Abstract
Objective
This study evaluated the complete patient experience of at-home self-collection for cervical cancer screening using the Teal Wand, the United States Food and Drug Administration (FDA)-authorized vaginal self-collection device, paired with a Health Insurance Portability and Accountability Act (HIPAA)-secure telehealth platform.
Introduction
The data assesses patients’ understanding, experiences, and engagement, particularly around results delivery. The findings inform telehealth strategies that can improve patients’ human papillomavirus (HPV) results experience, build HPV awareness, facilitate follow-up care when needed, and increase screening adherence.
Methods
A prospective, mixed-methods cohort usability study was conducted using structured surveys and in-depth interviews, supplemental to prospective clinical validation. Thirty participants used the Teal Wand and completed surveys after major touchpoints. A subset (n = 10) participated in semi-structured interviews. Surveys captured usability, satisfaction, comprehension, and overall experience. Interviews were analyzed using an abductive approach. Platform analytics provide further insight into utilization.
Results
All participants (30/30) completed self-collection, with 4.7/5 average satisfaction score. Most (93%) found the platform easily navigable and results easy to access and understand. All participants viewed results within 1.5 hours on average and understood next steps. Among those needing follow-up (n = 9), 100% scheduled follow-up telehealth appointments within hours of viewing results, and all attended those appointments. Qualitative data underscored the importance of supportive language and provider engagement in mitigating emotional distress, especially for HPV-positive results.
Conclusions
At-home cervical cancer screening supported by a tailored telehealth platform is usable and acceptable. Thoughtful results delivery can improve patient comprehension, reduce anxiety, and strengthen future adherence, supporting broader efforts to increase screening access and reduce cervical cancer burden.
Keywords
Introduction
Routine screening can prevent nearly all cervical cancers, which are almost always caused by persistent human papillomavirus (HPV) infections.1–3 Yet, in 2021, 26.1% of U.S. women aged 21–65 were not up to date with their cervical cancer screening, with more people falling behind each year. 4 Primary HPV testing is a recommended screening test for those above the age of 25 because it has a higher sensitivity than Pap tests alone, leads to fewer unnecessary follow-up procedures compared to co-tests, and, importantly, enables vaginal self-collection, which improves screening access as a preferred collection method among patients.2,5–9
The United States Food and Drug Administration (FDA) approved self-collection within healthcare settings in 2024, and in 2025, the Agency authorized an at-home self-collection device, the “Teal Wand,” a vaginal self-collection device for testing on an FDA-approved Primary HPV assay in the laboratory.10,11 In a nationwide clinical validation study (n = 609 participants, 16 clinical sites), self-collected samples using the Teal Wand achieved 96% sensitivity (ability to detect CIN2+, relative sensitivity of 1.00) and showed 95% positive percent agreement (PPA) for detecting high-risk HPV, performance equivalent to that of clinician-collected samples. 12
At-home self-collection (i.e., mailed HPV self-collection kits) offers patients equivalent clinical results to clinician-collection, a widely preferred collection experience, and a solution for barriers to in-clinic screening attendance.2,5,9,12–16 An at-home alternative can increase screening toward the achievable elimination of cervical cancer as a public health concern (fewer than 4 cases per 100,000).5,17 Reducing barriers to screening increases uptake, especially among underscreened populations (e.g., low-income communities of color, remote or rural areas).5,18–22 Those who prefer receiving health recommendations from providers or media tend to be more willing to use at-home self-collection, as are those willing to pay for this collection method. 23 Individuals who are unsure of how they want to receive an at-home self-collection kit and who prefer delivering the sample to the lab themselves (vs. via mail) are less willing to participate. 23
Telehealth technology-driven strategies for healthcare delivery have increased, particularly since the COVID-19 pandemic, and are an effective approach for delivering clinically validated at-home self-collection for HPV testing. 24 Digital telehealth platforms can enable tracking of at-home mailing logistics, virtual patient-provider engagement, asynchronous HPV results delivery, educational and instructional resources, and patient tracking and reminders for follow-up care and future screenings. This presents an opportunity to close gaps in awareness and access that keep one in four women from screening.
Still, telehealth strategies for results delivery and patient engagement can be optimized for patient understanding and adherence to the screening continuum. Gaps in HPV awareness, and how HPV can persist to cause cervical cancer, can lead to adverse emotional and psychosocial responses when individuals receive HPV-positive results.25–27 These include anxiety, fears about cancer, sexual distress, changes in one's body image, and concerns about the loss of reproductive functions.27,28 Many misinterpret the implications of HPV-positive results–often learning about sexual transmission and relation to cervical cancer for the first time–and thus struggle to navigate further information.25,26 Additionally, while patients may be directed to attend follow-up appointments with providers after receiving results, initial results anxiety and a lack of immediate provider contact can hinder their likelihood of engaging in critical follow-up care.26–28 Challenges around results delivery and interpretation are widespread and are particularly common among women with lower education, numeracy skills, and health literacy. 29
This study evaluates the usability of the end-to-end patient experience of at-home self-collection (using the Teal Wand device) and asynchronous results delivery on a HIPAA-secure telehealth platform paired with the device, supplemental to the clinical validation study. 12 We analyze the main needs and concerns patients raise upon receiving their results (both HPV-positive and -negative), to inform evolving strategies for designing telehealth approaches to deliver HPV results. Improving the patient experience around results delivery can contribute to better HPV awareness, engagement in future screenings, and adherence to essential follow-up care in the case of HPV-positive results.
