Abstract
Purpose
Remote patient monitoring (RPM) allows healthcare providers to monitor patient outcomes outside of a traditional healthcare setting, potentially supporting reductions in acute care utilization. This systematic review aims to assess whether RPM use among cancer patients reduces hospitalizations and length of stay (LOS).
Methods
A systematic review was conducted to identify articles published in PubMed and CINAHL between 2019 and 2024 that evaluated the impacts of RPM in cancer patients compared to the standard care. The primary outcomes examined were hospitalizations and LOS. Secondary outcomes were emergency department (ED) visits and hospital readmissions. Two reviewers screened and assessed the studies. Evidence strength was assessed using the Grading of Recommendations Assessment, Development and Evaluation approach. Risk of bias was assessed using the Newcastle-Ottawa Scale and the Cochrane risk-of-bias tool for randomized trials (RoB 2). While statistical tests were not conducted on results, outcomes were categorized as having increased, decreased, or no change.
Results
This review included one randomized controlled trial, seven cohort studies, and one case-control study. RPM was associated with reductions in healthcare utilization among cancer patients. There were significant reductions in hospitalizations, LOS, ED visits, and hospital readmissions in 67% (4/6), 67% (4/6), 75% (3/4), and 67% (2/3) of studies, respectively.
Conclusion
This systematic review builds upon existing literature that demonstrates the effectiveness of using RPM to manage acute conditions such as cancer. RPM may support management of various cancer-related conditions and, in turn, potentially reduce acute care use. However, study homogeneity and additional rigorous study designs are necessary to draw more definitive conclusions about the impacts of RPM in cancer care.
Keywords
Background
Cancer rates in the United States (US) have been rising, with cases climbing from 1.9 million in 2022 to over 2 million in 2023. 1 Innovative methods to diagnose, treat, and monitor cancer are needed to improve patient outcomes. Cancer patients often face adverse health events, leading to acute care visits. 2 Inappropriate symptom management is associated with excessive healthcare utilization including unplanned hospitalizations and longer LOS.3,4 Among a sample of cancer patients in the US, 65% experienced one hospitalization and 31% experienced two or more hospitalizations in the first year after diagnosis. 5 A 2023 study of cancer patients found that 15% of all hospitalizations were considered potentially preventable. 6 These types of acute care visits cause healthcare systems and patients significant financial strain, accounting for 48% of cancer care expenditures. 7 The total cost for cancer-related hospitalizations vary significantly based on the severity of a patient's condition and whether they have insurance. However, a brief published in 2021 reported that the most expensive hospital stays ranged between $$32,900 and $ $64,700$ for patients before insurance. 8 Acute care utilization could be prevented through more innovative outpatient approaches that manage cancer.
Remote patient monitoring (RPM) enables continuous management of cancer complications. RPM is a form of telehealth that records and transmits patient's biometrics, vital signs, and/or disease-related data outside of a conventional healthcare setting to a healthcare provider using information and communication technology. 9 RPM includes non-invasive devices like wearables that collect physiologic data, electronic patient-reported outcomes (ePRO) for manual symptom entry into a digital device 10 or invasive devices like cardiac monitors to regulate or track vital signs. 11 Using patient data, providers can monitor treatment response, identify complications, and intervene to manage symptoms beyond the clinical setting.11–13
Various factors influence the financial impacts of RPM programs on healthcare systems. In the US, hospitals with value-based care payment (VBC) models, such as the Centers for Medicaid and Medicaid Service's (CMS) 2023 Enhancing Oncology Model (EOM), compensate providers based on the quality of care delivered rather than volume of services provided.14,15 VBC approaches can potentially improve outcomes by prompting doctors to provide patient-centered, coordinated care. 14 Under CMS's EOM, participating oncology practices are financially incentivized to meet or exceed performance measures. 15 In contrast, hospitals using a fee-for-service (FFS) model reimburse providers for the volume of services provided. 16 To bill for RPM services, CMS uses a multitude of Current Procedural Terminology (CPT) and Healthcare Common Procedure Coding System (HCPCS) codes. Providers can bill for services, including RPM device setup and patient education (Code 98975), patient-provider communication related to RPM data (Code 99458), and therapeutic devices (Code 98980). 17
RPM is covered as a reimbursable Medicare program as long as healthcare practices meet a multitude of conditions set by CMS, including but not limited to: (1) an acute or chronic condition is being monitored; (2) monitoring is medically reasonable and necessary; and (3) the device used to collect and transmit the data meets the definition of a medical device as defined by the Food and Drug Administration. 17 Each state has its own billing and reimbursement policies for patients who are on Medicaid. 17 However, according to the Center for Connected Health Policy, RPM was covered through Medicaid in 42 states as of January 2025. 18 Furthermore, billing and reimbursement policies for private insurance may vary. In the case that a patient's insurance doesn’t cover RPM, a patient may have to pay out-of-pocket for services.
