Abstract
Background
Noninvasive remote monitoring can improve the home management of heart failure. However, the low specificity of current parameters limits their effectiveness. This study examined the association between respiratory rate stability (RRS), detected using a piezoelectric noncontact vibration sensor, and heart failure–related adverse events. We explored the relationship between the RRS measured during hospitalization and the occurrence of adverse events, including mortality and rehospitalization, within one year after discharge.
Methods
This prospective observational study was conducted between 2022 and 2024. Patients with heart failure were monitored using a piezoelectric sensor placed under their mattress; the RRS was quantified through frequency analysis. The primary endpoints were postdischarge events, including all-cause mortality, heart failure–related rehospitalization, all-cause rehospitalization, and composite events. Patients were stratified into the low (RRS < median) and normal (RRS ≥ median) RRS groups. Survival analysis was performed.
Results
A total of 57 patients were included in this analysis. The median composite event time was 203 and 477 days in the low and normal RRS groups, respectively. The log-rank test showed significant differences between the groups. The hazard ratio for event occurrence in the low RRS group compared with that in the normal RRS group was 3.25. Significant differences were observed for all individual postdischarge events, including mortality and rehospitalization.
Conclusions
Evaluating the RRS using a piezoelectric sensor identified patients at a high risk of mortality and rehospitalization within a year of discharge. These findings support the use of this noninvasive method for risk stratification and remote heart failure management.
Keywords
Introduction
Owing to its high recurrence rate, heart failure continues to impose significant health and economic burdens, highlighting the anticipated role of telemonitoring in managing symptoms within home settings.1,2 Noninvasive telemonitoring, a form of remote monitoring, leverages physiological parameters such as heart rate, blood pressure, and body weight and has gained attention for its ease of implementation. 2 However, its effectiveness remains inconclusive. While several meta-analyses and randomized controlled trials (RCTs) have demonstrated positive effects in reducing all-cause mortality and heart failure–related hospitalizations,3–6 other studies, including large-scale RCTs conducted under stringent conditions, 7 have shown no significant effects.7–10
The lack of consistency in the results has been attributed to the low heart failure specificity of general physiological parameters such as the heart rate and blood pressure, which are commonly utilized in noninvasive telemonitoring. By contrast, pulmonary arterial pressure monitoring via implantable hemodynamic monitoring systems has supported the management of patients with heart failure,2,11 underscoring the critical need for developing noninvasive, heart failure–specific indicators and measurement methods.
Therefore, we focused on respiratory instability, specifically central sleep apnea with Cheyne–Stokes respiration (CSA-CSR), as heart failure–specific parameters that can be measured noninvasively. The CSA-CSR, which is diagnosed using polysomnography (PSG), has a reported prevalence of 21–66% among patients with heart failure. 12 Its prevalence increases as cardiac function declines,13–15 and it is regarded as an independent prognostic factor. 16 Based on these findings, we hypothesized that respiratory instability, a characteristic of CSA-CSR, could serve as a potential indicator for remote home monitoring to detect heart failure exacerbations.
Several studies have demonstrated the utility of respiratory instability as an indicator in the management of heart failure.17–19 Asanoi et al. 17 developed the Respiratory Stability Index (RSI), which is defined as the inverse of the standard deviation of specific frequency bands extracted from the power spectrum of respiratory flow measurements. Their 85-month retrospective analysis indicated that the RSI could predict cardiovascular mortality. Building on this work, Sakoda et al. 18 aimed to adapt the RSI for use in home settings by measuring it using a piezoelectric noncontact vibration sensor placed on a bed. They reported that patients who experienced a heart failure exacerbation leading to hospitalization or sudden death showed a decline in the RSI to ≤20 s 7–28 days before hospitalization.
However, the study by Asanoi et al. was limited by its retrospective design and reliance on direct measurements of respiratory flow, rendering it unsuitable for direct application in home settings. Similarly, the study by Sakoda et al. had limitations, including a small sample size and lack of statistical significance in demonstrating the predictive value of the RSI for hospitalization.
Therefore, no study has prospectively reported the association between respiratory stability measurements obtained using a piezoelectric noncontact vibration sensor and the occurrence of heart failure events. Thus, this study is the first to measure respiratory instability in hospitalized patients with heart failure using a piezoelectric noncontact vibration sensor and to observe event occurrences over a one-year period based on these measurements.
This study aimed to prospectively investigate whether quantified respiratory stability values measured during hospitalization were associated with the occurrence of heart failure events after discharge. The clinical significance of this study lies in demonstrating the feasibility of using respiratory stability measurements for the remote monitoring of heart failure at home.
