Abstract
Background
Type-2 diabetes (T2D) is largely preventable through sustained lifestyle change, yet healthcare systems face challenges in implementing and sustaining lifestyle interventions at scale. Digital health technologies (DHTs), offering personalized risk assessments, remote monitoring, and behavior change support, may support T2D prevention. However, the systemic role of DHTs within the T2D prevention ecosystem remains poorly understood. This study examines (RQ1) What stakeholder incentives are associated with prevention engagement among payers, providers, and individuals? (RQ2) What incentive patterns are associated with DHT adoption in T2D prevention? (RQ3) How is DHT adoption associated with value exchange among stakeholders in the T2D prevention ecosystem?
Methods
We conducted a systematic literature review to identify existing incentives in preventive care (RQ1). Business model data from leading DHT companies in T2D prevention (via PitchBook and Crunchbase) were analyzed to examine emerging incentive patterns (RQ2). We conducted expert interviews (N = 26) and synthesized findings using the e3-value framework to map stakeholder relationships (RQ3).
Results
Our findings show that financial and non-financial incentives for prevention are often temporally misaligned. Engagement in lifestyle-based prevention is linked to short-term rewards, health, and convenience benefits for individuals and long-term cost savings for payers. DHT adoption for T2D prevention is associated with three key patterns: enhancing personalization and convenience for individuals, supporting value-based payment models for payers, and improving workflow efficiency for providers.
Conclusions
DHTs may help align stakeholder incentives by promoting (1) sustained engagement in lifestyle prevention programs (i.e., continuous glucose monitoring with real-time dietary or activity feedback) and providing individuals with micro-rewards (i.e., for behavior change and improved clinical outcomes). These coordinated feedback loops could be embedded within (2) outcome-based reimbursements for payers and linked to (3) automated workflows to improve provider efficiency (i.e., risk stratification). Realizing this potential requires updated reimbursement models, integrated stakeholder coordination, and supportive policy frameworks.
Keywords
Introduction
Type-2 diabetes (T2D) presents a growing global health and economic challenge, with nearly USD 966 billion spent on diabetes care in 2021. 1 T2D is largely preventable through sustained changes in modifiable risk factors, such as diet and physical activity. A large body of work has demonstrated the effectiveness of lifestyle interventions in reducing T2D incidence and delaying disease progression and complications2–5; as well as cost-effectiveness for healthcare systems.2–5 Despite this evidence, healthcare systems remain predominantly structured around acute care delivery. Current reimbursement models primarily incentivize treatment of diagnosed T2D—such as medication management, hospitalizations, and complication-related care—rather than lifestyle-based prevention, that is, proactive efforts to reduce the onset of disease through sustained lifestyle modification (i.e., low-glycemic diet, physical activity, and behavioral counseling) among individuals at-risk of T2D.4,5 As a result, preventive lifestyle programs often face limited funding and long-term sustainability within healthcare systems.6,7
This disconnection between empirical evidence supporting lifestyle programs for T2D prevention and their limited implementation and sustainability in healthcare systems, in part, reflects a fundamental misalignment in stakeholder incentives. Here, we define stakeholder incentives as any factor (i.e., financial, non-financial) designed to influence the decision-making or behavior of individuals, providers, or payers towards engagement in preventive care. 8 In many healthcare systems, incentives for prevention vary widely among stakeholders and may conflict in practice. Providers often operate under reimbursement models that reward service volume, particularly acute and procedural care, rather than preventive engagement.9,10 Meanwhile, individuals are expected to sustain lifestyle changes with minimal ongoing support after an intervention ends, which may limit their motivation to sustain behavior change. Payers, including insurers and government funders, interested in prevention, need to finance long-term costs but face difficulty attributing savings directly to specific interventions or actors.11,12 Specifically, fee-for-service reimbursement models reward the quantity of acute interventions, such as hospitalizations, emergency visits, or medication adjustments, rather than continuous preventive care, including ongoing lifestyle counselling, regular physical activity promotion, or dietary support programs.9,10,13 This reactive care bias could be further reinforced by payer's cost-containment pressures and the absence of effective policies to align incentives across multiple stakeholders, including patients, providers, payers, and regulators.14,15 This fragmented operational and financial landscape makes T2D prevention challenging to fund, coordinate, or scale16–18. The result is systemic underinvestment in precaution, even when prevention is clinically effective and economically justified. 19
Digital health technologies (DHTs), defined by the U.S. Food and Drug Administration (FDA) as “computing platforms, connectivity, software, and sensors used for healthcare and related purposes,” 20 have gained increasing attention for continuous lifestyle monitoring, risk assessments, and behavioral lifestyle support. 21 In T2D prevention contexts, DHTs often integrate remote monitoring and behavioral interventions, delivered through smartphones, or wearables (i.e., continuous glucose monitors (CGMs) or smartwatches) to support glycemic control—frequently leveraging advanced data analytics. 22 Research has examined factors influencing the adoption of DHTs by patients and healthcare providers, with studies identifying barriers, including technological interoperability and fragmented systems, as major impediments to widespread implementation.23,24
While adoption barriers at the individual and provider level have been documented,13–15 stakeholder incentives across the broader healthcare ecosystem,
25
comprising individuals (i.e., patients or individuals at-risk of T2D), providers, and payers, remains unclear.26,27 Building on recent studies that have begun to conceptualize T2D prevention from a system-level perspective28–30 and describe ecosystems in T2D management, this study aims to map stakeholder incentives for prevention and identify incentive patterns associated with DHT use
Theoretical background
Prevention as an ecosystem
Noncommunicable, chronic disease prevention often relies on a coordinated network of interdependent stakeholders, including individuals, healthcare providers, payers, and regulatory bodies. These actors collectively influence key underlying processes, including risk screening, the delivery of lifestyle programs, and their potential reimbursement.31–33 Ecosystem theory conceptualizes this network not as a linear sequence of care delivery, but as a dynamic system in which value is co-created through shared infrastructure and ongoing value exchange among stakeholders. 34 To operationalize T2D prevention from an ecosystem perspective, we adopted the e3-value ontology, a modeling framework that defines an ecosystem through three core components: actors, value exchanges, and value interfaces. 35 Actors represent distinct organizational or individual stakeholders.36,37 Value exchanges involve the transfer of goods, services, or information between two or more parties. Value interfaces define how exchanges are governed, that is, defining the transactional conditions under which exchanges occur. Applied to T2D prevention, the e3-value ontology allows us to describe associations between key stakeholders from a systemic (vs. single stakeholder) perspective, 25 with potential to identify stakeholder relationships and gaps in prevention pathways.