The CMS Hospital Outpatient Quality Reporting (OQR) Program include cancer quality metrics, the first of which is to reduce preventable acute care visits for chemotherapy-related symptoms. 19 The OP-35 and OP-36 rules penalize hospitals for high inpatient admissions and ED visits after chemotherapy or outpatient surgery, reducing payments by 2% for non-compliance. 19 RPM must reduce acute hospital use among patients receiving chemotherapy and surgical procedures for it to be a cost-effective method for health systems, thus exhibiting the need for evidence on the effects of RPM on care utilization outcomes. 20 While initial costs to implement RPM in healthcare organizations vary, an RPM program implemented for patients with hypertension reported that the average cost per enrolled patient cost the organization $330. 21 The program was reported to demonstrate a positive return on investment (ROI), with an average ROI of 22.2% or $1.22 made for every dollar that was invested into the program.. 21 In another RPM program used among COVID-19 patients, the cost-utility analysis found that the intervention was associated with an 87% reduction in hospitalizations and reduced per-patient costs of $11,472 over standard care. 22
RPM interventions that have been implemented to manage other disease-specific conditions such as heart failure, cardiovascular disease, and COVID-19 have successfully reduced hospitalizations and LOS.11,23 A systematic review conducted by Taylor et al. demonstrated that RPM was successful in reducing hospitalizations and LOS when implemented to manage chronic diseases. 11 In a study investigating the effects of RPM specifically among patients with cancer, Basch et al. found that RPM was associated with increased survival.24,25
However, more evidence is needed to demonstrate the effects of RPM on cancer care utilization. As of 2024, and to the best of the author's knowledge based on a comprehensive literature search, no reviews were identified that examined the impacts of RPM on healthcare utilization-specific outcomes among cancer patients. Hence, a review of the literature on these outcomes is warranted. This systematic review aimed to evaluate whether RPM is associated with reductions in hospitalizations and LOS among cancer patients. In this review, remote monitoring capabilities included ePROs for symptoms along with biometric monitoring using digital devices. The primary outcomes of interest were all-cause hospitalizations and LOS. Secondary outcomes were ED visits and hospital readmission.
Methods
A systematic review was conducted in June 2024 to evaluate whether RPM is associated with reduced hospitalizations among cancer patients compared to the standard of care. A protocol for this review was not prepared or pre-registered. No frameworks were used to formulate the research question or initiate the systematic review. Given the variability in study designs, objectives, cancer types, and populations in the literature, a meta-analysis was not conducted. Additionally, data analysis prioritized assessing broad categorizations of effects (e.g. increased, decreased, no change) rather than statistical aggregation, further warranting the decision to conduct a systematic review. CINAHL and PubMed were used to identify relevant studies since they were determined to be most aligned with the disciplines of medicine and health-related research. CINAHL was selected since it indexes the top nursing and allied health literature and includes search limiters specific to health, medicine, and evidence-based practice. 26 PubMed is considered a premier source for biomedical and life sciences literature, allowing for the retrieval of a comprehensive range of sources. 27 Detailed Boolean search strategies used a combination of medical subject headings and keywords related to remote monitoring, cancer, and healthcare utilization (Table 1). Reference lists of relevant systematic reviews and studies were consulted.
Boolean search strategy.

