Abstract
Purpose
This study explores global frameworks for healthcare data privacy, focusing on the General Data Protection Regulation (GDPR), the California Consumer Privacy Act (CCPA), and the Protection of Personal Information Act (POPIA). It examines the challenges of regional regulatory disparities, systemic vulnerabilities identified through major health data breach case studies, and the potential of advanced technologies to enhance privacy protections.
Methods
A qualitative research approach was adopted, incorporating corpus construction and comparative analysis of legal and technical frameworks. The study also utilized case studies of significant health data breaches to identify vulnerabilities and evaluate the role of emerging technologies, such as artificial intelligence (AI) and machine learning (ML), in mitigating risks and enhancing regulatory compliance.
Results
Findings indicate that GDPR, CCPA, and POPIA set high standards for data protection but reveal significant variability in enforcement and technological adoption across regions. Challenges include inconsistent definitions of sensitive data, semantic discrepancies, a lack of standardized protocols, and limited information technology infrastructure in certain jurisdictions. Advanced technologies like AI and ML promise to address these gaps by improving data harmonization and security.
Conclusions
Addressing healthcare data privacy challenges requires harmonized global regulations, advanced technological tools, and international collaboration. Strengthening frameworks, enhancing information technology infrastructure, and employing semantic models and ontologies are essential for protecting sensitive data, ensuring compliance, and fostering public trust in digital healthcare systems.
Keywords
Introduction
Data privacy in healthcare entails protecting sensitive patient information, including medical records, personal identifiers, and other health-related data, from unauthorized access, misuse, or disclosure.1,2 To clarify, “personal data” refers to any information that can identify an individual (such as names, addresses, and contact details), whereas “medical data” specifically pertains to information generated during healthcare delivery, such as diagnoses, treatment records, and clinical outcomes. Although these categories often intersect, medical data is intimately linked to patient care and is subject to additional ethical and professional safeguards imposed by the deontological codes of healthcare providers. 3 Accordingly, the term “personal health information (PHI)” is used in this review to denote data directly related to an individual's health status and care, warranting enhanced protection under both legal regulations and ethical standards.
The World Health Organization (WHO) defines healthcare data privacy as the implementation of measures that guarantee the confidentiality, integrity, and availability of patient information. 4 Similarly, the United Nations Educational, Scientific, and Cultural Organization (UNESCO) describes personal data as any information that directly or indirectly identifies an individual, emphasizing the secure collection, usage, storage, and transfer of such data. 5 In making this distinction, our review aims to underscore that while both personal and medical data require robust safeguards, medical data, by virtue of its connection to patient care, necessitates even stricter protection.
In North America, the US Department of Health and Human Services governs healthcare data privacy through the Health Insurance Portability and Accountability Act (HIPAA), which mandates measures such as access restrictions, encryption protocols, and breach notification requirements for electronically protected health information (ePHI).6,7 Similarly, Europe's General Data Protection Regulation (GDPR) emphasizes explicit consent, data minimization, and comprehensive technical and organizational safeguards, setting a global benchmark for data protection.8–10 In the Asia-Pacific region, frameworks like the Asia-Pacific Economic Cooperation (APEC) Privacy Framework advocate for interoperable and culturally tailored privacy protection mechanisms.11–13 Meanwhile, in sub-Saharan Africa, emerging policies endorsed by the Africa Centers for Disease Control and Prevention (Africa CDC) stress respecting cultural values, securing patient consent, and ensuring accountability in health data management.14–16 This review examines these diverse regulatory approaches and evaluates how they interact with information systems such as electronic health records (EHRs) and cybersecurity infrastructures to both protect and sometimes compromise PHI.
Despite the presence of robust regulatory frameworks, the increasing integration of EHRs and digital tools has significantly amplified the risk of data breaches and unauthorized access. Notable examples include the Anthem Inc. breach in the USA, which exposed the ePHI of 79 million individuals, and the WannaCry ransomware attack on the UK's National Health Service (NHS), which disrupted critical healthcare services.17–21 Similarly, the SingHealth breach in Singapore compromised the personal data of 1.5 million patients,22,23 while incidents in sub-Saharan Africa such as the compromise of the Ghana Health Service's COVID-19 test results portal and the South African medical data breach of 2020 highlight challenges related to inadequate encryption and resource constraints.24,25 These cases underscore that even with established regulations, systemic vulnerabilities and technological shortcomings can undermine data privacy.
Emerging technologies, particularly blockchain and artificial intelligence (AI), offer promising avenues for addressing these vulnerabilities. Blockchain technology, which operates as a decentralized and immutable digital ledger, can enhance data integrity and transparency by securely recording transactions and preventing unauthorized alterations. Similarly, AI and machine learning (ML) technologies enable real-time breach detection, predictive risk assessment, and automated compliance monitoring. Recent studies, such as those highlighted in Reflections on Blockchain in Health Data Sharing (10.3390/ijerph21020230), demonstrate the transformative potential of these innovations in strengthening healthcare data privacy practices. Balancing technological innovation with stringent privacy protections is, therefore, critical for safeguarding sensitive patient information in an increasingly digitalized healthcare landscape.
This review comprehensively analyzes global healthcare data privacy by examining legal, ethical, and technical dimensions across diverse regulatory frameworks. It emphasizes the need for harmonized global regulations that are adaptable to regional nuances and highlights innovative technological solutions that can bridge current security gaps. Through a detailed analysis of case studies and policies, the study aims to provide actionable insights into enhancing the protection of PHI while maintaining the integrity and trust essential to healthcare delivery.
