Abstract
Introduction
Varenicline is one of the most effective smoking cessation medications; however, non-adherence remains a significant barrier to successful quitting. Conversational agents have the potential to support medication adherence in home and community settings. However, generative AI models pose risks due to hallucinations, making them less reliable for this purpose. Rule-based chatbots provide a more transparent, theory-driven approach to patient support. Thus, we developed ChatV, a rule based chatbot grounded in the Behaviour Change Wheel framework, to enhance varenicline adherence.
Methods
ChatV was developed using a three-step process. First, we identified core determinants of varenicline adherence through a rapid review and qualitative interviews with healthcare providers and patients using the Theoretical Domains Framework. Second, we identified the intervention options through group discussions. Third, we identified intervention components using Behaviour Change Techniques (BCTs) Taxonomy v1. We applied the Acceptability, Practicability, Effectiveness, Affordability, Safety, and Equity (APEASE) criteria to determine the final intervention components.
Results
We identified 11 key domains relevant to behaviour change, including knowledge, beliefs about capabilities and consequences, memory, attention and decision-making processes, reinforcement, intentions, goals, social influences, environmental context and resources, behaviour regulation, and skill. Applying the APEASE criteria, we refined these to nine theoretical domains and identified 21 BCTs as core components of ChatV.
Conclusion
This study demonstrates a structured, theory-informed approach to chatbot development for medication adherence. By integrating evidence-based behaviour change principles with practical considerations, ChatV offers a model for designing rule-based conversational agents in healthcare.
Keywords
Introduction
The Canadian healthcare system is grappling with a multitude of issues including a shortage of healthcare providers (HCPs), financial constraints, and HCP burnout, 1 which have been exacerbated by the COVID-19 pandemic. 2 Primary care faces additional hurdles, including an aging physician population, declining interest in entering general practice, and rising rates of multi-morbidity amidst rapid population growth and aging.3,4
In this context, conversational agents, designed to generate responses that mimic human language, emerge as promising tools to address common healthcare challenges seen in primary care settings. 5 Conversational agents can streamline patient interactions, provide timely access to information, support decision-making processes, and enhance patient engagement. 6 Their potential to automate routine tasks, offer personalised healthcare recommendations and facilitate remote monitoring holds promise in optimising healthcare delivery and improving outcomes. These capabilities could help solve many of the challenges the healthcare systems around the world are facing, including reducing financial costs, 7 HCP burnout, 8 and enhancing patient experiences and outcomes. 9
However, implementing conversational agents within healthcare settings also poses several challenges. Several researchers have shown that generative artificial intelligence can augment our existing biases.10–12 Historically disadvantaged groups can be especially negatively affected by bias, leading to less accurate diagnoses and inadequate estimates of the need for medical care. 10 In addition, most generative artificial intelligence systems still operate as a ‘black box’, 13 where we lack transparency into the mechanisms governing their outputs, limiting our ability to comprehend their decision-making processes fully. This stands in contrast to the U.K. Medical Research Council (MRC) guidelines 14 which advocate for a systematic approach to intervention development and evaluation, placing greater emphasis on the supporting evidence base and the utilisation of theory.
In contrast to generative AI, rule-based chatbots provide a more transparent framework, where the decision-making processes are explicitly coded based on predefined rules and logical structures. 15 This transparency allows researchers and practitioners to understand exactly how the chatbot arrives at its conclusions and guides users through interactions. While this approach may limit the flexibility and adaptability of the chatbot compared to generative AI, it ensures a more predictable and controllable system. Moreover, rule-based chatbots can be more easily aligned with theoretical models and evidence-based practices, as their behaviour can be explicitly designed to follow intervention frameworks.