Eligibility criteria
Experimental and non-experimental studies published between January 1, 2019 and June 1, 2024 and conducted in the US were eligible. Insurance reimbursement policies surrounding RPM drastically changed in the US during the pandemic, increasing the adoption of RPM. Requiring studies to be published after 2019 ensures that this review is relevant to the current policies affecting oncology practices. Remote monitoring of clinical and/or physiological information for cancer-related conditions using non-invasive monitoring equipment, ePRO, and/or invasive implantable devices were eligible. Studies that investigated the following outcomes in cancer patients were eligible: (1) all-cause hospitalizations; (2) LOS; (3) emergency department (ED) visits; and/or (4) hospital readmissions. Table 2 provides definitions used to determine outcome eligibility.
Definitions used to determine eligibility of study outcomes.

Exclusion criteria
Studies were excluded if they (1) were theoretical studies, systematic and nonsystematic reviews, case studies, letters, conference abstracts, and commentaries; (2) monitored physical activity; (3) were not published in English; (4) were conducted outside of the US; and (5) collected ePRO prior to or at the time of a clinician visit. Billing and insurance reimbursement policies significantly impact the provision of RPM in the US, determining service coverage and data collection. 17 Since programs may base their RPM methods off federal policies, studies not conducted in the US were excluded to ensure relevance to the US healthcare system.
Study selection and quality assessment process
Two reviewers (CRB, JM) independently screened all of the titles and abstracts to determine if the studies met the criteria and independently assessed the full texts for eligibility. Conflicts between the two reviewers during the study selection process were resolved through discussion until consensus was reached. A third reviewer (SK) was available to settle unresolved conflicts, but this was not necessary. After excluding irrelevant articles, studies were selected for the review.
Risk of bias within studies was initially assessed by one reviewer (CRB), who manually reviewed every included article. The Newcastle-Ottawa Scale (NOS) was used to assess bias in cohort studies and case-control studies, with two different scales for each respective study type. 28 In order to assess each study, NOS uses a “star system” that is based on three broad perspectives: the selection of the study groups; the comparability of the groups; and the ascertainment of either the exposure or outcome of interest for case-control or cohort studies respectively. 28 The Cochrane risk-of-bias tool for randomized trials (RoB 2) was used to assess bias in randomized studies. 29 The tool focuses on different aspects of trial design, conduct, and reporting, structuring the assessment through a fixed set of bias domains. Each domain includes a set of questions to elicit information about features of the study that are relevant to bias. A proposed judgement arising from each domain is then generated based on answers to the domain questions, using ratings of “Low risk of bias,” or “High risk of bias.” Since the selected tools did not include domains to explicitly evaluate reporting bias, this was assessed separately by the reviewer. The criteria evaluated to assess for reporting bias included publication bias and selective outcome reporting.
After bias was assessed, the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach was used to assess the strength of evidence for outcomes (Table 3). 30 The quality of each outcome was rated by four levels of evidence across all studies, based on the five domains depicted in Table 3. Using the GRADE handbook, each study outcome was analyzed for a variety of criteria to develop a grade for each overall domain. 30
GRADE evidence profile on healthcare utilization outcomes.