Methods
Research design
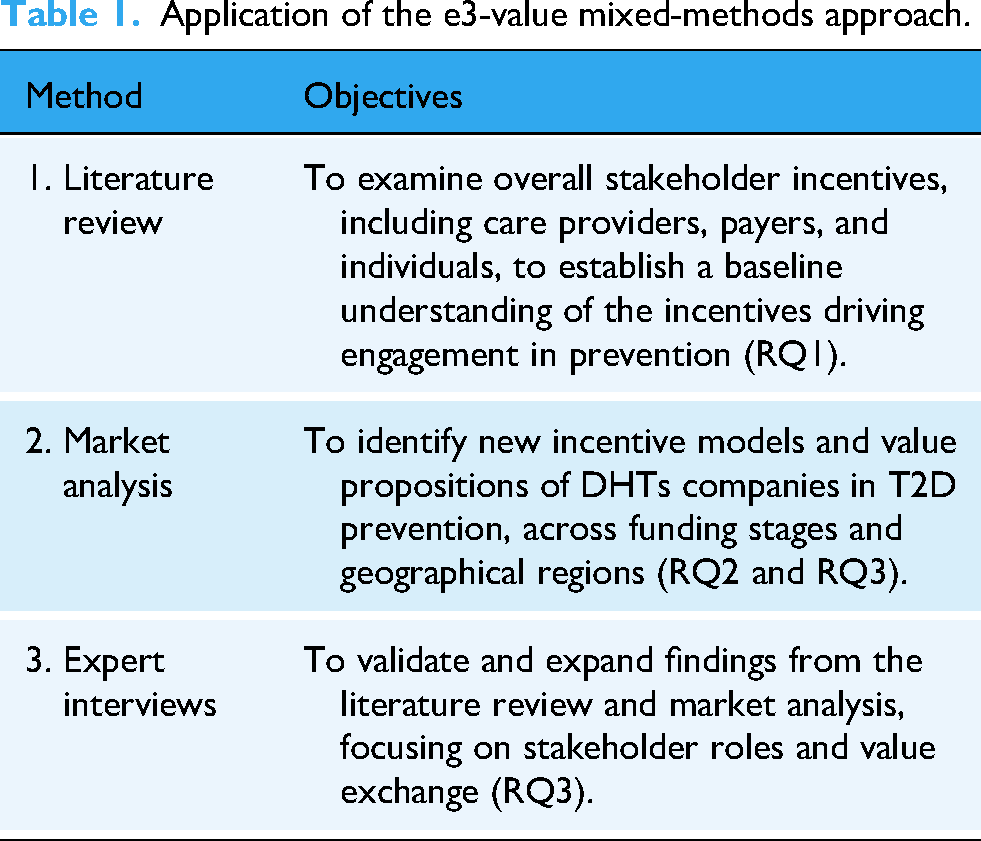
We adopted a mixed-methods design structured around the three key research questions. We integrated (1) a systematic literature review to identify existing incentives across payers, providers, and individuals (RQ1), (2) a market analysis of leading DHT companies to identify emerging incentive patterns linked to DHTs (RQ2), and (3) expert interviews to refine and validate incentive structures and explore stakeholder value exchanges (RQ3). The e3-value methodology35,36 served as the underlying modeling framework. Drawing on prior applications in digital health and T2D management,28,29 we used e3-value to map stakeholder incentives and highlight new patterns associated with the use of DHTs. Figure 1 illustrates our step-by-step application of the e3-value method, and Table 1 outlines each phase of the method, triangulating insights through a systematic literature review, market analysis, and expert interviews.28,29

A mixed-methods approach for investigating associations between stakeholder incentives and DHTs in T2D prevention.
Application of the e3-value mixed-methods approach.
Data collection
Systematic literature review
To examine how stakeholder incentives are associated with engagement in prevention among individuals, providers, and payers? (RQ1) We conducted a systematic literature review following the PRISMA 2020 guidelines, see Figure A1. 38 The review targeted peer-reviewed studies published between 2010 and 2023, a period previously marked by the onset of DHT adoption in chronic disease management and prevention.39,40 We searched five academic databases, ScienceDirect, EBSCOhost, Web of Science, IEEE Xplore, and the ACM Digital Library. We used the following search terms: search terms “business model” or “health ecosystem with keyword-based constructs representing populations, interventions, and incentive mechanisms related to preventive care, chronic disease, and digital health, including “preventive care,” “non-communicable disease,” “chronic disease,” “digital health,” and “electronic health.” Boolean operators and field restrictions were adapted to each database. The search strategy is detailed in Table A1. We performed a backward search to screen for additional relevant references cited in the identified articles. 39 Three researchers (Wasu Mekniran, Victoire Stalder, Wilma Diethelm) independently screened all titles and abstracts, reconciling discrepancies through discussion to ensure conceptual consistency. Given the qualitative focus of this review, thematic saturation was considered achieved when successive interviews no longer yielded new incentive categories or mechanisms, rather than based on statistical agreement metrics.
Studies were included if they (1) focused on stakeholder incentives in the abstract, (2) mentioned preventive care or DHTs within chronic disease management in the abstract, and (3) were peer-reviewed and published in English between 2010 and 2023. From each included study, we extracted publication characteristics, study design, stakeholder group, incentive type (financial or non-financial), and the reported mechanism linking incentives to prevention engagement. Extracted data were synthesized using thematic aggregation to derive recurring incentive categories. The literature screening and reviewing process was conducted using Covidence, 41 a web-based collaboration platform that streamlines the production of systematic literature reviews.
Market analysis
To examine the incentives associated with DHT adoption in T2D prevention (RQ2), we conducted a market analysis of DHT companies active in T2D prevention. This approach is critical as the business models of these firms inherently reflect the specific value propositions and exchange mechanisms designed to incentivize engagement from individuals, providers, and payers.30,42 Company-level data were retrieved and screened from PitchBook, a global private capital database, 43 on April 16, 2024, and the funding status of screened companies was updated on October 10, 2025. The initial search returned 602 companies, filtered using the following industry categories: “healthcare services,” “outpatient care,” and “digital health,” alongside the following keywords: including “diabetes” and “metabolic health.” Detailed search categories and search terms are presented in Table 2.
Search filter and search terms for the market analysis.

To capture companies with distinguishable market traction and scalability, we selected the top 100 companies ranked by total funding in PitchBook. This threshold was chosen to prioritize firms with publicly traceable operations and validated investment activity, thereby enhancing the comparability of business-model data while acknowledging potential survivorship bias. Three entries were excluded due to missing data, leaving 97 companies for full screening. Inclusion criteria comprised: explicit engagement in T2D prevention, provision of a digital health component within the core offering, and sufficient documentation in English to allow systematic business model assessment. Exclusion criteria included seed-stage companies. A total of 32 companies met all inclusion requirements. The selection and screening workflow is depicted in Figure A2.
