Abstract
Background
Regular monitoring of blood pressure (BP) provides early detection of hypertension. Automated cuff-based oscillometric devices are commonly used for simplicity and reduced observer subjectivity. However, these devices can be uncomfortable, prompting the development of non-invasive, cuffless BP monitoring in smartwatches. Current validation standards, including ISO 81060-2:2018 and IEEE 1708TM-2019, specify accuracy criteria, yet most studies focus on short-term performance and do not address their long-term stability across the calibration period.
Objective
This prospective single-arm study aimed to assess the long-term accuracy and stability of BP measurements obtained from a smartwatch throughout its recommended calibration interval.
Methods
Thirty-seven participants completed a 28-day protocol, consisting of an initial calibration on day 0 followed by 27 consecutive days of paired measurements of systolic BP (SBP) and diastolic BP (DBP). BP was simultaneously measured using a Samsung Galaxy Watch 5 and a validated reference sphygmomanometer Omron M4. Accuracy was assessed against ISO and IEEE standards, and the recalibration test consistent with European Society of Hypertension (ESH) guidelines was performed using the last three measurements before scheduled recalibration.
Results
The smartwatch met the accuracy criteria based on ISO and IEEE standards in all but one measure, with mean absolute differences, mean differences (MD), and standard deviations for SBP and DBP within acceptable limits. Bland-Altman analysis revealed negligible MD for SBP (–0.34 mmHg) and DBP (0.62 mmHg), with minimal drift over the calibration period (–0.19 mmHg for SBP and 1.02 mmHg for DBP). However, when reference BP was 10 mmHg away from the calibration point, MD was 3.4 mmHg for SBP and 5.1 mmHg for DBP.
Conclusions
The smartwatch demonstrated acceptable long-term stability and accuracy for BP monitoring and trend tracking. However, accuracy declined as values diverged from the calibration point. Cuff-based confirmation is advised when BP fluctuates substantially or when diagnostic or therapeutic decisions are planned.
Trial Registration
ClinicalTrials.gov NCT06098092
Introduction
Hypertension is one of the leading risk factors for developing cardiovascular disease. 1 Regular monitoring of blood pressure (BP) provides an early warning of hypertension. 2 While the auscultatory method is the gold standard for non-invasive BP measurement, 3 the automated oscillometric method is the most widely used to reduce subjective error and simplify measurement.4,5 A notable advantage of automated methods is the possibility of BP self-monitoring. Given the inherent variability of BP values over a day, depending on the mental and physical state of the individual, self-monitoring of BP by the patient is desirable. The phenomenon known as “white coat syndrome” is another rationale for BP self-monitoring, as BP readings may be higher in a hospital environment.6,7 One of the disadvantages of cuff-based oscillometric sphygmomanometers is the discomfort experienced by some patients when inflating the cuff, for example, due to high BP or during sleep.8,9 In some patients, the mere presence of the cuff can cause stress and an increase in BP. This problem is eliminated with automatic cuffless BP measurement based on photoplethysmography, which is currently implemented in many wearable devices such as smartwatches or smartphones.5,10 Other advantages of these devices include ease of use with minimal intervention and the associated ease of repeat BP measurements.
The accuracy of BP measurement of many smart devices currently available on the market remains unconfirmed. 9 A study of the Australian online marketplace revealed the existence of over 500 different cuffless BP measuring devices, the accuracy of which has not been validated using appropriate protocols. 11 The situation is further complicated by the absence of a universally accepted, standardized procedure for verifying the accuracy of automatic cuffless BP measurement. Consequently, most existing studies employ the ISO 81060-2:2018 standard (Non-invasive sphygmomanometers: Part 2: Clinical Investigation of Intermittent Automated Measurement Type). 12 This standard addresses the requirements and methods for clinical testing of a device used for intermittent non-invasive and automated estimation of arterial BP using a cuff. The standard indicates that a minimum of 85 participants shall be enrolled in the full validation study, a minimum of 255 paired BP values are recommended to be measured, and that a mean difference (MD) between the device under test and the reference used shall belong to an interval of ±5 mmHg, with a standard deviation (SD) of no more than 8 mmHg. Some studies refer to the IEEE Standard for Wearable, Cuffless Blood Pressure Measuring Devices (IEEE Std 1708TM-2014 and its first update, IEEE Std 1708aTM-2019), which provides guidelines for the objective evaluation of BP measurement accuracy by cuffless devices.8,13 The standard introduces the mean absolute difference (MAD) parameter as an accuracy assessment criterion, requiring a MAD of less than 7 mmHg for SBP and DBP analyzed separately.
Ambulatory blood pressure monitoring (ABPM) has frequently been applied to compare the accuracy of smart device BP measurement with cuffless devices, relying on data collected over a single day and therefore capturing only short-term accuracy.5,14–18 For example, Falter et al. evaluated the accuracy of the Samsung Galaxy Watch Active 2 (Samsung Electronics Co., Ltd., Suwon-si, South Korea) in 40 healthy subjects and reported MD ± SD of −2.05 ± 15.5 mmHg for systolic BP (SBP) and −5.58 ± 22.5 mmHg for diastolic BP (DBP). 5 Therefore, the smartwatch in this study only met the requirements of ISO 81060-2:2018 for the MD in SBP. The accuracy requirements of the standard were fully met in the study by Ahn et al. 14 in which Samsung Galaxy Watch 3 was verified in 56 patients with Parkinson's disease. The study reported that the MD ± SD in SBP and DBP were 0.4 ± 4.6 mmHg and 1.1 ± 4.5 mmHg, respectively. 14
A limited number of studies have focused on evaluating the long-term accuracy and stability of BP measurements in cuffless devices. A study by Han et al. 19 evaluated the stability of BP measurement in the Samsung Galaxy Watch 3 and Samsung Galaxy Watch Active 2 smartwatches. A total of 760 participants who provided data from at least 20 days of device use were included in the study. During the study period, 574 participants performed recalibration with the automated upper-arm cuff BP monitor, but no reference measurement was performed during daily routine measurements. Measurement stability was then evaluated as the difference between the 7-day average SBP before and after recalibration. For SBP, the difference was 6.8 ± 5.6 mmHg, and in hypertensive subjects it increased to 9.4 ± 7.1 mmHg. The study showed that 15 days after calibration, the difference in SBP became statistically significant, with this difference increasing linearly by 0.022 ± 0.002 mmHg for each additional day. A study by Liu et al. 20 evaluated the accuracy of cuff-calibrated and calibration-free smartwatch models from Huawei Technologies Co, Ltd. A total of 3077 participants were enrolled in the study, and each participant measured their BP three times a day over four days within one month (1st, 7th, 14th, and 21st day). A cuff-calibrated smartwatch model achieved lower variability than a calibration-free model, with the MD ± SD of 2.31 ± 9.57 mmHg for SBP and 1.33 ± 6.43 mmHg for DBP compared to an auscultatory reference sphygmomanometer. The MD ± SD for SBP decreased significantly to 1.97 ± 7.85 mmHg when the hypertensive subjects were excluded. For the model without calibration, the MD ± SD was calculated to be −0.71 ± 13.04 mmHg for SBP and −0.29 ± 8.78 mmHg for DBP. Liu et al. 20 found BP measurement by a Huawei cuff-calibrated smartwatch to be effective for measuring DBP in all participants and for measuring SBP in the normotensive population. Although the studies were conducted over multiple days and included many subjects, the measurements were not taken at regular intervals throughout the calibration period of the smartwatch or compared simultaneously with a reference method.
