Abstract
The intersection of digital health technologies (DHT) and real-world evidence (RWE) is redefining the landscape of clinical research and regulatory science, offering unique opportunities to improve drug development, patient monitoring, and evidence-based decision-making. The extensive use of digital technologies has facilitated continuous, real-time health data collection, while RWE helps bridge the gap between controlled clinical trials and real-world patient experiences. As healthcare systems progressively integrate these various data sources, they are reshaping regulatory approval processes and enhancing post-market surveillance of therapies. Furthermore, advancements in the integration of artificial intelligence and the application of pharmacogenomics results in more effective, efficient, and personalized healthcare delivery. Nonetheless, these developments are met with critical challenges, and the lack of standardized global regulatory frameworks creates disparities in how this data is utilized across jurisdictions. Addressing these limitations through robust validation frameworks, harmonized regulatory policies, and interdisciplinary collaboration is essential to realizing the complete potential of DHT and RWE in precision medicine applications. This review examines the opportunities, challenges, and future directions of merging DHT and RWE to enhance precision medicine, emphasizing the importance of collaboration among researchers, regulators, and healthcare providers.
Introduction
On average, it takes about 12–15 years for a new therapeutic product such as a drug or biologic to progress from development to commercialization. 1 This lifecycle usually involves identification of a therapeutic target, followed by preclinical studies, and then progresses through clinical phases of testing aimed at evaluating safety, efficacy, toxicity, and dosage of the potential drug in human participants. 2 The data from preclinical and clinical studies are then submitted to regulatory agencies for review and marketing approval. Clinical trial operations are strictly governed by legal and regulatory requirements, based on Good Clinical Practices 3 and require close interdisciplinary collaboration to ensure the treatments remain safe and effective even after market approval. 4
The healthcare industry has embraced economic globalization as a core aspect of its business model, particularly in clinical research where global clinical trials have become the norm. As of April 2025, ClinicalTrials.gov lists 29% registered studies with locations in the USA and 56% of study locations outside of the USA. 5 indicating a more favorable landscape for multinational trial sites. While conducting trials internationally offers significant advantages, it also presents challenges related to diverse regulatory frameworks, cultural and ethical differences among participant populations, and varying healthcare systems. 6 For regulatory affairs professionals and pharmaceutical companies, engaging with regulatory authorities worldwide means navigating distinct requirements from each region. This calls for a deep understanding of international regulations along with the ability to harmonize those requirements without compromising local compliance. At the same time, technological advancements add another layer of complexity, as the pharmaceutical industry increasingly integrates cutting-edge technologies into drug development. 7 In today's rapidly changing world, traditional problem-solving methods often struggle to address the complex challenges brought on by technological advances, global market changes, and evolving regulatory demands. This has created an urgent need for innovative, data-driven tools to manage the growing complexity of global clinical trials and to support timely and compliant drug development and approval. 8
To meet these challenges, organizations are turning to advanced tools, stronger data capabilities, and better platform integration. These efforts aim to give regulatory teams the insights they need to stay compliant and competitive. From leveraging Digital Health Technologies (DHT) for data collection, 9 employing artificial intelligence (AI) and machine learning (ML) to integrate and analyze datasets, 10 to utilizing real-world evidence (RWE) to inform everyday patient treatment decisions, 11 these emerging trends are reshaping clinical trial design and drug development worldwide. Moreover, evolving patient needs, advances in data analytics and sequencing technologies, alongside robust research funding and large-scale genomic initiatives, have elevated the prominence of precision medicine in recent years. 12 Among these technological innovations, DHT and RWE have emerged as particularly transformative forces in addressing the challenges of globalized clinical research while advancing personalized medicine.
In this article, we will focus our discussion on DHT and RWE as essential tools for achieving Precision Medicine, examining how these technologies are being integrated to drive therapeutic innovations.