Methods and study design
Following a large national clinical validation study, this mixed-methods usability study evaluated the telehealth and at-home collection end-to-end experience, incorporating surveys and interviews. 12 The clinical validation study was, by design and FDA agreement, a method comparison study simulating an at-home experience, so this subsequent usability study served to confirm that participants could navigate the entire at-home product offering, inclusive of the telehealth platform. This prospective cohort study evaluated all aspects of implementing at-home self-collection for cervical cancer screening using the Teal Wand and a secure telehealth platform specifically designed to pair with the device and assay. Participants also received personalized automated emails and texts for reminders (e.g., to complete their self-collection, view results, book necessary telehealth appointments), appointment details, and relevant resources. The telehealth platform facilitated patient communication, account management, results delivery, patient-provider appointments, and an extensive library of educational resources. The use of the Teal Wand, which at the time of this study was an investigational device (FDA-authorized in May 2025), and related data collection instruments were IRB approved (WCG IRB, 20244750); participants completed written informed consent forms before beginning any study-related activities. 10 All figures and study instruments included in this manuscript are original materials.
The study took place between November 2024 and March 2025, inclusive of participant recruitment and all study procedures. While study participants were based in California, participants did not physically visit a study site, as all study procedures were completed from their home (e.g., at-home self-collection using the Teal Wand, telehealth visits) and data collection took place digitally (e.g., Zoom interviews, Google Form surveys).
The researchers are employees of Teal Health, which is the designer and manufacturer of the Teal Wand, or Teal Medical Practice, which is the clinical practice under which medical providers prescribe the Teal Wand to patients and provide related patient care. The networks used to recruit participants were familiar to some employees at Teal Health and Teal Medical Practice. However, the researchers who interviewed participants (MM and LS) did not have a first-degree connection to the interviewees. Interviewers identified as women, which may be pertinent in building rapport and comfort when discussing shared topics of women's health experiences regarding HPV and cervical cancer. Survey data entry was completed by participants electronically (via Google forms); there was no personal interaction between researchers and respondents during survey completion. Researchers involved in study design and implementation have extensive experience in mixed methods research methods. No nonparticipants or nonresearchers were present during any study-related procedures.
Participants were recruited between November 2024 and January 2025, in two stages. First, a convenience sample was recruited from known screening-aged communities (e.g., parent groups within schools, recent retirees, professional groups), wherein emails were circulated with resources about the study and participation requirements. Second, word-of-mouth snowball sampling identified additional participants. Recruitment resources informed participants of the purpose of this study, exclusion criteria, study procedures and time commitments, compensation structure, nature of HPV results received after self-collection (i.e., not medically actionable, investigational medical device), background on Teal Health including existing Teal Wand safety data from preceding clinical validation study. 12 Recruitment was restricted to California for reasons of compliant telehealth delivery. Recruitment focused on cervical cancer screening-aged individuals (25–65 years old), concentrating on those aged 36–45, as this is typically a group behind on screenings and in whom cervical cancer diagnoses have recently been increasing (and who therefore are expected to be a key US target audience for this device and paired telehealth service). 30 In addition to age, recruitment considered participants’ race/ethnicity, as key demographic variables identified as being relevant to at-home self-collection and telehealth usage.
This usability study recruited a total of 31 participants, of which one participant dropped out due to becoming pregnant, comprising a final sample size of 30 participants. Sample size is based on FDA guidance on Human Factors testing (used for medical devices and platforms such as that studied herein), which recommends a minimum of 15 users to identify 90% of usability problems. This sample size was doubled to detect 97–99% of possible use errors (per FDA guidance) and to strengthen qualitative input. 31 Table 1 summarizes participant demographics.
Study participant race/ethnicity and age demographics.

Participants could complete all study procedures—that is, telehealth engagement, self-collection, surveys, interviews—from their home or any other location of their choice. After consenting, participants completed the eligibility screener and account registration process on the telehealth platform and scheduled an introductory telehealth appointment with a qualified medical provider. Consistent with guidelines from the American Society for Colposcopy and Cervical Pathology (ASCCP) and clinical practice, participants were included (considered “eligible”) if they were evaluated during the eligibility screener and introductory telehealth appointment as being at average risk for cervical cancer, within the ages of 25–65, with an intact cervix. Exclusion criteria included being pregnant or within 6 weeks of giving birth, experiencing abnormal vaginal bleeding that had not been evaluated by a medical provider, and having had a LEEP or cold knife procedure for moderate to severe precancerous changes in the past 25 years, a medical history of female reproductive cancers, HIV, in utero DES (diethylstilbestrol) exposure, or currently taking immunosuppressant medications.