One area where conversational agents could have an important role is in smoking cessation. Each year, over 46,000 Canadians die due to smoking-related illnesses. 16 Evidence-based treatments are available to aid people who smoke in quitting, with numerous clinical practice guidelines recommending pharmacotherapy and behavioural interventions to those ready to quit. 17 Varenicline is one of the most effective smoking cessation medications, that acts as a partial agonist of the α4β2 nicotinic acetylcholine receptor, reducing nicotine withdrawal symptoms and diminishing the rewarding effects of smoking. 18
Compared to other first-line pharmacotherapies such as nicotine replacement therapy or bupropion, varenicline has demonstrated higher abstinence rates in head-to-head trials and meta-analyses. 19 Despite its proven efficacy, and coverage on public and several private drug formularies, and being the most commonly prescribed smoking cessation medication in the United States, 20 poor adherence remains a significant barrier, undermining the potential for successful smoking cessation.21,22 Common reasons for poor adherence include side effects such as nausea and vivid dreams, lack of ongoing behavioural support, forgetfulness, early relapse to smoking, and limited understanding of the importance of full-course treatment.22–25 A rule-based chatbot offers a promising solution to this problem given its ability to provide consistent, scalable, and evidence-based behavioural support. By automating interactions that mimic those of human counsellors, a rule-based chatbot can guide users through medication routines, deliver reminders, and offer tailored feedback, all the while adhering closely to structured behavioural intervention frameworks. Its predictable and transparent nature makes it a reliable tool for ensuring fidelity to adherence strategies, while also being more accessible than counselling services to a broader audience.
While studies have shown that conversational agents can increase the engagement of smoking cessation apps 26 and a recent scoping review found preliminary evidence of their potential benefit for smoking cessation overall, 27 none of the reviewed interventions appeared to be directed specifically towards supporting smoking cessation medication adherence. The review also emphasised the need for researchers to provide more in-depth descriptions of chatbot theoretical underpinnings, and consistency in language and terminology was recommended to facilitate synthesis across studies. To help address these gaps, in this article, we detail the development of ChatV, a theory-informed, rule-based chatbot designed specifically to support adherence to varenicline and informed by the Behaviour Change Wheel (BCW). 28
The BCW offers a robust theoretical foundation for designing interventions and has proven successful across diverse settings.29–33 The BCW consists of the Capability, Opportunity, and Motivation (COM-B) model, which identifies the essential factors for behaviour change; the nine intervention functions, such as education, training, and incentivization that define the types of interventions needed; and seven policy categories, offering high-level strategies for implementing these interventions. In addition, interventions incorporate behaviour change techniques (BCTs), the ‘active ingredients’ specified in the BCTs Taxonomy v1 (BCTTv1), which enable precise content descriptions.28,34 These BCTs were later converted into BCT Ontology (BCTO) labels 35 and allow for improved standardisation, interoperability with other ontologies, and enhanced clarity in digital and automated intervention design. A BCW guide provides theory-based connections between COM-B behaviour influences, intervention types, and BCTs, aiding in the selection of intervention components. 28 The BCW aligns with other behavioural frameworks, including the Theoretical Domains Framework (TDF), which builds on COM-B by revealing specific barriers and facilitators to change. 36 The TDF synthesises key constructs from multiple behaviour change theories into 14 domains that help identify the underlying determinants of behaviour in a systematic and theory-informed way. 36
In addition to its structured approach, the BCW framework incorporates the Acceptability, Practicability, Effectiveness and cost-effectiveness, Affordability, Safety, and Equity (APEASE) criteria to evaluate potential intervention options. 37 This ensures that chosen interventions not only address the identified behaviour determinants but also align with real-world considerations such as resource constraints, stakeholder support, and ethical implications. By applying the APEASE criteria, researchers and practitioners can enhance the feasibility and impact of interventions while mitigating unintended consequences.
In our protocol papers,38,39 we detail our proposed approach to developing and testing a rule-based chatbot, ChatV, designed to improve adherence to varenicline. The literature review, which also helped inform the development, was previously published. 40 ChatV is a web-based chatbot that can currently be accessed by research study participants with log in credentials provided by research staff members. It can be used 24/7 to document medication adherence and smoking status, and use other functionalities such as asking questions about varenicline and cigarette smoking.
This article focuses on describing the initial steps of adopting a theory-driven approach in the development process and the rationale for selecting BCTs to develop ChatV.
Methods
Study design
This study describes the qualitative interviews and consensus discussion used in the development of the ChatV. It was conducted at the Centre for Addiction and Mental Health and the University of Toronto. We designed ChatV in three steps (Figure 1). The first step focused on identifying and situating the determinants of varenicline adherence and of using digital technology, and the use of digital technology within the COM-B model. 28 In the second step, we identified intervention functions using the BCW's structured approach. The intervention options are chosen from a set of nine potential functions (e.g. education, persuasion, incentivization, training, environmental restructuring) based on their relevance to the identified behavioural determinants. The third step involved specifying the active components of the intervention using the BCTTv1, and later converted to BCTO, 35 and finalising components we would include in ChatV based on the APEASE criteria.

Design process.
