Abstract
Background
Advancements in clinical medicine depend on state-of-the-art healthcare analytics, which require patient data from electronic health record (EHR) systems and electronic data capture (EDC). We developed and implemented an independent, yet integrated EDC system (designated IMI-EDC) in a tertiary hospital. This system ensures that clinical data are available for patient care and research while complying with regulatory standards. In this study, we compared the first cohort of IMI-EDC users against those using the predecessor paper-mediated system, measuring patient comfort and satisfaction, data quality, and facilitation of workflow facilitation.
Methods
Until July 2025, more than 5000 patients were able to submit their medical history and health-related quality of life questionnaires via the IMI-EDC. All patients using tablets were surveyed regarding their user experience. Additionally, a subset of 100 EDC submissions was compared to 100 submissions via the paper-based system with respect to data completeness, correctness, clarity, and readability. Finally, the time to transfer data to the Heidelberg Pancreas Registry (HPR) research database was measured.
Results
IMI-EDC was successfully implemented at our center. Approximately three-quarters (75%) of patients using tablets were comfortable with the system. Tablet users were significantly more likely to submit completed documents than those using paper (98/100 vs. 66/100), and among submitted forms, fully completed questionnaires were more frequent in the tablet group (70/98 vs. 24/66). Data transfers to the HPR were quicker for tablet submissions than for paper submissions in a small pragmatic subsample (4.2 minutes vs. 9.9 minutes; 7 tablet vs. 7 paper cases).
Conclusions
IMI-EDC is satisfactory for patients and staff in terms of user experience, data completeness, and workload reduction in the setting of a high-volume surgical center.
Keywords
Background
Background and significance
Advancements in healthcare analytics have significantly improved clinical outcomes in recent decades. These advancements, which are based on electronic health records (EHRs) and electronic data capture (EDC) systems, offer insights into hospital processes and significantly broaden the breadth and depth of data available for healthcare and clinical research. 1 However, the translation of these developments into real-world clinical routines is often hindered by fragmented infrastructures, a lack of standards compliance, and a disconnect between data collected for care versus research.2,3 Patient-reported outcome measures (PROMs) are essential to understanding treatment impact and long-term quality of life, yet they are rarely captured in a structured, reusable form. 4 Most current EDC tools, such as REDCap, are tailored for research settings and not integrated into hospital EHRs. 5 Conversely, many large commercial EHR systems offer patient portals, but these often lack regulatory audit trails, study governance, and export standards such as CDISC ODM, thus limiting their suitability for multi-use documentation. Furthermore, even in advanced research environments where retrospective EHR data are harmonized to models like OMOP for secondary use, 6 real-time PROM integration and patient-facing digital documentation remain largely absent from routine workflows. To date, no vendor-independent, EHR-integrated EDC solution exists that allows structured patient-reported data to be collected once at the point of care and seamlessly reused across routine documentation, quality assurance, and research. This gap is especially critical in hospitals that lack advanced EHR functionality or encounter regulatory constraints that make direct research use of EHR data difficult.
In response, the Institute of Medical Informatics (IMI) at Heidelberg University Hospital developed a system-independent, yet EHR-integrated electronic data capture platform, IMI-EDC. Its architecture supports the triple use of data from a single source: integration into patient care via the EHR (1), quality monitoring via clinical registries (2), and secondary use for research, including AI-driven analytics (3). The system's design was based on input from 11 clinical disciplines and aims to overcome common limitations by enabling standards-based PROM capture, semantic annotation, and scalable governance across institutions. 7
Additionally, a dedicated development team at the IMI and at the European Pancreas Center (EPZ) of Heidelberg University Hospital created a direct data transfer interface between the IMI-EDC and the Heidelberg Pancreas Registry (HPR) research database, promoting an efficient information infrastructure that can increase the data flow between hospitals, laboratories, researchers, patients, and auditors.
In this article, we present its successful implementation and initial evaluation at the EPZ high-volume surgical center as part of the wider roll-out across the hospital. 7
General objectives
The objective of IMI-EDC implementation at the EPZ is to include high-quality, standards-based patient-reported outcome (PRO) data into the EHR and subsequently into a longitudinal study database. This system architecture will support medical staff in care processes, facilitate research on essential clinical questions, and provide patient-based data for quality management processes, thus enabling the reuse of routinely captured data for care, quality management, and research in line with established frameworks for secondary use of clinical data. 8 In this article, we describe the implementation in order to facilitate further expansion in similar settings.