A structured content analysis was conducted using the value proposition dimension of Foss and Saebi's business model innovation framework, see Table A2 for the codebook. 44 Two researchers (Wasu Mekniran, Victoire Stalder) independently reviewed PitchBook profiles, Crunchbase entries, 45 and official websites to extract three elements: (1) core market segment (i.e., diagnostics, therapeutics), (2) target customer (i.e., employer, payer, diagnostic center), and (3) value object delivered (i.e., intervention, data, convenience). Discrepancies were resolved by a third researcher (Wilma Diethelm). These elements were synthesized to identify recurring incentive logics, which informed the subsequent ecosystem modeling using the e3-value approach. 37
Expert interviews
To validate the stakeholder incentives identified through the systematic literature review and market analysis, we conducted 26 semi-structured interviews with experts directly involved in the delivery, financing, or governance of T2D prevention in their respective local healthcare setups. Participants represented diverse local healthcare systems across 13 countries, primarily from Europe (n = 16; Switzerland n = 11, Hungary n = 1, the Netherlands n = 1, France n = 1, Poland n = 1, and the United Kingdom n = 1), followed by Asia-Pacific (n = 5; Singapore n = 1, Indonesia n = 1, Thailand n = 1, the Philippines n = 1, and Pakistan n = 1), Australia (n = 1), and the Middle East/Africa (n = 1; Egypt n = 1). Participants represent seven stakeholder groups, including payers (n = 10), healthcare providers (n = 7), investors or consultants (n = 3), policy leaders (n = 2), manufacturers (n = 2), academic researchers (n = 1), and the support community (n = 1); as detailed in Table A3. Interviews were conducted via video call between January and June 2024 (mean duration, 32 min), guided by a standardized protocol that included interview questions aligned with RQ1 and RQ2, as shown in Table 3. The interview protocol consisted of nine questions grouped into three analytical domains: (1) preventive care incentives, (2) ecosystem dynamics, and (3) business model innovation.
Interview themes, questions, codes, and definitions.

Interview transcripts were coded in ATLAS.ti 46 tool using a deductive coding framework, drawing on the Business Model Innovation (BMI) concept. BMI examines how business models are transformed in response to internal and external drivers 44 and business model patterns. 47 Codes covered antecedents, moderators, outcomes, and business model elements, as defined in the coding framework in Table 3. Two researchers (Wasu Mekniran, Wilma Diethelm) coded the interview transcripts independently, with discrepancies resolved by a third researcher (Victoire Stalder). Thematic saturation was reached after 26 interviews, see Table A3. This study was granted a formal Letter of Exemption by the Ethics Committee of the University of St Gallen (September 30, 2025), under institutional guidelines. All interview participants provided verbal informed consent prior to participation. No compensation was offered, and all transcripts were anonymized and securely stored.
The systematic literature review and market analysis relied exclusively on publicly available data and therefore did not require ethics approval. From the included studies, we identified 39 unique incentives driving engagement in prevention across stakeholders (17 financial and 22 non-financial). For instance, financial incentives included performance-based payments for providers and premium reductions for individuals, while non-financial incentives encompassed enhanced professional reputation and access to personalized health coaching.
Results
RQ1: Incentives associated with engagement in prevention among payers, providers, and individuals
From these included studies, we identified a variety of different incentives associated with prevention engagement across individuals, payers, and providers in the literature (39 unique incentives; 17 financial and 22 non-financial in total). Examples of financial incentives included performance-based payments for providers and premium reductions for individuals, while non-financial incentives included enhanced health outcomes, recognition, and access to personalized lifestyle coaching. Drawing on prior value exchange evaluation studies,29,35 we further categorized incentives by temporal orientation: short-term incentives were defined as benefits realized within six months, and long-term incentives as benefits accruing beyond six months. Among individuals, the majority of identified incentives (8 of 9) were short-term, emphasizing immediate, tangible benefits such as short-term health improvements, financial rewards, convenience, or personalized health feedback. In contrast, the majority (4 of 5) payer incentives and a large number (9 of 16) of provider incentives were long-term, focusing on population health outcomes, operational efficiency, and cost containment.
This temporal divergence reflects a persistent misalignment of incentive horizons and perceived value: individuals prioritize immediate personal gains, whereas institutions emphasize delayed systemic benefits. For example, individuals are likely to be more responsive to short-term health benefits tied to prevention programs, such as weight loss or improved glucose metrics, while providers and payers value long-term improvements in population health and cost savings, that is, related to T2D incidence. Table 4 summarizes the typology and key points of divergence.
Typology of incentives in prevention, categorized by stakeholder, financial/non-financial type, and temporal horizon.
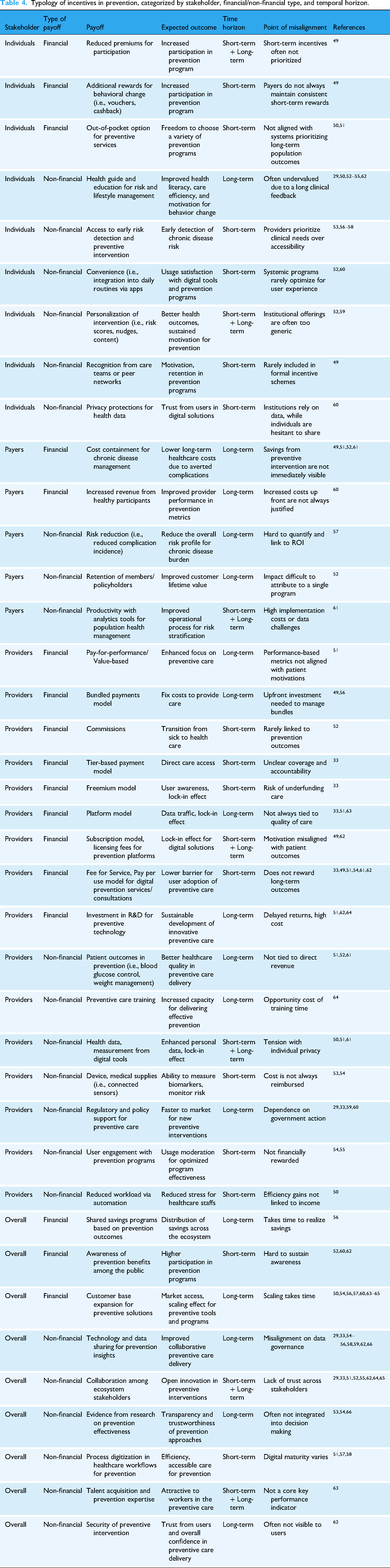
Individuals
Our literature review suggests that both financial and non-financial incentives are important factors to motivate individuals to engage with lifestyle prevention programs. Financial incentives, specifically, reduced insurance premiums, have been shown to increase short-term and sustained participation in preventive lifestyle programs by lowering direct monthly costs in healthcare systems. 48 Short-term incentives such as vouchers, cashback, or small monetary rewards have also been shown to encourage short-term behavior change and initial engagement with lifestyle interventions. 49 Additionally, the option to pay out of pocket allows individuals to access a broader range of prevention services, such as specialized dietary coaching or personalized exercise programs, thereby promoting consumer autonomy.50,51
Our review suggests that non-financial incentives are similarly important for individuals. Access to lifestyle guidance, delivered through digital coaching, personalized reports, or community seminars, has been shown to improve health literacy and motivation for lifestyle behavior change.50,52–55 Further, personalization of interventions through tailored risk scores, nudges, or content delivery has also been found to foster sustained motivation and the likelihood of behavior change.53,56–59 Convenience, such as integrating coaching, reminders, or glucose feedback into existing mobile routines (i.e., via smartwatch notifications or calendar-linked nudges), is also a significant driver of engagement with preventive interventions.52,60 Additionally, support and recognition from care teams and peer networks can improve user adherence and satisfaction with prevention programs. 49
Payers
Payers, including both public and private insurers, have financial incentives to invest in prevention, as shared savings programs may allow payers to fund preventive interventions and later benefit from avoided costs associated with acute disease management.49,51,52,61 Such programs often aim to tie medical cost savings to the performance metrics of prevention programs, thereby aligning financial incentives with providers. In certain healthcare contexts, such as revenue opportunities for payers, bundling preventive services with supplementary insurance offerings, such as wellness, dental, or vision packages, with the potential to enhance customer acquisition and retention. 60
Non-financial incentives include reduced portfolio risk across population segments. By lowering the prevalence of chronic conditions, payers may experience lower overall claims, long-term savings, lower claim volatility, and improved actuarial predictability. 57 Customer retention is another strategic benefit: policyholders are more likely to remain loyal to insurers that offer proactive, value-added services. 52 With the help of advanced analytics and segmentation tools, payers may also support more efficient customer targeting, resource allocation, and preventive service personalization, 61 reinforcing long-term customer value and operational sustainability.