Methods
Design and setting
This single-center prospective cohort study included adult patients hospitalized for decompensated heart failure. Enrollment was conducted at Nagano Chuo Hospital in Japan between April 2022 and May 2024 and follow-up continued until October 2024. A noncontact monitoring sensor was installed by the research team solely for physiological data collection. The sensor did not alter patient care or treatment decisions, and no control group or randomization was involved.
This study, including the study objectives, social significance, and ethical considerations, was registered with the University Hospital Medical Information Network (ID: UMIN000052143). This study was designed and conducted in accordance with the Declaration of Helsinki on Ethical Principles for Medical and Biomedical Research. All participants were provided with an explanation of the purpose and details of the study as well as the use of their data, and written informed consent was obtained. This study was approved by the Shinshu University Ethics Committee in November 2021 (Approval No. 5401).
This observational study was designed and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. A completed STROBE checklist is provided in Supplemental File 1.
Study participants and eligibility criteria
Patients aged ≥20 years who were admitted to the cardiology ward at Nagano Chuo Hospital during the study period were consecutively screened for eligibility. Patients with initial and recurrent heart failure were included without distinction. The exclusion criteria were as follows: 1) Patients with excessive nocturnal body movements owing to cognitive impairment or other neurological conditions; 2) those on assisted ventilation devices such as adaptive servo ventilation (ASV) or noninvasive positive pressure ventilation (NPPV); 3) those with acute coronary syndrome, chronic obstructive pulmonary disease, pneumonia or other infections, central nervous system disorders, chronic renal failure requiring hemodialysis, or symptomatic malignancies within three months prior to enrollment; and 4) those requiring intensive care unit (ICU) management at the time of admission.
Additionally, owing to the limited availability of monitoring devices, some eligible patients who provided consent could not be enrolled at the time of admission. These logistical constraints were nonselective and unrelated to the clinical characteristics of the patients.
Data collection procedure
In this study, a piezoelectric sensor (Vital Sensor, Z-Works Inc., Tokyo, Japan) and transmitter (Gateway, Z-Works Inc.) were installed on the beds of participants who provided informed consent. The piezoelectric sensor was placed under the bed mattress, enabling continuous nighttime respiratory monitoring during hospitalization. It continuously recorded respiratory data until discharge, and the transmitter sent the analyzed data to a server.
The Vital Sensor is a piezoelectric vibration-based, noncontact monitoring device that incorporates a Piezola® element (Mitsui Chemicals, Inc., Tokyo, Japan) within a patented structure (Japanese Patent No. 7602222). Although detailed technical specifications are not publicly available, a general product description is provided on the manufacturer's website (https://liveconnect.jp/product). This sensor was selected owing to its high sensitivity to subtle physiological vibrations such as respiration and heartbeat, motion-tolerant signal acquisition, and practical feasibility in overnight monitoring.
Baseline data, including demographic information, were collected at the time of participant enrollment, and clinical data were extracted from electronic medical records within three days after discharge. Data were primarily obtained from medical records for the follow-up assessment of key outcomes. If sufficient information was unavailable, follow-up was conducted via telephone interviews with participants or their designated caregivers.
Observational items
Demographic and clinical characteristics
Patient demographics and clinical characteristics, including age, sex, height, weight, and body mass index, were collected from the patients’ medical records. Heart failure stages at discharge were classified as Class C or D according to the 2022 American Heart Association/American College of Cardiology/Heart Failure Society of America guidelines.² Medical history, including comorbidities such as atrial fibrillation, hypertension, coronary artery disease, and diabetes, was also extracted. The use of medications for major cardiovascular treatments was also recorded.
Blood and biochemical tests, as well as echocardiographic and chest radiography examinations, were performed upon admission, and the results closest to the admission date were analyzed. Blood and biochemical tests included renal function markers, electrolytes, hemoglobin, and brain natriuretic peptide (BNP) levels.
Echocardiographic data were used to assess left ventricular ejection fraction (LVEF) and classify patients into those with heart failure with reduced ejection fraction (LVEF ≤ 40%), mildly reduced ejection fraction (LVEF 41–49%), and preserved ejection fraction (LVEF > 50%). 2 Cardiothoracic ratio was measured from chest radiographs.
Hospitalization data, including daily blood pressure and pulse, were collected from medical records, and average values were calculated. Physical findings related to heart failure, such as edema, peripheral coldness, and dyspnea (at rest and on exertion), were assessed, and their time to improvement was recorded. Symptoms at admission and discharge were graded I–IV using the New York Heart Association (NYHA) classification.² The length of hospital stay was determined from medical records.