The quality of evidence was assessed by one reviewer (CRB) based on a variety of factors. Through manual review of articles, studies were rated down if there was within-study risk of bias, indirectness of evidence, inconsistency of results, imprecision in effect size estimates, and publication bias. A bias rating was determined for each overall outcome based on the risk of bias scores that were gathered from individual studies, and the respective outcomes that they reported. Outcomes were rated up if there was a large effect size, dose-response relationship, and antagonistic bias. Per GRADE standards, prior to conducting the assessment, randomized controlled trials (RCTs) automatically received a rating of high but were downgraded depending on the presence of the five factors. 30 Furthermore, observational studies were graded as low quality initially but could be upgraded depending on the presence of the five factors. 30
Analysis
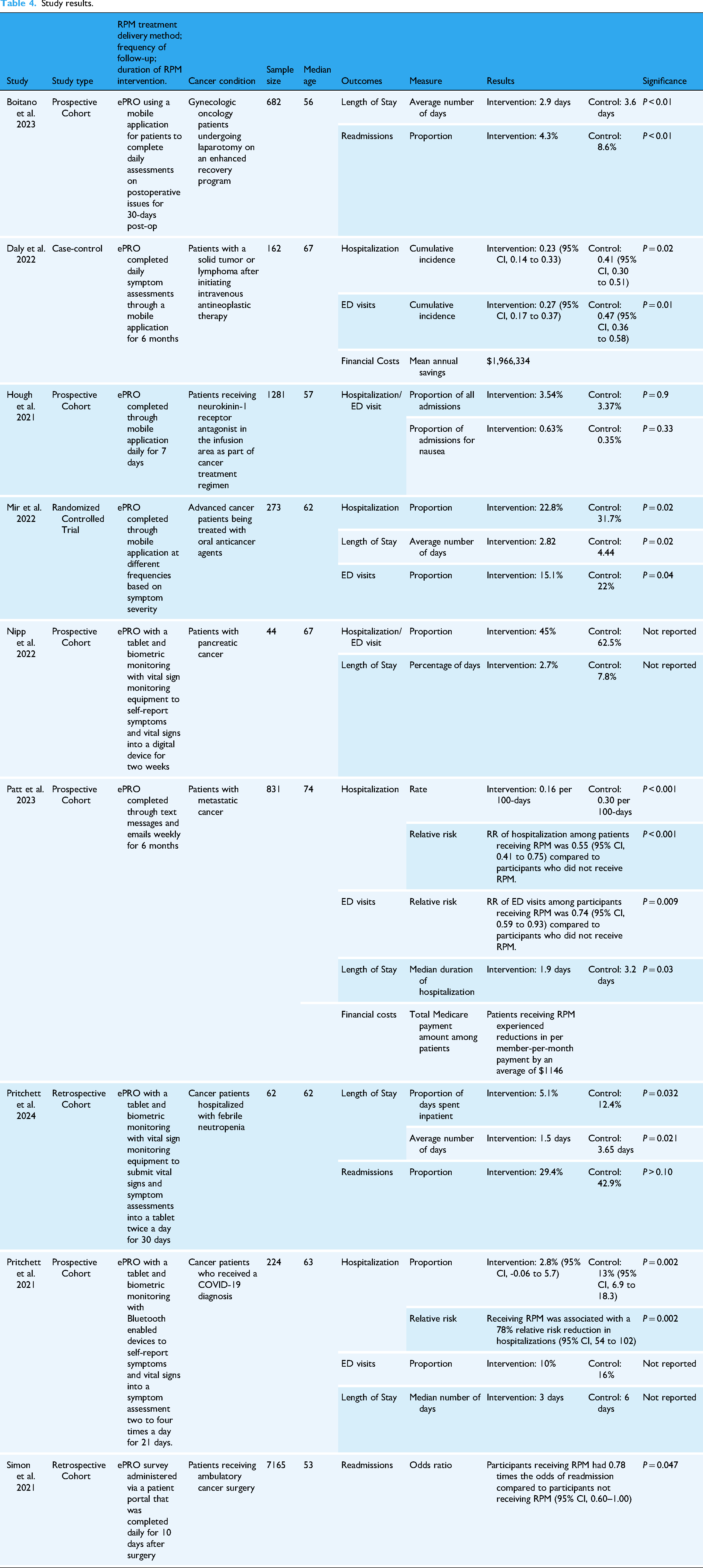
Information on multiple variables from studies was extracted manually by one reviewer (CRB) through a full-text review of each included article (Table 4). Each study was read in-depth to determine which outcome data was collected (hospitalizations, LOS, ED visits, and/or hospital readmissions) and how data were measured and reported. Findings from selected articles were then organized by outcome and categorized based on the reported effect on care utilization (increased, decreased, or no change). Outcomes that weren’t statistically significant were categorized as having no change. RPM intervention details, outcomes, measures used, statistically significant outcome results, and p-value were identified and extracted to be reported in this review (Table 4). While financial data were extracted if included, studies were not excluded if they did not report this. No methods were used to conduct sensitivity analyses or explore causes of heterogeneity.