Study objectives
The objectives of this study were to evaluate the successful implementation of IMI-EDC in clinical routines at the EPZ and to compare a pilot cohort of IMI-EDC users against those using the pen-and-paper-mediated predecessor system, as we hypothesized that the tablet version would be superior to the paper version. Therefore, the systems are compared in terms of (1) patient comfort and satisfaction, (2) data quality, and (3) workflow facilitation.
Methods
Institutional setting
Heidelberg University Hospital treats more than a million patients each year across more than 50 clinical departments. This trial was conducted at the affiliated EPZ in the Department of General, Visceral, and Transplantation Surgery. The EPZ includes five hospital locations with more than 140 surgeons and conducts over 700 pancreas surgeries and 3500 outpatient visits each year. The center's research registry includes clinical data from more than 10,000 surgical patients, from first encounter to postoperative follow-up. 9
Clinical setting
Pancreatic surgery is a routine and often essential therapy for various pathologies that are increasingly common at high-volume centers. 10 However, its success is limited by severe underlying diseases and risks of postoperative morbidity, mortality, and reduced quality of life.11,12 Regardless, the quality of pancreatic surgery has improved over recent decades, becoming safer and more effective.
These developments have been critically supported by retrospective analyses of clinical health records that identify risk factors related to patient attributes, diseases, and techniques.13–15 Such efforts demand the optimal capture, management, and analysis of relevant data on potential prognostic factors to enhance the translation of clinical practice into focused analyses and trials.3,9,16–18
System characteristics
The EPZ's patient data registry HPR, started in 2002, is managed and analyzed by a dedicated data manager with training in clinical informatics. It is an SQL-based relational database accessed via a forms-based front end. For 20 years, entries into the database were based only on a pen-and-paper-mediated system, utilizing reports filled out by patients (medical history and health-related quality of life) and clinicians (e.g. anesthesia protocol and discharge letter). Although all the paper documents were archived, paper-based information was introduced manually into the database by a departmental clerk only for selected patients and forms (primarily medical history). The workload demand of this procedure limited data transfers to selected patients and data forms (only medical history). Available paper data are mostly unstructured free-text data that were not amenable to immediate analysis. Furthermore, archived documents are difficult to retrieve. Thus, despite the value of the EPZ's study database, which is one of the most comprehensive datasets for this type of disease, its data management has been laborious with limited efficiency. Although the hospital uses Cerner i.s.h.med as its primary EHR, this system does not offer a patient-facing module for structured medical history and PROM capture, nor does it allow regulatory-compliant, study-specific data exports. Consequently, data capture relied on paper forms and manual transcription, a process prone to omissions, delays, and limited reusability.
IMI-EDC is a versatile data management solution designed for clinical use, research, and quality control.7,19 In practice, IMI-EDC resembles a graphical application for hosting questionnaires and entering data, with interfaces to clinical information systems. It integrates the hospital's existing EHR system, i.s.h.med (Oracle Cerner), with a vendor-independent customized open-source EDC platform (OpenEDC)19,20 that might also be used in other clinical locations. Ultimately, IMI-EDC ensures efficient data exchange and integrity across interoperable platforms to support triple primary data flows: (1) streamlining routine clinical data collection and decision-making, (2) facilitating researchers to use and supply hospital data, and (3) enabling auditors to trace data to its origin (Figure 1(a)).

Schematic representations of the data flow in (a) the general case of IMI-EDC and (b) this pilot trial in which patient-reported outcomes were collected. The arrows represent the flow of data between components (green: inputs; red: outputs; blue: data transfers between OpenEDC and i.s.h.med, black: generic data transfers). The grid background represents structural metadata attached to data objects within the system. eCRF: electronic case report form; IMI-EDC: Institute of Medical Informatics and electronic data capture.
While IMI-EDC functionality is geared toward the needs of clinicians and researchers, it especially provides opportunities to collect PROMs as the architecture operates as a client-based web application. This flexibility allows patients to complete questionnaires directly via intuitive interfaces on any mobile device, with identification through their respective patient ID barcodes. The web-survey format offers interactive features such as mandatory fields to prevent omissions, and skip-logic tailors questions based on prior responses. Moreover, the system can automatically calculate variables such as body mass index from patient inputs, while free-text responses are easily converted into structured formats. Finally, the digital format provides improved accessibility in terms of language and readability since it can host multilingual translations and allow adjustments to the typeface. All data fields can be semantically annotated.