Across all three steps, we used the same dataset, comprising findings from a rapid systematic review that examined the determinants of varenicline adherence (methodology and results reported elsewhere 40 ) and interviews conducted with HCPs and patients, the details of which are presented in this article.
Ethics approval
Ethical approval was granted by The Centre for Addiction and Mental Health (CAMH) Research Ethics Board (REB No. 050/2022).
Step 1 – Understanding the behaviour
Sampling and recruitment of patients and HCPs
The target sample size for the development of ChatV was 20 HCPs and 20 people who had used varenicline (patients). This target was based on prior research indicating that data saturation in qualitative studies is typically achieved with 9–17 interviews. 41 We employed purposeful, non-probabilistic sampling 42 to ensure a diverse sample. For patients, we aimed for a diverse group of participants across different genders and ages; for HCPs, we prioritised gaining diversity by purposefully sampling by specialty, clinic type, geographic location, and population size. We monitored for data saturation throughout the analytic process. Saturation was determined when no new codes, domains, or themes were identified in consecutive interviews.
Eligibility criteria for patients and HCPs
Patients were eligible if they:
Were currently using varenicline or had taken it within the last six months. Self-reported difficulty adhering to their prescription. Were at least 18 years old. Could read and speak English.
HCPs were eligible if they:
Had seen at least five patients prescribed varenicline within the last two years. Could read and speak English.
Recruitment methods for patients and HCPs
Both HCPs and patients were recruited through The CAMH Smoking Treatment for Ontario Patients (STOP) Program, an Ontario-wide smoking cessation initiative offering free counselling and nicotine replacement therapy to individuals interested in quitting smoking. 43 In addition, patients were recruited through Community Boards and Online Platforms: These included Rainbow Health Ontario, the CAMH Nicotine Dependence Clinic website, and clinics offering smoking cessation services in the Greater Toronto Area. HCPs were also recruited through the monthly STOP Program teleconferences, the Nicotine Dependence Clinic at CAMH, and the Training Enhancement in Applied Counselling and Health Project Listserv, which is a tobacco cessation education programme. 44
Procedures for patient and HCP interviews
After confirming eligibility, an MSc student researcher (mentored by NM) conducted the informed consent discussion over the phone, explained the study, and answered questions. We emailed the consent forms to all participants, who signed them electronically. One MSc student conducted all patient interviews between October 2022 and January 2023, and a different MSc student conducted all HCP interviews between January and May 2023. Both students explained that the project was funded by the PI (NM) and would form part of their MSc theses, and that we were developing a chatbot where participants’ input would directly shape its design and functionality, reflecting a co-creation approach. For patients, this ensured the chatbot met their needs, while for HCPs, it helped identify features that would make the chatbot easier and more appealing to recommend to their patients. Only the interviewer and the interviewee were present during each session. Both interviewers received training in qualitative interviewing from NM and KM. After each interview, the interviewers kept detailed notes, which were discussed with NM or KM on a weekly basis along with the interview content. Both interview guides probed for determinants of varenicline adherence and usefulness of digital technologies (conversational agents for varenicline adherence in particular), intervention functions, and BCTs that should be included in a digital intervention (interview guides available in Supplemental Material). The interview guide had been pilot tested among research staff before use. Patient interviews lasted between 27 to 66 min, and HCP interviews lasted between 30 to 60 min. Interviews were conducted over the phone, audio-recorded, transcribed verbatim, and cross-checked for accuracy by the student researcher. Transcripts were not returned to participants for comment and/or correction.
Analysis of patient and HCP interview data
Step 1: Identification of determinants of varenicline adherence. We employed framework analysis, a method well-suited for applied health research that allows for systematic categorisation of data into predetermined and emergent themes.
45
The data was analysed by:
Transcription: A professional transcriptionist transcribed all interviews verbatim, and a researcher cross-referenced the transcripts with audio recordings to ensure accuracy. All transcripts were de-identified and securely stored in CAMH's server. Familiarisation: Researchers read and re-read the transcripts to immerse themselves in the data and gain an overall understanding. Coding: Initial coding was conducted by organising data into themes based on the TDF and BCTs, along with any emerging concepts from the interviews. Developing an analytical framework: An analytical framework was developed by refining the codes and categories based on the initial coding process. Applying the framework: The framework was systematically applied to all transcripts to ensure consistency in coding. Charting data into a framework matrix: The data were summarised and organised into a matrix, facilitating comparison of themes across participants. Interpreting the data: Patterns and relationships between the themes were identified, and findings were synthesised to draw meaningful conclusions.