The study aims to delineate the distinctions between personal and medical data, emphasizing that while both demand robust protection, medical data, being intrinsically tied to patient care, requires additional ethical and professional safeguards. It seeks to examine global regulatory frameworks by analyzing the legal landscapes governing healthcare data privacy in regions such as North America, Europe, Asia-Pacific, and sub-Saharan Africa, thereby evaluating the nuances of compliance and enforcement in diverse contexts. The research further endeavors to identify systemic vulnerabilities by investigating prominent data breaches, which illuminate the inherent weaknesses in information systems and cybersecurity infrastructures. Moreover, the study evaluates best practices and explores the potential of emerging technologies, including AI, ML, and blockchain, to mitigate these vulnerabilities and enhance data privacy. Finally, by anchoring its analysis in established theoretical frameworks, the study aspires to propose tailored solutions and policy recommendations that promote harmonized, yet locally adaptable, approaches to safeguarding sensitive patient information in an increasingly digitalized world.
Theoretical framework
The study draws on multiple theoretical perspectives to analyze the complex dynamics of healthcare data privacy, providing a structured foundation for understanding patient trust, regulatory compliance, and the management of privacy among healthcare stakeholders. The rationale for integrating these theories is to capture both the human and technological dimensions of privacy challenges. Specifically, these theories help explain how trust is built and maintained, how innovations such as blockchain and other technologies can be adopted, and how privacy boundaries are negotiated in digital environments, a critical aspect given the multifaceted nature of healthcare data.
Social exchange theory, developed by George Homans and Peter Blau, highlights the critical role of trust in social interactions. Within healthcare data privacy, this theory underscores that patients are more likely to share sensitive health information when they are assured that their data will be protected.26,27 In emphasizing trust as a currency in the patient–provider relationship, this theory supports our analysis of how ethical safeguards and transparent practices contribute to secure data sharing.
Everett Rogers's innovation diffusion theory provides insights into how new technologies and practices are adopted within social systems. Although first introduced in the early 1960s, its relevance persists today as it illuminates the processes by which privacy-enhancing technologies such as encryption, blockchain, and AI are embraced by healthcare organizations. This theory helps us understand barriers to innovation and suggests strategies for overcoming resistance, thereby facilitating the integration of advanced data protection measures. 28
Travis Hirschi's social control theory emphasizes the importance of institutional and social mechanisms in deterring deviant behavior. In healthcare, this translates into the necessity for robust governance structures and organizational policies that ensure compliance with privacy regulations and prevent unauthorized access to sensitive patient data. 29 This theory underscores the need for regulatory oversight and internal controls in maintaining data integrity.
The ethical decision-making theory, influenced by scholars such as Lawrence Kohlberg, serves as a guide for navigating moral dilemmas in healthcare data privacy. This framework stresses the importance of balancing patient privacy rights with the imperatives of data sharing for research and treatment. It grounds our discussion in ethical principles of autonomy, beneficence, and justice, thereby guiding stakeholders in making informed decisions that respect patient rights while advancing healthcare objectives.30,31
Helen Nissenbaum's contextual integrity theory enriches the analysis by emphasizing the importance of maintaining privacy norms and expectations that are specific to social and professional contexts. This theory is particularly useful in explaining how healthcare settings, with their unique norms and relational dynamics, must adapt their data-handling practices to preserve confidentiality and trust. 32
Finally, Elena Karahanna and Detmar Straub's information boundary theory explores how individuals establish and manage boundaries around their personal information in digital environments. This theory elucidates patient preferences regarding the control of access to their medical records and underscores the challenges of maintaining privacy in an era of pervasive digital connectivity. 33
Together, these theories not only provide a comprehensive framework for analyzing the multifaceted challenges of healthcare data privacy but also directly inform our study's focus on the integration of emerging technologies and the management of privacy among healthcare stakeholders. In linking trust-building, innovation adoption, ethical decision-making, and privacy management, the theoretical framework underpins our recommendations for harmonized and adaptable data protection strategies.
Methodology
This study is a review that examines the multifaceted challenges and solutions associated with data privacy in healthcare. It employs a qualitative approach, integrating corpus construction and thematic analysis to explore global healthcare data privacy frameworks, identify vulnerabilities, and propose actionable recommendations.
Research design: corpus construction
The research adopts a corpus construction approach as a substitute for traditional sampling methods. Corpus construction involves selecting a set of relevant documents to form a representative body of knowledge on the subject matter. This approach is functionally equivalent to sampling but differs in structure, allowing the research to delve into various secondary data sources. The study constructed a robust corpus comprising regulatory documents, case studies, and scholarly articles. The corpus included key legal frameworks such as the GDPR in Europe, the HIPAA in North America, the APEC Privacy Framework, and sub-Saharan Africa's emerging data protection policies. These documents were sourced from reputable databases (e.g. PubMed, Scopus, Web of Science, Google Scholar, Wiley, and official organizational websites), official publications, and reports by regulatory authorities.6,8,11,14 Case studies of significant health data breaches, such as the Anthem Inc. breach in the USA, 17 the WannaCry ransomware attack in the UK, 21 and the SingHealth breach in Singapore, 22 provided additional contextual insights.