EDC systems like IMI-EDC and EHR systems serve fundamentally different purposes. EDCs are built for clinical trials and research, ensuring that study-specific data are collected, cleaned, and managed in compliance with regulatory standards like Good Clinical Practice (GCP). They associate every data field with detailed study events (e.g. “pre-operative assessment”) rather than broader clinical encounters. Because of this, EDCs capture more granular, study-specific data than EHRs and are equipped with audit trails tailored for research purposes.
By contrast, routine-care EHR systems with their associated patient portals are designed for routine clinical care. Their focus is on documenting patient treatment, ensuring continuity of care, managing billing, and supporting flexible, encounter-based documentation. While EHRs may include some compliance features, they often lack ways for managing study-specific data, such as structured, de-identified exports in CDISC ODM format, which are required for regulatory submissions.
Importantly, IMI-EDC operates independently of the underlying EHR system. This separation allows the system to maintain study governance, protocol enforcement, and audit trail capabilities—regardless of which EHR is used at a site.
System developmental stage
The IMI implemented the customized IMI-EDC client-server infrastructure on the hospital's intranet, which has been used in the EPZ since September 2022. Patient data can be captured via a web-based graphical user interface on PCs or iPads (Apple, Inc.). Questionnaire responses hosted on IMI-EDC can be saved in portable document format (PDF) and loaded into the i.s.h.med interface via the Health Level 7 (HL7, version 2 21 ) communication server (Orchestra version 4.10, x-tention Informationstechnologie GmbH). Structured data can be directly uploaded from IMI-EDC to the existing HPR database by using the application programming interface (API) of IMI-EDC and realizing a script-based ETL (extract, transform, and load) process, enabling data transfer controlled by the EPZ clerks. While this combination of HL7 v2 messaging, PDF summaries, and API-based ETL was chosen pragmatically for the present implementation, our long-term goal is to extend IMI-EDC towards contemporary interoperability standards (e.g. HL7 FHIR resources and mappings to common research data models such as CDISC ODM and OMOP) to facilitate reuse across different clinical settings (see Future Developments). The electronic case report forms (eCRFs) used for data entry were obtained from the Medical Data Models (MDM) portal. 22 The MDM-Portal is a metadata registry in a standardized format for creating, analyzing, sharing, and reusing medical forms for research.
Implementation of IMI-EDC in routine clinical practice and the evaluation phase
We conducted a roll-out of IMI-EDC across the various settings of the EPZ (Figure 2). Validation of data capture, quality, and management via the IMI-EDC system was conducted by the clerk, a data manager, and two physicians from September 2022 to April 2023.

Study flowchart representing two implementation phases from July 2022 until July 2025.
Before the introduction of the tablets, patients received pen-and-paper forms of medical history and HRQoL questionnaires, which were manually entered into the scientific database by the departmental clerk if the patient was undergoing surgery; otherwise, they were archived. HRQoL paper questionnaires were archived for analysis later if needed. The workflow protocol for this trial began with the secure login of patients into the system, followed by the collection of patient-reported data. The data were uploaded to i.s.h.med in PDF format and to the local HPR database in XML format via manual validation by the clerk (Figure 1(b)), a process requiring several minutes per patient because of identity checks, visual verification of responses, and manual triggering of upload scripts. This explains the observed transfer times for both digital and paper-based data.
The first phase of the implementation occurred from 7 September 2022 to 13 March 2023 and involved the use of a single tablet running IMI-EDC. Upon hospital registration, all patients received hardcopy barcodes linked to their identity in i.s.h.med. A physician visited eligible patients while they awaited consultation and, after obtaining informed written consent, scanned barcodes via a tablet camera to log the patient into the IMI-EDC. After data entry, patients underwent consultation, where the attending physician validated the responses before the data were uploaded as PDFs into the EHR. Throughout this first phase, 336 outpatients used the IMI-EDC.
The tablet presented patients with two questionnaires: (1) a comprehensive medical history questionnaire covering 180 questions across 12 categories (e.g. demographics, reason for visit, medical history) and (2) a 30-item health-related quality of life (HRQoL) questionnaire (EORTC QLQ-C30 v3.0 23 ).