After the interviews were transcribed and checked for accuracy, they were imported to qualitative analysis software (NVivo V. 12). To ensure the reliability of the coding process, two independent researchers coded 25% of the transcripts. Interrater reliability was assessed using Cohen's kappa, yielding a kappa coefficient of 0.90 and a 99.6% agreement for patient interviews and a kappa coefficient of 0.94 and a 99.7% agreement for HCP interviews. After achieving this high level of agreement, a single researcher coded the remaining transcripts. The transcripts were coded based on the TDF to identify determinants of varenicline adherence. Participants did not provide feedback on the findings.
Step 2 – Identification of intervention options
The framework matrix, compiled from Step 1, was reviewed and discussed by the research team to establish criteria for determining the relevance of each domain to varenicline adherence. The summaries were then assessed to determine whether participants explicitly connected the domain to the target behaviours. Using a consensus-based approach, the multidisciplinary research team (researchers, clinicians, and the tech vendor) selected key theoretical domains for the intervention. Selection decisions were informed by identified barriers and facilitators within the relevant domains and were grounded in the feasibility of addressing these factors within the constraints of digital intervention and the available project resources.
Step 3 – Identification of intervention content and implementation outcomes
The final step involved mapping theoretical domains to BCTs. Three main sources of data were used to identify BCTs:
Two researchers (NM and KM) reviewed all BCTs mapped to the key domains across both sources and reached a consensus on which BCTs to include in the intervention by applying the APEASE criteria. Each behavioural change technique identified was assessed not only for its potential impact on varenicline adherence but also for its feasibility and relevance in the real world:
For the purpose of this article, and to ensure consistency with the latest terminology, we converted the identified BCTs to their corresponding BCTO 35 labels and Behaviour Change Intervention Ontology (BCIO) identifiers. The BCTO was released after we had initiated our study; therefore, we report both the original BCT labels, used during intervention development, and the updated BCTO labels and numbers. This dual reporting approach enhances transparency, reproducibility, and alignment with evolving standards in behavioural science and digital health research. By adopting the BCTO framework, we contribute to the growing movement towards more precise, computable representations of behaviour change interventions, enabling better cross-study comparisons, meta-analyses, and implementation in digital systems.35,47
Results
Participant characteristics
We reached 89 of the 139 patients who we attempted to contact to participate in the interview. Of these, 27 declined participation and 62 were screened for eligibility. Thirty-six patients were found to be eligible to participate (reasons for ineligibility usually included not taking varenicline in the last six months or not experiencing issues with adherence), and 26 people provided informed consent, out of which 20 patients completed the interview. Twenty HCPs reached out to participate in the interviews, out of which 19 met eligibility criteria, provided informed consent and completed the interviews. Table 1 provides the socio-demographic characteristics of the patient and HCP participants.
Patients’ and healthcare providers (HCPs)’ socio-demographic characteristics.

Summary of interviews
Data saturation was reached after 16 patient interviews and 15 HCP interviews; additional interviews were conducted to ensure robustness and confirm saturation.
Analysis of patient interviews identified four overarching themes related to barriers and facilitators to varenicline adherence.
Analysis of HCP interviews identified six overarching themes that shaped their decision to recommend or not recommend a digital health tool such as a ChatV.
Summary of key determinants
As can be seen in the themes above, various factors were identified as barriers and facilitators to medication adherence as well as to use of digital technologies When we mapped these determinate to the TDF we identified eleven key TDF domains for behaviour change :
Key theoretical domains identified in patient and healthcare provider (HCP) interviews.

Identification of intervention options (Step 2)
The findings from the rapid systematic review and interviews identified similar domains, with one exception of pessimism, where patient participants reported that their confidence in the effectiveness of varenicline diminished over time.
40
While both sources highlighted comparable barriers and facilitators to varenicline adherence, the interviews offered richer context through personal and HCP narratives. The research team determined that all domains, except for
Identification of BCTS (Step 3)
A total of 24 BCTs were identified from the rapid systematic review 40 and interviews with patients and HCPs and considered for inclusion in ChatV. These BCTs were systematically evaluated for inclusion, guided by the APEASE criteria. Of the 23 BCTs, 20 met the APEASE criteria and were endorsed for implementation. Two BCTs, problem solving and social support (unspecified), were partially implemented based on participants’ suggestions, while other proposed features were excluded (see Tables 3 and 4 for details). Although most BCTs contained elements that aligned with the inclusion criteria alongside others that did not, the excluded BCTs posed more substantial challenges, ultimately warranting their exclusion.