Measurement of respiratory rate stability using a piezoelectric noncontact vibration sensor
The respiratory rate stability (RRS) is a quantitative index used to assess respiratory stability. It was calculated by modifying the frequency bandwidth definition from standard deviation–based computation, following the RSI calculation method described by Asanoi et al. 16 The measurement system consisted of a Vital Sensor, a piezoelectric noncontact vibration sensor placed beneath the bed mattress, and a Gateway, a transmitter that sent data via an LTE network.
The sensor passively detected subtle body movements, including respiration and heartbeats, without direct contact with the patient. The pressure variation data collected by the sensor were processed in the transmitter before transmission to the Z-Works servers.
The sensor was installed beneath each participant's bed mattress as early as possible after hospital admission, usually within 1–2 days. Nighttime respiratory data were collected continuously throughout the hospitalization. For RRS analysis, the average value was calculated using all valid nighttime data (7:00 PM to 7:00 AM) recorded from the day of installation until discharge. This approach was consistently applied to all participants. Device installation was postponed in environments where radio-emitting devices were restricted (e.g., ICUs and high care units), when bed movement was required for treatment, when temporary ventilatory support (mechanical ventilation, ASV, NPPV) was used, or when measurement devices were unavailable.
The system incorporated proprietary preprocessing to automatically exclude segments with artifacts such as excessive movement (≥10% of the 24-h maximum fast Fourier transform [FFT] power), bed nonuse, or low signal-to-noise ratio. Although the precise filtering algorithm is not publicly available, these procedures ensured that only physiologically meaningful and stable segments were included in the analysis.
Respiratory rate stability calculation
The RRS was calculated from the frequency domain of the waveform data extracted by the Vital Sensor. First, the waveform data were sampled at 50 Hz. Fast Fourier transform was applied to a sliding window of 16,384 data points, equivalent to approximately 328 s of data. A 30-point moving average was applied to the FFT spectrum to reduce noise and smooth fluctuations in the frequency domain.
A peak was identified within the frequency range of 0.1–0.8 Hz. The width of the spectral curve was measured at a distance of 1/e from the maximum amplitude of the peak, and its reciprocal was calculated as the RRS value (Figure 1).

Respiratory rate stability (RRS) calculation. A fast Fourier transform (FFT) is applied to a sliding window of 16,384 data points. Within the frequency range of 0.1–0.8 Hz, the peak of the spectral curve is identified. The width of the spectral curve at a distance of 1/e from the peak's maximum amplitude is measured, and its reciprocal is defined as the RRS.
The sliding window was advanced by 90 data points, with the oldest 90 points removed and 90 new points added. The FFT and RRS calculations were repeated. A higher RRS value indicated a narrower spectral curve width and more stable respiration, whereas a lower RRS value suggested a wider spectral curve width and less stable respiration.
Endpoints
The primary endpoints were the occurrence and duration of composite events, including all-cause mortality and unplanned heart failure–related rehospitalization within 365 days after discharge. The secondary endpoints included the time-to-event for each event: all-cause mortality, unplanned all-cause rehospitalization, and unplanned heart failure–related rehospitalization within 365 days of discharge. Additionally, the number of composite events was assessed 90, 180, and 270 days postdischarge. The follow-up period was determined based on the longest available data. Patients who survived beyond 365 days were followed until their last confirmed date.
Rehospitalizations were identified from electronic medical records, and heart failure–related rehospitalization was defined according to the discharge diagnosis codes or, for admissions at other hospitals, referral documents explicitly indicating heart failure.
Sample size estimation
The required sample size was determined based on reports indicating that the hazard ratio (HR) for survival rates in patients with heart failure with and without CSR ranges from 2.1 to 5.7.12,16 Using Schoenfeld's formula, an HR of 2.1 was assumed, with a two-sided significance level of α = 0.05 and a statistical power of 80% (1−β = 0.8). This resulted in an estimated requirement of 15 events. Two variables were included in the model for the Cox proportional hazards analysis. The number of events per variable was set to 10, necessitating a minimum of 20 events. Furthermore, based on a report indicating that rehospitalization for heart failure occurs in approximately 35% of cases, 20 the total required sample size was calculated to be 60.
Statistical analysis
The baseline characteristics were analyzed by calculating the median RRS for all participants, including those who died during hospitalization. Participants were classified into the normal (RRS ≥ median) and low (RRS < median) RRS groups. Continuous variables are summarized as medians with interquartile ranges (IQRs), whereas categorical variables are presented as frequencies and percentages. For the primary and secondary endpoints, the number and percentage of occurrences as well as the median time to event were reported at 90, 180, 270, and 365 days postdischarge. Differences between the RRS groups were evaluated using the Wilcoxon rank-sum test for continuous variables and Fisher's exact test or the chi-square test for categorical variables.