In the second phase of the implementation, an additional seven tablets were introduced to the clinic for routine implementation of IMI-EDC. From 14 March 2023 until July 2025, all outpatients received tablets upon registration. The registration staff logged patients into the IMI-EDC, after which patients completed the questionnaires independently and returned tablets to consulting physicians. The data was automatically uploaded into the EHR, and the tablets were redistributed at reception. During this period, 5315 patients used IMI-EDC, increasing the total number of interactions with the system from September 2022 until July 2025 to 5651 subjects.
Evaluation of patient satisfaction and preference
Patients who used tablets during the trial were asked to respond to two survey questions regarding their user experience: (1) Did you have any difficulties answering the questions via the tablet? (Not at All/Somewhat/Moderately/Very Much), (2) Do you prefer to answer such questions handwritten on paper or on the tablet? (Prefer Handwritten/No Preference/Prefer Tablet). Response frequencies were tested against the effects of patient age and gender via χ2 analysis. Confidence intervals (95%) for the proportion of responses in each age and gender group were calculated using the Wilson score method for binomial proportions. We decided to only use these two questions instead of a longer usability questionnaire in order to keep our clinical routine study design and avoid a study bias while obtaining a high response rate.
Evaluation of data quality
Since the evaluation of data quality was explorative, we chose a sample size of 100 subjects, who were retrospectively analyzed. We selected the first 100 subjects who used a tablet from September to November 2022 as our pilot cohort (tablet group). For comparison, we randomly selected 100 patients who did not use tablets from those patients attending the clinic during on the same days and being in the same age groups (paper group). The paper group included those individuals not attend by the study physician during the first tablet period, those who declined to use a tablet, or those unable to use tablets (e.g. tablet availability, minor technical issues, and health issues). These subjects completed medical history and HRQoL questionnaires using pen-and-paper, submitting documents to reception per prior procedure. We compared baseline patient variables between groups via a ranked, two-sided Mann‒Whitney U-test for median age and χ2 or Fisher's exact tests for categorical variables (latter used for gender and when any cells had <5 expected cases). Wilson score intervals with Sidak & Bonferroni corrections were used to calculate 95% confidence intervals for each group in all categorical comparisons except gender (provided intervals correspond with the expected proportions in the paper group. The interval for gender was calculated from the conditional maximum likelihood estimation and corresponds with the odds ratio. The 95% confidence interval for the median difference from the Mann-Whitney U-test was calculated with the Hodges-Lehmann estimator.
The number of medical history submissions from both groups were counted and graded according to the following criteria: (1) completeness (complete/small number of gaps/large number of gaps), (2) correctness (proper/small number of mistakes/large number of mistakes), (3) clarity (fully comprehensible/slight uncertainty/large uncertainty), and (4) readability (all readability and some text illegible). A third person reviewed the results. Groups were compared for each variable via the χ2 test or Fisher's exact test. The Wilson score interval was used to calculate 95% confidence intervals for each group (with respect to the paper group).
Indicative evaluation of work facilitation
The time to upload data from the IMI-EDC to the HPR SQL database was recorded for seven randomly selected cases. Since a clear trend toward the superiority of the tablet group was already apparent at the beginning of the evaluation, we decided to stop the quantitative collection and only pursue with giving an indication. Transferred data were prefilled by the IMI-EDC but visually confirmed and manually submitted by the clerk. All seven cases included medical history responses, whereas 6/7 included HRQoL responses.
For comparison, seven paper medical history forms were selected from patients who presented on the same days and were manually entered into the HPR SQL database by a departmental clerk. Medical history forms were included in all seven submissions, but HRQoL responses were included in only 4/7 transfers.
The volume of data transferred per patient was up to 180 items for medical history and 30 items for HRQoL. The average time taken to transfer the data was compared by t-test, and the variance was tested with the F-test.
Results
IMI-EDC was successfully implemented into clinical practice at the EPZ.
Patient characteristics
Subjects coming to the outpatient clinic presented with a range of pathologies, including pancreatitis, intraductal papillary mucinous neoplasms, cysts, carcinomas, and adenomas (Table 1). Most patients attended outpatient consultation and follow-up, with some receiving preoperative assessment. Patients using paper forms tended to be slightly older (median age = 67, range = 16–85) than tablet users (median age = 62, range = 19–85), based on a Mann-Whitney U-test (Z = 2.435, p = 0.015, 95% CI for median interval = 0.89–8.12). This difference may be related to patient preferences (see below) and capacities. Age group analysis indicated more paper users in the 70–79 range and more tablet users under 60, although the overall differences were not statistically significant according to χ2 tests. No other significant differences were observed between groups in terms of gender, diagnosis, presentation time, or reason for presentation.