Accordingly, during the introductory appointment, the provider confirmed eligibility according to medical guidelines and prescribed the Teal Wand at-home screening device (contained within a screening kit that includes all materials needed for sample self-collection and mailing to the laboratory).5,32 Once the kit was delivered to their home (mailed via United States Postal Service), the participant self-collected with the Teal Wand using the instructions provided and mailed their sample via USPS to a CLIA-certified laboratory, where it was processed per standard laboratory protocols on the FDA-approved Roche cobas HPV test. Results management is compliant with protected health information, where results are delivered to the patient's secure electronic health record, reviewed by providers, and delivered electronically to the participant via the HIPAA-compliant telehealth platform. Participants were notified to view their results via text and email. When participants requested, results were shared with their regular in-clinic provider.
Those with HPV-positive results or a recent history of HPV were prompted to schedule a follow-up telehealth appointment with a provider to discuss next steps for on-guideline testing and screening frequency. Per self-collection guidelines, providers also referred participants to local clinics for in-person follow-up, sharing results and coordinating care with referred providers as required; however, adherence to in-clinic follow-up was not studied here. 32
The majority of participants were given test results based on the laboratory's analysis of their sample, but some results were simulated to ensure enough data for both “normal” and “abnormal” care pathways (Table 2). The variations of results were as follows: “normal” (HPV-negative), “abnormal” (HPV-positive) along with HPV genotype(s) detected, “normal” results with recent HPV-positive history (within the past 5 years), and invalid test outcome (e.g., inadequate sample). If a participant's sample was invalid, results were simulated as “normal.” This study was focused on usability rather than clinical outcomes, which have been reported elsewhere. 12 Offering users (n = 2) who had invalid outcomes a simulated normal result allowed these participants to complete the usability journey and provide feedback on their experience without having to re-collect a sample, which was required for the study's usability objectives. Further, the HPV results produced were not medically actionable as the device was still under FDA review (“investigational”) at the time of the study, so participants were not relying on these results for any clinical next steps. Similarly, some patients (n = 4) were also randomly preselected to receive simulated abnormal results, as the sample set was expected to consist of fewer “true” HPV-positive results. Preselecting a subset of participants to receive simulated abnormal results ensured that the study would be able to capture sufficient feedback regarding the abnormal results pathway. Participants were informed of the possibility of simulated results when consenting to the study; those who had simulated results were informed of this before results delivery and provided a PDF of their actual laboratory results, but were instructed to engage with the follow-up experience based on their simulated abnormal HPV outcome.
Summary of HPV results delivered to participants.

Note, results delivery via the telehealth platform refers to HPV-positive results as “abnormal” for patient comprehension; earlier usability tests of the platform revealed that “abnormal” terminology was easier to understand and was more likely to drive action on the patient's part, compared to “HPV” terminology, about which awareness is often lacking. 25 Accordingly, when analyzing results in this paper, we reflect the terminology that patients saw in their experience.
Data about participants’ experiences and feedback were collected through structured surveys emailed to participants after they completed key stages of the at-home screening experience. Surveys were sent to participants’ email addresses electronically, via Google Forms, and managed through a Google Workspace with a Business Associate Addendum (BAA) that is HIPAA compliant. Collected data was populated into a CSV spreadsheet for descriptive statistical analysis. Analyzed data was aggregated, deidentified, and verified by at least two other members of the research team. The surveys asked multiple-choice questions about usability, accessibility, comprehension, preferences, provider engagement, experience, and emotions at each part of the journey. There were open comment fields to provide further details. Surveys were distributed after each of the following touchpoints:
Survey 1: Eligibility and Account Registration–20 questions about account usability and provider care-establishment telehealth appointment. Survey 2: At-home Self-collection using the Teal Wand–30 questions about unboxing the kit, using instructions to self-collect, and sample mailing to the laboratory. Survey 3: Results delivery via secure telehealth platform
“Normal” HPV-negative results–21 questions about telehealth platform accessibility, results understanding, and overall experience. “Abnormal” HPV-positive results or history—9 questions about telehealth platform accessibility, results understanding, and platform usability to schedule a follow-up telehealth appointment. Survey 4 (only for those with HPV-positive results/history): Follow-up telehealth appointment with medical provider—9 questions about results comprehension, provider appointment, and overall at-home screening experience.