Study results.
Results
Study selection
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram (Figure 1) depicts the citations reviewed, included, or excluded during the systematic review process. 31 After screening 499 studies, ten were identified as relevant. However, a study conducted by Doolin et al. ended up being excluded as RPM was only conducted once during the intervention. 32 The study included one RCT, seven cohort studies, and one case-control study.

PRISMA diagram of study selection process.
Risk of bias assessment findings
The scores from the risk of bias assessment conducted for each study are detailed in Table 5. Among all studies, bias tended to be low for selection and outcome domains. In regard to selection bias, studies generally had an intervention cohort that was representative of the general population. However, some studies relied on participants’ willingness to partake in receiving RPM, potentially introducing selection bias. Additionally, many cohort studies did not select the non-exposed participant group from the same population as the exposed group, resulting in a lower selection rating. Most studies leveraged data that was integrated into healthcare systems, such as patient EHR data or payer claims, enabling accurate detection of outcomes and adequate follow up with participants, thereby preventing information bias. Multiple studies reported shorter length to follow up times when evaluating healthcare utilization outcomes, which influenced the bias rating in the outcome domain. This may have affected statistical significance of outcomes, biasing results towards the null. Reporting bias was not present in any studies.
Risk of bias assessment.

There was high comparability bias in many of the cohort studies mainly due to the lack of controlling for potential confounding factors that could influence the relationship between RPM and healthcare utilization outcomes. Confounders that may have been introduced in studies included cancer type, stage of cancer, income, or race. The observational nature of these studies and small sample sizes could have influenced the ability to control for confounding through design or analysis.
Evidence strength assessment: GRADE findings
Per the GRADE assessments, the quality of evidence for hospitalizations was low and LOS was moderate (Table 3). ED visits and hospital readmissions received a very low and moderate evaluation, respectively. Since confounding factors and selection bias introduced bias into multiple interventions, particularly in the cohort studies, all outcomes received a lower bias rating. All outcomes reported relatively large effect sizes across studies, as demonstrated by significant p-values, resulting in low imprecision ratings. Furthermore, two of the six studies reporting hospitalization measurements combined proportions of ED visits and hospitalizations. As a result, hospitalization and ED visit outcomes were determined to have moderate indirectness. Given that only three of the six studies that reported ED visit outcomes included p-values or collected ED visit data alone, the quality of evidence significantly decreased for ED visit outcomes. Since reporting bias was low across individual studies, publication bias was determined to be undetected for all outcomes.
Study participant characteristics
Study characteristics are detailed in Table 4. The total population across studies was 10,724 participants and the median age ranged between 53 and 74 years old. RPM was used for cancer patients who had undergone a surgical procedure (n = 2), received therapeutic cancer treatment (n =5), or were diagnosed with an additional illness (n = 2). Interventions conducted with biometric monitoring equipment (n = 3) used devices such as blood pressure cuffs and monitors, pulse oximeters, heart rate monitors, thermometers, and scales to collect physiological measurements and vital signs. ePRO interventions (n =9) utilized mobile applications, tablets, and emails to assess symptoms. Details on data collection and RPM delivery methods are included in Table 4.
Program intervention characteristics
Given the variability of RPM intervention methods included in the review, the authors categorized the key program features into eight groups (Table 6). Categories were not mutually exclusive, with some programs incorporating multiple features. However, each program utilized either a primary care team or centralized care team—never both. Five programs linked RPM platforms or applications with patient's electronic records, including electronic medical and health records.33–37 All studies incorporated structured algorithms, using predefined, technology-embedded decision trees to generate alerts of adverse patient-generated data to the clinical team.
RPM program characteristic categories and definitions.