As noted by Bauer and Aarts (2000, see Bauer and Aarts, Chapter 2 in this volume), “sample size does not matter in corpus construction as long as there is some evidence of saturation. Corpus construction is an iterative process where researchers continuously refine and expand their collection until no new insights emerge, thereby ensuring that the reliability and validity of the qualitative analysis are maintained.” Furthermore, “in this sense, corpus construction and the representative sampling of textual and multimedia data enable researchers to capture the complexity of social phenomena, thereby providing a robust foundation for qualitative analysis. In short, we contend that corpus construction typifies an iterative and reflexive process in which disparate data sources are systematically identified, selected, and organized into a coherent body of evidence. Secondly, we demonstrate corpus construction in the field by carefully curating documents from a range of reputable sources, ensuring that the resulting corpus accurately reflects the multifaceted dimensions of the research topic” (Bauer and Aarts, 2000).
To enhance clarity, a flowchart summarizing the methodological steps is provided in Figure 1.

Coding process for deriving theme. This diagram illustrates the coding process: starting with the compilation of a comprehensive table of studies, the three authors independently coded the data to identify recurring patterns. Discrepancies were resolved through consensus-based discussions, leading to the grouping of codes into thematic categories and ultimately deriving four main themes (2025).
Data sources
To ensure comprehensive coverage of the topic, we used databases such as PubMed, WHO databases, Africa CDC repositories, and government health ministries’ websites. Additional reports and articles were sourced from PubMed, Scopus, Web of Science, Google Scholar, Wiley, and official organizational websites using the search terms: “healthcare data privacy,” “regulatory frameworks,” “data breaches in healthcare,” “HIPAA,” “GDPR,” “APEC Privacy Framework,” and “sub-Saharan Africa data protection.”
Keywords were combined using Boolean operators (AND/OR) to ensure comprehensive retrieval of documents relevant to healthcare data privacy.
Inclusion and exclusion criteria
The inclusion and exclusion criteria were applied systematically during corpus construction to ensure that only documents meeting the study's relevance and quality thresholds were selected. To ensure rigor and relevance, this study employed clearly defined inclusion and exclusion criteria for selecting documents to be included in the analytical corpus. The inclusion criteria were as follows:
Documents published between 2010 and 2024 to reflect both historical context and the most recent developments in healthcare data privacy. Peer-reviewed journal articles, policy briefs, legal frameworks, regulatory reports, and official communications from authoritative bodies such as the WHO, Africa CDC, European Data Protection Board (EDPB), and national data protection agencies. Case studies or reports involving notable healthcare data breaches or the implementation of major data protection regulations (e.g. GDPR, HIPAA, Protection of Personal Information Act (POPIA), APEC Privacy Framework). Materials addressing technical, regulatory, or ethical dimensions of data privacy within healthcare systems, including those focused on low-resource and high-resource contexts.
Documents were excluded if they:
Were published before 2010, as these may not accurately represent current technological capabilities or regulatory environments. Provided only editorial opinions, news articles, or non-empirical content without substantive data or legal grounding. Focused exclusively on non-healthcare sectors or covered highly localized or outdated regulatory instruments with limited applicability to the broader study objectives.
These criteria were developed through iterative discussion among the research team, with consensus reached on ambiguous cases. The selected timeframe (2010–2024) was justified based on the proliferation of global data protection policies, the emergence of AI and digital health innovations, and the increasing frequency of high-impact data breaches during this period.
Data collection
Data collection followed a multipronged approach, emphasizing the triangulation of secondary sources. Legal texts, policy documents, and publicly available reports from organizations such as the EDPB, 9 the Office for Civil Rights, 7 and the Africa CDC 14 formed the backbone of the analysis. Peer-reviewed journal articles and industry reports supplemented these sources to provide a comprehensive perspective. This systematic approach ensures that the data collected is both comprehensive and directly relevant to the study's objectives.
Thematic analysis
A thematic analysis was conducted to systematically identify and extract recurring patterns from the corpus of selected documents. Initially, the research team compiled a comprehensive table summarizing each study by author(s), year, title, type of paper, and key findings. This table provided a structured foundation for our iterative coding process. During coding, emerging patterns were identified and grouped into distinct categories.
Through this process, four main themes emerged: (a) regional vulnerability in data privacy challenges; (b) technological vulnerabilities and systematic weaknesses; (c) best practices and proactive responses; and (d) innovative solutions and the role of advanced technology. These themes were derived inductively from the data and are directly aligned with the regulatory, technical, and strategic dimensions of healthcare data privacy identified in our review.
The coding was performed manually by three researchers, with any discrepancies resolved through consensus-based discussions. This iterative and collaborative approach ensured the transparency and rigor of the analysis, as well as a clear linkage between the emergent themes and the underlying data.
Addressing bias
The reliance on secondary data introduced the potential for selection bias. To mitigate this, the study incorporated diverse data sources, including regulatory texts, case studies, and academic research, ensuring a balanced representation of regional and global perspectives. Peer-reviewed articles were prioritized to ensure credibility, and the corpus construction and analysis were conducted independently by multiple researchers, with disagreements resolved through iterative consensus discussions. This approach enhanced the objectivity and reproducibility of the findings.
Comparative analysis
The study conducted a comparative analysis of healthcare data privacy regulations across North America, Europe, Asia, and sub-Saharan Africa. For example, North America's HIPAA Privacy Rule emphasizes legal obligations for protecting ePHI, while Europe's GDPR mandates comprehensive consent and accountability measures.6,8 In contrast, the APEC Privacy Framework focuses on interoperability and voluntary cooperation, reflecting its member economies’ diverse legal and cultural contexts. 11 Sub-Saharan Africa's policies, such as those endorsed by the Africa CDC, emphasize capacity building and incorporating cultural norms into privacy frameworks. 14 This comparative approach allows for a nuanced understanding of how different regulatory environments impact data privacy practices.
