in machine learning and digital health, encompassing accuracy, precision, recall, F1-score, inference latency, and usability scores.
Model configuration and baseline performance
A 1D-CNN was developed and trained using the TensorFlow/Keras framework to classify breath samples based on sensor data from eight input channels. The network architecture comprised two convolutional blocks, each consisting of a Conv1D layer followed by batch normalization and dropout layers for regularization, and concluded with fully connected dense layers. The final output layer utilized a softmax activation function to perform four-class classification across the labels: COPD, Healthy, Smoker, and Ambient Air. Model training was conducted over 300 epochs with a batch size of 32, using the Adam optimizer with a learning rate of 0.005. Early stopping was employed based on validation loss to mitigate overfitting. The dataset was partitioned into 64% for training, 16% for validation, and 20% for testing. To address class imbalance and ensure fair representation across all categories, a weighted loss function was applied during training. Class weights were computed using the formula wi = N/C⋅ni, where N denotes the total number of samples, C is the number of classes, and ni represents the sample count for the i-th class. The Performance of the models as reported in Table 4.
Evaluation of machine learning models for COPD classification.

COPD: chronic obstructive pulmonary disease; 1D-CNN: one-dimensional convolutional neural network.
Sensor configuration versus performance trade-off
To determine the most efficient sensor configuration, the diagnostic model was independently retrained and evaluated using various subsets of the original eight-sensor array, specifically configurations consisting of 1, 3, 5, and 7 sensors. Each configuration was benchmarked in terms of classification accuracy and inference latency to assess the trade-offs between diagnostic performance and computational efficiency as shown in Table 5.
Sensor count vs accuracy, and cost.
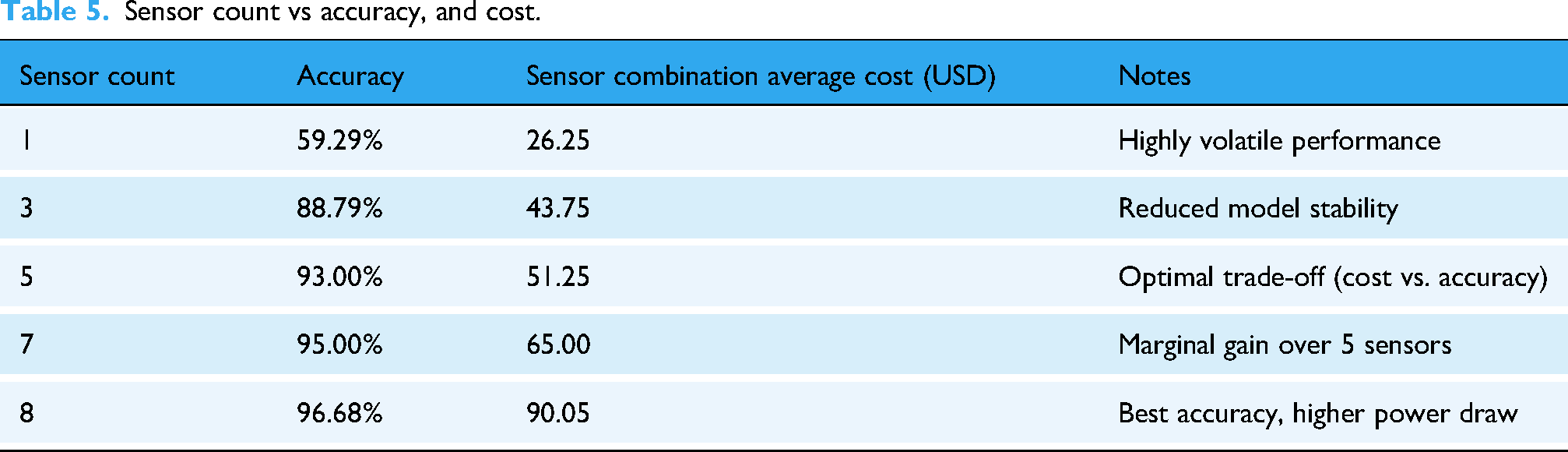
Results summary
See Table 5.
Cost efficiency insight
The five-sensor configuration retained approximately 98.6% of the diagnostic performance achieved with the full eight-sensor array, while simultaneously reducing hardware costs by an estimated 30% and decreasing inference computational load by 21%. These trade-offs position the five-sensor setup as a highly practical and cost-effective option for large-scale deployment in resource-constrained environments. The comparison in cost and benefit in Table 6 below helps to clearly show the influence of the cost of COPD diagnostic kit on industry workers and health facilities. Here, the long traditional procedures rob both the doctors and patients of precious working time which could have saved another patient or increase economic value for the patient at work. This ripple effect goes a long way to reduce labor input as COPD patients become weaker and incapable of continuing with work. This turn to rob African countries like Ghana of foreign exchanges as they are heavy on exploiting and refining natural resources, a double casualty reliving the tragic $568 M plus $32 M lost wages due to lost of 9.6 million working days for miners also in south Africa 23
Device types and its’ economic impact on patients.