The aim of our study was to verify the long-term accuracy and stability of smartwatch BP measurements at regular intervals throughout the designated calibration period of the smartwatch.
Methods
Ethical considerations
This interventional prospective single-arm study was approved by the Ethics Committee of the Faculty of Biomedical Engineering of the Czech Technical University in Prague on May 15, 2023 (No. C41/2023). The study was registered on the ClinicalTrials.gov database (No. NCT06098092). All participants provided written informed consent before their enrollment in the study and could withdraw from participation at any time without reprisal. Participation in the study was voluntary, and no compensation was provided to participants. The data of all the participants were de-identified before the final processing and publication.
Participants
We determined the minimum sample size for the study based on the ISO standard 12 to be 35 participants. This number was slightly increased to prepare for possible withdrawals. Participants were recruited via personal networks of the authors. Any volunteer over 22 years of age was eligible for the study. The exclusion criteria were mainly based on the recommendations of the smartwatch manufacturer (Samsung Galaxy Watch 5 User Manual, Samsung Electronics Co., Ltd., Rev. 1.0, August 2022). These included acute illness, diabetes, 21 pregnancy, 22 and end-stage renal disease. 23 Neurotic disorders associated with involuntary movement (e.g., tremor or shivering), post-traumatic upper limb conditions, and visible skin alterations (e.g., tattoos, birthmarks, injuries, or dermatological disease) on the left wrist or hand were also considered exclusion criteria, as these factors can interfere with the smartwatch's optical sensor. Exclusions related to cardiovascular health 24 included cardiac arrhythmias, circulatory or peripheral vascular disease, valvular or other structural heart diseases (including previous myocardial infarction), cardiomyopathy, hemostatic disorders, and current use of anticoagulant medication. Three participants were excluded during the enrollment for the following reasons: one for diabetes (in accordance with the smartwatch manufacturer's instructions), one for pregnancy, and one for tattoos in the wrist area of the left hand. A total of 42 participants (21 men, 21 women) aged 22–80 years participated in the study, a number consistent with the European Society of Hypertension Working Group on Blood Pressure Monitoring and Cardiovascular Variability recommendations for the validation tests (ESH recommendations). 25 Five participants discontinued daily smartwatch measurements during the calibration period and were therefore excluded from the final analysis. The characteristics of the study participants are shown in Table 1.
Characteristics of the study participants.
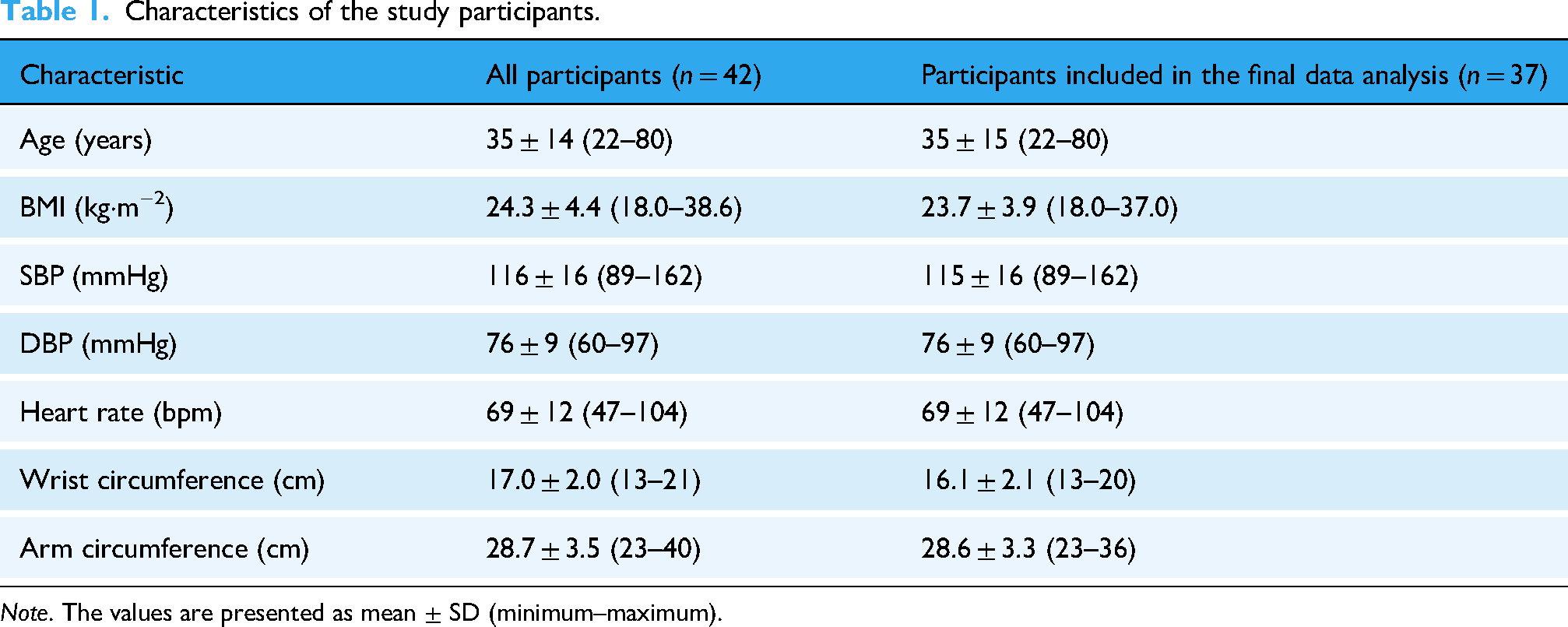
Note. The values are presented as mean ± SD (minimum–maximum).