Behaviour change techniques (BCTs) integrated into ChatV and their alignment with APEASE criteria.

BCTs that did not fulfil APEASE criteria.

The excluded BCTs were as follows ( Monitoring outcome(s) of behaviour by others without feedback (2.1); (BCIO: 007020): This involved providing HCPs access to patient profiles without offering feedback. Concerns were raised regarding this approach's cost, practicability and acceptability, particularly around data privacy and patient autonomy. Problem solving (1.2) (BCIO: 007008): This proposed providing information unrelated to varenicline or smoking but potentially helpful (e.g. locations of nearby restaurants to enable patients to have food before taking medication). This BCT was excluded due to its limited relevance to the intervention's primary objectives and questions about its effectiveness. Social support (unspecified) (3.1) (BCIO Social support (emotional) (3.3) (BCIO: 007036): This proposed linking ChatV to unrelated apps for meditation or music to provide emotional support. Affordability and integration challenges were primary reasons for exclusion. Punishment (14.2) (BCIO: 007241): This involved emphasising guilt to promote adherence. This BCT was excluded due to safety concerns, as it could exacerbate stress or shame, particularly among users facing adherence challenges due to personal or environmental barriers. Such an approach risked reducing motivation, increasing anxiety, and potentially leading to avoidance of ChatV, thereby undermining the intervention's purpose and causing unintended harm.
Table 3 provides a detailed overview of the 21 BCTs that met the APEASE criteria, including their theoretical underpinnings within the TDF and how they were operationalised in ChatV. Table 4 summarises the five BCTs that did not fulfil the APEASE criteria, along with the specific reasons for their exclusion.
Figures 2–5 illustrate ChatV's visual layout, tone of messaging, and how selected BCTs are operationalised within the conversation flow.

ChatV's menu.

Example of Commitment (1.9) BCTO: affirm commitment BCT (BCIO:007015).

Example of Feedback on outcome of behaviour (2.7) BCTO: provide feedback on outcome of behaviour BCT (BCIO:007027).
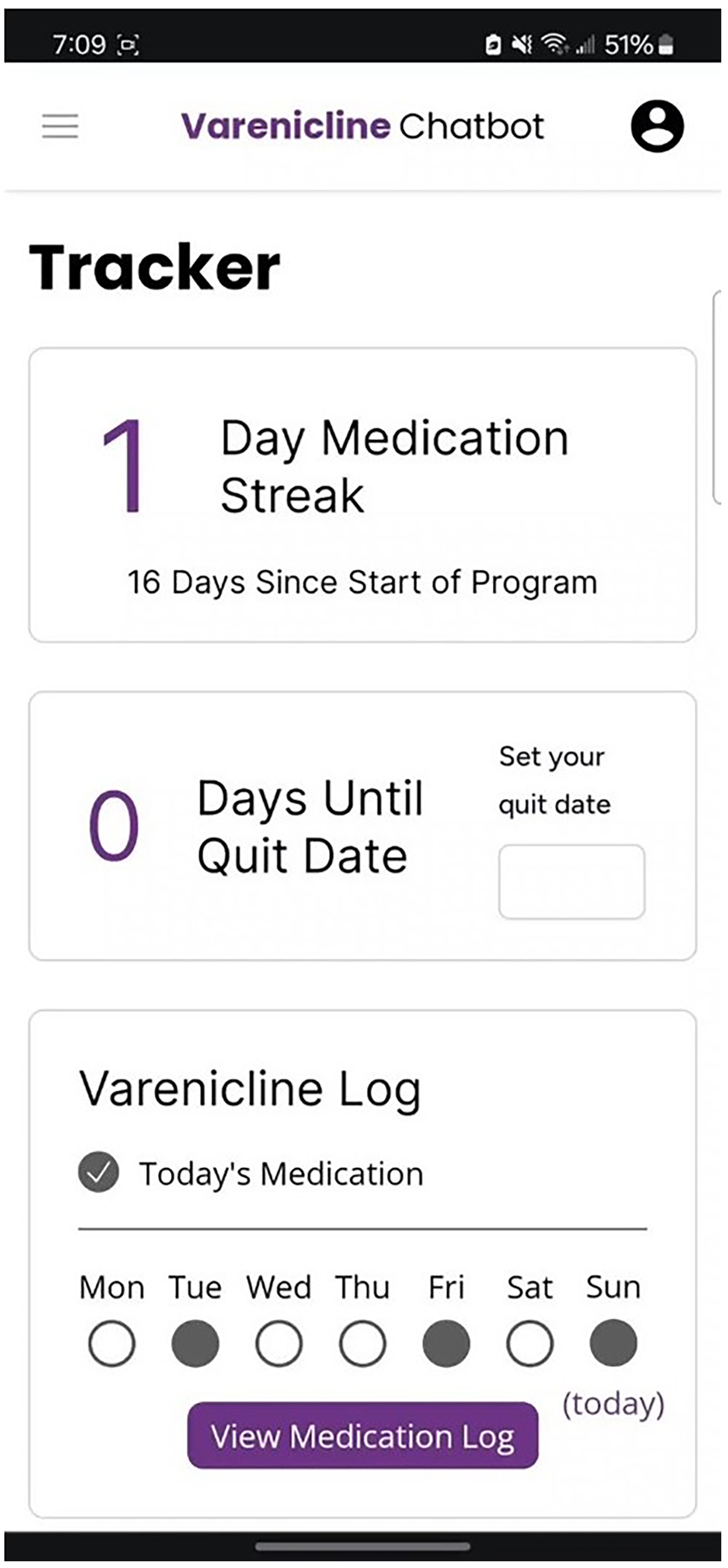
Example of Self-monitor outcomes of behaviour (2.4) BCTO: self-monitor outcome of behaviour BCT (BCIO:007025).
Discussion