Two studies examined the financial landscape of the healthcare system in which their programs were implemented. Patt et al. reported that their intervention occurred within a VBC infrastructure, where prior investments had been made to hire staff, establish triage pathways, and set standards on response times to address patient symptoms. 38 While Daly et al. did not specify whether their intervention operated in a VBC or FFS model, they conducted a cost analysis of RPM using FFS-based pricing. Applying Medicare and commercial payer rates, they assessed ROI. While the annual cost of the program was $928,800, the mean annual savings were estimated at $1,966,334 due to reductions in acute care visits, yielding an estimated ROI of 2:1. 34 The total Medicare payment amount was reduced by an average of $1146 per member-per-month. 34
Hospitalizations
Of studies measuring hospitalizations, 67% (4/6) demonstrated significant reductions among participants receiving RPM. The cumulative incidence of inpatient admission among participants receiving intravenous antineoplastic therapy was 0.23 compared to 0.41 in control participants (P = 0.002). 34 RPM received among patients with COVID-19 was associated with a 10% absolute risk reduction and a 78% relative risk reduction in hospital admissions (P = 0.002). 35 Mir et al. reported that 22.8% of RPM participants were hospitalized compared to 31.7% of control participants (P = 0.02). 39 Additionally, the hospitalization rate among participants undergoing therapeutic treatment for metastatic cancer was 0.16 and 0.30 per 100-days in the ePRO intervention and control groups, respectively (P < .001). 38 Two studies reported no statistically significant differences in combined measurements of hospital admissions and ED visits among RPM participants.36,40 No studies reported negative associations between hospitalizations and RPM.
Length of stay
Of studies measuring LOS, 67% (4/6) demonstrated significant reductions among participants utilizing RPM. The mean LOS in the Mir et al. study was 2.82 days among participants compared to 4.44 days in control participants (P = 0.02). 39 RPM participants hospitalized with febrile neutropenia had a reduced LOS compared to controls (1.5 days versus 3.65 days, P = 0.021). 33 Gynecologic oncology patients also had a shorter LOS compared to the control group (2.9 days vs 3.6 days, P < 0.01). 37 Metastatic cancer patients had a reduced LOS compared to controls (1.9 days vs 3.2 days, P = 0.03). 38 While two studies reported no statistically significant differences in the LOS, there were no negative results reported among RPM recipients compared to control recipients.35,40
ED visits
Of studies reporting ED visit data separately from hospitalization data, 75% (3/4) indicated significant reductions associated with RPM. In Mir et al., the number of visits to the ED was significantly lower among participants receiving RPM compared to control participants, at 41 (15.1%) and 63 (22.0%) total visits, respectively (P = 0.04). 39 The cumulative incidence of an ED visit between intervention and control participants receiving intravenous antineoplastic therapy was 0.27 and 0.47, respectively (P = 0.01). 34 Metastatic cancer patients who completed ePRO were 0.74 times as likely to visit the ED compared to controls (P = 0.009). 38 While Pritchett et al. reported that the proportion of ED visits among RPM participants (10%) was lower than control participants (16%), findings were insignificant. 35
Readmissions
Of the studies reporting readmissions, 67% (2/3) indicated significant reductions among RPM participants. Simon et al. indicated the RPM participants had 0.78 times the odds of urgent care readmission within 30 days of ambulatory cancer surgery (P = 0.047). 41 Participants who completed ePRO after gynecologic surgery were significantly less likely to be readmitted to the hospital compared to the control group (4.3% vs 8.6%, P < 0.01). 37 Pritchett et al. reported decreased readmissions among RPM participants, it was not statistically significant. 33
Discussion
Principal findings
This review examined the relationship between individuals with cancer using RPM and patient care outcomes, namely, hospitalization and LOS. Eight of the nine studies described interventions that significantly decreased healthcare utilization, with 67% (4/6) and 67% (4/6) reporting significant reductions in hospitalizations and LOS, respectively. Significant reductions were exhibited in 75% (3/4) of studies that measured ED visit data alone and 67% (2/3) measuring hospital readmissions among RPM participants. No studies reported that RPM was associated with an increase in any of the four care utilization outcomes. Our findings supplement the current evidence-base, suggesting that RPM can help to reduce acute care utilization. However, there are some methodological gaps and differences across the studies in this review that make us unable to draw the conclusion that RPM directly contributes to reduced hospitalizations and LOS.