The primary analysis involved a time-to-event survival analysis of the primary endpoint. Kaplan–Meier survival curves were generated to visualize survival differences between the two groups, and comparisons were performed using the log-rank test. Additionally, the impact of the RRS classification on the primary endpoint was estimated using a Cox proportional hazards model, with results reported as HRs and 95% confidence intervals. The event-per-variable ratio was set to 10, and participant age and RRS groups were included as covariates. Model fit was evaluated using concordance, the likelihood ratio test, and the Wald test. To further assess the robustness of the findings, a sensitivity analysis was conducted using an alternative RRS cutoff derived from the Youden Index of the receiver operating characteristic (ROC) curve. Based on this cutoff, patients were reclassified into low and normal RRS groups, and the primary analyses were repeated to verify consistency with the original results.
In the secondary analysis, time-to-event survival analyses were conducted for all-cause mortality, all-cause rehospitalization, and heart failure–related rehospitalization within 365 days of discharge. Kaplan–Meier survival curves were generated to visualize the difference in survival rates between the two groups, consistent with the primary analysis, and comparisons were performed using the log-rank test. Missing data were handled using listwise deletion; participants with missing values for any of the variables used in the respective analysis were excluded. All statistical analyses were performed using R version 4.4.2, with a significance level of 5% applied to all tests.
Results
Participant characteristics
A total of 95 patients met the inclusion criteria. Of these, 32 were excluded, leaving 63 (66.3%) for observation. The reasons for exclusion included the inability to obtain RRS data owing to mechanical issues (20 patients) and transfer to another hospital or department owing to the onset of other diseases (12 patients). Among the remaining patients, those who died during hospitalization (three patients) were excluded from follow-up. Additionally, three patients for whom event occurrence within 365 days after discharge could not be confirmed, and five patients whose survival status was unknown were excluded. Follow-up was completed in the remaining patients (Figure 2). The median follow-up period was 393 days (IQR: 177–520 days). The baseline characteristics of excluded and lost-to-follow-up patients are summarized in Supplemental Table 1.
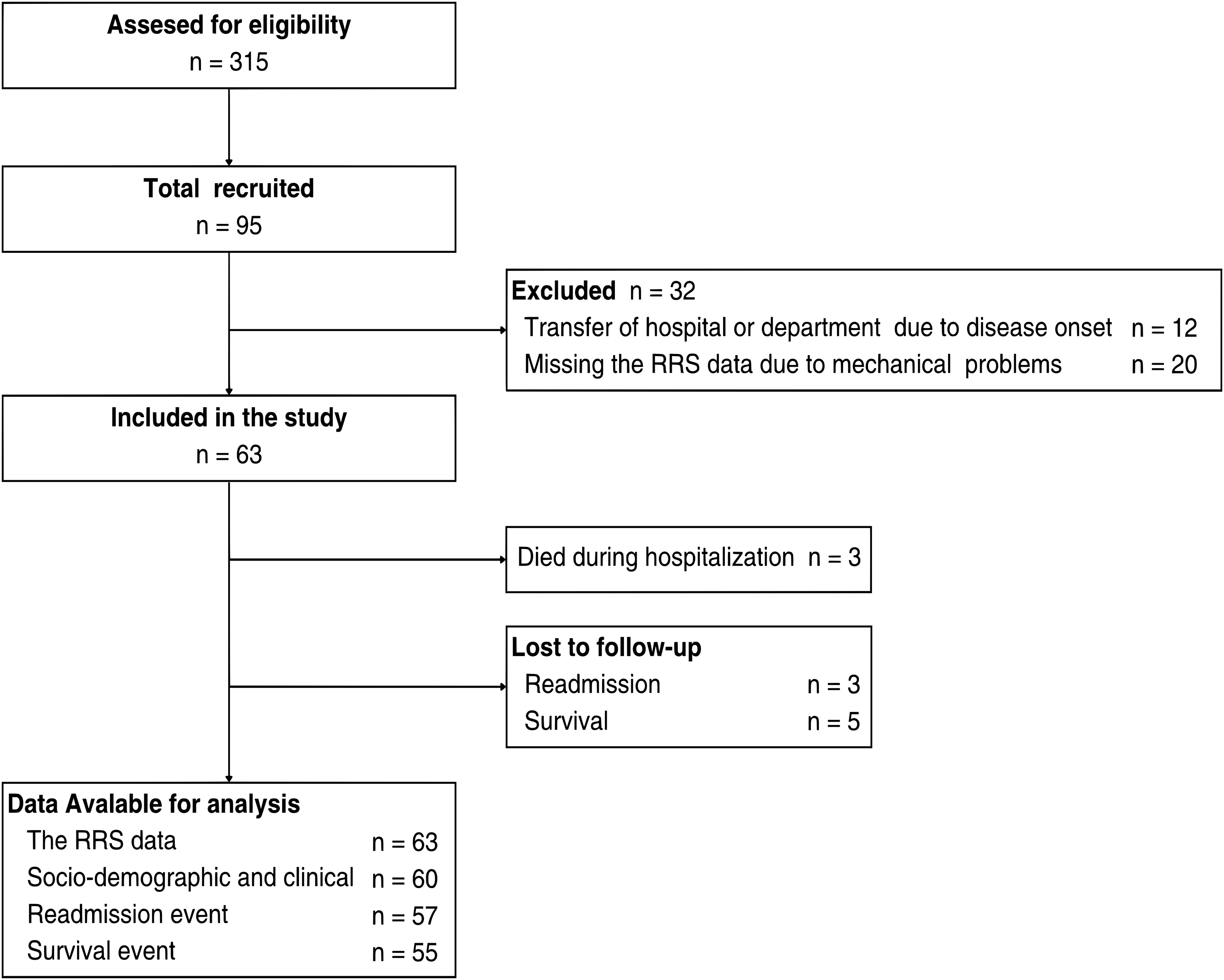
Recruitment and follow-up flow diagram. RRS, respiratory rate stability.