Providers
Healthcare providers, including general practitioners, specialists, and integrated care teams, play a crucial role in enabling preventive care. Pay-for-performance models often reward measurable outcomes, such as weight loss, medication adherence, or clinical improvements (i.e., glycemic control), aligning revenue with health improvements. 51 By participating in bundled or subscription-based pay-for-performance models, providers can be incentivized to perform early risk assessments, monitor lifestyle behaviors, and coordinate care effectively to ensure patients access to appropriate lifestyle and clinical decision support.49,56,62 While fee-for-service structures are still common, especially in systems without mature prevention reimbursement, pay-for-performance models can enable providers to monetize preventive interventions.33,48 For example, digital business models, such as subscription-based or freemium apps, may incentivize healthcare providers to offer preventive care and improve health outcomes33,51,63; thereby aligning immediate clinical action with long-term health outcomes.
Non-financial incentives encourage healthcare providers to engage in preventive care by supporting both professional effectiveness and patient impact. Providers gain satisfaction from improving patient outcomes and strengthening relationships, while early interventions reduce administrative burden and improve clinical efficiency.8,61 Access to real-time biometric and behavioral data further enhances decision-making, enabling providers to triage visits effectively and allocate time and resources where they are most needed. Training, continuing education, and participation in quality improvement initiatives help providers adopt evidence-based practices and advance their professional development. 64 Finally, involvement in collaborative research programs, recognition from peers, leadership opportunities within care teams, and contributions to organizational population health goals reinforce providers’ sense of expertise and long-term impact, motivating sustained engagement in preventive care.
Incentive misalignment
Despite growing interest in prevention, misalignments persist across stakeholders along both financial and temporal dimensions. Our results suggest that individuals are primarily motivated to engage with prevention through short-term, tangible benefits such as financial rewards for sustaining healthy behaviors (i.e., vouchers or app-based cashback), ease of use of lifestyle interventions and low burden (i.e., frictionless app onboarding), and access to personalized feedback (i.e., real-time glucose trends, tailored lifestyle advice via app notifications and or/ digital coaching), and short-term or immediate tangible health improvements.49,52,59 However, these short-term individual-level incentives are often undervalued in system-level designs, which prioritize long-term population-level outcomes, such as cost avoidance from avoided hospitalizations or risk reduction for population-level indicators like T2D incidence.51,60,61
Misalignments also occur in data governance. For example, providers may use risk assessment scores derived from EHR data to stratify patients into personalized prevention pathways, while insurers may use engagement data from prevention programs or claims analytics to provide financial rewards. 48 Yet individuals are often reluctant to share their personal health data without strong assurances of privacy and transparency. 60 Moreover, while payers and providers may aim to optimize for efficiency and scalability, individuals prioritize trust and convenience. Relatedly, there remains a disconnect between individual and population-level incentives. Short-term individual incentives (such as motivating individuals to engage in prevention with cashback rewards in exchange for daily steps) remain disconnected from long-term systemic returns (i.e., evidence of a reduction in population-level chronic disease incidence as a function of step count), potentially undermining the systemic sustainability of lifestyle prevention programs.
RQ2: Incentive patterns and adoption of DHTs in T2D prevention
The included sample of selected top-funded companies reflects a diverse set of DHT providers founded between 2008 and 2022, with the most active founding years between 2010 and 2019. A summary of the founding years is presented in Table 5. Funding status search and classifications were distributed across early-stage venture (n = 15), private equity (n = 7), late-stage venture (n = 4), post-IPO (n = 3), M&A (n = 1), seed (n = 1), and closed (n = 1); as of October 2025. Headquarter locations show that most included companies were based in the United States (n = 22), followed by India (n = 3), with the remainder distributed across seven other countries, including Australia (n = 1), Finland (n = 1), France (n = 1), Germany (n = 1), New Zealand (n = 1), Switzerland (n = 1), and United Kingdom (n = 1).
Companies included in the market analysis: country, funding status, market segment, value object, and target customer.
