Abstract
Objectives
The selection of appropriate outcome measurement instruments (OMIs) in neurodegenerative disease (NDD) researches remains complex and often inconsistent. This study aims to consolidate knowledge on OMIs applied in NDD over the last two decades and to develop outcome measurement instruments in neurodegenerative diseases (NDDOMI), a web-based knowledge platform for OMIs selection.
Methods
We collected clinical trials from the past two decades across six prevalent NDDs from ClinicalTrials.gov. OMIs were manually extracted, categorized, and normalized by experts. We analyzed OMI utilization patterns, cross-disease applicability, and temporal trends. Additionally, we developed NDDOMI, a knowledge resource platform, using Django, ECharts, and JavaScript. The usability of NDDOMI was assessed using the System Usability Scale and Net Promoter Score.
Results
The NDDOMI platform provides an interface covering 4619 clinical trials and 2494 OMIs. On average, 4.4 instruments were utilized per trial. Fifteen instruments are shared across six NDDs. 76.75% of the trials employed more than one instruments, often in combination. A positive correlation was observed between the number of trials and the diversity of OMIs employed. The top 10 frequently employed instruments for each disease were identified, and their utilization has increased over time. The usability assessment demonstrated excellent user satisfaction with an average SUS score of 88.19.
Conclusions
This study introduces a comprehensive analysis of OMI usage trends in NDD research and introduces NDDOMI platform for OMI navigation and selection. NDDOMI serves as a valuable resource for optimizing clinical research design and standardizing outcome measurement in NDD management. The NDDOMI platform is available at http://bioinf.org.cn:3134/index/.
Keywords
Introduction
Neurodegenerative diseases (NDDs) are a group of neurological disorders that have a detrimental impact on millions of people worldwide.1,2 NDDs involve the progressive loss of neurons, resulting in a decline of memory, cognition, behavior, sensory and/or motor function. 3 As the global population ages, the proportion of individuals being diagnosed with NDDs continue to increase.4–6 In recent decades, thousands of clinical trials have been conducted to enhance profound understanding and provide better health management for NDDs. 7 Among the trials, various outcome measurement instruments (OMIs) are utilized to evaluate the effectiveness of interventions, treatments, or programs on specific outcomes of interest in individuals with health conditions. 8 With the growing number of clinical trials focused on NDDs, there has been increasing interest in enhancing measurement tools to better assess disease progression and treatment efficacy.9,10 Various OMIs have been developed and employed by clinicians and researchers across diverse fields to evaluate cognitive function, neuropsychiatric symptoms, motor symptoms, daily function, and quality of life in individuals with NDDs. 11
OMIs comprise various assessment instruments, such as clinical scales,12–14 imaging tests, 15 patient questionnaires, 16 and performance-based tests. 17 OMIs can be reported by patients, family members, caregivers or clinicians.10,18–20 OMIs used in clinical research can provide valuable evidence on the progression of diseases and the efficacy of drugs.21,22 Selection of appropriate instruments in clinical trials is challenging due to vast array of options and the lack of standardization. 23 Some experts, based on their expertise in developing and evaluating OMIs, have reached consensus-based outcome measurement standards in several NDDs.24–27 Some recommended instruments in NDDs include the Mini-Mental State Examination (MMSE), 28 the Montreal Cognitive Assessment (MoCA), 29 Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-Cog) 30 for cognitive function in AD, and the Unified Parkinson's Disease Rating Scale (UPDRS) 31 for motor function in PD. However, the development of core outcome set is time-consuming, 32 and lacks information support of OMIs utilization from massive amount of real-world clinical trials. OMIs is widely scattered in numerous clinical trials which lack standardization, integration, and curation. It is challenging for clinicians to quickly browse, choose, analysis, visualize, and examine OMIs utilization in clinical trials.
To address these challenges, we aimed to synthesize trial spanning over two decades and develop a knowledge-guided platform for OMIs in NDDs (NDDOMI).
Methods
In this original research study, we systematically consolidated knowledge on OMIs used in NDD clinical trials and developed a web-based platform to assist clinicians in selecting appropriate instruments.
Search strategy and selection criteria
For each NDD, we collected entry terms from Medical Subject Headings (MeSH) database (Supplemental Table S1). The MeSH terms and MeSH entry terms were employed as keywords to retrieve clinical trials registration website up to January 2023. An overview of the data inclusion process is noted in Supplemental Figure S1. We established several filtering criteria, including: (a) trials submitted before 2023; (b) trials related to the corresponding disease; and (c) trials with a description text of outcome measurement. A custom Python Script-based automatic pipeline was constructed to retrieve clinical trials, download registration files and extract information. The extracted information contains national clinical trial (NCT) identifier, title, date, condition or disease, outcome measurements. With the information, we removed trials that not matched the above criteria.
OMIs annotation
To determine whether a trial applied any instruments, we manually annotated instrument entities within the description text of outcome measurements. The instruments annotation was performed using brat, 33 a widely used web-based and user-friendly tool for text annotation in natural language processing research. Two annotators, the one is neurology clinician and the other is neuroscience researcher, collaborated to annotate the data.
Initially, they surveyed the definitions and literatures of the OMIs, and reviewed various expressions within clinical trials text. Then, the data was divided into two parts and labeled separately. Finally, they cross-checked each other's annotations to ensure consistency, and discussed contradictions until consensus was achieved. Based on the annotation results, we removed trials that did not apply any instruments in the outcome measurements. For instance, some outcome descriptions, such as “status of progression—changes in language processing” indicated the intention to measure language ability but did not reference any related assessment tools. Therefore, these trials were not included in our analysis.