The representative results of patient characteristics for the low (RRS < 4.43) and normal (RRS ≥ 4.43) RRS groups are summarized in Table 1, and the complete dataset is provided in Supplemental Table 2. The median patient age was 83 years, with 48% female and 52% male. Heart failure stages at discharge were 90% in Stage C and 10% in Stage D (Supplemental Table 1). At discharge, the NYHA classification was grade I in 41%, II in 41%, III in 17%, and IV in 0%. During hospitalization, the low RRS group had a median RRS of 3.83 and respiratory rate (RR) of 14.7, while the normal RRS group had a median RRS of 5.19 and an RR of 14.78. The median time to symptom resolution, including dyspnea, was 16 and 9 days in the low and normal RRS groups, respectively.
Baseline characteristics of the study population, according to the RRS group.

RRS: respiratory rate stability; BMI: body mass index; BNP: brain natriuretic peptide; LVEF: left ventricular ejection fraction; HFpEF: heart failure with preserved ejection fraction; HFmrEF: heart failure with mildly reduced ejection fraction; HFrEF: heart failure with reduced ejection fraction; CTR: cardiothoracic ratio; NYHA: New York Heart Association; NA: not available; Median (Q1, Q3); n (%).
Missing endpoints
Three patients had an unconfirmed event occurrence within 365 days, and five patients had an unconfirmed survival status (Table 2).
Number of missing data points for outcomes.

n (%).
Endpoints
The number and duration of events at 90, 180, 270, and 365 days are presented in Table 3. During the 365-day follow-up period, 27 composite events (45%) were identified as the primary endpoints.
Time to event and event frequencies at 90, 180, 270, and 365 days by RRS groups.
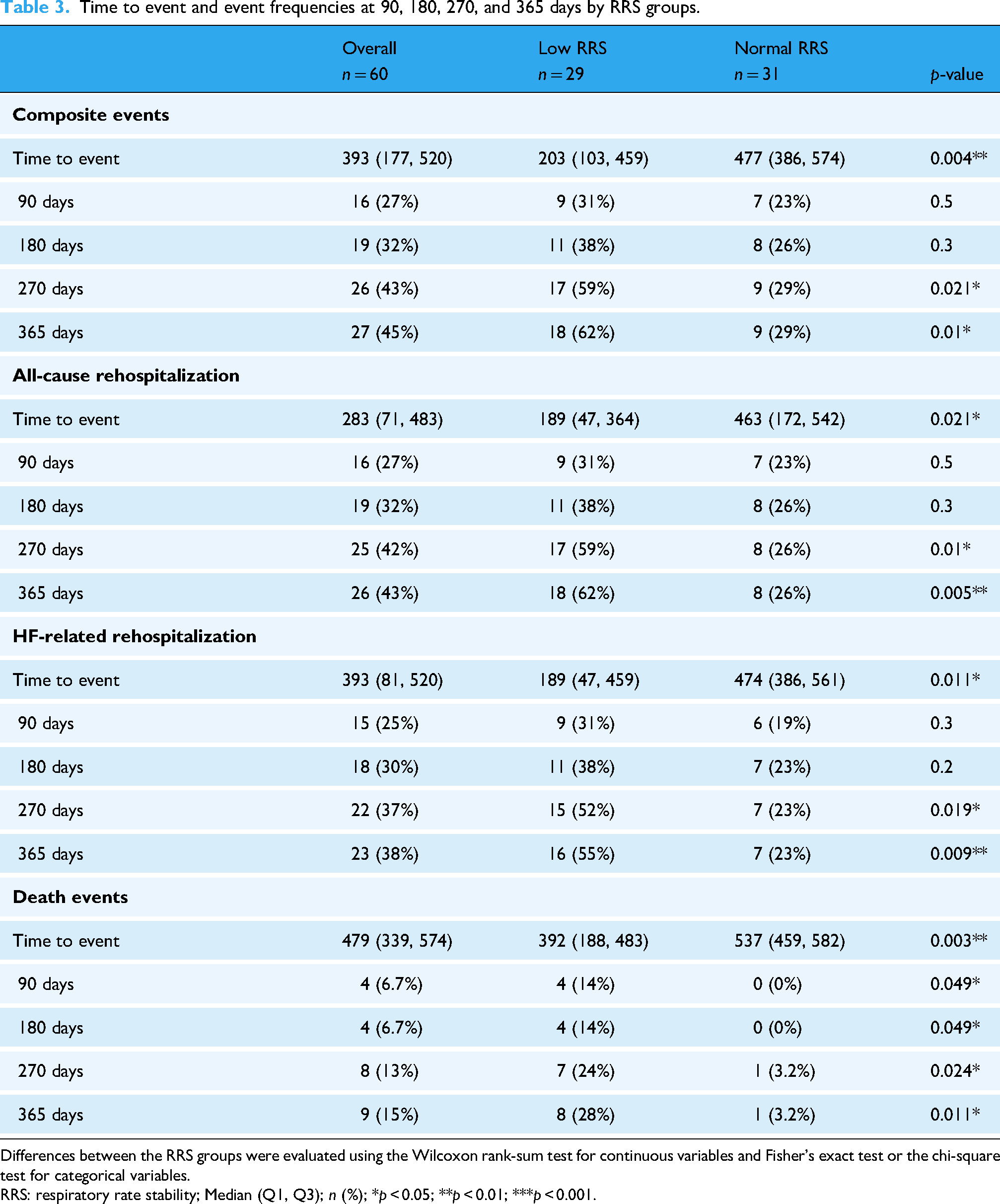
Differences between the RRS groups were evaluated using the Wilcoxon rank-sum test for continuous variables and Fisher's exact test or the chi-square test for categorical variables.
RRS: respiratory rate stability; Median (Q1, Q3); n (%); *p < 0.05; **p < 0.01; ***p < 0.001.
For the secondary endpoints, 26 cases (43%) of unplanned all-cause rehospitalization, 23 cases (38%) of unplanned heart failure–related rehospitalization, and 9 cases (15%) of all-cause mortality were observed. Group comparisons based on the RRS classification revealed significant differences in mortality across all time periods after 90 days. Additionally, significant differences were observed in composite events, all-cause rehospitalization, and heart failure–related rehospitalization after 270 days (Table 3).