Procedures
Our study employed the Samsung Galaxy Watch 5 smartwatch (Samsung Electronics Co., Ltd., Suwon-si, South Korea) with all participants. The device estimates BP from a wrist photoplethysmography signal using pulse-wave analysis calibrated to a cuff-based reference. The measurement protocol consisted of two phases, both of which took place in the participants’ home environment. The first phase (day 0) included verification of the interarm BP difference—referred to as the lateral difference (LD) 12 —and calibration of the smartwatch BP measurement. This phase was carried out in the presence of a specialist in the field, who also provided training in the correct practice of BP measurement, explained the measurement protocol to study participants, and provided a predefined document to record all measured data. To obtain the LD, the participants were seated comfortably with both hands placed on the table in front of them and underwent three simultaneous measurements on opposite arms using two validated Omron M4 Intelli IT digital sphygmomanometers (Omron Corporation, Kyoto, Japan). 26 LD was less than 15 mmHg for SBP and less than 10 mmHg for DBP in all participants, meaning that no participant was excluded from the study on this basis. 12 Mean LD for SBP and DBP for each participant is available as Supplementary Material.
For smartwatch BP measurement calibration, a cuff of the Omron M4 Intelli IT digital sphygmomanometer was placed on the participant's right arm. This digital sphygmomanometer with an accuracy of ±3 mmHg was used as a reference. The smartwatch was placed on the participant's left forearm. The participant was asked not to consume alcohol, caffeine, or nicotine products, not to exercise or take a bath for at least 30 min before the calibration. The participant was then instructed to remain quiet and sit still with their arms comfortably placed on a table, at approximately heart level. They were told to breathe normally during the measurement. The BP calibration was performed according to the user manual and the instructions of the Samsung Health Monitor application (Samsung Electronics Co., Ltd., Suwon-si, South Korea); the measurement on the smartwatch and the reference sphygmomanometer were started simultaneously, and when the smartwatch finished the measurement and the reference sphygmomanometer displayed SBP and DBP, the values were entered into the Samsung Health Monitor application. The calibration was completed after three successful BP measurements.
In the second phase, the participant measured BP regularly every morning and evening for 27 days using the smartwatch and the reference sphygmomanometer, following the same instructions as for the calibration procedure in the first phase. The time delay between the smartwatch measurement and the corresponding reference sphygmomanometer measurement should be less than 60 s to ensure their consistency, in line with the IEEE standard. 8 The obtained SBP, DBP, and heart rate (HR) values were recorded on a predefined document. Thus, each participant was measured for 28 consecutive days, including the first phase (day 0) followed by 27 consecutive days of paired BP measurements, which is the valid calibration period of the smartwatch.
Data analysis
Data from the participants who completed the entire measurement protocol were included in the final data analysis. All data were analyzed in Matlab R2021b (MathWorks, Natick, MA, USA), with a p-value of less than 0.05 considered statistically significant.
First, we evaluated compliance with the selected ISO/ESH/IEEE validation requirements8,12,25 and classified each criterion as fulfilled, partially fulfilled, or not fulfilled. Then, MD, SD, and MAD were calculated for SBP, DBP, and HR and the difference between the smartwatch and the reference sphygmomanometer was assessed by a paired t-test.
Next, two criteria of BP measurement accuracy were tested according to the ISO standard. 12 According to Criterion 1, the required MD of all measurements for both SBP and DBP must be less than or equal to ±5 mmHg and corresponding SD must be no more than 8 mmHg. Criterion 2 evaluates the variability in the bias of the tested device across participants. It is calculated as the SD of averaged individual differences in SBP or DBP. The ISO standard specifies that the maximum acceptable value of this SD decreases as the MD of all measurements increases, ranging from 6.95 mmHg for an MD of ±0.0 mmHg to 4.79 mmHg for an MD of ±5.0 mmHg. We also classified smartwatch performance using the MAD grading: A ≤ 5 mmHg, B > 5–6 mmHg, C > 6–7 mmHg, and D ≥ 7 mmHg (unacceptable).
To validate the stability of cuff calibration accuracy over time, we performed the recalibration test according to the ESH recommendations. 25 Three valid paired BP measurements must be obtained for each participant before the recalibration for the test. Thus, we used the last three measurements of the calibration period (i.e., the evening of day 26 and the morning and evening of day 27).
For graphical assessment of the measured data, the Bland-Altman analysis was conducted to evaluate the agreement between the smartwatch and the reference sphygmomanometer for the obtained SBP and DBP values, as recommended by the IEEE standard.8,13 The 95% limits of agreement were calculated as the mean difference ± 1.96 standard deviations.
Next, we used least-squares linear regression to (1) to evaluate the long-term stability of the smartwatch measurements by fitting the difference between the smartwatch and the reference sphygmomanometer as a function of the day of measurement and (2) to evaluate the difference between the smartwatch and the reference sphygmomanometer as a function of the difference between the calibration point and the reference sphygmomanometer. Finally, we calculated the cumulative distribution function (CDF) of the differences between the smartwatch and reference sphygmomanometer for different levels of the difference between the calibration point and the reference sphygmomanometer.
Results
The study was conducted in the participants’ home environment in the Czech Republic from October 2023 to October 2025. Of the original 42 participants enrolled in the study, 37 participants completed the entire experiment according to the instructions in the measurement protocol with no missing data, resulting in a total of 1998 paired values (smartwatch vs. sphygmomanometer) for each of SBP, DBP and HR. Based on the subject selection requirement of the IEEE standard, 8 21 participants were normotensive, 13 prehypertensive, and 3 hypertensive. Compliance with the selected ISO/ESH/IEEE validation requirements is summarized in Table 2.