This study outlines the systematic development of ChatV, a conversational agent designed to enhance adherence to varenicline, guided by the BCW as a theoretical framework. Using a previously published rapid systematic review 40 and semi-structured interviews with HCPs and patients, we identified 20 BCTs from nine TDFs for potential incorporation into a varenicline-adherence app. Compared to other conversational agents focused on health behaviour change, ChatV incorporates a greater number of BCTs. A recent scoping review found that the average number of BCTs used in ‘lifestyle-change’ conversational agents was nine, 48 which is considerably lower than what we included in ChatV. Some of the BCTs in ChatV, such as problem-solving, goal setting, and self-monitoring, align with those commonly identified in the scoping review. 48
While there were many similarities in the preferences expressed by patients and HCPs regarding features (or BCTs) for ChatV, we identified a few differences. Patients emphasised the importance of immediate, accessible, and personalised support, whereas HCPs prioritised evidence-based, transparent features that align with clinical guidelines. In addition, one patient participant mentioned the BCT of punishment as a desirable feature in the chatbot. However, the HCPs did not report a similar BCT, nor does the literature report such features. Indeed, based on the APEASE criteria, this feature was excluded due to the acceptability and safety for other patients.
A strength of this study is its deliberate inclusion of input from individuals who face social and structural disadvantage. Many, participants represented groups that are often underrepresented in digital health design, including women, people with low income, and low formal education.49,50 This demographic diversity enhanced the relevance, acceptability, and potential equity impact of ChatV, particularly in tailoring behavioural strategies to users at higher risk of tobacco-related harms who are often excluded from clinical and digital innovation efforts. 51
To be able to provide tailored evidence-based support to users, we programmed ChatV from December 2023 to May 2024 based on the data collected to answer user questions, aligning with instructions on how to perform the behaviour and problem-solving. In order to build trust, we include the source of the evidence. This feature provides tailored, evidence-based information addressing user doubts about taking varenicline and managing side effects. For instance, if a user is unsure how to handle a missed dose or manage a specific side effect, ChatV delivers actionable advice informed by the product monograph, clinical guidelines, and user input. According to self-determination theory, 52 addressing user questions in this way enhances autonomy by helping individuals feel informed and confident in their decisions.