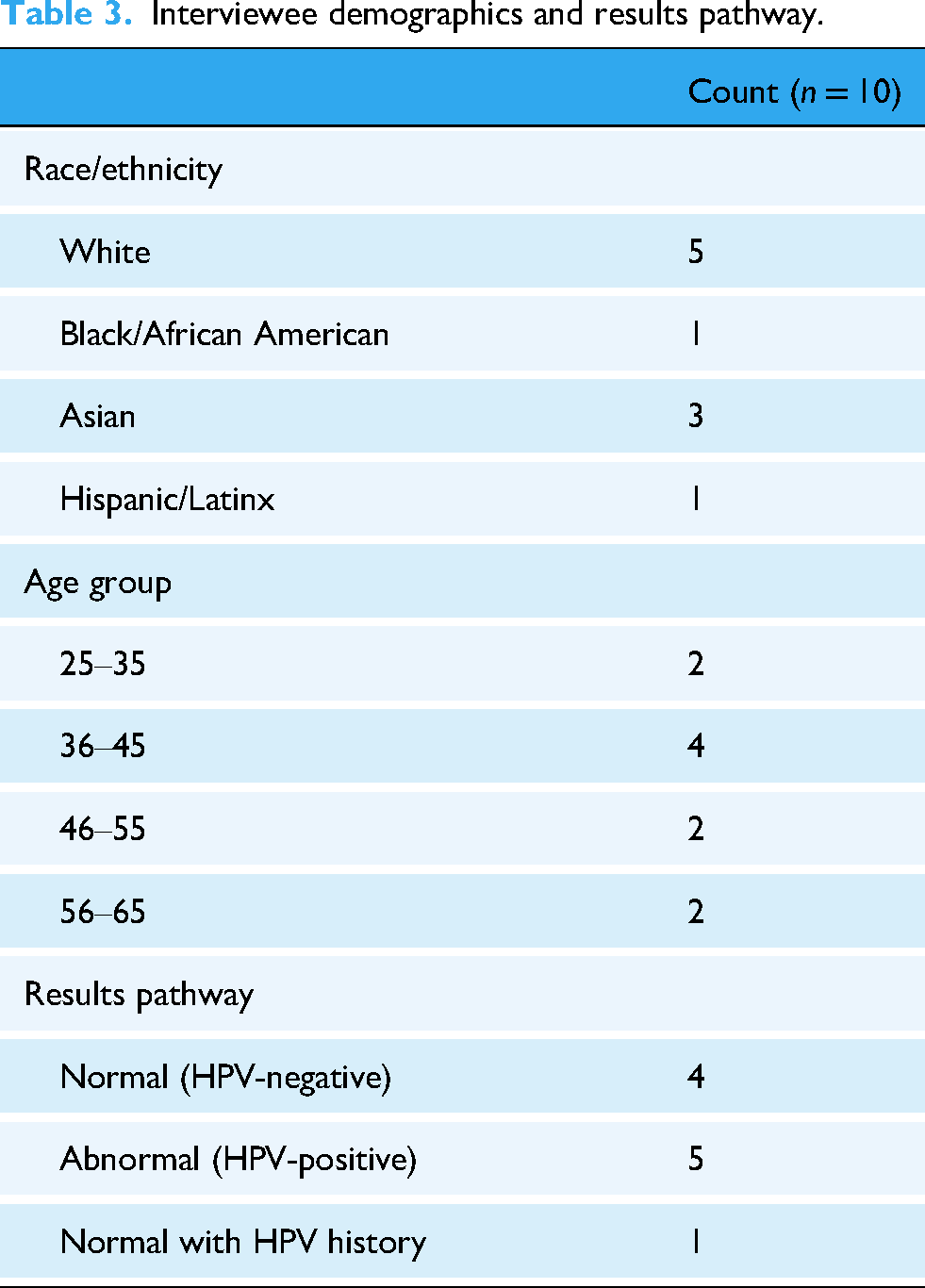
After completing surveys, select participants were invited to in-depth interviews about their results delivery experience. Researchers designed interview guides using themes from secondary literature, tracing each touchpoint in the screening and telehealth experience, while also tailoring interview guides based on each interviewee's experiences as reported in completed surveys to achieve deeper qualitative insights. Interviews were semi-structured, conducted virtually over Zoom, and lasted around 1 hour each, asking about results comprehension, accessibility, usability, experiences and emotions, provider engagement, and preferences. Interviewers (LS, MM) took notes during each interview, in addition to obtaining audio recordings for transcription. Interviews were audio recorded on Zoom, auto-transcribed by the recording platform, and manually deidentified; transcripts were not returned to participants for comment, and participants did not request their transcripts. Coded transcripts were reviewed by at least two other members of the research team. Transcripts were coded using an abductive approach, integrating both inductive insights from the data and deductive themes identified in the existing literature. 25 Analytical themes were developed from established findings in secondary literature and iteratively refined as new patterns and insights emerged from the transcripts, allowing the analysis to balance theoretical grounding with data-driven discovery. Two researchers conducted the interviews jointly, then individually analyzed each transcript and subsequently compared and discussed their analyses to reach a consensus. Interviewees (n = 10) were selected based on experiences reported in surveys and HPV results to reflect a range of feedback. Selection also considered various age groups, as a proxy for familiarity with technology and telehealth (Table 3). Data saturation was achieved upon completion of surveys and interviews.
Interviewee demographics and results pathway.
Participants were compensated nominally for their involvement in various parts of the testing, with additional compensation for those who participated in interviews. Minimal compensation was provided after completion of all surveys and, when relevant, an interview. We infer that this compensation structure (payment post-surveys and interview completion) may have contributed to the high rate of participant follow-through on study procedures reported below.