Compliance with the selected ISO/ESH/IEEE requirements for BP measurement accuracy validation.
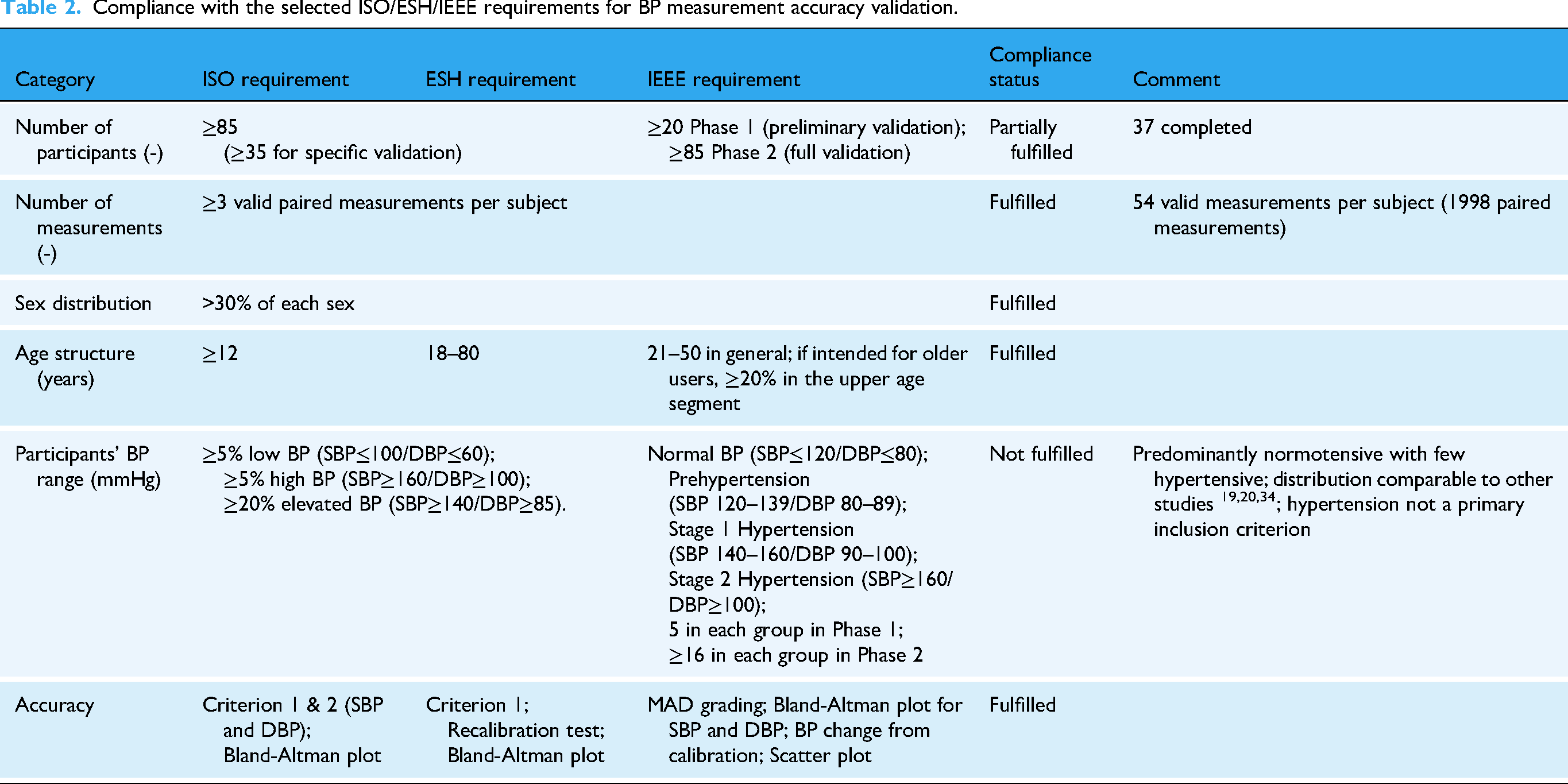
The MD ± SD between the smartwatch and the reference sphygmomanometer were −0.34 ± 8.10 mmHg for SBP, 0.62 ± 7.07 mmHg for DBP, and −0.01 ± 3.71 bpm for HR. The paired t-test indicated a statistically significant difference between the smartwatch and the reference sphygmomanometer for DBP, whereas no significant differences were observed for SBP and HR. The MD and SD values practically met Criterion 1 of the ISO standard, 12 with only a marginal deviation in SD for SBP. The MAD was less than 7 mmHg for SBP and DBP, corresponding to grade C for SBP and grade B for DBP. The results, including MAD values, are presented in Table 3.
Comparison of readings between the smartwatch and sphygmomanometer across all participants and days of measurement.

A statistically significant difference in MD (paired t-test) between smartwatch and the reference sphygmomanometer.
For Criterion 2 of the ISO standard, the SD of averaged individual differences was 4.67 mmHg for SBP and 4.59 mmHg for DBP, thus meeting the requirements. Individual MD ranged from −10.43 mmHg to 11.63 mmHg for SBP and from −10.63 mmHg to 9.04 mmHg for DBP. Individual SD of the difference ranged from 3.55 mmHg to 11.26 mmHg for SBP and from 3.07 mmHg to 8.80 mmHg for DBP.
In the recalibration test according to the ESH recommendations, the MD ± SD between the smartwatch and the reference sphygmomanometer were −0.08 ± 7.09 mmHg for SBP (p = 0.90) and 0.71 ± 6.06 mmHg for DBP (p = 0.22), meeting Criterion 1 again. The 95% confidence intervals for the MD were (−1.42, 1.25) mmHg and (−0.43, 1.85) mmHg, respectively.
Figure 1(a) and (b) presents Bland-Altman plots that evaluate the MD and limits of agreement between the smartwatch and the reference sphygmomanometer, using pooled simultaneous SBP and DBP readings. The MD was −0.34 mmHg for SBP and 0.62 mmHg for DBP, with 95% limits of agreement ranging from −16.22 to 15.54 mmHg for SBP and from −13.24 mmHg to 14.48 mmHg for DBP.