RPM was used for different purposes in the cancer care continuum, indicating that the type of RPM employed may depend on the type of cancer-related condition being monitored. Two of the three studies utilizing ePRO and biometric monitoring collected participant vital signs to manage secondary conditions, namely COVID-19 and febrile neutropenia.33,35 Given the effects that multiple illnesses can have on the immune system, these patients may be at greater risk of adverse health events and could benefit more from biometric monitoring.
Differences in study participant demographics, such as cancer stage, cancer type, and age, may have influenced RPM utilization behaviors and therefore, impacted outcomes. Patients could be more motivated to adopt RPM to prevent complications if their cancer was more severe. However, the severity and type of cancer could have reduced physical capacity and motivation to engage in RPM. Given the range of cancer types, study objectives, and stages of participants across studies, it is difficult to assess how these characteristics impact effectiveness of RPM.
RPM program elements may differentially affect participant behaviors and attitudes, such as self-efficacy and motivation, indirectly affecting healthcare utilization. Some characteristics of programs could enhance RPM, such as centralized care teams, participant education, and communication pathways. Integrating pathways to communicate with clinical teams may also increase a sense of connection with the clinical team, improving adherence to RPM. Developing structured algorithms appeared to be a crucial component of RPM based on the methods included in all our studies. As reported by Simon et al., alert-triggered interactions with the care team may have been most beneficial for managing symptoms. 41 Furthermore, there are various implications of integrating platforms with electronic records. Patients could potentially assess health trends more quickly, empowering them to manage symptoms at home. The incorporation of patient-generated data into health records may improve clinical workflow efficiencies by supporting real-time data access to drive decision-making and care coordination.
Centralized and primary teams provide various benefits in cancer care. Two studies included centralized teams who collaborated to manage reported symptoms, education, specialty referrals, and/or prescribing supportive medications.34,35 Daly et al. indicated that a centralized team could be resource intensive due to investment costs and shifting workflow processes. 34 However, care coordination could be enhanced across a centralized team if staff bring specialized expertise in distinct RPM programmatic areas. In contrast, a primary care team may promote continuity of care since the same clinician managing the patient's treatment plan would also handle symptom monitoring. Intervention methodologies influence healthcare utilization rates differently, making it difficult to conclude what impact healthcare utilization the greatest.
The median age among all studies was older, suggesting an age-dependent digital divide that could have affected RPM adoption. Mir et al. was the only study to discuss the implications of implementing digital interventions in an older group, describing that they may be less inclined to participate in RPM due to a limited understanding of its use or lack of access to the appropriate technology. 39 Digital literacy is a crucial factor that should be considered when developing programs, especially with complex interventions that use biometric monitoring equipment.
Several factors limit the expansion of RPM in cancer care, including concerns around reimbursements and the cost-effectiveness of RPM. 42 In addition, adoption of RPM has been modest due to concerns around liability to monitor and intervene in the continuous flow of data from RPM devices. 42 Lack of RPM best practices around data governance, patient engagement, and patient-clinician relationships are current obstacles to RPM expansion in cancer. 43 To advance policies that accelerate the adoption of RPM, there are two requirements: (1) data-driven evidence around the cost-effectiveness of RPM in cancer care; and (2) consensus around key quality indicators and metrics for the implementation of RPM in cancer care to demonstrate value to payers and policymakers.
Two studies discussed intervention costs, exhibiting a clear gap in the existing literature. If operational processes and care models are in place, such as those demonstrated by Patt et al., the VBC infrastructure of organizations could optimize RPM programs. 38 Providers in VBC arrangements could leverage real-time data offered through the different program characteristics highlighted in Table 6, such as structured algorithms, trend assessments, and communication pathways, to proactively manage cancer complications, thereby improving patient outcomes and reducing healthcare costs. Institutions working under FFS arrangements may forego revenue generated from providing care to hospitalized patients. However, there seems to be a shift towards application of VBC in oncology, reflected by the increased use of at-risk contracts and bundled payments along with the recent implementation of EOM. 34
While financial outcomes vary by payment model, RPM could help healthcare organizations meet CMS OQR program rules. Several CMS CPT/HCPCS billing codes could also improve the provision of RPM services. Specifically, code 98975 allows providers to bill for setting up RPM services and providing patient education, potentially reducing patient and/or caregiver burden of having to troubleshoot obstacles. 17 Education could improve digital literacy, potentially increasing patient satisfaction and retention in utilizing RPM. Providers could potentially receive financial renumeration for integrating these patient education and communication pathways at the benefit of patient outcomes.