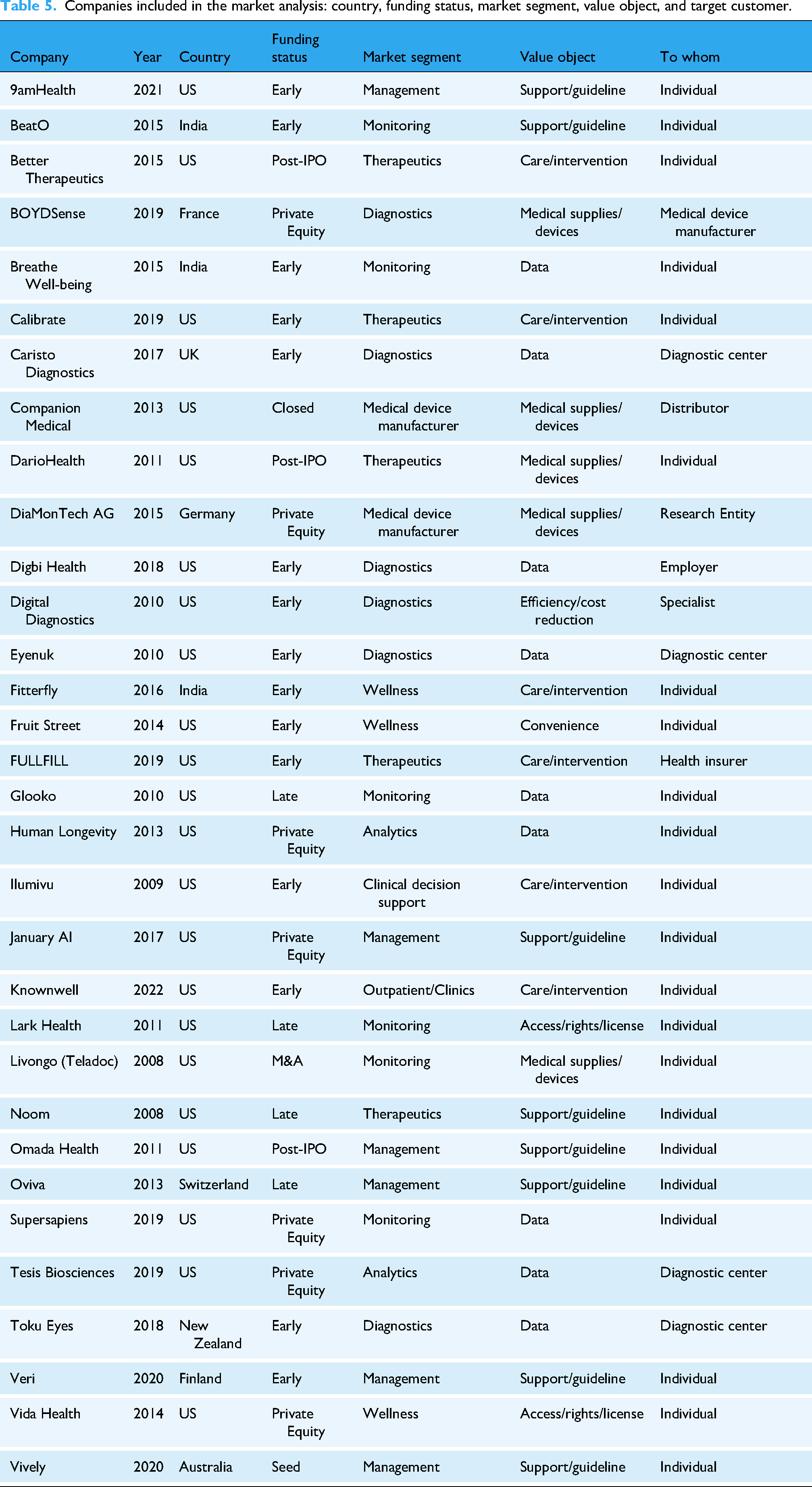
Based on the market analysis, we identified three dominant incentive patterns associated with the use of DHTs in the T2D prevention ecosystem: (1) Among individuals, DHTs are associated with enhance individual access to personalized health insights, (i.e., biometric T2D risk screening and recommendations), and motivation to engage in lifestyle interventions (i.e., digital coaching, wearable feedback). Among payers (2), DHTs are associated with support value-based payment for payers, via outcome-linked reimbursement and engagement-based pricing. Among payers (3), DHTs are associated with provider organizational workflow efficiency, that is, through remote continuous monitoring, care team coordination, and patient stratification and triage.
Individuals: Sustained engagement in digital prevention programs through personalized health insights and continuous monitoring in daily life
Of the 32 companies analyzed, 23 companies target individuals with value propositions centered on personalized guidance for T2D prevention and management, delivered through digital coaching, remote monitoring, and mobile app platforms. A majority of these services fall into the care guiding (n = 8), intervention (n = 6), or monitoring (n = 6) market segments. Companies like Noom (2008), Omada Health (2011), and Oviva (2013) deliver coaching programs designed to sustain healthy behavior change, while platforms like Supersapiens (2019) and Glooko (2010) offer continuous glucose monitoring insights with risk assessments. January AI (2017) and Lark Health (2011) embed behavioral nudges and AI-driven coaching into consumer apps, aiming to sustain engagement over time.
These DHT-based solutions aim to align short-term individual motivations (convenience, feedback, community) with long-term outcomes (reducing T2D incidence, improving app use adherence). The resulting generation of continuous, individual-level health and engagement data may provide a new basis for realigning incentives across the T2D prevention ecosystem. For instance, providers may leverage individual-level health and DHT engagement data to gain actionable insights for personalized lifestyle recommendations and decision support, while payers may leverage individual-level data for risk stratification, to measure evidence for cost containment, and to develop value-based contracts for prevention. Yet, while in theory promising, health and cost improvements linked to DHT use were rarely reported in our expert interviews, underscoring the lack of available longitudinal health (i.e., glycemic) and cost measures in combination.
Payers: Healthcare cost containment through value-based models
Only two companies explicitly target payers or employers, but this represents a growing strategic shift, as companies such as Lark Health (2011), Livongo (2008), Omada Health (2011), and Oviva (2013) are providing reimbursable T2D prevention programs backed by payers in their respective healthcare systems. In the case of FILLFILL (2019), for example, payment to payers is tied to reductions in glycated hemoglobin (HbA1c)—a clinical measure of long-term glycemic control and a key indicator of T2D progression. 73 In principle, such models may help align incentives across stakeholders: subsidized access to preventive programs may support uptake among individuals; outcome-linked reimbursement may promote performance incentives for providers; and payers may benefit from long-term cost containment, supported by T2D prevention programs. However, our analysis reveals that few companies have demonstrated evidence for sustained real-world cost savings at scale (i.e., BeatO, Lark Health, Noom, Omada Health). A persistent gap remains between the long-term (i.e., over 15–20 years) realization of prevention-related cost-saving and the typical short-term (i.e., 1–5 years) contracting cycles of many payers. Moreover, evidence around cost-effectiveness and clarity regarding reimbursement mechanisms for DHT-supported programs remains limited.
Providers: Workflow integration and diagnostic support
Approximately one-third of the companies in our sample focus on supporting clinicians and diagnostic providers in optimizing their workflows to enhance T2D screening, diagnosis, and related complications. Companies like Caristo Diagnostics (2018), Eyenuk (2010), and BOYDSense (2015) offer AI-enhanced screening and early detection of T2D and its complications, aiming to streamline clinical workflows through automated data capture and analysis presented in integrated provider and user-facing dashboards. For example, Digital Diagnostics (2010) incorporates clinical decision support into practice, potentially reducing missed diagnoses and enhancing efficiency. However, based on our interviews, providers remain cautious about widespread DHT adoption, citing integration costs, unclear liability pathways, and a lack of reimbursement as major deterrents to their integration into routine practice and scalable implementation.60,67,68
While these DHT-based solutions offer automation benefits, their adoption depends heavily on alignment with clinical incentives and workflow compatibility. When successfully integrated, provider-focused DHTs may potentially enhance clinical efficiency, leading to significant organizational benefits. For instance, automated retinal screening for diabetic retinopathy, as offered by Eyenuk, can reduce the need for specialized ophthalmologist visits, potentially saving costs per screening in some settings by freeing up physician time and accelerating time-to diagnosis.