Frequency of baseline characteristics for patients in the paper and tablet groups.

a Intraductal papillary mucinous neoplasm.
b Pancreatic neuroendocrine neoplasm.
Patient satisfaction and preference
A total of 463 patients were evaluated regarding their satisfaction and preferences. Difficulties using tablets varied significantly across age groups, with older patients reporting greater difficulty than younger patients did (Figure 3 and Supplemental Tables S1 to S5). Preferences for tablets versus paper also varied by age group, with patients under 50 showing a stronger preference for tablets, whereas older patients (age group 70–79) preferred the paper version. However, many patients expressed no clear preference. Usability did not significantly differ by gender, although males showed a greater preference for tablets than females did. Notably, preferences were closely linked to usability issues; patients who preferred paper often reported greater difficulties using tablets.

Difficulties with the tablet (n = 462 patients) and preference for tablet/paper (n = 463 patients) varied by age, gender, and by each other among all patients using tablets.
Data quality
Tablet users submitted their medical history questionnaires significantly more frequently than did paper users (Figure 4 and Supplemental Tables S6 to S10). Only 66/100 subjects from the paper group submitted their medical history documentation, compared to 98/100 subjects from the tablet group. Furthermore, tablet users provided significantly more complete documentation compared to paper users. Specifically, 71% (95% CI: 62%–79%) of the tablet cases were complete, with only 5% (95% CI: 2%–11%) containing large gaps. In contrast, only 36% (95% CI: 26%–48%) of submitted paper forms were complete, and 24% (95% CI: 16%–36%) contained large gaps. Overall, 70/100 tablet cases were both submitted and complete, whereas only 24/100 paper cases met both criteria. The grading system used was pragmatic and not formally validated; however, the majority of submissions from both groups were classified as correct, clear, or readable, with no significant differences found between groups according to Fisher's tests (Figure 4). The third person who reviewed the comparison results received no disagreements to be solved, thus no inter-rater reliability was performed.

Data quality assessment for medical history submissions from paper and tablet groups. The proportions given for completeness, correctness, clarity, and readability are relative to the number of submitted questionnaires and not the total sample.
Work facilitation
In a small pragmatic subsample of seven tablet and seven paper cases, the transfer of data from completed submissions to the HPR SQL database was, on average, approximately twice as fast for tablet forms as for paper forms. Tablet transfers took a mean of 4.2 ± 0.7 minutes, compared with 9.9 ± 1.1 minutes for manually entered paper transfers (median 4.3 vs. 10.3 minutes). Given the modest sample size, these observations should be interpreted as indicative evidence of workflow facilitation rather than as precise quantitative effect estimates. Notably, the quicker transfer of tablet forms even included HRQoL responses in 6/7 cases, compared with 4/7 cases for the paper group.
Discussion
Principal results
IMI-EDC was successfully integrated into the outpatient setting of a high-volume pancreas center. The implementation process was smooth, and staff accepted the workflow within a month. Initially, physicians and administrative staff provided close support during the single-tablet pilot phase, but personal support became less necessary as the multi-tablet phase progressed. This indicates a trend toward unsupervised and spontaneous institutional adoption, demonstrating the feasibility of implementing such procedures and devices in a controlled clinical setting. Rollout of IMI-EDC in further clinical domains showed similar implementation processes and results.
Following this introduction, the EPZ can utilize a single-source triple-flow information system to support the center's surgeons and staff effectively while also providing usability for researchers and regulatory auditors.
An essential feature of the IMI-EDC is its ability to gather PROMs that are crucial for evaluating diagnoses, interventions, and checking biases22,23 while offering benefits over pen-and-paper methods because of its interactive and user-friendly digital forms. This ease-of-use might likely result in higher completion rates and more data, as paper forms are time-consuming, and patients often forget or lose them. Before the IMI-EDC, most patient-reported documents, including HRQoL questionnaires, were archived without being used, as similarly reported in literature from other sites.24,25 Currently, medical history and HRQoL documentation are available for most patients. Although some patients may need assistance or prefer paper forms, data capture with IMI-EDC can establish robust, patient-reported baselines and trajectories.