We used the SRQR reporting checklist when editing, included in Supplement A. 33
Results
All 30 participants completed their required surveys, and 10 participants completed interviews. There is no missing data. In total, 77% (23/30) reported “yes” when asked whether they would use this at-home self-collection device along with the paired telehealth platform for their next cervical cancer screening, and the remaining 23% (7/30) reported “maybe.” Overall, when asked to rate their satisfaction with this end-to-end telehealth and screening experience on a scale of 1 (least satisfied) to 5 (most satisfied), the average satisfaction score was 4.7.
Ninety-three percent (93%, 28/30) found that eligibility and account registration on the telehealth platform were easy to navigate and complete, and 97% (29/30) reported that providing all the required information during this registration stage was easy. All (100%, 30/30) reported they felt cared for and supported by the provider during their introductory telehealth visit to review screening and medical history before prescribing the at-home self-collection device, and 100% (30/30) knew what to expect with regard to the at-home self-collection after this visit.
On average, the screening kits were delivered to participants’ homes in 4.5 days (σ = 1.6), and participants took 8.8 days (σ = 9.4) after kit receipt to complete the sample collection and mail it to the laboratory. They required 2.4 reminders (range 0–6) on average to complete self-collection. The telehealth platform is designed to deliver 10 reminders total, every few days for the first 2 weeks, followed by a monthly reminder for 3 months. All participants (30/30) completed the at-home self-collection using the Teal Wand, and all reported that they could open the kit and see and identify all components easily. Samples took on average 3.9 days to reach the laboratory (σ = 1.6), which is well within the established sample stability timeframe.
Turning to results delivery, the focus of this analysis, all participants (30/30) viewed their results within an average of 1.5 hours (σ = 1.5) from time of receipt. Participants were notified via text and email that results were ready to view within their telehealth patient portal. No one (0/30) required reminders to view results or schedule virtual follow-ups; however, the telehealth platform is designed to deliver four reminders over 10 days, prioritizing high-touch engagement (e.g., phone calls) for those with “abnormal” results. All participants (30/30) knew the next steps to take after receiving their results, and 97% (29/30) agreed that they were provided helpful options to discuss results further as needed. All those who needed to schedule a telehealth follow-up appointment (i.e., “abnormal” HPV-positive results, recent HPV history; n = 9) with the provider to discuss next steps did so, and all attended the scheduled appointment. On average, it took 2.4 hours (σ = 3.4) for participants to schedule necessary telehealth follow-up appointments from the time of viewing results, in which it was indicated they needed to schedule a follow-up.
In terms of usability and access on the telehealth platform, 93% (28/30) said it was easy to find and access test results within their secure account, and 93% (28/30) reported that the language used to communicate results was easy to understand. Further, 93% (28/30) agreed that the written note from the provider accompanying their results included helpful and supportive information to process what their results mean (Figure 1). Participants’ survey comments further illustrate these findings: Empathetic note from doctor acknowledging I would be scared but also making me feel less alone that this happens (made me feel supported)l; (I feel) empowered that I can take action right away to book a follow up. (Study participant, ‘abnormal’ results) The words ‘may be at risk’ brought in some doubt that the abnormal results [sic] was going to be really scary–but not much. […] I felt better about the message from the doctor. It would have been nice to see her message first […]. (Study participant, ‘abnormal’ results) I liked that they were processed quickly and that they included what my next steps are. (Study participant, ‘normal’ results) I appreciated that the results were accompanied by a personalized note from a physician. It made me feel like there was less room for error. (Study participant, ‘normal’ results)

Teal health at-home self-collection kit for cervical cancer screening, showing all components included within the screening kit.

Example telehealth delivery of “normal” HPV-negative results on paired platform, including a results summary card, results PDF download option, and a note explaining the results from the medical provider.

Example telehealth delivery of “abnormal” HPV-positive results on paired platform, showing a results summary card, follow-up appointment booking options, results PDF download option, and a note explaining the results from the medical provider.
All those who completed follow-up telehealth appointments (100%, 9/9) to discuss next steps based on their “abnormal” results (e.g., in-clinic Pap smear or colposcopy, per current self-collection guidelines) reported that they had a deeper understanding of their “abnormal” HPV-positive results and the associated risks after their telehealth appointment, that the provider addressed all their questions and concerns, they trusted the provider, and felt cared for and supported during the appointment. 32
The surveys also asked users to report on feelings experienced at the various steps in the at-home self-collection experience. Table 4 summarizes these emotional experiences during the results delivery stage (participants could select all that apply).
Participants’ reported feelings experienced during telehealth results delivery.