Case studies
Prominent case studies were analyzed to ground the findings in real-world scenarios. Examples include the Anthem Inc. breach, which exposed systemic weaknesses in cybersecurity and risk management in North America, and the WannaCry ransomware attack, which revealed vulnerabilities in outdated information technology (IT) systems in Europe. In the Asia-Pacific region, the SingHealth breach demonstrated the need for robust EHR systems. In contrast, sub-Saharan African examples, such as the Ghana Health Service's COVID-19 test results breach, underscored challenges related to encryption and governance.17,21,22,24 These case studies were selected based on their ability to illustrate systemic vulnerabilities and inform the thematic analysis.
Ethical considerations
This study strictly adhered to ethical research practices appropriate for qualitative research involving document analysis and corpus construction. The selection and analysis of documents were conducted with a commitment to transparency, integrity, and accountability. All sources were obtained from reputable databases, official publications, and credible peer-reviewed journals, ensuring the validity and reliability of the findings. The collaborative approach among the authors further minimized potential biases.
Results
Through iterative coding and thematic analysis of the corpus, four primary themes emerged from the review of global healthcare data privacy frameworks:
Regional variability in data privacy challenges
The analysis revealed significant differences across regions:
North America: Although the HIPAA Privacy Rule provides robust protections, enforcement challenges persist, as evidenced by breaches such as the Anthem Inc. and Premera Blue Cross incidents. Europe: The GDPR establishes strict data protection standards; however, events like the WannaCry ransomware attack on the UK's NHS expose vulnerabilities stemming from outdated IT systems and suboptimal patch management. Asia-Pacific: Integration of cross-border data privacy measures remains problematic. The SingHealth breach in Singapore, for example, underscores the gaps in data governance and the need for enhanced employee training. Sub-Saharan Africa: Resource limitations and inconsistent policy enforcement exacerbate vulnerabilities, with incidents such as the breaches in Ghana and South Africa highlighting region-specific challenges.
Technological vulnerabilities and systemic weaknesses
Across all regions, there is a consistent pattern of systemic shortcomings, including outdated IT infrastructures, insufficient encryption practices, and gaps in cybersecurity protocols. These technological vulnerabilities contribute significantly to the risk of data breaches and impede effective data protection.
Best practices and proactive responses
Despite these challenges, several proactive strategies have been adopted:
North America: In response to high-profile breaches, organizations have implemented comprehensive risk assessments and upgraded to advanced encryption protocols. Europe: The GDPR has catalyzed the development of disaster recovery plans and the institution of robust employee training programs, thereby strengthening data protection practices. Asia-Pacific: Regular cybersecurity audits, along with the implementation of stricter access controls, have been effective in mitigating risks, as seen in Singapore's response to the SingHealth breach. Sub-Saharan Africa: Initiatives by bodies such as the Africa CDC and the introduction of regulatory frameworks like POPIA have facilitated capacity-building and improved cybersecurity practices in the region.
Innovative solutions and the role of advanced technologies
The study also highlights the promising potential of emerging technologies to address existing gaps.
AI and ML: these technologies involve algorithms and computational models that can learn from data, detect patterns, and make predictive decisions. In the context of healthcare data privacy, AI and ML can be leveraged to enhance breach detection systems, optimize data encryption algorithms, and automate compliance monitoring. Semantic ontologies: this refers to structured frameworks that define and relate concepts within a domain. In employing semantic ontologies, healthcare systems can improve data interoperability and understanding by establishing standardized relationships between data elements. This approach facilitates better data harmonization and supports more effective regulatory compliance and patient trust (Figure 2).

Thematic framework for global healthcare data privacy. This diagram illustrates the four main thematic areas derived from our review: (1) regional variability in data privacy challenges, (2) technological vulnerabilities and systemic weaknesses, (3) best practices and proactive responses, and (4) innovative solutions and advanced technologies.
Discussion
The present study synthesizes a broad spectrum of literature on global healthcare data privacy, unveiling four interconnected thematic areas that not only reflect distinct challenges and opportunities but also provide avenues for policy evolution, technological innovation, and future research. In synthesizing over 90 studies (see Tables 1–5 and Tables A1–A5 in the Supplementary material), our analysis reveals significant regional disparities, systemic technological vulnerabilities, proactive regulatory responses, and the emerging role of advanced technologies in shaping this field. This comprehensive overview lays the groundwork for an in-depth discussion of how these themes inform current practices and future directions in healthcare data privacy.
Thematic framework for healthcare data privacy analysis.