Prior work has shown that systematic capture and feedback of PROMs in routine oncology care can improve symptom control and even survival. 4 In our context, more complete and rapidly available medical history and HRQoL data could support earlier risk stratification, better informed shared decision-making, and more targeted follow-up of high-risk patients after pancreatic surgery. However, our study was not designed or powered to test such downstream effects, and we did not analyze endpoints such as postoperative morbidity, mortality, readmissions, or longitudinal HRQoL trajectories. These hypotheses should, therefore, be addressed in future prospective studies building on the infrastructure described here.
Future developments
The success of the data collection process involving IMI-EDC and API transfer to the SQL database has prompted the initiation of an extension to the integrated collection of structured data from the anesthesiologic anamnesis, imaging findings, and the inpatient clinical course. After the pilot trial, the utilization of IMI-EDC for the HPR database has grown to include over 5000 patient encounters within 2 years (Figure 5).
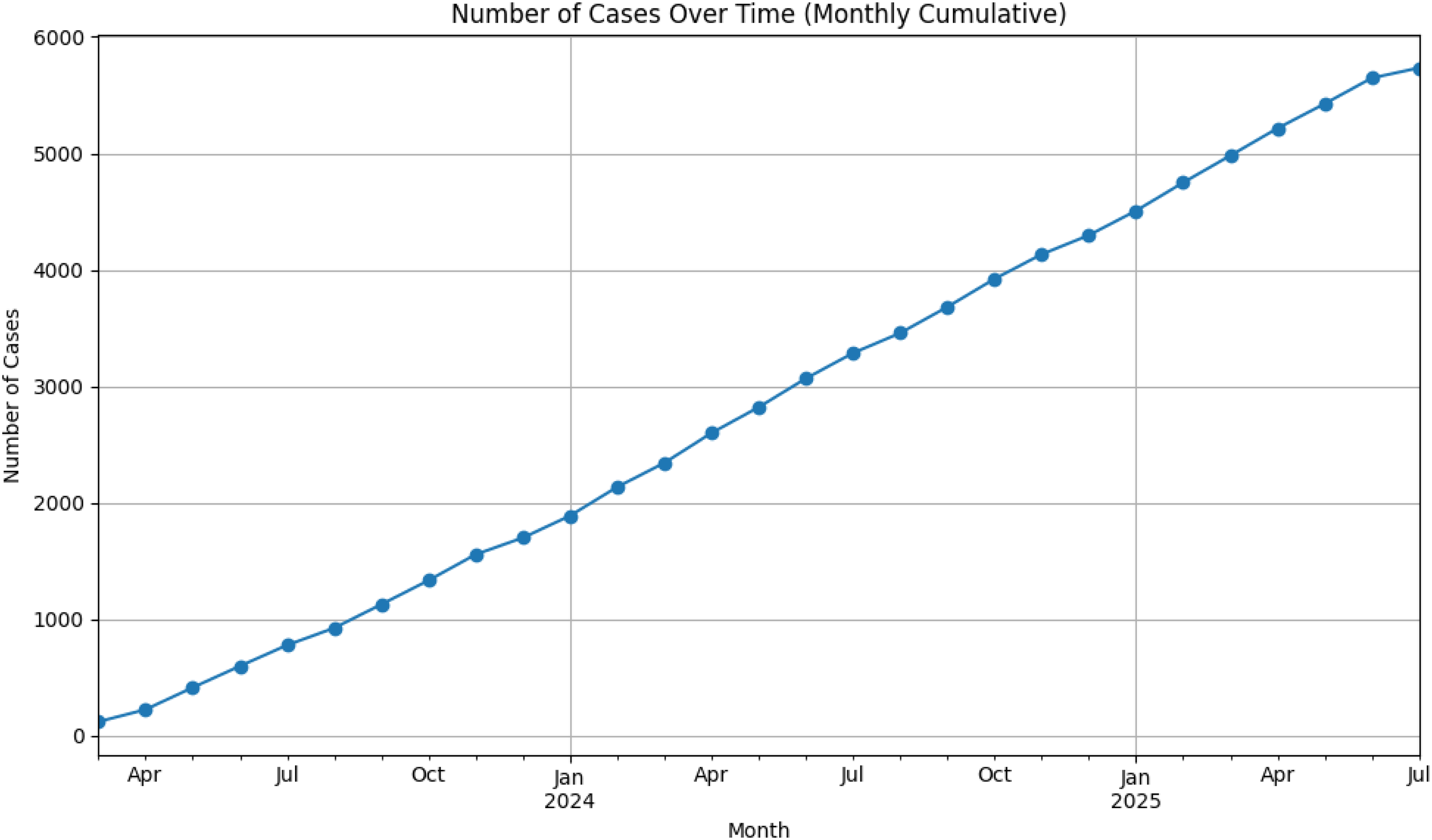
Pancreatic outpatient cases in the Institute of Medical Informatics and electronic data capture (IMI-EDC) over time from April 2023 to July 2025.