Figures 3 to 6 present the results of the log-rank test and Kaplan–Meier survival curves. Significant differences in time-to-event outcomes were observed between the low and normal RRS groups, including differences in the composite primary endpoint, unplanned all-cause rehospitalization, unplanned heart failure–related rehospitalization, and all-cause mortality, which were the secondary endpoints.

Kaplan–Meier curve and log-rank test for composite primary endpoint according to the RRS group. RRS, respiratory rate stability.

Kaplan–Meier curve and log-rank test for unplanned all-cause rehospitalization according to the RRS group. RRS, respiratory rate stability.

Kaplan–Meier curve and log-rank test for unplanned heart failure–related rehospitalization according to the RRS group. RRS, respiratory rate stability.

Kaplan–Meier curve and log-rank test for all-cause mortality according to the RRS group. RRS, respiratory rate stability.
The results of the Cox proportional hazards analysis are presented in Table 4. Age and RRS were identified as statistically significant predictors of survival time. The HRs for age and low RRS were 1.051 and 3.25, respectively. The concordance index was 0.712, and both the likelihood ratio test and Wald test were statistically significant (p < 0.001), indicating good predictive performance of the model (Table 4).
Effect of age and RRS category on composite primary endpoint: A Cox proportional hazards analysis.

RRS: respiratory rate stability; Coeff: coefficient; HR: hazard ratio; SE: standard error; CI: confidence interval; Concordance: 0.712; Likelihood ratio test: p < 0.001; Wald test: p < 0.001.
Sensitivity analysis
When patients were reclassified using the ROC-derived cutoff (RRS = 4.455; Supplemental Figure 1), both the Kaplan–Meier survival analysis and the Cox proportional hazards model yielded results consistent with those obtained using the median cutoff. These findings suggest that the results were robust to the choice of threshold. Detailed outcomes are presented in Supplemental Table 3 and Supplemental Figure 2.