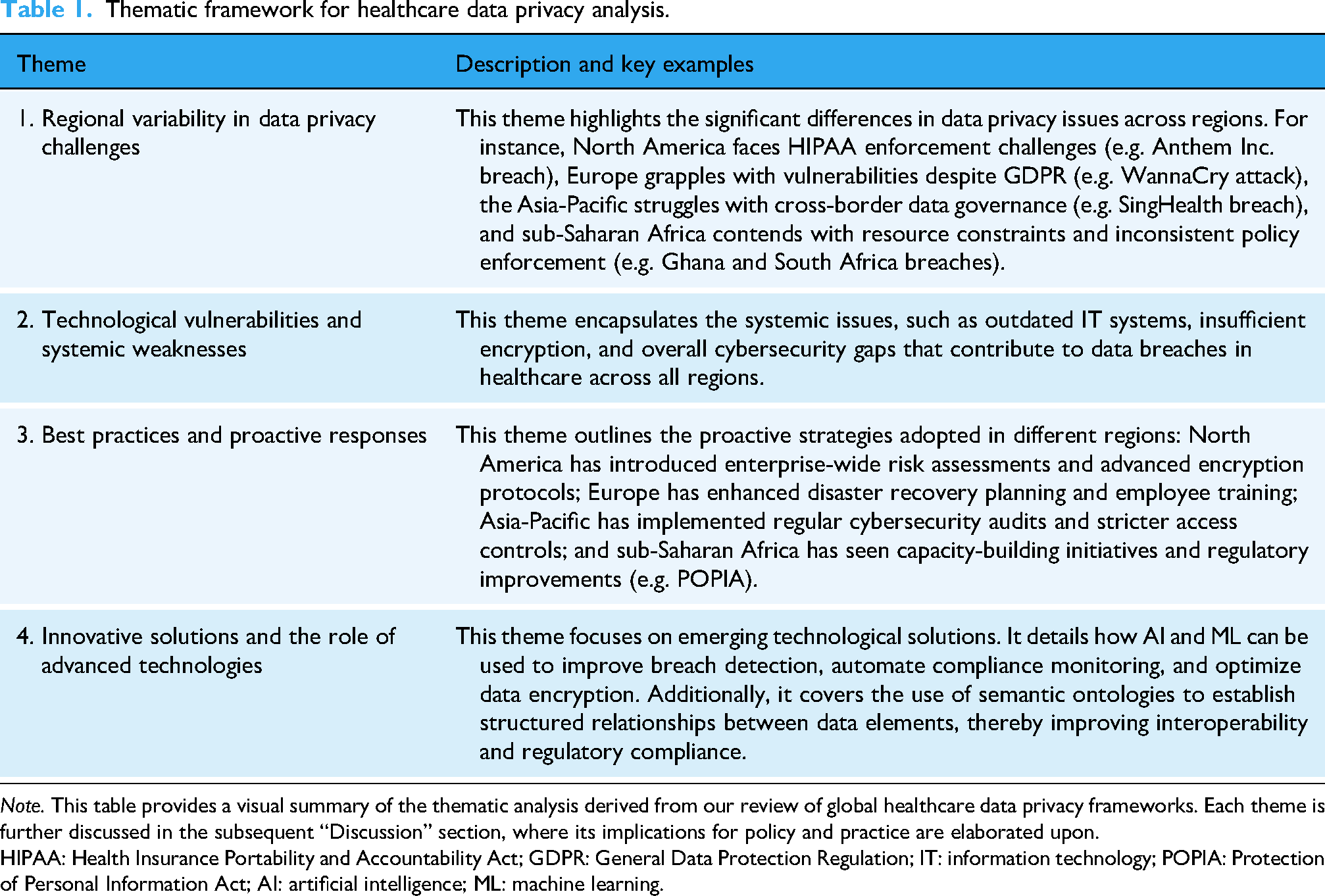
Note. This table provides a visual summary of the thematic analysis derived from our review of global healthcare data privacy frameworks. Each theme is further discussed in the subsequent “Discussion” section, where its implications for policy and practice are elaborated upon.
HIPAA: Health Insurance Portability and Accountability Act; GDPR: General Data Protection Regulation; IT: information technology; POPIA: Protection of Personal Information Act; AI: artificial intelligence; ML: machine learning.
Regional variability in data privacy challenges.

Note. This theme captures how different regions face distinct challenges in healthcare data privacy, reflecting variations in regulatory frameworks, enforcement practices, and contextual factors. This table illustrates the distinct challenges faced by different regions in healthcare data privacy. It highlights variations in regulatory frameworks, enforcement practices, and contextual factors that influence the protection of sensitive patient information. EHR: electronic health record; HIPAA: Health Insurance Portability and Accountability Act; GDPR: General Data Protection Regulation; NHS: National Health Service.
Technological vulnerabilities and systemic weaknesses.

Note. This theme focuses on technical gaps, outdated systems, and cybersecurity flaws that underlie many data breaches across healthcare systems.
This table focuses on the technical gaps, outdated systems, and cybersecurity flaws that underlie many data breaches in healthcare systems. It summarizes the key findings related to the inherent vulnerabilities that compromise data security. IT: information technology; EHR: electronic health record.
Best practices and proactive responses.

Note. This theme outlines the proactive measures, regulatory frameworks, and policy-driven responses aimed at improving healthcare data privacy. This table outlines the proactive measures, regulatory frameworks, and policy-driven responses implemented to improve healthcare data privacy. It details best practices and governance strategies that mitigate privacy risks and enhance data protection. GDPR: General Data Protection Regulation.
Innovative solutions and the role of advanced technologies.

Note. This theme focuses on emerging technological innovations, such as blockchain, AI, ML, and semantic ontologies, that promise to enhance data privacy through improved integration, automation, and interoperability. This table synthesizes emerging technological innovations, such as blockchain, artificial intelligence (AI), machine learning (ML), and semantic ontologies, which promise to enhance data privacy through improved integration, automation, and interoperability. It provides a comprehensive view of forward-thinking strategies to address current challenges in healthcare data privacy. IT: information technology; XML: extensible markup language.