Differences between pooled simultaneous BP readings from the smartwatch and the reference sphygmomanometer for SBP (a) and DBP (b). The solid line is the MD of the measurements. Dashed lines are the 95% limits of agreement. The markers in the graph are sized according to the frequency of observation of paired BP readings.
The time course of the difference between the smartwatch and the reference sphygmomanometer shows that the trendline for SBP (Figure 2(a)) deviates insignificantly from the difference of 0 mmHg (calibration on day 0) by −0.01 mmHg per day (p = .53), resulting in an average bias of −0.19 mmHg at the end of the calibration period. For DBP (Figure 2(b)), the trendline deviates from 0 by 0.04 mmHg per day (p < .01), resulting in an average bias of 1.02 mmHg at the end of the calibration period.

Time course of the difference between the smartwatch and the reference sphygmomanometer for SBP (a) and DBP (b). The crosses represent the MD for the given time of the measurement and the circles in the graph are sized according to the frequency of observation of paired BP readings.
When the BP, as measured by the reference sphygmomanometer, deviated from the calibration point, the difference between the smartwatch and the reference sphygmomanometer increased with the distance from the calibration point for both SBP (Figure 3(a)) and DBP (Figure 3(b)). For example, if SBP was 10 mmHg different from the calibration point during a BP measurement, the difference between the smartwatch and the reference sphygmomanometer was 3.4 mmHg on average (y = 0.34x). For DBP, the difference was larger, with a 10-mmHg deviation from the calibration point resulting in a mean difference of 5.1 mmHg (y = 0.51x). Correlation coefficients of 0.43 for SBP and 0.55 for DBP indicate a moderate relationship negatively affected by significant outliers.
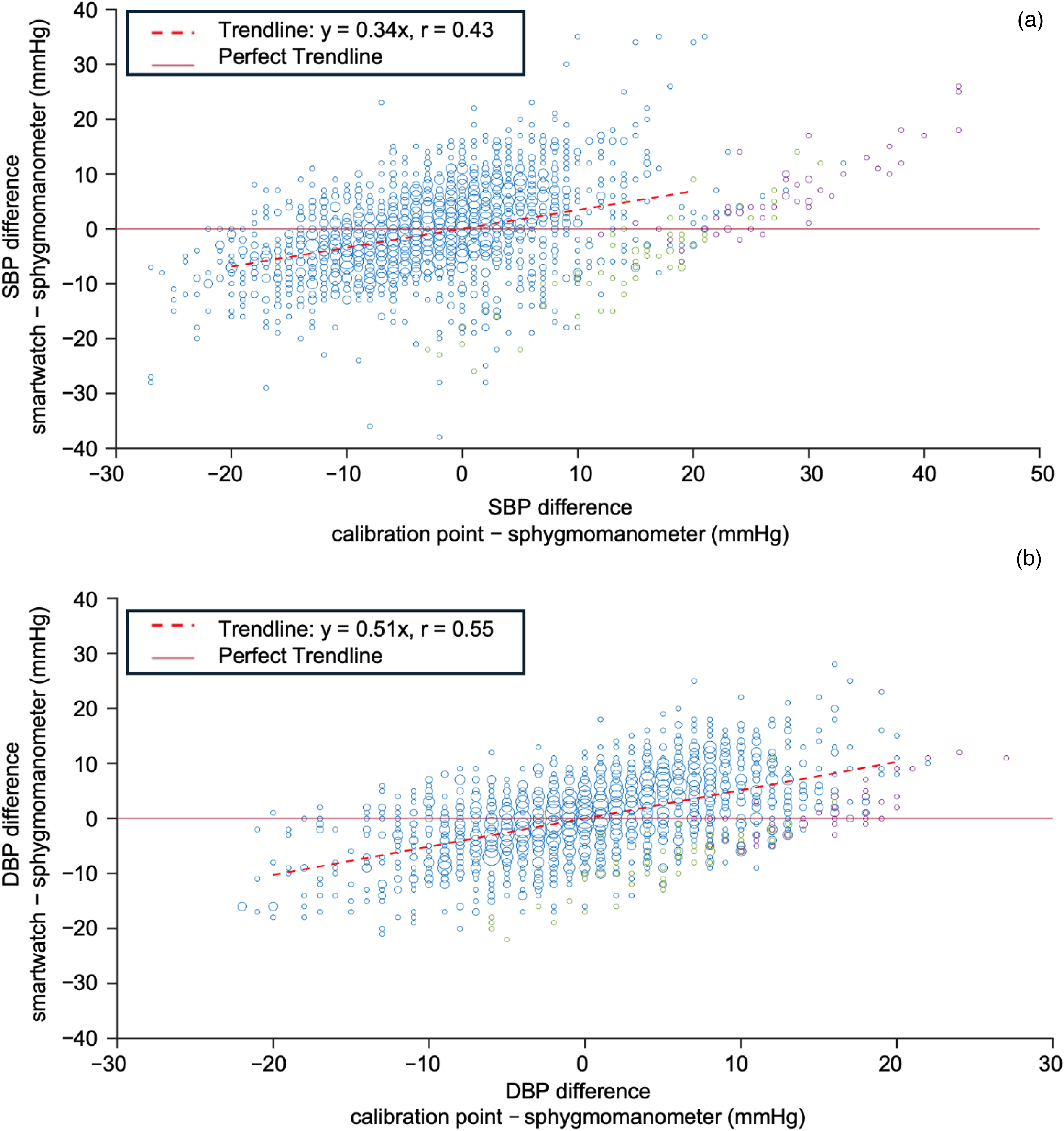
The difference between the smartwatch and the reference sphygmomanometer readings depends on the difference between the actual BP and the calibration point for both SBP (a) and DBP (b). Ideally, the bias between the smartwatch and the reference sphygmomanometer should always be zero (perfect trendline), regardless of how far the measured BP is from the point at which the smartwatch was calibrated. The solid line represents the perfect trendline and the dashed line the determined trendline. The markers are sized according to the frequency of observation of paired BP readings. Green and purple markers represent the group of outliers for two participants identified in the post-processing.