Several BCTs incorporated into ChatV aim to reduce cognitive load, which, according to Cognitive Load Theory,
53
enhances decision-making by optimising working memory capacity. ChatV supports users in developing clear, actionable strategies for medication adherence, for example, taking their medication after breakfast with a glass of water (action planning)
Both patients and HCPs emphasised the importance of providing positive reinforcement to users, whether through rewards (e.g. stars, streaks, and accomplishments), encouraging messages, or feedback on progress. This emphasis on positive reinforcement aligns with a growing body of evidence from other digital behaviour change interventions, which frequently incorporate reward systems and positive feedback as effective mechanisms to drive adherence and sustain behaviour change.48,55 Research highlights that such strategies not only enhance user engagement but also create a sense of intrinsic motivation and satisfaction, further reinforcing the desired behaviours.48,55 This emphasis on positive reinforcement aligns with findings from other digital interventions, which frequently incorporate gamification elements and positive feedback as effective strategies for sustaining behaviour change.56,57
Another core focus of ChatV is building self-efficacy and promoting self-management. Techniques such as goal setting, action planning, and problem-solving were incorporated to empower users to take ownership of their adherence journey. Based on patient interviews, personalised goal-setting features were designed to integrate adherence behaviours into users’ daily routines while addressing their unique challenges. For example, users can set specific goals, such as taking their medication at the same time each day, and receive tailored prompts to help them achieve these objectives. Evidence suggests that self-management strategies are particularly effective in fostering adherence, especially in the context of smoking cessation and chronic disease management. 58 The personalisation implemented in ChatV reflects the successful use of self-management techniques in digital interventions, as documented in systematic reviews.
Several participants expressed a desire for ChatV to provide social support. While we could only implement a limited number of the requested features, we included options such as encouraging users to share their progress and behavioural contracts with friends, family, or research staff, to harness the accountability and motivation derived from social networks, which are well-documented as key factors in sustaining long-term behaviour change. 59 Some suggested features, such as communal chat boards, were excluded. Although these could have facilitated peer-to-peer support, they were considered impractical and potentially unsafe. Concerns about long-term sustainability, risks such as cyberbullying, misinformation, and inadequate moderation posed significant barriers to their inclusion.
ChatV differs in several important ways from existing smoking cessation and medication adherence chatbots. First, whereas most chatbots have been developed without an explicit theoretical foundation,27,60 ChatV was intentionally designed using the BCW, ensuring theory-driven, evidence-based content and structure. Compared to existing smoking cessation chatbots, which often focus broadly on motivation or behaviour change without addressing medication adherence specifically, 26 ChatV was uniquely designed to support adherence to varenicline, one of the most effective smoking cessation pharmacotherapy, by targeting known barriers and facilitators of adherence identified through interviews with both patients and HCPs. When compared to medication adherence chatbots, ChatV is also distinctive. Many such tools rely primarily on reminders and basic educational prompts, 61 whereas ChatV integrates a broader range of BCTs.
This study contributes to the growing body of literature demonstrating how the TDF and the U.K. MRC's guidance can be applied to the development of digital health interventions, with a novel focus on their application in the context of a rule-based chatbot. In doing so, it addresses key gaps identified in a recent scoping review,
27
which highlighted the lack of detailed descriptions of chatbot functionality, delivery mode, and theoretical underpinnings in existing smoking cessation interventions, as well as the need for greater consistency in terminology. Importantly, our work documents not just
By detailing these considerations and challenges, this study offers insights into the nuanced process of translating behavioural science frameworks into actionable components for rule based conversational agents. As detailed in our protocol manuscript, 39 we are currently conducting a feasibility study to evaluate whether ChatV is used, whether it is used as intended, and whether it warrants testing in a randomised controlled trial.
Several limitations should be acknowledged in the design of ChatV. While we aimed to make the process of choosing BCTs as transparent and reproducible as possible, we encountered several challenges.
First, using the APEASE criteria to select BCTs required subjective judgement in evaluating techniques across multiple dimensions. For instance, assessing the ‘acceptability’ of a technique can vary significantly based on individual user experiences or cultural contexts. Similarly, what is considered ‘practicable’ for a chatbot may depend heavily on the available technical infrastructure. These subjective interpretations introduce variability in how BCTs are selected and implemented within a digital intervention.
Second, many BCTs overlap conceptually, making it difficult to distinguish between them and select the most appropriate one for a given scenario. For example, techniques related to ‘goal setting’ and ‘action planning’ often share similar features but may be classified separately depending on their emphasis. This overlap can complicate efforts to maintain consistency and clarity in intervention design. To mitigate these issues, in this article we also report on the updated BCTO, 35 which offers more granular and clearly defined categories, improving precision, standardisation, and interoperability in the classification and application of BCTs within digital interventions.