Interviews
A recurring theme for all those interviewees with “normal” results (n = 5) was wanting to know whether their HPV-negative outcome may have been due to a self-collection error (i.e., whether an incorrect sample collection could lead to a “normal” result, rather than an invalid outcome). These individuals desired more reassurance about the self-collection process, correct device use, and accuracy of their results. Furthermore, these participants wanted clarity on whether they could rely on future screening reminders from the telehealth platform several years after their current “normal” results. On this note, they expressed wanting a positive and conclusive tone in their results delivery, which would emphasize the lack of current concern and their next screening due date, without mention of cervical cancer risk, as this felt irrelevant to their current situation. Interview excerpts elaborate: Ideally, (the results) would be like ‘you have collected the sample correctly,’ at the end of the note, ‘cells observed, everything is fine.’ […] I'd want confirmation that they saw what they were expecting to see. […] I was just worried that I'm not doing this right […]. Did I just twirl it around, but I didn't actually get the cells that I was supposed to? (Study participant, ‘normal’ results) For me personally, I don't care what (infection) it is, I want to know how it's going to impact me. So, ‘what does it mean to get an abnormal result’ is more important to me […]. And, I want something in here that says ‘how this impacts your husband.’ (Study participant, ‘abnormal’ results) It was education needed on my side because I'm used to when tests come back, there's a range. […] So, if I have an abnormal, is there a range or is it only, it's a yes or no answer? […] I wanted more information immediately than just “abnormal.” I wanted something in writing, a printout of the test results or something that I could have to […] do a little research or to help me prepare before I meet with a nurse or the doctor. (Study participant, ‘abnormal’ results) What does it mean to have an abnormal (HPV) result? How long it's going to take (to develop into cancer). Is there a cure? Those are the main things (I want to know right away). […] I guess preventative measures too. I know there's a vaccination, but there's some recommendations around when you can take it […]. Can you treat (HPV)? (Study participant, ‘abnormal’ results)
Interviewees across both result outcome types expressed different preferences in terms of how they would like to receive this additional information, in terms of the type of resource (e.g., long narrative-style document, links to external resources, shorter frequently asked questions), but all agreed that these main themes should be addressed in a telehealth communication from the provider delivered alongside the results. They specifically asked for this resource to come from within the telehealth platform, as a part of results delivery, to preclude undirected broader online searches that can be distressing or misleading.
The shared preference was to have an informational resource provided alongside asynchronous telehealth results delivery, which patients could use to learn more about their results, seek reassurance, and prevent panic in the immediate moment of learning their results. Interviewees specifically raised that this resource should not replace an interaction with the provider to follow; rather, they sought a basic level of understanding to bridge the time in between results delivery and their virtual provider appointment, wherein they could receive a more thorough consultation.
Discussion
Participants in this study reported high levels of usability and accessibility with the Teal Wand at-home self-collection device and its paired telehealth platform. High device usability is notable, with 100% (30/30) completing their at-home collection and reporting ease with doing so, which is consistent with other reports of larger studies using this device.12,16 Regarding the telehealth platform, 93% (27/30) easily found and accessed test results within their secure telehealth account, and 100% (30/30) successfully viewed their results on the platform within an average of 1.5 hours (σ = 1.5) from time of results posting. Participants preferred and were satisfied with this at-home approach to screening, with all participants to some extent reporting they would repeat this process for their next screening (76% “yes,” 24% “maybe”). Preference for at-home screening among U.S. women is echoed in multiple reports and reflected in draft USPSTF cervical cancer screening guidelines (December 2024).5,13–16,18,21,22 An at-home screening and paired telehealth approach is likely to increase patient engagement in screening and follow-up care.5,34 Supporting evidence shows that patients within an active online account, or those receiving reminders and communications via telehealth, tend to participate more in the cervical cancer screening continuum.35,36
Participants’ experience of receiving “normal” results was generally positive, which is to be expected as results do not imply health concerns. For example, when receiving “normal” results, most participants report feeling at-ease/comfortable, cared for/supported, and confident/empowered. Some participants shared in survey comments and interviews that they sought more reassurance that their self-collection was conducted correctly, and that “normal” results were not due to device use errors (i.e., that incorrect sample collections would lead to invalid outcomes, rather than “normal” results). Ideally, this reassurance would come within a communication from the provider that accompanies their results. This information is worth integrating into how HPV-negative results are delivered via telehealth following self-collection. FDA review and authorization of the Teal Wand, along with published clinical results, can also provide reassurance in device performance and a user's ability to perform self-collection successfully. 12
In contrast, negative emotions of feeling confused/overwhelmed, nervous/hesitant, scared/fearful were more prevalent among those who received “abnormal” HPV-positive results. Without adequate contextual information about the health implications of their results, overall HPV education, and clear next steps, those who receive HPV-positive results can feel immediate panic or worry when reading their results. Notably, these negative emotions resolved after these participants met with the provider virtually and had their questions and concerns addressed. After this follow-up appointment, these participants report feeling largely positive emotions of being at-ease/comfortable, cared for/supported, and confident/empowered, with negative emotions no longer reported. This highlights the importance of enabling provider engagement as a part of telehealth results delivery following HPV self-collection, while also providing some of the information that providers will explicate upfront, upon initial results delivery.
Our data show that HPV-positive results delivery should incorporate information addressing patients’ key questions and concerns regarding how HPV is reported, general HPV awareness, reassurance that HPV is common and does not necessarily equate to a cervical cancer diagnosis, and that follow-up steps can ensure appropriate treatment or prevention. Addressing these areas as part of the telehealth results experience can mitigate the negative emotions of panic and worry during initial results reception. This information, however, should not replace the recommended provider-patient interaction during a follow-up appointment to discuss results in more depth and encourage in-person follow-up where indicated.