IMI-EDC will undergo further technical improvement to integrate EDC and EHR systems through data mapping and transformation of the interoperable format of IMI-EDC. This includes the capacity to directly call specific data elements between systems, enabling clinicians to source patient data from studies and enabling researchers to populate datasets from qualified hospital records. Other refinements will improve the user experience and expand interoperability beyond EHR and EDC applications, offering customized exports for third-party software.
An integrated, standardized, and enhanced data management system significantly advances research capabilities. While IMI-EDC supports common statistical software, its greatest utility lies in serving well-structured data at scale for modern and future data science applications. Modern machine learning algorithms are set to transform healthcare analytics and surgery. 26 An advanced database leveraging AI can answer important clinical questions, particularly in the context of pancreatic diseases and their treatment at the EPZ. Most healthcare organizations currently lack the infrastructure necessary for training and utilizing AI. 27 However, the IMI-EDC and its data management reforms are designed to streamline data preparation for advanced AI research. Additionally, all the data handled by IMI-EDC are of high quality and align with FAIR principles for scientific data management and stewardship, which emphasize data Findability, Accessibility, Interoperability, and Reusability. 28 These design choices are aligned with ongoing European initiatives around the European Health Data Space (EHDS), which aim to enable secure reuse of interoperable health data for both care and research (Regulation (EU) 2025/327; available at: https://eur-lex.europa.eu/eli/reg/2025/327/oj).
Limitations
This pilot study has several limitations. First, it was conducted in a single high-volume pancreatic surgery center at a university hospital with a specific EHR environment. Extrapolation to other hospitals, especially those with different EHR systems or more limited informatics support, may therefore be restricted, and the generalizability of our findings is limited.
Second, allocation to tablet versus paper completion was not randomized. Although we sampled a comparison group of paper users matched by visit date and age group, unmeasured differences in digital literacy, health status, staffing, or clinical context may have influenced both the choice of documentation mode and the quality of submitted data. This convenience sampling introduces potential selection bias and limits causal interpretation of the observed between-group differences.
Third, the usability assessment relied on two short survey questions rather than a standardized instrument such as the Technology Acceptance Model (TAM) or System Usability Scale (SUS). We deliberately chose this minimal approach to avoid overburdening patients who already completed extensive medical history and HRQoL questionnaires in a real-world clinical setting. While this enabled a very high response rate, it provides only a coarse measure of user experience. More detailed usability testing, including qualitative interviews and validated scales, is warranted in future studies. In addition, stratification of patient-specific confounders was limited to age and gender; we did not capture socioeconomic variables or detailed comorbidity indices that may affect digital tool use. Patients with severe visual impairment were supported by staff, but usability and autonomy in this subgroup could not be evaluated systematically.
Finally, the indicative evaluation of workflow facilitation was based on a small sample of seven tablet and seven paper cases. This pragmatic sample reflects the real-world nature of the study and the substantial time investment required from the departmental clerk. The large effect size observed in transfer times suggests that the qualitative conclusion that IMI-EDC can reduce documentation time is robust; nevertheless, the small sample size precludes precise estimates and subgroup analyses. Future work should therefore include larger samples and prospective time-motion studies to confirm the magnitude of the workflow benefit.
From a technical point of view, there are also limitations, especially when it comes to data exchange. For data exchange and archival, IMI-EDC relies on HL7 Version 2 messaging and PDF-based document formats. While more recent standards, such as FHIR and models, such as CDISC ODM, OMOP, or openEHR, offer richer semantic interoperability, they were not adopted in this work because the HL7 V2 TCP-based API of the institutional communication server (Orchestra) represents the main mechanism for integrating third-party applications with the hospital information system at our site. The communication server is directly integrated with the Hydmedia archive, enabling reliable storage and retrieval of PROM documents as PDFs within existing clinical workflows. Acknowledging the limitations of text-only document-based approaches, we have already started developing a solution that combines structured, standardized FHIR data with human-readable documents by embedding FHIR resources into PDF/A-2 files. However, this approach has not been implemented or evaluated as part of the present work.