RQ3: Associations between DHT adoption and value flows among stakeholders in the T2D prevention ecosystems
Combining interviews and insights from the literature review, spanning 1005 coded excerpts derived from 14 h of expert interviews in Table A3 and Table A4, 32 DHT companies, and 21 peer-reviewed articles, we identified three interrelated mechanisms through which DHTs are associated with potentially changing value flows and stakeholder relationships in the T2D prevention ecosystem.
First, (A) DHTs may enable the emergence of new collaborative networks by expanding the roles of non-traditional providers, such as digital health coaches delivering personalized lifestyle interventions. Second, (B) digital platforms make it increasingly possible to automatically link payments to engagement or preventive outcomes through data-driven, conditional reimbursement mechanisms, potentially changing financial relationships in the T2D prevention ecosystem. Third, (C) accountability among individuals, providers, and payers may also shift as DHT-based models challenge existing regulatory and governance frameworks and redistribute responsibility. For example, individuals become more accountable for self-monitoring and lifestyle management through apps or wearable devices that track activity, diet, or glucose. Providers may face increased accountability via dashboards and analytics that show patient engagement and adherence, allowing performance monitoring at the individual or population level.
Furthermore, payers may be increasingly held accountable for supporting effective preventive programs through outcome- or engagement-linked reimbursements, such as paying only when users reach target risk thresholds or achieve measurable improvements. Figure 2 depicts how DHTs may serve as enabling infrastructure for a transition toward more integrated, data-driven T2D prevention. However, despite the potential structural change, the long-term clinical and economic benefits of these transformations remain to be validated.

Diagram illustrating value exchange between key stakeholders in the digital diabetes prevention ecosystem, highlighting (3) potential DHT-enabled transformations.
Emerging collaborative networks and the expanding role of non-traditional providers
DHTs are associated with shifts in how preventive care is organized, with non-traditional providers increasingly occupying roles that have historically been concentrated within clinical settings. Consumer-oriented digital health platforms, in particular, are observed to operate across both wellness and clinical domains. Hybrid platforms such as January AI (2017), Digbi Health (2018), and Caristo Diagnostics (2017) illustrate this pattern, positioning themselves at the intersection of self-management and clinical decision support and interacting with both providers and payers. Supersapiens (2019), for example, integrates real-time glucose tracking with performance analytics to serve both personal health optimization and clinical use cases, functioning as an intermediary between individuals and providers. These developments coincide with broader institutional adjustments across the healthcare landscape. Established actors appear to be redefining their roles in response to the growing presence of DHTs.
Medical associations, for instance, are positioning themselves as trust brokers by evaluating and endorsing digital tools. As one association founder explained, “We would almost be a trust layer … vet a product and say this is good based on research” (ID 1). Primary care physicians also describe expanded activities beyond traditional clinical encounters, including public education and digital outreach. One community-based physician noted, “My initiative is to educate the people through social media as my responsibility” (ID 2). Some academic researchers similarly report increased involvement in applied, community-based DHT prevention programs, such as co-developing strategic frameworks and health literacy materials with schools and public health institutions (ID 3). Insurers are likewise reported to be broadening their scope beyond claims administration. Through investments in DHT startups and partnerships with technology firms, insurers articulate aspirations to engage more directly in prevention and care coordination. As one insurer explained, “We want to play a role as a healthcare partner for our customers,” while also noting the growing influence of technology companies that “are super active with their platforms and collect valuable prevention data” (ID 6). In parallel, wellness platforms and workplace coaching programs (ID 9) are cited as mechanisms through which insurers participate in more integrated, service-oriented prevention arrangements.
Potential realignment of financial ties via programmable incentive architectures
DHTs are associated with changes in how preventive care is financed by embedding near-real-time behavioral and physiological data into adaptive incentive systems. Unlike traditional reimbursement models that reward one-off, reactive treatments, emerging DHT-enabled models promote continuous engagement and outcomes-based payments. Platforms such as BeatO (2015), Glooko (2010), and Lark Health (2011) exemplify this transformation. These DHT-based example companies use biometric feedback loops—integrating glycemic control metrics, lifestyle patterns (such as physical activity), and medication adherence—combined with AI-driven behavioral nudges and real-time coaching to guide users toward healthier behaviors. This approach aims to replace static benefit structures with dynamic financial models where outcomes and payments are closely linked. FULLFILL (2019), for instance, ties its revenue to improvements in glycemic markers, while Veri (2020) and Supersapiens (2019) monetize real-time adherence, aiming to turn behavioral compliance into a billable asset.
Insurers are beginning to adopt similar models. For example, some insurers in Belgium and Switzerland currently offer digital programs that reward preventive behavior with micro-incentives, as one insurer described: “We promote and reward healthy behavior through our app” (ID 6). Others link provider payments to patient adherence: “We remunerate the doctor … if he makes sure that some people adhere to the standards” (ID 7). Additional strategies include loyalty-based engagement: “If you use this app, you collect points … discounts, vouchers” (ID 9), and ecosystem positioning “We now shift towards being a companion for all aspects of health” (ID 8). However, as one CIO noted, “Investments in prevention typically do not take place where the benefits accrue” (ID 9), highlighting a potential structural misalignment: payers funding preventive programs (i.e., such as short-term cashback programs) may not directly capture the long-term returns in investment, as individuals may switch private insurance providers in different healthcare contexts, such as in Swiss context.
New actors are also entering the T2D DHT prevention ecosystem e by experimenting with decentralized incentive architectures. For example, a pharmacist-led longevity platform uses blockchain to reward users with utility tokens for engaging in healthy behaviors, such as a low-glycemic diet and sharing their meals: “Upload your meals for today. You earn a few utility tokens” (Content Strategist, ID 23). Yet, two of our interviewees further suggested that technical innovation alone is not enough for sustainable T2D prevention. As one digital health entrepreneur emphasized, “For prevention to really make an impact, we need structural and legislative changes,” warning that “poor data leads to inconclusive results” (ID 26). Others point to scalability barriers: “Only the bigger teams … with a lot of funding … can succeed in this ecosystem” (DHT Developer, ID 5), underscoring that the promise of data-driven incentive models depends not just on technological feasibility, but on institutional coordination and equitable access, the bigger teams … with a lot of funding … can succeed in this ecosystem” (DHT Developer, ID 5).