Regional variability in data privacy challenges
The literature indicates that healthcare data privacy challenges differ significantly by region due to variations in regulatory frameworks, enforcement practices, and local contexts. In North America, for instance, the enforcement challenges of HIPAA have led to high-profile breaches, as noted by Cohen 6 and exemplified in the Anthem incident. 39 In contrast, European healthcare systems benefit from the stringent requirements of the GDPR9,10,40 but are not immune to systemic vulnerabilities, as evidenced by the WannaCry attack. 21 Similarly, studies from Asia-Pacific and sub-Saharan Africa11–16 reveal that resource constraints and diverse legal interpretations further complicate the implementation of robust data protection measures. This regional heterogeneity suggests that while international frameworks provide a useful baseline, tailored approaches are necessary to address local challenges.14,36 Such variability reinforces the need for policies that are both globally informed and locally adaptable.
Technological vulnerabilities and systemic weaknesses
A substantial portion of the literature underscores the technical deficiencies inherent in many healthcare systems. Outdated IT infrastructures, insufficient encryption methods, and gaps in cybersecurity measures contribute significantly to data breaches. For example, Mertens et al. 8 highlight how hidden data leaks can occur even in advanced systems, while Foreman et al. 42 and Goodday et al. 43 emphasize vulnerabilities in data capture and management. Furthermore, systematic reviews and case studies17,37,44 consistently reveal that these technical shortcomings are compounded by systemic weaknesses, such as poor integration of new technologies and inadequate training of staff. The theoretical framework proposed by Nissenbaum 32 helps explain these contextual flaws by emphasizing the role of “contextual integrity” in maintaining privacy standards. Thus, addressing technological vulnerabilities requires a two-pronged strategy: immediate technical upgrades and long-term systemic reforms.
Best practices and proactive responses
In response to the multifaceted challenges of healthcare data privacy, a growing body of evidence highlights the significance of best practices and proactive regulatory interventions. Frameworks such as the GDPR, 49 the California Consumer Privacy Act, and South Africa's POPIA 51 have established rigorous benchmarks for data governance. These standards not only delineate legal obligations but also promote institutional accountability, secure data handling, and enforcement mechanisms that are increasingly viewed as models for global adoption.
Empirical studies from South Africa15,16 illustrate how local implementation of such frameworks coupled with capacity-building and stakeholder engagement has led to measurable improvements in data privacy and cybersecurity infrastructure. Similarly, regulatory instruments in the USA, including the HIPAA 53 and the Gramm-Leach-Bliley Act, 52 have shaped privacy practices through stringent compliance requirements and post-breach corrective mechanisms.17,21 These interventions underscore the value of regulatory foresight combined with technical resilience, particularly when deployed through cross-sectoral collaboration.
Across regions, proactive responses to breaches have become instrumental in evolving healthcare privacy norms. In North America, high-profile incidents such as the Anthem Inc. data breach prompted organizations to adopt comprehensive enterprise-wide risk assessments, implement advanced encryption standards, and provide identity theft protection services.17,20 These efforts reflect a shift from reactive crisis management to preemptive risk mitigation and continuous system audits.
In Europe, the implementation of GDPR has catalyzed sector-wide changes, particularly following cyber incidents like the WannaCry ransomware attack. Institutions responded by strengthening disaster recovery protocols, investing in ongoing employee training, and deploying real-time vulnerability detection systems. 21 These practices have cultivated a culture of compliance and adaptability, where digital vigilance is integrated into operational workflows.
The Asia-Pacific region, guided by the APEC Privacy Framework, has emphasized interoperability and multilateral cooperation. In the aftermath of the SingHealth breach, Singapore undertook a comprehensive review of its EHR systems, adopting stricter access controls, enhanced audit trails, and regular cybersecurity audits.22,23 These reforms illustrate the potential of combining regional policy alignment with technological innovation to build resilient health data ecosystems.
In sub-Saharan Africa, where resource limitations and fragmented policy enforcement present enduring obstacles, regional actors have pursued strategic responses. The Africa CDC, for example, has championed governance reforms, professional training, and regulatory harmonization across member states. Legislative instruments such as POPIA, along with targeted interventions in Ghana and South Africa, have advanced encryption use, fostered data protection awareness, and improved breach response mechanisms.14,24,25 While challenges persist, these efforts demonstrate the efficacy of context-sensitive approaches that balance international standards with local capabilities.
Despite these advances, our review reveals persistent variability in implementation, technological readiness, and legal enforcement across jurisdictions. Discrepancies in the definition of sensitive health data, semantic inconsistencies in regulatory language, and infrastructural deficits continue to undermine standardization efforts. In response, there is a pressing need to develop interoperable frameworks and shared protocols for encryption, breach detection, and compliance auditing that are adaptable to both high-resource and low-resource settings.
Innovative technologies, including AI, ML, and semantic ontologies, offer transformative potential in addressing these gaps. AI and ML can automate compliance monitoring and enhance breach detection, while semantic ontologies support data harmonization by standardizing how information is interpreted across systems. Together, these tools contribute to scalable, context-responsive models of healthcare data protection that uphold both security and patient trust.
Drawing on insights from over 90 scholarly and policy documents, this analysis affirms that best practices in healthcare data privacy are most effective when they are proactive, technologically integrated, and regionally adaptable. Regulatory evolution, institutional investment in IT security, and cross-regional knowledge sharing will be critical to building secure and equitable digital health systems globally.