The declining accuracy of the smartwatch with increasing deviation from the calibration point is further demonstrated by the CDFs in Figure 4, which show an increasing proportion of measurements with a larger absolute difference between the smartwatch and the sphygmomanometer when measurements are grouped according to their distance from the calibration point. As illustrated in Figure 4(a) for SBP, in the 0–10 mmHg range, 55% of the absolute differences were under 5 mmHg, compared to 47% in the 10–20 mmHg range and 42% in the >20 mmHg range. Similarly, 85% of the values in the 0–10 mmHg range had absolute differences below 10 mmHg, while this proportion decreased to 80% and 72% in the 10–20 mmHg and >20 mmHg ranges, respectively. The 90th percentile of the absolute differences between the smartwatch and sphygmomanometer was 12 mmHg for the 0–10 mmHg range, 13 mmHg for the 10–20 mmHg range, and 17 mmHg for the >20 mmHg range. For DBP (Figure 4(b)), the analysis revealed a similar trend to SBP. Specifically, in the 0–5 mmHg range, 66% of the values had the absolute difference of less than 5 mmHg, compared to 46% in the 5–10 mmHg range and 50% in >20 mmHg range. Additionally, 93% of values in the 0–5 mmHg range were within 10 mmHg, while 82% and 72% were within 10 mmHg in 5–10 mmHg range and >10 mmHg range, respectively. The 90th percentile of the absolute differences between the smartwatch and sphygmomanometer was 9 mmHg for the 0–5 mmHg range, 12 mmHg for the 5–10 mmHg range, and 16 mmHg for the >10 mmHg range.
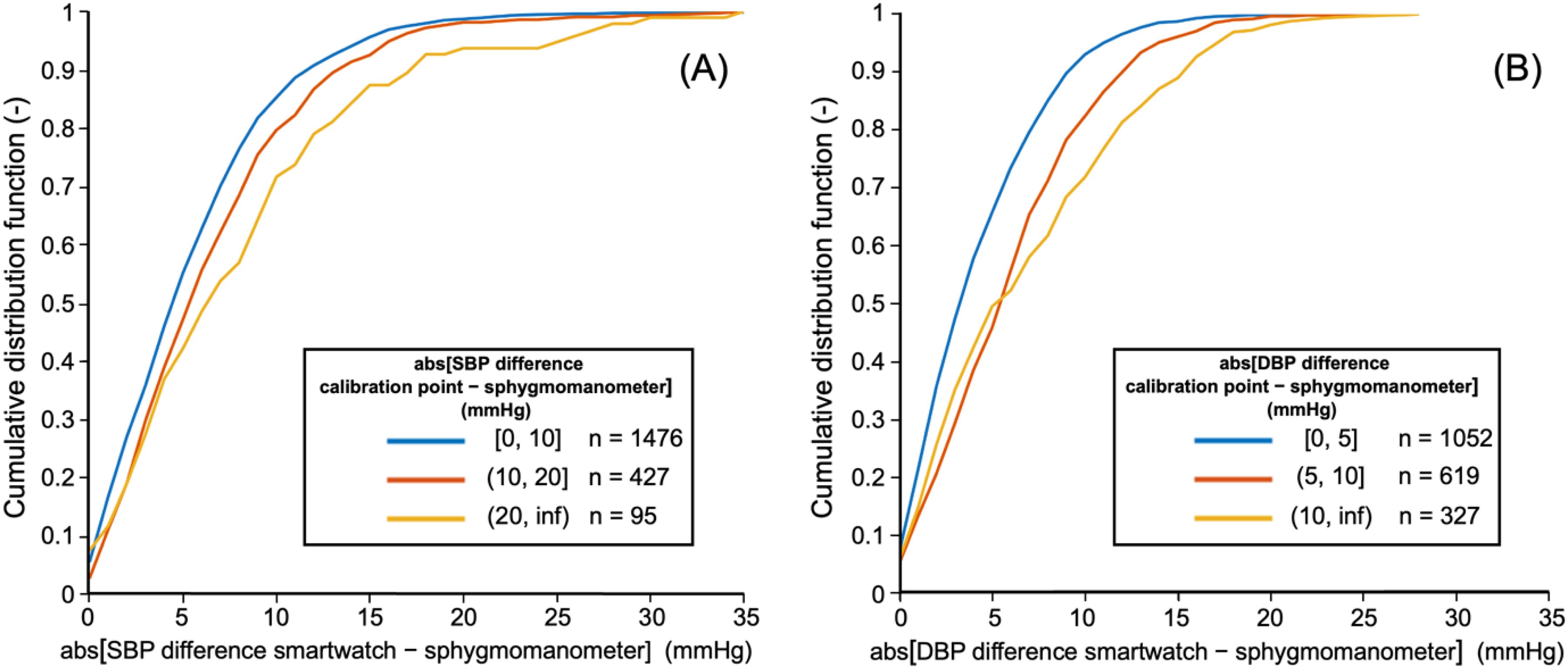
Cumulative distribution of smartwatch differences to sphygmomanometer readings for different levels of BP deviation from the calibration point. As the deviation of the actual BP from the calibration point increases (three different CDFs), the proportion of measurements with larger differences between the smartwatch and the sphygmomanometer increases.
Discussion
In this study, we found that, on average, the Samsung Galaxy Watch 5 smartwatch did not substantially differ from the reference BP monitor (sphygmomanometer) when measuring BP, and this agreement did not change over 28 days until the next calibration according to the manufacturer's recommendations. However, we also showed that this agreement depends on how the measured BP changes against the BP at which the smartwatch was calibrated. The more different the BP is from the calibration pressure, the more the smartwatch differs from the sphygmomanometer on average, thus its accuracy (or better, trueness) decreases.
As shown by the Bland-Altman plots in Figure 1 and Table 3, the MD and SD mostly complied with Criterion 1 of the ISO 81060-2:2018 standard 12 (i.e., MD values no more than 5 mmHg and SD of no more than 8 mmHg), with SD for SBP narrowly missing the specified threshold, and MAD with the IEEE Standard for Wearable, Cuffless Blood Pressure Measuring Devices8,13 (i.e., MAD of less than 7 mmHg). This criterion was also met in short-term studies using the Samsung Galaxy Watch 3 14 and the InBody Watch, 15 but not in a short-term study using the Samsung Galaxy Watch 2, 5 as summarized in Table 4. A long-term study using the Samsung Galaxy Watch 2 19 likewise did not meet this criterion.
Summary of short-term studies on the accuracy of BP measurement by cuffless devices over the course of a single day.