COPD: chronic obstructive pulmonary disease; AI: artificial intelligence; VOC: volatile organic compound; MOS: metal oxide semiconductor; GC/MS: gas chromatography–mass spectrometry.
Edge AI deployment and system performance
The optimized model was converted into TensorFlow Lite format and deployed on the Raspberry Pi 5, where inference was executed locally through a custom Python script resulting in a performance shown in Table 7.
Edge deployment benchmarks.

These benchmarks validate the feasibility of deploying complex AI models at the edge, even without cloud assistance.
User experience and usability evaluation
A pilot usability study involving 31 participants—comprising 18 patients and 13 clinicians—was conducted at Ashesi University's Health Center and in surrounding community settings with results reported in Table 8.
System performance on scalability.

The chatbot interface was rated favorably for its multilingual support and clear explanations of results.
As reflected in participant feedback, the system's transparency and offline functionality were highly valued. One patient noted, “I trust the machine more when it explains why it said I have COPD” (Patient 003), highlighting the importance of explainability in fostering user confidence. A clinician emphasized its utility in clinical workflows, stating, “It helps me screen patients before I request further tests” (Clinician 009). Similarly, a community nurse remarked on the practical benefits of offline capability, stating, “I would use this if it doesn’t require internet all the time.”
Privacy and security evaluation
The system was subjected to a simulated breach audit and stress testing of its access control mechanisms to evaluate data security and system resilience. The results demonstrated robust protection against unauthorized access, with no data reads or writes occurring under scenarios involving misconfigured or spoofed credentials. Additionally, attempts to reverse-engineer or decompile the mobile application failed to expose encrypted data, confirming the effectiveness of the implemented end-to-end encryption protocols. The chatbot component also maintained a high standard of reliability, achieving an estimated factual accuracy rate of approximately 83.6% within the operational boundaries of the RAG context.
Discussion
Principal findings
This study demonstrates the feasibility of a low-cost, AI-powered BBD system for the early detection of COPD in resource-limited environments. The proposed solution achieved a diagnostic accuracy of 96.68% and 100% precision for COPD classification, with inference latency as low as 0.02 ms on a Raspberry Pi 5. Importantly, a five-sensor configuration preserved nearly 98% of diagnostic accuracy while reducing hardware costs by 30%. Usability testing confirmed high user acceptance, with a System Usability Score (SUS) of 86.3/100 and strong trust in the multilingual chatbot interface. Together, these findings indicate that BBD, when coupled with edge AI and human-centered design, can provide an affordable and scalable alternative to conventional diagnostic modalities in underserved regions.
Relation to existing literature
The findings build on and extend prior research in breathomics and E-nose technologies. Earlier work has established VOCs as reliable biomarkers for pulmonary diseases, but traditional methods such as GC/MS remain costly, non-portable, and unsuitable for point-of-care use in low-resource contexts.7,9 Low-cost sensor-based systems have been proposed as alternatives, yet their diagnostic accuracy has typically remained moderate. For instance, Binson et al. 10 reported 76.7% accuracy for COPD detection using a five-sensor Arduino-based E-nose, while 11 achieved 96.9% accuracy in controlled laboratory conditions using eight MOS sensors on a Cortex-M4 microcontroller. However, both systems relied heavily on synthetic or simulation data and lacked robust validation under real-world conditions. By contrast, the present study validates performance on both a public dataset 15 and a real-world dataset collected using the deployed prototype, demonstrating generalizability under practical conditions. The integration of augmentation strategies to simulate sensor drift and environmental variability further strengthens robustness, addressing limitations commonly cited in earlier E-nose studies.10,14 In addition, while previous research has largely focused on laboratory validation, this study advances the field by embedding the model into an edge device and evaluating usability among patients and clinicians, aligning with calls in recent reviews for real-world validation of AI-enabled diagnostic systems.13,24 The system's performance also compares favorably with existing AI-based diagnostic models for respiratory diseases. Recent studies employing machine learning for breathomics have reported accuracies ranging between 85% and 95% for COPD and lung cancer detection.9,12 Our CNN-based model not only exceeds these benchmarks but does so within the constraints of low-power edge deployment, achieving inference at <2.5 W. This efficiency is significant when contrasted with cloud-dependent approaches, 16 which may not be feasible in rural areas with poor connectivity.
Contribution to AI-enabled diagnostic innovation
The novelty of this study lies in its integration of three complementary innovations. First, it introduces a cost-efficient hardware optimization strategy, demonstrating that five sensors can achieve near-equivalent diagnostic performance compared with larger arrays. This trade-off directly addresses cost and power constraints noted in earlier work,10,14 making the system more practical for scale-up. Second, the system employs edge AI for real-time inference, eliminating dependence on continuous internet connectivity, a limitation frequently highlighted in global health settings.3,4 Third, the inclusion of a RAG-based multilingual chatbot represents an advance beyond diagnostics, enabling culturally relevant patient engagement and education. To the best of our knowledge, this is one of the first studies to combine breathomics, edge AI, and explainable natural language processing into a unified diagnostic platform.