Third, we did not have a working prototype of ChatV, which may have made it harder for participants to fully envision how specific techniques would function within the chatbot. Without direct interaction with a prototype, participants had to rely on descriptions and hypothetical scenarios, which could have influenced their judgements about the feasibility and usability of different BCTs. To address this, we conducted a Wizard of Oz (WoZ) study, where human operators simulated the chatbot's responses to assess how users interacted with the intervention. The findings from the WoZ will be described in a separate manuscript.
The use of a systematic approach to ChatV development is feasible and should be followed before testing the chatbot in a feasibility trial. Despite the limitations, the MRC framework helps locate the stages of the project, and the COM-B model helped make decisions about the inclusion or exclusion of features in the chatbot. It remains to be proved that such efforts translate into better patient outcomes at scale.
Conclusion
This study describes the systematic, theory-informed development of ChatV, a rule-based conversational agent designed to improve adherence to varenicline, a highly effective smoking cessation pharmacotherapy. We selected a diverse set of BCTs based on input from patients and HCPs and applied the APEASE criteria to ensure each BCT was evidence-based, feasible, equitable, and acceptable for implementation within a digital intervention.
To enhance transparency, reproducibility, and alignment with emerging standards, we reported both the original BCT labels and their corresponding BCTO labels and BCIO identifiers. This dual reporting ensures our work remains accessible and relevant to both current and future behaviour change researchers and digital health developers.
Importantly, ChatV incorporates more BCTs than most existing health behaviour chatbots and addresses several documented gaps in the literature, including limited use of theory, poor reporting of chatbot functionality, and lack of consistency in terminology. By integrating theoretical frameworks with practical design decisions, we offer a replicable model for others developing digital adherence tools.
Our findings contribute to the growing evidence base on how to design rigorous, user-informed digital health interventions. Results from our ongoing feasibility study will determine whether ChatV is acceptable, usable, and ready for full-scale evaluation in a randomised controlled trial. Ultimately, ChatV represents a scalable, evidence-informed approach to improving medication adherence, increasing quit success, and reducing tobacco-related harms at the population level.
Supplemental Material
sj-pdf-1-dhj-10.1177_20552076251380324 - Supplemental material for Using the behaviour change wheel framework to develop a rule-based chatbot to support varenicline adherence for smoking cessation
Supplemental material, sj-pdf-1-dhj-10.1177_20552076251380324 for Using the behaviour change wheel framework to develop a rule-based chatbot to support varenicline adherence for smoking cessation by Nadia Minian, Kamna Mehra, Jonathan Rose, Scott Veldhuizen, Laurie Zawertailo, Matt Ratto, Ryan Ting-A-Kee, Osnat Melamed, Victor Tang and Peter Selby in DIGITAL HEALTH
Footnotes
Acknowledgements
We gratefully acknowledge Sowsan Hafuth and Mackenzie Earle for their valuable contributions to this project, particularly for their support in conducting interviews. Their dedication and assistance were instrumental in advancing this work.
Ethical considerations
The Research Ethics Board of the Centre for Addiction and Mental Health approved this study (REB No. 50/2022).
Author contributions
Nadia Minian: conceptualisation, funding acquisition, methodology, project administration, supervision, visualisation, writing – original draft preparation, and writing – review and editing; Kamna Mehra: formal analysis, investigation, methodology, project administration, visualisation, writing – original draft preparation, and writing – review and editing; Jonathan Rose, Scott Veldhuizen, Laurie Zawertailo L, Matt Ratto and Peter Selby: funding acquisition, methodology, and writing – review and editing; Ryan Ting-A-Kee, Osnat Melamed and Victor Tang: methodology, and writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by a Proof of Concept Intervention Grant in Primary Prevention of Cancer (Action Grant) of the Canadian Cancer Society and the Canadian Institutes of Health Research-Institute for Cancer Research (Grant No. 707218) and a Canadian Institutes of Health Research Project Grant: Funding Reference No. PJT 180405.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NM reports receipt of funding from Canadian Institutes of Health Research, Canadian Cancer Society, and from the Discovery Fund of CAMH. PS reports receipt of funding from Canadian Institutes of Health Research, Canadian Cancer Society, Pfizer Inc., Pfizer Canada, and Ontario Lung Association. PS also reports that through an open-tender process, Johnson & Johnson, Novartis, and Pfizer Inc. are vendors of record for having provided smoking cessation pharmacotherapy for research studies at free or discounted rates.
Guarantor
Nadia Minian
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