Discussion
The results of this study support the hypothesis that the RRS, measured noninvasively during hospitalization using a piezoelectric noncontact vibration sensor, is associated with adverse event occurrence within one year after discharge. The analysis revealed statistically significant differences in the time to occurrence of all-cause mortality, all-cause rehospitalization, heart failure–related rehospitalization, and composite events between the two groups divided by the median RRS value. Additionally, Cox proportional hazards analysis estimated that a low RRS increased the risk of event occurrence by 3.25 times compared with the normal RRS. These findings indicate that the RRS is an effective predictor of event occurrence risk.
Respiratory rate stability was associated with both rehospitalization and mortality in the analyses of each event, which is consistent with reports on the association between CSR detected by PSG and prognosis. Hanly et al. reported that during a two-year follow-up period, the mortality rate was 86% in patients with heart failure with CSR and 56% in those without CSR. 21 Furthermore, multiple studies have reported that CSR increases mortality rates, with HRs of 2.1–5.7.12,16 In the present study, the 365-day mortality rate was 28% in the low RRS group and 3.2% in the normal RRS group, demonstrating an association between the RRS and mortality, consistent with previous studies.
Previous studies have reported postdischarge rehospitalization rates of 11% within 90 days, 27% within 180 days, and 35% within 365 days in patients with heart failure.20,22 In the present study, rehospitalization rates in the low RRS group were 31% within 90 days, 38% within 180 days, and 65% within 365 days, all of which were significantly higher. These findings indicate that respiratory pattern analysis using the RRS measured using a piezoelectric noncontact vibration sensor is a useful tool for identifying patients with heart failure with a high risk of adverse events, comparable to CSR or CSA detected using PSG.
The RRS, which is theoretically based on the detection of CSA-CSR, may be associated with heart failure events owing to mechanisms such as circulation time delay and blood-fluid factors underlying CSA-CSR. Takagawa et al. highlighted the relationship between the RSI and pulmonary congestion. 19 The time to resolution of physical findings such as dyspnea and edema differed between the low (16 days) and normal (9 days) RRS groups in our study, suggesting a potential association with these heart failure–related symptoms. Previous studies have demonstrated that monitoring and managing pulmonary arterial pressure in patients with heart failure is effective in improving prognosis and have recognized the importance of monitoring indicators reflecting blood-fluid factors. 11 Respiratory rate stability may also be an effective indicator of these blood-fluid factors. However, CSA-CSR is influenced by various factors, including age, sex, and sympathetic nervous system activity, all of which affect the respiratory center and breathing. 12 Therefore, definitive conclusions cannot be drawn based solely on the results of this study.
In previous studies on the calculation method for respiratory stability, Asanoi et al. used the standard deviation to define the frequency width, 17 whereas the present study adopted the 1/e method. This difference arises from the method of acquiring respiratory pattern information. Previous studies used respiratory airflow, whereas the present study utilized a piezoelectric sensor. Unlike the respiratory airflow, piezoelectric sensors continuously capture vibration data, and their power never reaches zero. Therefore, the standard deviation could not be defined. In this study, the 1/e width of the peak was used as the standard for defining the frequency width, based on a calculation method for the attenuation point of a Gaussian distribution.
Additionally, the selection of the 1/e threshold was informed by empirical observations. Because the respiratory frequency spectra in this study did not consistently exhibit Gaussian distributions, the use of full width at half maximum was considered less appropriate owing to its lower sensitivity to spectral variability. By contrast, the 1/e threshold provided a more robust and sensitive indicator of respiratory rhythm instability, better capturing physiologically meaningful fluctuations.
This methodological difference is related to variations in the frequency components reflected in the indices, suggesting that different respiratory patterns may have been captured in previous studies and the present study.
Furthermore, the RRS was not externally validated against gold-standard sleep assessment tools such as PSG. As a result, it remains unclear whether the RRS specifically reflects CSA, CSA-CSR, or other factors such as obstructive sleep apnea or body movements. Although the RRS demonstrated an association with clinical outcomes, its physiological and clinical validity remains uncertain. Future studies comparing the RRS with PSG-derived respiratory metrics are warranted to establish its external validity
The RRS showed no significant prognostic differences at 90 and 180 days in this study, suggesting that it may not be a reliable short-term prognostic indicator. The OPTIMIZE-HF study reported a 30% rehospitalization rate within 60–90 days postdischarge, 23 indicating that the RRS was not associated with this high-risk period following acute-phase treatment. This limitation highlights the need for further research on alternative calculation methods or the combination of the RRS with other indicators to enhance predictive accuracy.