Public health implications
The underdiagnosis of COPD remains a major public health concern in sub-Saharan Africa, where spirometry and radiographic imaging are scarce.1,3 The World Health Organization emphasizes the need for accessible, affordable, and context-adapted digital health solutions to reduce this diagnostic gap. The BBD system directly addresses these recommendations by combining non-invasive breath analysis with offline-first operability and multilingual support. Its potential deployment in community health centers, mobile clinics, and rural hospitals could decentralize respiratory disease screening, alleviating the burden on tertiary hospitals and enabling earlier interventions. Moreover, the low-cost design (estimated below USD 100) aligns with affordability targets for point-of-care devices in LMICs, suggesting strong scalability for public health programs.
Ethical, regulatory, and data privacy considerations
Adherence to HIPAA, HL7, and FHIR standards distinguishes this system from many prior E-nose prototypes, where data privacy and interoperability have been underexplored.10,14 The integration of role-based access control, AES-256 encryption, and consent-driven data sharing ensures compliance with international standards and prepares the system for integration into national health information infrastructures. By incorporating explainable AI outputs and transparency features in the chatbot, the system also addresses ethical concerns about patient trust in AI-based diagnostics, which have been highlighted in previous reviews.13,16
Limitations
Despite promising results, several limitations must be acknowledged. The MOS sensors used are broad-spectrum and lack specificity for individual COPD biomarkers, a limitation also noted in earlier sensor-based studies.10,14 While environmental compensation was applied, real-world deployments will introduce additional confounders such as smoke, dust, and perfumes, which may influence accuracy. The training dataset, though augmented, remains relatively small and geographically localized, limiting generalizability across populations. These constraints mirror challenges identified in previous breathomics research,7,12 underscoring the need for larger, multi-center datasets. Finally, while usability testing confirmed positive perceptions, formal clinical trials are needed before regulatory certification can be pursued.
Conclusion
Summary of contributions
This study developed and validated a low-cost, AI-powered BBD system for the early detection of COPD in resource-limited environments. By integrating an array of MOS sensors with a lightweight 1D-CNN deployed on a Raspberry Pi 5, the system achieved 96.68% accuracy and 100% precision for COPD classification, while maintaining ultra-low inference latency (0.02 ms) and power consumption (<2.5 W). Importantly, the five-sensor configuration preserved nearly all diagnostic performance while reducing hardware cost by 30%, underscoring the system's scalability for widespread use.
Beyond technical performance, the platform incorporated a multilingual mobile and web interface, supported by a RAG-based chatbot for patient engagement. Usability testing demonstrated strong acceptance, with a SUS of 86.3/100 and a trust rating of 4.4/5. Ethical and regulatory compliance were ensured through role-based access control, AES-256 encryption, and adherence to HIPAA and FHIR standards, positioning the system for integration into national healthcare infrastructures.
Implications
The results highlight the potential of BBD as a viable alternative to conventional COPD diagnostic tools, which remain inaccessible in many LMICs. The BBD system not only addresses the limitations of spirometry and imaging—cost, infrastructure dependence, and specialist requirements—but also extends current E-nose and breathomics research by demonstrating high diagnostic accuracy under real-world conditions. Its offline-first design and multilingual chatbot directly respond to the challenges of limited connectivity, linguistic diversity, and low health literacy, thereby aligning with WHO's call for inclusive and context-sensitive digital health solutions.
Future directions
Future research will prioritize large-scale clinical validation across diverse populations to establish the system's generalizability and secure regulatory approvals from appropriate. In parallel, efforts will focus on expanding the diagnostic scope beyond COPD to encompass other respiratory diseases, including asthma, tuberculosis, and lung cancer, through the integration of multimodal data sources such as cough acoustics and nanomaterial-enhanced sensors. To safeguard patient privacy while enabling continual model improvement, federated learning approaches will be explored, allowing decentralized on-device training without the need for centralized data sharing. Hardware optimization will also be pursued through printed circuit board miniaturization, incorporation of solar charging, and ruggedized enclosures to enhance portability and resilience in rural environments. Finally, successful deployment will require building community trust and capacity through tailored training programs for nurses and frontline health workers, participatory evaluation frameworks, and multilingual support resources to ensure usability and adoption in low-literacy and linguistically diverse populations.
Footnotes
Ethical considerations
This study was approved by Ashesi IRB Protocol Number: 2432026.
Author contributorship
EK: Conceptualization, implementation, and validation. TTM: Conceptualization, visualization, and implementation. JKA: Initial draft, methods, and validation. DEA-Y: Supervision, methods, resources, and final review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