Comparison with prior work
Several groups have reported EDC implementations in outpatient and perioperative settings, often demonstrating good usability and data quality. 29 However, these systems are typically deployed within individual research projects and are not tightly integrated with the hospital EHR. REDCap (Research Electronic Data Capture), for example, is widely used for research data collection and has been evaluated with respect to user preferences and usability.5,29 Although REDCap provides APIs and web services that can be used to interface with EHR systems in some institutions, these connections usually require substantial local customization and are not part of routine clinical documentation in many hospitals. Conversely, commercial EHRs frequently offer patient portals for questionnaire completion, yet they often lack study-centric governance, granular audit trails, and standards-based export formats such as CDISC ODM that are required for multi-center research and regulatory audits. In contrast, IMI-EDC was designed from the outset as a vendor-independent, EHR-embedded infrastructure that systematically feeds structured patient-reported data into both routine documentation and quality registries, thereby operationalising the “single source–triple flow” concept within a clinical specialty context.
Conclusions
This study showed the successful implementation of a vendor-independent, yet EHR-integrated EDC system that captures structured patient-based data in a user-friendly manner and thus includes the patient's voice into clinical care and research. IMI-EDC facilitates seamless transfer from a unified data source to databases accessible to surgeons, clinical researchers, and regulatory auditors without causing additional workload to clinical staff.
Supplemental Material
sj-pdf-1-dhj-10.1177_20552076261416337 - Supplemental material for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients
Supplemental material, sj-pdf-1-dhj-10.1177_20552076261416337 for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients by Max Blumenstock, Fleur Fritz-Kebede, Niels Siegel, Nathalia Giese, Ulrike Heger, Christian Niklas, Tobias Dittrich, Nelly Zental, Matthias Ganzinger, Christoph U Lehmann, Martin Loos, Ulf Hinz, Martin Dugas and Thomas M Pausch in DIGITAL HEALTH
Supplemental Material
sj-pdf-2-dhj-10.1177_20552076261416337 - Supplemental material for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients
Supplemental material, sj-pdf-2-dhj-10.1177_20552076261416337 for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients by Max Blumenstock, Fleur Fritz-Kebede, Niels Siegel, Nathalia Giese, Ulrike Heger, Christian Niklas, Tobias Dittrich, Nelly Zental, Matthias Ganzinger, Christoph U Lehmann, Martin Loos, Ulf Hinz, Martin Dugas and Thomas M Pausch in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076261416337 - Supplemental material for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients
Supplemental material, sj-docx-3-dhj-10.1177_20552076261416337 for Single source – triple flow: Structured electronic data capture for pancreatic surgery patients by Max Blumenstock, Fleur Fritz-Kebede, Niels Siegel, Nathalia Giese, Ulrike Heger, Christian Niklas, Tobias Dittrich, Nelly Zental, Matthias Ganzinger, Christoph U Lehmann, Martin Loos, Ulf Hinz, Martin Dugas and Thomas M Pausch in DIGITAL HEALTH
Footnotes
Abbreviations
Acknowledgements
This publication was supported through state funds approved by the State Parliament of Baden-Württemberg for the Innovation Campus Health + Life Science alliance Heidelberg Mannheim. EPZ departmental clerk: Andrea Teichgräber. Medical writing consultation for text edits and visuals: Stephen Heap (https://www.drstevilphd.com).
Ethical approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the medical faculty at the University of Heidelberg (S301/2001; S708/2019; S083/2021). Written informed consent to participate was obtained from all ents by medical staff during the clinical process.
Consent for publication
Not applicable.
Author contributions
MB, FF-K, UHi, and TMP: conceptualization; MB, FF-K, UHi, CN, and TMP: methodology; NS, UHeg, TD, NZ, MG, NG, and CUL: validation; MB, FF-K, UHi, and TMP: formal analysis; MB, FF-K, UHi, and TMP: investigation; ML and MD: resources; UHi and TMP: data curation; MB and TMP: writing–original draft preparation; MB, FF-K, MD, NS, UHeg, TD, NZ, MG, NG, CUL, ML, UHi, MD, and TMP: writing–review and editing; TMP: visualization; ML and MD: supervision; MD: project administration. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data that is not part of the manuscript may be obtained from the corresponding author upon reasonable request.
Clinical trial number
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