This study has several limitations. First, the generalizability of the study may be limited because it was a single-center study. Compared with a representative large-scale observational study targeting chronic heart failure in Japan, 24 the participants in this study were older, had a lower proportion of males, and exhibited a higher prevalence of valvular disease and atrial fibrillation. As age and sex are known to increase the prevalence of CSR, 12 and patients with CSR have a higher prevalence of atrial fibrillation, 15 further investigations are needed to evaluate how these characteristics influence the results. Large-scale, multicenter studies are required to address the limitations of generalizability.
Second, this study was limited by the exclusion criteria and missing follow-up data. Mortality follow-up was incomplete in 9.6% of patients, and some were excluded due to ICU admission or transfer. Although screening was conducted consecutively, not all eligible patients were enrolled due to challenges in obtaining consent and limited device availability. In addition, technical issues such as sensor disconnection or removal led to data loss. These operational factors may have introduced selection bias, and the excluded and lost-to-follow-up patients tended to be older compared with those included in the analysis.
Third, owing to the limited number of events, only age and RRS were included in the Cox model. However, the exclusion of other clinical variables such as the BNP levels, LVEF, or comorbidities may lead to residual confounding, which should be considered when interpreting the results.
Fourth, sensor accuracy may have been influenced by environmental noise and mattress characteristics. Although internal testing demonstrated stable performance under standard conditions, these factors were not systematically evaluated in the present setting
The RRS enables the identification of patients with heart failure at high risk of adverse events using a noninvasive approach. The device employs a piezoelectric sensor that is already widely used in Japan's caregiving sector and can be utilized by integrating the measurement algorithm. By simply placing the sensor beneath a bed mattress, ubiquitous monitoring becomes possible, offering a potentially useful method for remote management of heart failure.
Future research should investigate whether this approach can detect heart failure exacerbations in home-based patients with heart failure and evaluate its effectiveness as a supportive tool for remote heart failure management. Further studies are needed to assess the external validity and generalizability of these results.
Conclusion
The RRS analysis method using a piezoelectric noncontact vibration sensor was shown to potentially identify patients with heart failure at high risk of mortality and rehospitalization within one year after discharge. The present findings remain exploratory, and further research is required for external validation and to evaluate its applicability in home-based settings.
Supplemental Material
sj-png-1-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-png-1-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-docx-2-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-xlsx-3-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-xlsx-3-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-xlsx-4-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-xlsx-4-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-xlsx-5-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-xlsx-5-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-docx-6-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-docx-6-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Supplemental Material
sj-jpeg-6-dhj-10.1177_20552076251388137 - Supplemental material for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study
Supplemental material, sj-jpeg-6-dhj-10.1177_20552076251388137 for Association between heart failure events and nighttime respiratory stability measured using a piezoelectric noncontact vibration sensor: A prospective observational study by Tetsuroh Tamaru, Makoto Ogawa, Yasuyuki Kurasawa, Minato Hayashi and Yoshiharu Yokokawa in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to express their gratitude to the staff of Nagano Chuo Hospital for their cooperation in data collection. The authors would also like to thank the members of the Yokokawa Seminar at Shinshu University for their critical review and valuable insights. The authors are especially grateful to Mr Ayoub Kasdaoui of Z-Works Inc., whose technical support and expertise contributed significantly to the development and clarification of the RRS calculation methodology. Additionally, the authors appreciate Editage's (![]() ) help with English language editing. Furthermore, the authors acknowledge the use of ChatGPT (OpenAI) for refining the English translation and enhancing the clarity of expressions in this manuscript.
) help with English language editing. Furthermore, the authors acknowledge the use of ChatGPT (OpenAI) for refining the English translation and enhancing the clarity of expressions in this manuscript.
Ethical approval
This study was approved by the Ethical Committee of Shinshu University (Approval No. 5401). The authors affirm that all procedures conducted in this study adhere to the ethical standards of the relevant national and institutional committees on human experimentation, as well as the 1964 Declaration of Helsinki and its latest revision in 2013.
Consent to participate
All participants received a detailed explanation of the study's purpose, procedures, and data usage. Written informed consent was obtained.
Contributorship
TT played a central role in conceptualization, data curation, formal analysis, investigation, project administration, and writing – original draft. MO contributed to methodology consultation, resources, and writing – review & editing. YK was responsible for formal analysis and writing – review & editing. MH contributed to investigation and writing – review & editing. YY was involved in conceptualization, project administration, supervision, and writing – review & editing.
Funding
The authors received no specific grants from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
MO is the CEO of Z-Works Inc., which supplied the equipment used in this study. However, MO's role in this study was limited to methodology consultation, provision of resources, and manuscript review. Z-Works Inc. had no involvement in the study's data collection, statistical analysis, interpretation, manuscript preparation, or decision to publish.
Data availability statement
The data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