Digital health technology
Digital Health Technology is defined as the use of wearable devices, mobile applications, and/or software by patients to monitor their health in real time. These tools can also be used by the physicians to monitor patient's health and make targeted decisions regarding diagnosis, treatment, or prevention of diseases outside of conventional clinic settings. 13
DHT has significantly enhanced clinical trials by facilitating the collection of real-world data (RWD) beyond the conventional clinical setting and adopting more patient-centered methods. Utilizing DHTs to collect data from trial participants remotely might enable more regular or even ongoing data gathering compared to scheduled trial visits that necessitate participants traveling to a facility.9,14 DHT could also offer chances to gather data from individuals engaging in daily activities (e.g., walking, sleeping, routine tasks) regardless of their location (e.g., home, school, workplace, outdoors). Additionally, they might aid in gathering information from participants who cannot communicate their experiences (e.g., infants, individuals with cognitive impairments). All of these contribute to easing the strain on patients, thus enhancing patient recruitment and retention rates.
The following are some examples of DHT tools used to capture patient data:
Real-world evidence
RWE refers to clinical evidence regarding the usage and potential benefits or risks of medical products derived from the analysis of RWD. 11 RWD encompasses various types of data related to patient health status and healthcare delivery, including EMR, claims and billing activities, product and disease registries, patient-generated data from home-use settings and data gathered from mobile devices and social media. 20 Unlike data from randomized controlled trials (RCTs), which are conducted in controlled environments and often involve homogeneous patient populations, RWE reflects the actual use and performance of products in diverse real-world settings. This broader scope provides insights into how treatments work in routine clinical practice, capturing a wider range of patient experiences and outcomes. 11
RWE plays a crucial role in supplementing data obtained from RCTs, offering insights into the long-term effectiveness and safety of treatments, and informing clinical guidelines and policy decisions. 21
A few key examples of RWD sources are as follows:
RWD and DHT are reshaping regulatory approval processes by offering insights into treatment efficacy and safety through diverse data sources. Data collected using these tools also enables optimization of healthcare including product value assessment, pricing and reimbursement, and patient access infrastructure. This data is widely used to inform policy changes and health technology assessment (Figure 1). These sources enhance the efficiency of healthcare delivery to make medicine more personalized and precise.

Integration of digital health technologies and real-world data in healthcare optimization data collected using digital health technologies (green circles) and real-world evidence tools (blue circles) help inform regulatory decisions including product value assessment, pricing and reimbursement, and changes to health policies. This data is also used in many aspects of the drug development and regulatory authorization.
From data to decision: how DHT and RWE enable precision medicine
Precision medicine is an advanced approach to healthcare that tailors treatments to individual patients based on their unique genetic, environmental, and lifestyle factors. It aims to identify specific biomarkers and uses them to predict the most effective treatments for particular patient subgroups. 28 Unlike traditional “one-size-fits-all” approach, precision medicine seeks to optimize therapy based on individual differences, resulting in more accurate treatment outcomes. This treatment considers genomics, biomarkers, electronic health records (EHRs), demographics, and patient's medical history and involves the use of new technologies such as AI, ML, Big Data, etc. to identify the precise medicine and exact dose for the patient. 12
DHTs can be used to deliver precision medicine to patients by getting real-time data from devices, self-reporting, or web-based platforms. 14 RWE further supports precision medicine by offering insights from clinical practice and patient-reported data, refining treatment decisions based on diverse populations (Figure 2). 29
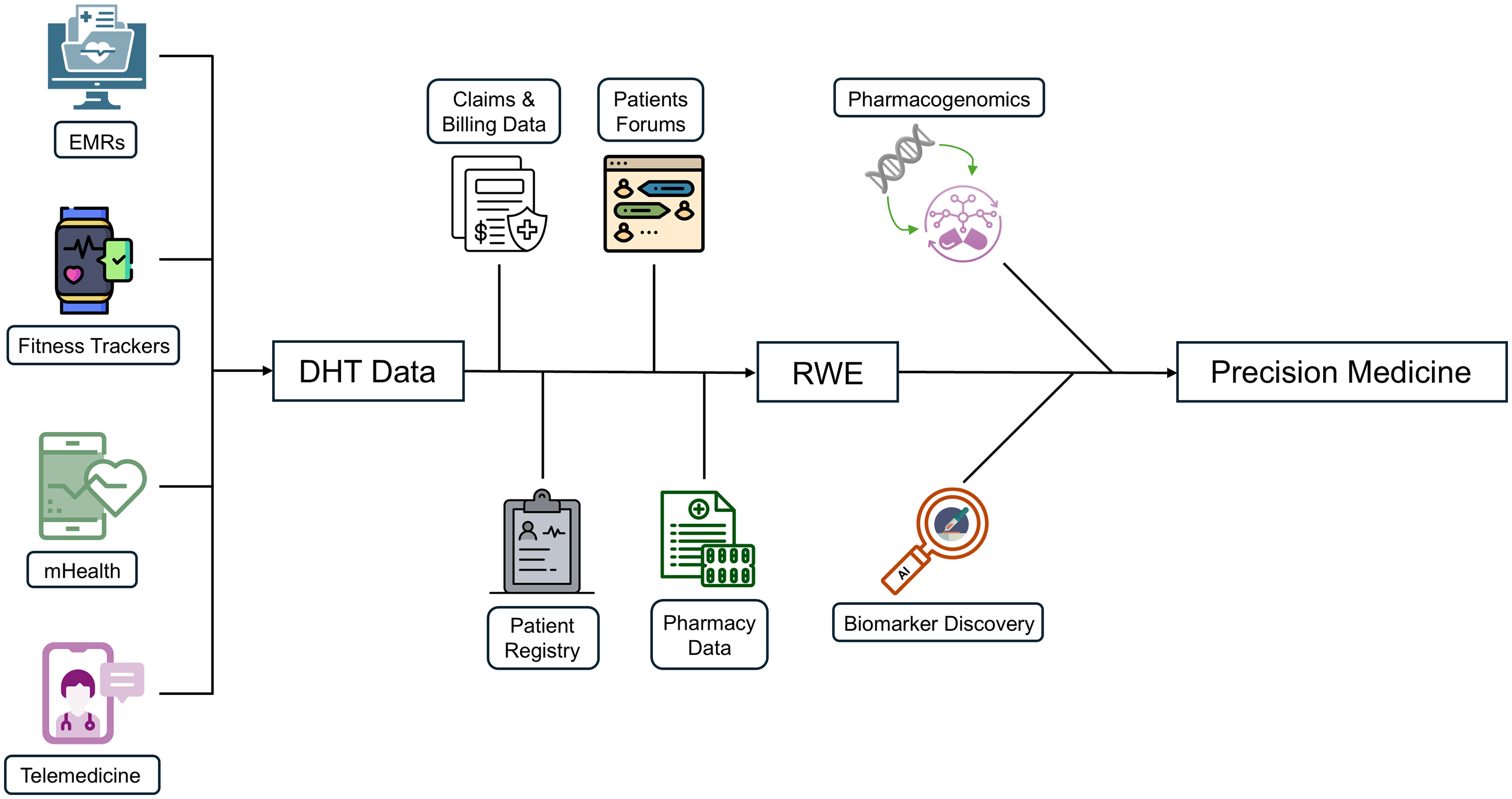
Data pathway toward precision medicine the figure illustrates the progression of data flow from the initial to the final stage. Data collection begins with the acquisition of patient health information from various electronic sources, termed Digital Health Technology data. This is enhanced by Real World Evidence data, including patient forums, insurance claims, patient registries, and pharmacy records. The integration of this dataset with pharmacogenomics, aided by artificial intelligence in biomarker discovery, enables the implementation of Precision Medicine.
As precision medicine continues to evolve, two key trends are playing a pivotal role in advancing its effectiveness: the integration of AI for biomarker discovery and the application of pharmacogenomics to tailor drug therapies.