While those with HPV-negative and positive results had distinct questions and concerns, the need for an informational resource to accompany asynchronous results following HPV self-collection is consistent. The informational resource accompanying self-collected HPV results can take various formats, and need not be extensive, but should provide patients with more context about interpreting their results and a reprieve in the interim between results delivery and their follow-up provider appointment. Further, while those with HPV-positive results benefit from a higher provider touchpoint (e.g., telehealth appointment), those with HPV-negative results also require provider communication to some extent (e.g., provider note accompanying results). These considerations should be accounted for in a telehealth results experience.
This study and these findings served to influence how the telehealth platform was modified for commercial use following FDA-authorization of the Teal Wand, informing how the at-home screening experience using the Teal Wand is designed to respond to data trends and emerging patient and provider needs. Accordingly, adjustments were made to the platform tested herein, based on the obtained feedback. Changes included reducing overly alarming references to cervical cancer risk when delivering HPV results, reassurance of correct sample collection and processing, and including several informative “frequently asked questions” as a part of the results delivery to educate about HPV, the meaning of “normal” or “abnormal” results, next steps for screening, prevention, or follow-up care, and implications for sexual partners. The platform's extensive resource library has also been expanded to include several additional educational articles about HPV and cervical cancer. Further, the telehealth platform was optimized for provider engagement, readily enabling follow-up virtual appointment booking as recommended. Not only can providers refer patients to local clinics for follow-up and coordinate with referred providers, but the telehealth system also consistently reminds patients to schedule their referral and offers scheduling assistance via navigators when needed.
There are several advantages to intentionally designing telehealth configurations around at-home self-collection, which can facilitate high-quality, accessible care, convenience, and patient education and engagement, thereby extending access and improving health outcomes. 37 In addition to supporting patients individually, an informative results delivery experience can also facilitate a broader culture of HPV awareness and stigma reduction over time. Further, this approach helps to prevent misinformation and unnecessary health scares, and can support providers in making use of appointments to address patients’ questions and next steps from a shared place of information.
Furthermore, studies show at-home self-collection paired with sustained telehealth engagement (e.g., reminders, patient navigation) can lead to greater participation in cancer screening and high adherence (85–95%) to follow-up consultations and procedures–often higher than HPV-positive results from clinician-collection.34,38,39 Related specifically to the use of the Teal Wand device studied herein, existing studies show that the device is a clinically accurate and a widely preferred screening option and particularly valuable to groups at higher risk of cervical cancer and who face additional barriers to screening that often position them among those underscreened in the USA, including those with higher BMI, of nonheterosexual sexual orientation, of peri- and postmenopause status, rural, lower income, and with prior HPV diagnoses.12,16,40 As such, this screening alternative presents a significant public health opportunity at the intersection of at-home self-collection and telehealth models.
Conclusion
This usability study, supplemental to the large national clinical validation study, evaluated all components of the at-home self-collection experience for cervical cancer screening, including a user-friendly, now FDA-authorized at-home vaginal self-collection device, virtual patient-provider engagement, and use of a telehealth platform designed to offer clinically validated cervical cancer screening, education, and follow-up support. The data illuminate patient experiences regarding usability and accessibility of the collection device and telehealth platform, as well as detailed insights into key themes and concerns in results delivery for both HPV-positive and HPV-negative outcomes using mixed methods surveys and interviews. The feedback obtained from this investigation has been incorporated into the telehealth platform studied, which continues to evolve as commercial use expands and more data are obtained.
Complete and in-depth data are presented. Limitations include that the sample could have been more representative in terms of race and geographical demographics, and the analysis did not go beyond results delivery to evaluate adherence to recommended in-clinic follow-up care (however, the commercial product is designed to capture adherence to follow-up). Additionally, although data saturation was achieved using the interview guide and surveys used in this study, these were not validated or pilot-tested (aside from Survey 2 “At-home Self-collection using the Teal Wand,” which was piloted in the clinical validation study). Overall, this was a usability study, focusing on whether the specific device and paired telehealth platform were usable and accessible to a range of participants, since the preceding clinical validation study was, by design, conducted in a healthcare setting and did not include complete use of the telehealth platform or actual at-home testing. As such, this study did not center on large-scale implementation of the device, varying participant contexts, or possible biases that patients may have when opting into at-home self-collection (although clinical data about the device's implementation have been previously published). However, given recent FDA authorization of at-home self-collection for cervical cancer screening, followed by the implementation of the device and platform studied herein (as of July 2025), real-world data are presently being generated, and strategies are being continuously adapted to optimize engagement in screening and follow-up as well as the accessibility of the paired telehealth platform across various patient populations.