Potential to reshape accountability: Regulatory tensions in the digital health era
DHTs are evolving faster than the regulatory systems designed to oversee them.8,68,69 By integrating behavioral and physiological data with AI-driven feedback loops, some DHT tools are increasingly challenging established frameworks for classifying medical devices, reimbursement pathways, and data governance legislation. While some platforms, such as Better Therapeutics (2015) and Noom (2008), have aligned themselves with behavioral CPT codes to gain reimbursement in the U.S., more complex hybrid models, like Companion Medical (2013) and BOYDSense (2019), often fall outside conventional regulatory categories. Their integration of sensing, behavior modification, and predictive analytics resists straightforward mapping onto existing billing and certification taxonomies.
These tensions play out across multiple regulatory layers, including platform access, coding and reimbursement, and data use restrictions. Developers often encounter commercial barriers before they ever reach clinical regulation: “Apple and Google Play store policies constrain smaller teams,” one engineer explained, adding that “only bigger teams with funding succeed” (DHT Developer, ID 5). Insurers, too, face legal and structural constraints: “We are not allowed to use our customer data for marketing” (Insurer, ID 6), and “Prevention is not our mandate. But the potential is there. It is just blocked by law and credibility gaps” (Insurer, ID 12). Even diagnostic laboratories face rigid pricing models that disincentivize innovation: “You can’t charge more than standard, even with better [technological tools]” (Diagnostics, ID 17), illustrating how outdated tariff structures penalize performance improvements. These governance challenges extend into the investment landscape for T2D prevention.
To avoid regulatory scrutiny, investors tend to favor “partly software” models that bypass FDA pathways (Investor, ID 24). Yet this risk aversion has structural consequences: the most scalable DHTs for T2D prevention may be the least embedded in traditional clinical practice. Our data suggests that entrepreneurs seeking to establish sustainable preventive health infrastructure may face a paradox. As one founder put it, “Prevention eliminates that recurring revenue” (Digital Health Entrepreneur, ID 26), suggesting that efforts to integrate DHT-based solutions in routine medical practice may be at odds with short-term financial incentives. Unless regulatory and reimbursement systems evolve alongside DHT innovation, high-impact technologies risk remaining marginal to formal healthcare delivery. Our analyses suggest that of the many of the identified DHTs with potential to advance T2D prevention, often operate at the edges of current regulatory and reimbursement frameworks (i.e., in standalone patient-facing wellness contexts), potentially highlighting the need for more adaptive governance models that can support innovation while ensuring accountability, patient safety, and value.
Discussion
Despite well-established scientific evidence for the prevention of T2D through sustained lifestyle change,2–5 preventive programs are seldom integrated and sustained within traditional healthcare systems. How is the uptake and systemic implementation of prevention associated with stakeholder incentives, specifically between individuals, payers, and providers? Grounded in an ecosystem theory8,34 and the e3-value ontology,36,37 this is the first study, to our knowledge, that examines incentives among individuals, payers, and providers and the associations between DHT adoption and stakeholder value flows in the digital T2D prevention context.
Our findings support theoretical perspectives that prevention is contingent on a dynamic network of interdependent actors.33,34 We identify three main insights. First, financial and non-financial incentives for lifestyle-based prevention are often temporally misaligned: engagement in lifestyle-based prevention may provide individuals with short-term benefits such as convenience, rewards, and improved health benefits, while payers are often motivated to support prevention programs as a function of long-term cost savings. Second, we find that DHT adoption for T2D prevention is associated with three main incentive patterns: personalization and convenience for individuals, value-based payment models for payers, and improving workflow efficiency for providers. DHTs may help support the realignment of stakeholder incentives through three key interrelated mechanisms: (i) sustaining individual engagement in prevention programs through personalized micro-incentives, (ii) supporting value-based payment models for payers, and (iii) automating workflows for providers. Using the e3-value framework, we visualize these reconfigured value exchanges (Figure 2), illustrating how DHTs may support a shared data-driven infrastructure to enhance individual engagement, performance tracking, and shared accountability across the ecosystem. 28
Building on the stakeholder incentive literature,10,13our identification of varying financial and non-financial is consistent with perspectives that prevention incentives remain highly fragmented across stakeholder groups As detailed in Table 4, this fragmentation is particularly evident in the temporal dimension: individuals are primarily motivated to engage in lifestyle programs and prevention efforts based on short-term, tangible rewards such as cashback, gamified tokens, or convenience-enhancing lifestyle tracking apps (i.e., “Upload [photos of] your meals for today. You earn a few utility tokens,” ID 23), In contrast, insurers and providers are more likely to prioritize delayed benefits like reduced readmissions or loyalty-based retention to their services and plans over extended periods of time, and at the population level (Prior work examining incentives has specifically focused on provider reimbursement models and patient adherence to treatment plans, finding fragmented financial incentives among various healthcare stakeholders (i.e., pay-for-service models disincentivizing preventive care). 17
Here, we also identified perceived trust in data fairness as an additional non-financial incentive for individuals to engage with DHTs for T2D prevention. This finding reframes data privacy, not as a compliance issue, 70 but as a potential behavioral motivator, potentially associated with willingness to engage with DHTs. For providers and payers, data privacy primarily functions as a regulatory compliance incentive, crucial for avoiding penalties and maintaining public trust. Furthermore, strong data privacy can also serve as a non-financial incentive for patient and customer acquisition or retention.
DHTs as potential incentive bridges
Building on prior research that positions DHTs as key enablers of value-based care,21,71 our results suggest that DHTs may modify traditional fee-for-service payment models by linking reimbursement to continuous behavioral and clinical signals rather than discrete treatment encounters. 72 Early implementations of such models include engagement-contingent payments in digital prevention programs, shared-savings contracts between payers and provider organizations, and remote monitoring models rewarding individuals for sustained adherence to behaviors such as dietary logging, physical activity, or medication adherence. 73 For instance, providers may receive payments based on adherence metrics, while payers may gain earlier risk stratification and reduced uncertainty in future claims. Individuals can also be incentivized to maintain healthy behaviors through gamified feedback loops that reward sustained engagement.74,75
DHTs may enable real-time incentive mechanisms through gamification, biometric feedback loops, and micro-reward systems, illustrated by platforms such as Lark Health, Veri, and Supersapiens. Such micro-incentives may work by strengthening individuals’ perceived benefits and self-efficacy to comply with lifestyle changes, as proposed by the Health Belief Model. However, these early, integrative cross-stakeholder models remain experimental and context-dependent, as healthcare systems frequently lack standardized data governance and near real-time interoperability across payers, providers, and technology vendors. Contractual frameworks that allow individual-level health and DHI engagement data to trigger automated reimbursement or benefit adjustments—capabilities that are essential for scaling prevention-oriented payment models,24,70,76–78 are often lacking.