Innovative solutions and the role of advanced technologies
Emerging technologies offer promising avenues for overcoming current challenges in healthcare data privacy. The application of blockchain technology, as discussed by Nelson, 7 illustrates the potential for enhancing data integrity and transparency. Likewise, advances in AI and ML have been reviewed by Meng et al. 63 and Zhang et al., 64 showing that these technologies can facilitate real-time breach detection, predictive analytics, and automated compliance monitoring. Semantic ontologies introduced by Tao et al. 67 and further developed by Marwadi 69 and do Espírito Santo and Medeiros 70 provide a framework for achieving improved data interoperability and standardization across heterogeneous datasets. Such innovative solutions not only address existing technical vulnerabilities but also enable a more integrated and adaptive approach to data privacy. The theoretical insights from Karahanna and Straub 33 on managing information boundaries further support the integration of these advanced technologies into existing systems.
Implications for research, policy, and practice
The findings of this review have direct and far-reaching implications for research, policy, and practice. For policymakers, the study emphasizes that effective data privacy management requires a multilayered, adaptive, and region-sensitive framework. Regulatory instruments must be grounded in globally accepted standards, such as GDPR, HIPAA, and POPIA, yet must remain flexible enough to accommodate local resource limitations and contextual differences. This approach ensures both international interoperability and contextual relevance.
Healthcare organizations must invest not only in technical enhancements—including encryption, blockchain, and AI—but also in operational reforms that prioritize continuous risk assessment, staff training, and internal governance. These investments should be viewed not merely as compliance obligations but as strategic imperatives for ensuring patient trust and institutional resilience.
For researchers, the review identifies a critical gap in evidence from underrepresented regions, notably Latin America and the Middle East. Future studies must incorporate primary data collection, particularly in low-resource settings, and assess the longitudinal impact of regulatory interventions and advanced technologies. In addition, exploring how AI, ML, and semantic ontologies can be integrated into legacy systems remains a key research frontier.
Policy recommendations
Effective data privacy management requires a multifaceted approach integrating technical, operational, and legislative measures. The following recommendations reflect both the universal standards and context-specific adaptations necessary for implementation.
Enhancing technical and operational requirements
Healthcare organizations should invest in advanced technologies like encryption, blockchain, and AI to secure patient data. Blockchain supports decentralized and tamper-proof data management, while AI strengthens breach detection and response capabilities. In resource-constrained settings, this may involve incremental adoption supported by donor agencies or international partnerships. Operationally, regular staff training, adoption of standardized data management protocols, and investment in secure digital infrastructure are critical to minimizing human error and improving accountability.
Establishing comprehensive legislative frameworks
Policies should align with global standards like GDPR, HIPAA, and POPIA while addressing regional realities. For instance, sub-Saharan Africa requires tailored legislative models that factor in limited digital infrastructure, enforcement capacity, and socio-economic diversity. Support from multilateral institutions could help build capacity for implementation and compliance. Harmonizing data privacy laws across jurisdictions will foster cross-border cooperation and mutual accountability.
Promoting governance structures and privacy awareness
Robust governance structures, including the institutionalization of data protection officers, are essential for ensuring compliance. Continuous education and awareness programs for all staff levels must be institutionalized, with particular attention to frontline health workers who often handle patient data. These initiatives are crucial in building a culture of privacy vigilance, especially in low-resource healthcare environments.
International collaboration and continuous adaptation
Policymakers and healthcare providers must work together to develop standardized protocols, share cross-border best practices, and foster international dialogue on evolving threats and technological solutions. This also includes regional cooperation in surveillance, compliance audits, and emergency response coordination. In adapting to emerging threats and innovations, healthcare systems must adopt iterative learning models that regularly update privacy protocols and risk frameworks.
To enhance clarity and guide policy implementation, Figure 3 illustrates the key domains that should inform healthcare data privacy policy development and execution.

Key considerations to inform policy. Adopted from Health Information and Quality Authority, 2022. 62
Directions for future research
As data harmonization continues to evolve through the integration of new technologies, methodologies, and collaborative frameworks, the field is poised for transformational advancements. These emerging trends highlight both the progress made and the dynamic, adaptive nature of healthcare data integration in addressing complex data privacy challenges.
The ability to scale
With the exponential growth of health data, the scalability of harmonization procedures is becoming increasingly essential. 80 Organizations and institutions are now collecting data at unprecedented volumes and velocity, necessitating harmonization frameworks capable of managing vast, heterogeneous datasets without compromising efficiency or data quality. Future research should prioritize the development and testing of scalable, high-capacity harmonization systems that preserve performance across diverse platforms and settings.
Mechanization
Automation represents a promising avenue for reducing the manual labor traditionally required in data harmonization processes. 80 Tasks such as data matching, cleaning, and integration can be significantly accelerated through AI and ML algorithms. Research should explore the extent to which automation can enhance accuracy and efficiency in harmonizing disparate datasets while also identifying safeguards to mitigate automation-related biases or errors.
Combining new and emerging data sources
The integration of real-time data streams and Internet of Things devices introduces both opportunities and complexities for harmonization. 80 These novel data types require adaptive frameworks that can accommodate rapid data flows and maintain interoperability across devices and platforms. Future studies should investigate how harmonization strategies can be dynamically restructured to accommodate evolving data environments without sacrificing coherence or security.