At-home self-collection for cervical cancer screening will be increasingly implemented due to its clinical validation, convenience, and high preference among screening-aged patient populations in the USA. Data demonstrate that women have greater engagement with at-home self-collection options and greater adherence to follow-up of HPV-positive results when they understand the meaning of those results. Thus, while at-home access is poised to substantially improve screening engagement, it is equally important to ensure patients receive results communications that are clear, informative, considerate of their emotional state, and that promote health literacy. A paired telehealth platform designed for women's health and the specific screening experience is an effective strategy for patient engagement, results delivery, and necessary education and support around follow-up actions. Increased screening will improve health outcomes, and enabling more access through user-friendly at-home telehealth while facilitating follow-up steps is essential to preventing cervical cancer diagnoses and mortality.
Telehealth-supported at-home self-collection will be able to increase engagement in cervical cancer screening by increasing access, facilitating timely reminders, encouraging adherence to follow-up, and creating more accessible touchpoints for care. When results delivery is thoughtfully designed within the telehealth experience, it has the potential to not only improve follow-up rates but also serve as a powerful opportunity for patient education and broader awareness around HPV and cervical cancer prevention. Healthcare services should take into consideration evidence from patient-centered studies to deliver results that meet the unique needs of those who receive HPV results.
Footnotes
Ethical considerations
The use of the Teal Wand, which at the time of this study was an investigational device (FDA-authorized in May 2025), and data collection instruments related to self-collection were IRB-approved by WCG Institutional Review Board (WCG IRB 20244750). Surveys and interviews related to the use of the telehealth experience were exempt per 45 CFR 46.104(b)(3); however, informed consent was obtained for these activities.
Consent to participate
All participants completed written informed consent forms before beginning any study-related activities.
Author contributions statement
Elizabeth Swenson, MD, FACOG, MSCP, contributed to investigation and writing–review and editing. ES carried out the study investigation as a medical provider supporting study participants through the digital health aspects of at-home self-collection for cervical cancer screening and results delivery. ES was also involved in providing feedback during the writing and revision stages of this manuscript. Elizabeth VonderHaar, MD, FACOG, contributed to investigation and writing–review and editing. EV carried out the study investigation as a medical provider supporting study participants through the digital health aspects of at-home self-collection for cervical cancer screening and results delivery. EV was also involved in providing feedback during the writing and revision stages of this manuscript. Joslin Ruiz, FNP-C, contributed to investigation and writing–review and editing. JR carried out the study investigation as a medical provider supporting study participants through the digital health aspects of at-home self-collection for cervical cancer screening and results delivery. ES was also involved in providing feedback during the writing and revision stages of this manuscript. Polina Segalova, PhD, contributed to conceptualization, data curation, formal analysis, investigation, methodology, project administration, supervision, and writing-review and editing. PS was involved in all aspects of designing and conceptualizing the study, establishing the selected methodology, and implementing the study. PS was also responsible for project administration and supervision, overseeing the investigation and data management and analysis. PS provided feedback during the writing and revision stages of this manuscript. Victor Su contributed to data curation, formal analysis, investigation, and writing–review and editing. VS was involved in quantitative data management and analysis, as well as carrying out study investigation procedures related to the engineering required of the digital health platform studied. VS also provided feedback during the writing and revision stages of this manuscript. Charlie Mouton contributed to conceptualization, investigation, methodology, project administration, and writing–review and editing. CM was involved in conceptualizing the study, establishing the selected methodology, and carrying out all aspects of the investigation, including user-facing design of the digital health platform studied. CM also supported project administration in getting the study underway and provided feedback during the writing and revision stages of this manuscript. Helena Lee contributed to investigation, project administration, and writing–review and editing. HL supported the investigation procedures and project administration, specifically leading operational support for the medical providers involved in this study and managing operational systems around patient-provider interactions. HL also provided feedback during the writing and revision stages of this manuscript. Lisa Santos contributed to investigation, formal analysis, and writing–review and editing. LS supported the investigation and analysis procedures, specifically creating materials needed for participant recruitment, co-creating interview protocols, interviewing participants, as well as supporting the analysis of interview transcripts. LS also provided feedback during the writing and revision stages of this manuscript. Meghna Mukherjee, PhD, contributed to conceptualization, data curation, formal analysis, investigation, methodology, project administration, writing-original draft, and writing-review and editing. MM was involved in conceptualizing and designing all aspects of the study, including establishing the selected methodology and managing participant recruitment. MM led project administration and investigation, including participant engagement, designing and carrying out surveys and interviews, data collection, data management, and analysis of qualitative and quantitative data. MM wrote the original draft of this manuscript and solicited and incorporated feedback from co-authors during the revision stages.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded entirely by Teal Health, Inc. [N/A grant number]. The authors (PS, VS, CM, HL, LS, and MM) are employees of the funder. The employees of Teal Medical Practice (ES, EV, and JR) were not compensated in addition to their clinical work for their contribution to this study. The authors were responsible for data collection, interpretation, and reporting.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors (PS, VS, CM, HL, LS, MM) are employees of Teal Health, Inc., the designer and manufacturer of the Teal Wand vaginal self-collection device and paired HIPPA-secure platform evaluated in this study, or Teal Medical Practice (ES, EV, JR), which is responsible for prescribing the Teal Wand and managing patient care.