In turn, DHT-based companies increasingly aim to foster individual engagement and support performance-linked reimbursement models. Yet, such architectures also often run into legacy reimbursement structures. Recent evidence on AI-driven nutrition systems79,80 similarly highlights the friction between technological implementation and institutional adaptability, underscoring the need for more responsive reimbursement frameworks that can accommodate algorithmic personalization, evolving data-governance requirements 81 (i.e., GDPR and HIPAA compliance). These examples show how digital advances challenge existing regulatory and payment structures, underscoring the need for adaptive governance and incentive models to sustain innovation.80,82 Consistent with research on sociotechnical systems,23,24 our findings suggest that successful implementation of prevention hinges not only on technological affordances but on stakeholder governance. DHT companies often lack alignment with payer timelines and face difficulty integrating into incumbent provider workflows, leading to implementation friction even when DHTs may be clinically validated.28,31
Overall, our findings extend the prior concept of intertemporal optimization in behavioral economics in health and payment models for chronic disease management10,51,83 by uncovering temporal misalignments, where short-term user rewards drive engagement, yet long-term payer savings are insufficiently captured. By integrating a systematic literature review, market analysis, and expert interviews into a unified e3-value model, we provide: (1) a typology of stakeholder-specific incentives grounded in empirical data; (2) the identification of system-level misalignments at the intersection of business model design and incentive logic; and (3) a mapping of DHT-association-induced shifts in stakeholder roles and value.
For policymakers, the results suggest that hybrid models combining immediate user-facing incentives (i.e., usage-based rewards) with long-term shared savings arrangements may warrant further consideration. For developers, the evidence indicates that factors such as trust, perceived fairness, and adaptive recognition are not merely ancillary but essential for sustaining user engagement, particularly in direct-to-consumer models. Future research could examine how real-world responses are affected by implementing the recommended incentives alignment.
Limitations and future directions
This study has several limitations. First, while our 26 expert interviews offer deep insights into stakeholder incentives, the sample is geographically concentrated in Switzerland (11 out of 26), which may constrain the generalizability of findings to regions with distinct regulatory, financing, or cultural contexts, see Table A5 and Table A6. For example, although the sample included experts from diverse regions, representation from several high-growth DHT markets, particularly representation from Asia, was limited. Second, our market analysis focused primarily on English-language materials from firms operating in high-income countries, which may underrepresent models and dynamics from major innovative low- and middle-income health systems, particularly those of China and India. As a result, our findings may not fully capture regional innovations or alternative approaches to T2D prevention. Third, while the study identifies patterns and theoretical associations between DHTs and stakeholder incentives, it does not establish causal effects.
Future research may experimentally evaluate the impact of DHT-enabled incentive mechanisms, such as outcome-based reimbursement, gamified adherence rewards, or real-time biometric feedback, on measurable endpoints, including user engagement, clinical outcomes (i.e., average blood sugar reduction), long-term adherence in preventive lifestyle programs, and long-term cost-savings. In addition to intervention studies, future work may fruitfully explore system-level simulations of the cost-effectiveness (both direct and indirect costs) of DHT adoption under various incentive configurations and policy regimes. In particular, comparative analyses across health systems should examine how incentive patterns identified in high-income settings translate to low- and middle-income countries, where fragmented financing structures, limited data infrastructure, and different reimbursement logics may constrain the implementation of value-based payment models and require localized adaptations of DHT-based prevention strategies. 84 In such settings, engagement-focused or community-based incentives may be more salient than payer-driven outcome contracts, suggesting that the observed patterns are context-dependent rather than universally transferable.
Conclusions
Scalable T2D prevention is most likely to succeed within ecosystems where individual, payer, and provider incentives are aligned. We find persistent misalignment of temporal incentives, which may hinder large-scale implementation. DHTs may support stakeholder alignment by directing personalized engagement in preventive interventions, promoting value-based reimbursement, and improving operational efficiency for providers. Sustained preventive impact, however, is likely associated with critical enablers: empirical evidence linking DHT prevention to health and cost outcomes, policy frameworks that reward preventive value creation, and DHT designs that prioritize trust and transparency. Realizing the potential of digital T2D prevention likely requires improved cross-sector coordination to align stakeholders and ensure these enabling conditions are met.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261425402 - Supplemental material for Digital health technologies and stakeholder incentives in type-2 diabetes prevention
Supplemental material, sj-docx-1-dhj-10.1177_20552076261425402 for Digital health technologies and stakeholder incentives in type-2 diabetes prevention by Wasu Mekniran, Wilma Diethelm, Victoire Stalder, Elgar Fleisch, Tobias Kowatsch and Mia Jovanova in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to thank all interview participants who participated and supported in data collection for this study. This article is partially based on a master's thesis, “Healthcare ecosystems for preventive care – role of Swiss health insurance companies” by C. Cathomas, University of St Gallen, which has provided us with a part of the data collection, explicitly interviews data with health insurers.
Ethical approval
This study was conducted in accordance with the Checklist for Assessing the Ethical Soundness of Research Projects at the University of St Gallen and was exempt from a formal review and approval by the Ethics Committee of the University of St Gallen.
Author contributions
Wasu Mekniran: conceptualization, methodology, investigation, formal analysis, data curation, software, writing–original draft, and writing–review and editing. Wilma Diethelm: methodology, investigation, formal analysis, data curation, and writing–review and editing. Victoire Stalder: methodology, investigation, formal analysis, data curation, and writing–review and editing. Elgar Fleisch: investigation, supervision, writing– review and editing, and funding acquisition. Tobias Kowatsch: conceptualization, resources, supervision, writing–review and editing, and funding acquisition. Mia Jovanova: conceptualization, methodology, investigation, formal analysis, resources, supervision, writing–review and editing, and funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by the Swiss health insurer CSS Health Insurance.
Declaration of conflicting interests
Wasu Mekniran, Wilma Diethelm, Victoire Stalder, Elgar Fleisch, Tobias Kowatsch, and Mia Jovanova are affiliated with the Centre for Digital Health Interventions (CDHI), a joint initiative of the Institute for Implementation Science in Health Care, University of Zurich; the Department of Management, Technology, and Economics at the Swiss Federal Institute of Technology in Zürich; and the Institute of Technology Management and School of Medicine at the University of St Gallen. CDHI is funded in part by CSS, a Swiss health insurer, and MavieNext, an Austrian healthcare provider. Elgar Fleisch and Tobias Kowatsch are co-founders of Pathmate Technologies, a university spin-off company that creates and delivers digital clinical pathways. However, neither Pathmate Technologies nor MavieNext were involved in this research.
Data availability
The data sets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Guarantor
Mia Jovanova.
Supplemental material
Supplemental material for this article is available online.
Appendix
The appendices support the study's methodology and findings, including search protocols, coding framework, interviewee data, detailed transformation analyses, and funding distributions that expand on the market analysis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