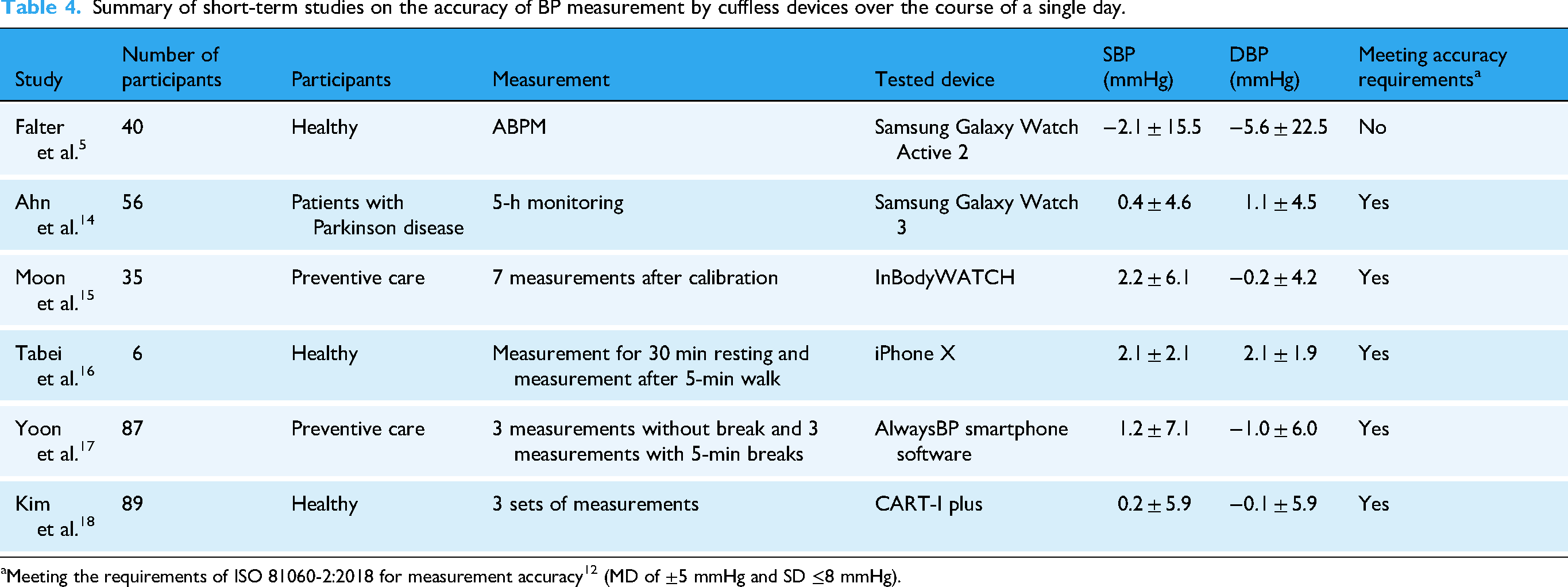
Meeting the requirements of ISO 81060-2:2018 for measurement accuracy 12 (MD of ±5 mmHg and SD ≤8 mmHg).
Although the MD values were different from zero (–0.34 mmHg for SBP and 0.62 mmHg for DBP), from a clinical perspective, this is a negligible MD below the accuracy of BP monitors and far below the thresholds (≥5–10 mmHg) that guide treatment decisions.27,28 A small MD indicates low systematic bias in the average; however, it does not guarantee tight agreement for individual paired readings. The ISO/ESH standards8,12 are relatively permissive regarding the requirement for the SD of MD (≤8 mmHg). In our dataset, the SDs reached 8.10 mmHg for SBP and 7.07 mmHg for DBP. Consequently, approximately one third of paired readings fell within ±3 mmHg, about one half within ±5 mmHg, and roughly 95% within ±14–15 mmHg. These limits of agreement indicate that individual smartwatch readings can differ from the reference value by an extent that could cross diagnostic or treatment thresholds,27,28 even though the device formally meets the acceptance criteria.
The comparison of HR values between the smartwatch and the reference sphygmomanometer showed no statistically significant difference with almost no MD and a very low SD. However, this result is not surprising. 29
Although Criterion 2 of the ISO standard was met, marked differences in the smartwatch bias across individuals were observed. In five participants, the individual MD was greater than ±5 mmHg for both SBP and DBP. We speculate that this may be because the smartwatch was calibrated to a BP that was not usual for some of the participants, for example due to white coat syndrome. 6 If the actual BP is temporarily high during calibration for various reasons, all subsequent measurements until the next calibration may be affected. This could explain the results in Figure 3, where the values of two participants (marked in green and purple) were found to be significantly shifted from the other measurements. The data were nevertheless retained in the statistical analysis because they were obtained in accordance with study protocol and no measurement problem was detected during their collection.
At the time of study conceptualization, the ESH recommendations for the validation of cuffless BP measuring devices were not yet available. Nonetheless, we evaluated BP measurements obtained immediately prior to recalibration, in accordance with their recalibration test. The results confirmed the stability of cuff calibration accuracy throughout the prescribed calibration interval. The narrow confidence intervals for the mean difference (MD)—within ±2 mmHg—indicate that any potential bias associated with the smartwatch measurements at the end of the calibration period not only fulfills Criterion 1 of the ISO standard but is also clinically negligible. This documents the stability of measurements across the calibration interval, a fundamental requirement for home monitoring.
The scatter plots in Figure 2 showed that there were no substantial shifts during the calibration period. Over the calibration period, there was a shift of −0.19 mmHg for SBP and 1.02 mmHg for DBP, which is clearly clinically insignificant. A very similar finding was made in the study by Han et al., 19 where they concluded that if recalibration is performed every 28 days as recommended by the manufacturer, only a difference of 0.6 mmHg in SBP can be expected during the period of use before the next recalibration, with a similar tendency for DBP.