Cutting-edge semantic and analytical technologies
Emerging semantic technologies and advanced analytics are reshaping how harmonized data can be interpreted and utilized. Techniques such as natural language processing and semantic ontologies play a crucial role in aligning the context and meaning of data from multiple sources.61,77 Further research is needed to enhance these tools for greater semantic precision, allowing for more insightful, context-aware analyses, and reducing the risk of misinterpretation.
Real-time analysis and processing
There is a growing demand for real-time data processing and decision-making capabilities, especially in high-stakes sectors like healthcare and finance. The ability to assess and act on harmonized data in real time or near real time is becoming critical for timely clinical interventions, risk assessments, and operational efficiency. Research should focus on the development of frameworks that support real-time harmonization and analytics, including the necessary technical infrastructure and algorithms. 80
Security, privacy, and ethical issues
As harmonization frameworks become more sophisticated, ensuring the security, privacy, and ethical use of sensitive data grows more complex and urgent. Future research must address the development of privacy-preserving methods and governance structures that comply with ethical standards and global regulatory requirements. 81 This includes exploring privacy-enhancing technologies, consent frameworks, and ethical auditing systems that ensure responsible data use while supporting integration and innovation.
Strengths and limitations of the study
This study is grounded in a robust review of 90 documents, including scholarly articles, regulatory frameworks, and case studies spanning four major world regions: North America, Europe, Asia-Pacific, and sub-Saharan Africa. The study's methodological strength lies in its systematic thematic analysis, which enabled the distillation of nuanced patterns and responses to healthcare data privacy across diverse contexts.
Additionally, the inclusion of detailed case studies (e.g. Anthem, WannaCry, SingHealth, and responses from Ghana and South Africa) provides empirical depth and contextual grounding for the thematic framework presented. The use of a rigorous coding process, a consolidated criteria for reporting qualitative research (COREQ) checklist, and visual aids (such as thematic diagrams) further enhances the transparency and reproducibility of this review.
However, the study acknowledges several limitations. First, the reliance on secondary data restricts the ability to capture emergent, localized dynamics that primary data could uncover. This includes real-time stakeholder experiences, evolving regulatory nuances, and the lived consequences of data breaches. Second, while the study covers all major global regions, it does not provide an in-depth analysis of Latin America and the Middle East, which limits the geographic generalizability of some conclusions.
Furthermore, although the literature selection was comprehensive, potential publication and language biases cannot be entirely ruled out. Some localized best practices or grey literature may not have been captured due to database constraints or language limitations. Finally, the fast-evolving nature of digital health technologies and privacy laws means that some of the findings may require periodic updating to remain current.
That said, the strength of this review lies in its methodological triangulation drawing from multiple case examples, thematic synthesis, and globally relevant regulatory frameworks to mitigate these limitations and produce generalizable, policy-relevant insights.
Conclusion
This review provides a critical synthesis of healthcare data privacy challenges and strategies across North America, Europe, Asia-Pacific, and sub-Saharan Africa. It highlights a global shift toward automated, intelligent data systems driven by regulatory reforms, technological innovation, and cross-border collaboration. While frameworks like GDPR, HIPAA, and POPIA offer strong legal foundations, disparities in implementation reveal the importance of adapting policies to local realities, especially in low-resource settings.
The findings emphasize that effective healthcare data protection requires multistakeholder engagement, combining government oversight, technological advancements such as AI and blockchain, and civil society participation. Although emerging technologies offer new opportunities for breach detection and interoperability, they also introduce ethical concerns regarding consent, autonomy, and data ownership that must be addressed.
Overall, this study underscores the need for an adaptive, equity-oriented approach to health data governance, balancing innovation with ethical safeguards. It offers practical, policy-relevant insights for strengthening global digital health systems, aligning with the WHO's vision for resilient, inclusive health governance in an interconnected world.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251343959 - Supplemental material for Data privacy in healthcare: Global challenges and solutions
Supplemental material, sj-docx-1-dhj-10.1177_20552076251343959 for Data privacy in healthcare: Global challenges and solutions by Andrew Kweku Conduah, Sebastian Ofoe and Dorothy Siaw-Marfo in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251343959 - Supplemental material for Data privacy in healthcare: Global challenges and solutions
Supplemental material, sj-docx-2-dhj-10.1177_20552076251343959 for Data privacy in healthcare: Global challenges and solutions by Andrew Kweku Conduah, Sebastian Ofoe and Dorothy Siaw-Marfo in DIGITAL HEALTH
Supplemental Material
sj-docx-3-dhj-10.1177_20552076251343959 - Supplemental material for Data privacy in healthcare: Global challenges and solutions
Supplemental material, sj-docx-3-dhj-10.1177_20552076251343959 for Data privacy in healthcare: Global challenges and solutions by Andrew Kweku Conduah, Sebastian Ofoe and Dorothy Siaw-Marfo in DIGITAL HEALTH
Footnotes
Acknowledgements
The corresponding author acknowledges the mentorship and quiet support received from academic colleagues whose encouragement made the completion of this manuscript possible.
Ethical approval
Not applicable. This study is based on publicly available secondary sources and did not involve direct interaction with human participants.
Consent to participate
Not applicable. No participants were recruited or surveyed for this review.
Author contributions
AKC led the thematic coding process and provided all financial resources. The emerging codes and categories were reviewed collaboratively with SHO, with discrepancies resolved through discussion.
DS-M assisted with both technical formatting and the literature review. AKC took primary responsibility for the manuscript's content and revisions, with all authors approving the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