The findings of this review are consistent with those of other studies that have examined outcomes in populations with other chronic conditions, including heart failure, diabetes, and COPD, where RPM was found to reduce healthcare utilization.11,44 As demonstrated through this review, RPM may have offer similar benefits to oncology patients that chronic disease patients have experienced. To the authors’ knowledge, this is the first systematic review that synthesizes the impacts of RPM on specific care utilization outcomes among cancer patients. The majority of systematic reviews conducted thusfar on RPM have examined the effects on patients with acute conditions more broadly, while cancer patients have been included as one subset of the population.11,45 Studies that have previously been conducted among oncology patients specifically have often focused narrowly on feasibility, adherence, and broadly defined patient outcomes related to satisfaction or survival.46,47 This review expands upon the existing literature by narrowing in on specific care-related outcomes, exhibiting the impact that RPM could have on cancer care delivery and healthcare system burden through the reduction of preventable high-cost visits.
Limitations
The review's limited number of studies and intervention heterogeneity make it difficult to determine where RPM is most impactful in reducing healthcare utilization along the cancer care continuum. While a meta-analysis would strengthen findings by providing a more precise estimate of the effect of RPM on outcomes, the results produced may have been misleading based on the varying quality of study methodologies and sample sizes included in the literature. The study authors aimed to limit study heterogeneity in the systematic review by creating narrow eligibility criteria. However, a low number of studies were included as a result. Only one RCT was included, limiting the strength of evidence and ability to draw causal inferences. The frequency and duration of RPM in selected studies also varied significantly. Among studies with shorter duration and low monitoring frequency, there may not have been an opportunity for irregular symptoms to be identified, therefore limiting the intervention's effect. Population size and age varied significantly across studies, which could have had differing effects on outcomes. Among studies that reported participant characteristics by race, the proportion of White participants was often significantly greater, limiting the generalizability of our findings to other races and ethnicities. Additionally, distance to healthcare systems and digital literacy may have influenced care seeking and adherence behaviors, biasing study findings. This review was not prospectively registered on PROSPERO because it was conducted prior to registration; however, we have adhered to established systematic review guidelines, and all methods, inclusion criteria, and analyses are reported in full to ensure transparency and reproducibility.
Conclusion
This systematic review builds upon existing literature that demonstrate the effectiveness of using RPM to manage acute conditions such as cancer. The current evidence suggests that RPM has the potential to reduce healthcare utilization among cancer patients, including hospitalizations and LOS, by enhancing symptom management and enabling timely interventions. However, the evidence base remains limited by study heterogeneity and methodological variability, underscoring the need for rigorous, well-designed studies to establish more definitive conclusions. Overall, RPM represents a promising yet evolving strategy to optimize cancer care delivery and improve patient outcomes.
Footnotes
Acknowledgements
Thank you to Jamie Conklin, Clinical Nursing Librarian in the Health Sciences Library at the University of North Carolina at Chapel Hill, for assisting in developing the search strategy used in this review.
ORCID iDs
Ethical considerations
Ethics approval was not required to conduct this systematic review.
Author contributions
The scope of this research was conceptualized by CRB, JM, MC, and SK. CRB and JM designed the methodology used and conducted the investigation, including the search, screening of record abstracts, and review of texts of all selected articles. A draft of the literature review was written by CRB under guidance from JM, MC, JG, and SK. Supervision was provided by SK throughout the writing of the draft. The draft was reviewed and edited by all authors. A final copy was approved by all authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Center For Advancing Translational Sciences of the National Institutes of Health (NIH) under Award Number RC2TR004380. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of NIH, nor does mention of department or agency names imply endorsement by the US government.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