Leveraging DHT and RWD in clinical trials and regulatory approvals
The increasing use of DHT and RWD has revolutionized clinical research and regulatory processes, with healthcare systems integrating diverse data sources to monitor patient outcomes. RWD identifies patient subgroups that benefit most and aids in trial modifications to reflect real-world settings whereas DHT applications enhance data collection, thus improving trial relevance and outcomes. 33 Furthermore, post-market monitoring of therapies using RWD and DHT ensures their long-term safety and effectiveness, which is critical in regulatory decision-making. 34
There are several instances where use of DHT has been successfully implemented to either detect or diagnose medical conditions. In 2017, the U.S. Food and Drug Administration (FDA) approved the first “digital pill,” called Abilify Mycite. This innovative pill contains a tiny tracking system that helps track adherence to aripiprazole treatment, an atypical antipsychotic commonly prescribed for schizophrenia. 35 The Norwegian ECG247 Smart Heart Sensor, a user-friendly wireless continuous ECG monitoring device that includes a web portal, a smartphone app, a back-end cloud service, a reusable sensor, and an electrode patch is another example of how DHT is used. According to the EU Medical Device Directives (93/42/EEC), the system is certified as a medical diagnostic device and complies with the General Data Protection Regulation's (GDPR) standards. 36 An example of the use of DHT in clinical study is the trial being conducted in the Netherlands. Radboud University Medical Center is conducting a study to create a comprehensive Parkinson's disease dataset using continuous wearable sensor data from 520 patients. In this trial, participants will wear the Verily Study Watch to collect high-resolution movement and other data, which will be correlated with clinical outcomes to assess disease progression and treatment responses. 37
RWD has been successfully applied in regulatory approvals, as seen with Entresto (sacubitril/valsartan). Initially approved by the FDA through RCTs, RWD analysis from EHRs and patient registries demonstrated its effectiveness across broader populations, leading to expanded use. 38 Similarly, Ibrance (palbociclib) was supported by RWD during its approval process by the European Medicines Agency, 22 confirming the drug's efficacy across diverse patient subgroups. 39 In clinical trial design, precision medicine has increasingly relied on RWD to improve trial outcomes. For example, the integration of RWD in oncology trials has allowed more accurate targeting of therapies based on real-world patient data, enhancing trial designs and results. 40
Precision Medicine is also being investigated for its utility and viability in treating specific conditions through clinical trials. One example is the Precision Medicine Study, sponsored by the Icahn School of Medicine at Mount Sinai, which will be a 2-year observational trial assessing cancer sequencing in multiple myeloma patients to improve tumor characterization and identify novel therapeutics. The study will involve advanced genomic analyses, reporting findings to participants and their physicians with a focus on the total number of somatic single-nucleotide variants per patient. 41 Another study, the NSIGHT2 trial, will investigate whether rapid genomic sequencing improves outcomes for critically ill infants by comparing whole-genome sequencing and whole-exome sequencing. This study aims to quantify the diagnostic benefits, clinical efficacy, and cost-effectiveness of these methods in managing acutely ill infants 42 (Table 1).
Shows selected ClinicalTrials.gov studies highlighting the use of digital health technology (DHT) and real-world evidence (RWE) in personalized medicine (PM) approach.

Sample sizes presented as rounded values (nearest 10 or 1000) for clarity.
Regulatory frameworks guiding DHT and RWE
Challenges in regulating these trends
While precision medicine holds enormous promise, regulating its application poses significant challenges due to the complexity of data and treatment personalization.
To improve the applications of precision medicine regulatory agencies are evaluating frameworks for algorithmic transparency, emphasizing the need for explainability in AI-based RWE assessments.
56
The increasing use of AI to analyze RWD introduces concerns about algorithmic bias—systematic errors in AI models resulting from imbalanced or incomplete training data.
57
Bias in AI-driven decision-making can lead to disparities in healthcare outcomes,
58
particularly for underrepresented populations, underscoring the urgent need for transparent algorithm development and equity-focused oversight mechanisms.
59
Ensuring diversity in training datasets and adopting bias-mitigation techniques are critical for the ethical integration of AI in regulatory science.
60
Conclusion
The integration of DHT and RWD into precision medicine is transforming clinical research by providing personalized healthcare solutions. Despite existing challenges, precision medicine continues to evolve, and regulatory bodies are gradually adapting to ensure that these innovations are accessible to patients while maintaining safety and efficacy standards. This will require collaboration among regulators, industry, healthcare providers, and patients to create clear guidelines and sustainable reimbursement models. Additionally, investing in regulatory education and pursuing global harmonization will support timely approvals and wider patient access to precision medicine.
Footnotes
Author contributions
SM and PS conducted investigation and wrote the manuscript. SM and PS are equal contributors. EJS conceptualized the content and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