The interpretation of the results based solely on paired data from the smartwatch and the reference sphygmomanometer could be biased and the accuracy of smartwatch BP measurements remains questionable when the actual BP changes from the calibration point. Ideally, the difference between the smartwatch and reference sphygmomanometer should not depend on the change in BP compared to calibration. However, the results showed (Figures 3 and 4) that the difference between the smartwatch and the reference sphygmomanometer increases as the difference between the BP measured by the reference sphygmomanometer and the calibration point increases. The smartwatch responded to the change in BP, but the larger the change in BP compared to the calibration, the less accurate the smartwatch measurement. This observation is consistent with an earlier study that used an older model of the smartwatch. 5
Contribution
Sensors for continuous monitoring of vital signs in wearable devices continue to improve and are gaining potential for early detection of deteriorating health status and for reducing financial costs in healthcare by monitoring patients in their home environment. The accuracy of HR measurement29,30 or the reliability of wearable devices to detect hypoxemia has been demonstrated.31,32 These studies have shown that HR and SpO2 are measured accurately over a wide range of values, even when significant deviations from baseline occur. However, based on our study, we cannot make the same claim for BP measurement. While smartwatches are likely to detect changes from the baseline (presumably represented by the calibration pressure) even after some time has passed since calibration, they may not quantify these changes accurately. While the smartwatch detects the direction of change from the calibration pressure, it may misestimate the magnitude, leading to underestimation at higher BP or overestimation at lower BP. Accordingly, smartwatch BP can be used to follow trends, when pressures are relatively stable. In patients with marked short-term fluctuations, 33 absolute values should be confirmed with validated home or ambulatory monitoring to avoid missing clinically important extremes. Although current wearables are not intended to replace BP measurement by certified medical devices, common consumers may underestimate this difference in capability and should be made explicitly aware of the specific limitation of smartwatch BP measurement.
Limitations
This study has several limitations. First, the distribution of measured BP was unbalanced, with predominantly normotensive and younger participants and only a few individuals with hypertension. However, similar participant distributions have been reported in other studies,19,20,34 and hypertension was not a primary inclusion criterion, as the study rather focused on assessing the long-term accuracy and stability of the smartwatch technology.
Second, a clinically validated automated oscillometric device was used as a reference, consistent with prior long-term work by Han et al. 19 Nonetheless, ISO/ESH/IEEE standards require an auscultatory reference when possible.8,12,25 Because the measurements were performed twice daily over 27 days in a participants’ home environment, obtaining physician-performed auscultatory reference measurements at every session were not feasible. At the same time, it has been found that the oscillometric method may be preferable to the auscultatory method when examining ABPM results for more valid successful measurements. 35
Next, the ISO/ESH/IEEE standards8,12,25 recommend at least 85 participants and 255 paired BP measurements for full validation. Smaller cohorts are acceptable for specific or preliminary validation, with minimum sample sizes of 3512,25 and 20 participants. 8 In our study, 37 of the enrolled 42 participants completed the entire experimental measurement and we obtained 1 998 paired values, which is almost eight times the minimum number of values required. For each participant, the measurements took almost a month to complete, and a larger number of participants would have led to an excessive prolongation of study with results that may no longer be relevant at today's rate of technological development. It would be advisable to perform measurements on a larger and broader population using multiple smartwatch manufacturers. However, even in many other studies, the population sample was less than 85.5,14–16,34
Although the ISO standard 12 allows simultaneous measurements on opposite arms, interarm BP differences may not be completely suppressed during calibration. Using the same arm would avoid this issue but prevent truly simultaneous reading. This would lead to temporal bias and conflict with the smartwatch manufacturer's instructions. The mean lateral difference was less than 1 mmHg for both SBP and DBP, as documented in the Supplementary material.
Participants were recruited voluntarily through personal and university networks. This approach could have preferentially attracted motivated, technology-familiar individuals with high adherence to the protocol and may reduce the diversity of measurement conditions. However, even with fully random recruitment, participation would remain voluntary, and a similar self-selection pattern would likely persist. Moreover, this should not bias the paired comparisons, as simultaneous BP measurements were obtained using the same standardized procedure.
Lastly, there was no supervision of the participants to respect the methodology of measurements after calibration with the professional in the field. But smartwatches, as well as oscillometric BP monitors, are intended for the general public, and we intended to test them in home conditions, without the intervention of other people.
Conclusion
This study demonstrated that the Samsung Galaxy Watch 5 exhibits sufficient long-term accuracy and stability in BP measurements over the 28-day calibration period and fulfills nearly all the ISO/ESH/IEEE accuracy criteria in predominantly young and normotensive adults. However, the accuracy decreased when BP values deviated from the calibration point. Thus, changes in BP may not be accurately quantified, resulting in an under- or overestimation of the absolute value. Smartwatch BP can be used to follow trends in users with relatively stable BP. When BP fluctuates substantially, or when therapeutic decisions are required, absolute values shall be confirmed with validated monitoring.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261415923 - Supplemental material for Long-term accuracy and stability of blood pressure measurements from a smartwatch: Prospective validation study
Supplemental material, sj-docx-1-dhj-10.1177_20552076261415923 for Long-term accuracy and stability of blood pressure measurements from a smartwatch: Prospective validation study by Simon Walzel, Hana Sebestova, Veronika Rafl-Huttova, Martin Rozanek and Jakub Rafl in DIGITAL HEALTH
Footnotes
Abbreviations
Ethical approval
The Ethical Review Board of the Faculty of Biomedical Engineering, Czech Technical University in Prague approved the study on May 15, 2023 (No. C41/2023). The study was registered on the ClinicalTrials.gov database (No. NCT06098092).
Informed consent
All participants provided written informed consent before their enrollment in the study.
Authors’ contributions
S.W., V.R.-H., M.R. and J.R. conceptualized the study. All authors prepared the methodology of the study. S.W. and H.S. administered, conducted the investigation, and curated the data. S.W., H.S., V.R.-H. and J.R. carried out formal analysis, interpretation, and visualization. The original draft of the manuscript was prepared by S.W., M.R., and J.R., while all authors participated in reviewing and editing the manuscript. All authors revised and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant Agency of the Czech Technical University in Prague (grant numbers: SGS25/110/OHK4/2T/17, SGS25/184/OHK4/3T/17) and by a grant from the Programme Johannes Amos Comenius under the Ministry of Education, Youth and Sports of the Czech Republic [CZ.02.01.01/00/23_021/0010653].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Use of generative AI
Generative AI tools were used solely for grammar correction and language refinement. All content, including study design, data analysis, interpretation, and conclusions, was developed by the authors based on cited resources.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