OMIs normalization
Generally, an instrument comprises of multiple subscales, and maybe has various versions. To address this issue, we proposed a two-level instrument name normalization pipeline aimed at transforming original instrument expressions in the text into their corresponding normalized full names. First, we accounted for variability in writing styles, including punctuation marks, singular and plural numbers, among others, and we standardized various instruments expressions to formal expressions. Second, we considered the diversity of versions and modules, and standardized the formal expressions to higher level instrument. For example, as depicted in Supplemental Figure S2, the terms “Alzheimer's Disease Assessment Scale—Cognitive Subscale” and “ADAS-cog” are different expressions, but actually refer to same instrument “Alzheimer's Disease Assessment Scale—Cognitive Subscale.” We defined it as a Level 2 instrument and further mapped to the Level 1 instrument “Alzheimer's Disease Assessment Scale.” Similarly, “Neuropsychiatric Inventory-Questionnaire” and “NPI-Q” are both normalized as instrument “Neuropsychiatric Inventory.”
Platform implementation
These trials underwent a rigorous screening process, and relevant OMIs information was carefully collected and organized. This included baseline information, clinical data and OMI details. To determine what information to include in the platform, we consulted clinicians and researchers during the development phase to understand their needs, focusing on providing quick access to frequently used OMIs, analyzing utilization patterns, and tracking temporal trends. The platform emphasizes empirical evidence from large-scale clinical trials to support OMI selection. Furthermore, we manually recognized and normalized OMIs from the trials, which were then analyzed to understand their utilization patterns and temporal trends. To facilitate user interaction and data visualization, we developed web pages using an open-source framework Django (https://www.djangoproject.com/). Data tables and figures were generated and visualized using ECharts (https://echarts.apache.org).
Moreover, we used the System Usability Scale (SUS)34,35 and the Net Promoter Score (NPS) 36 to assess usability and satisfactory of NDDOMI platform. We recruited 18 potential users to use and evaluate the NDDOMI platform. We recruited 18 potential users from five institutions in China to use and evaluate the platform: West China Hospital of Sichuan University (four participants: three clinicians specializing in neurosurgery, urology, and rehabilitation, and one nurse), First Affiliated Hospital of Xinjiang Medical University (three neurosurgery clinicians), Qingyang People's Hospital (one neurosurgery clinician), Southwest Hospital (one orthopedics clinician and one nurse), and Tongji University (eight researchers focused on NDDs). These participants were identified as potential users during the platform development phase and contributed to early discussions about user needs. We collected feedback and calculated the scores of SUS and NPS based on scoring formula.
Results
Data statistics
The major sources of data in NDDOMI were derived from NDD clinical trials. The initial search yielded 9515 trials. Following the data filtering process, instrument annotation, and eligibility assessment, a final data set of 4619 trials were retained for analysis. The submission date of these trials ranged from January 2001 to January 2023. As shown in Supplemental Table S2, the AD has the highest number of trials with 2163 (46.83%), followed by PD (2013, 43.58%), ALS (396, 8.57%), HD (132, 2.86%), LBD (90, 1.95%) and FTLD (69, 1.49%). When a trial mentioned more than one NDD in its conditions, we considered each of them separately in the analysis. For example, there were 69 trials with both AD and PD in their conditions, which we included in AD dataset and PD dataset separately for analysis.
A total of unique 10,679 original instrument entities were annotated, which were further mapped to 2494 normalized instruments. Supplemental Table S2 shows the number of clinical trials, employed instruments, unique instruments, and the average number of instruments used in each trial. The overall average number of instruments employed in trials is 4.4003, while the minimum number is 1 and maximum number is 31 (Supplemental Figure S3). AD has the highest number of unique instruments of 1647 with an average of 4.5603 instruments used per trial. FTLD has the highest average number of instruments per trial at 5.3188.
Most popular instruments
To display the prevalence of the OMIs in the platform NDDOMI, we have examined the data from NDD trials. There are totally 2494 unique instruments applied within the trials of NDD, including AD (n = 1647), PD (n = 1134), ALS (n = 223), FTLD (n = 185), LBD (n = 181), and HD (n = 166). The most frequently used instruments and their temporal usage trends of each disease are shown in Figure 1.

The top popular instruments and their usage frequency in temporal trends of each neurodegenerative disease. (a) Alzheimer disease. (b) Parkinson’s disease. (c) Amyotrophic lateral sclerosis. (d) Frontotemporal lobar degeneration. (e) Lewy body disease. (f) Huntington’s disease.
In AD trials, the MMSE, NPI, and ADAS are the most commonly employed instruments (Figure 1(a)). In PD trials, the UPDRS and Parkinson's Disease Questionnaire (PDQ) are most frequently used (Figure 1(b)). In ALS trials, the Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS) is the predominant instrument (Figure 1(c)), while in HD trials, the Unified Huntington's Disease Rating Scale (UHDRS) is the most applied (Figure 1(f)). In contrast, FTLD (Figure 1(d)) and LBD (Figure 1(e)) have fewer trials overall, with disease-specific instruments being more limited. The NPI and MMSE are frequently utilized in these conditions.
Shared instruments among NDDs
The number of shared and unique instruments among the NDDs are shown via an UpSet diagram in Figure 2(a). The AD had the most specific instruments with 1090, followed by PD with 649. Among the numerous instruments, 203 were shared between AD trials and PD trials, and 15 were shared across 6 NDDs.
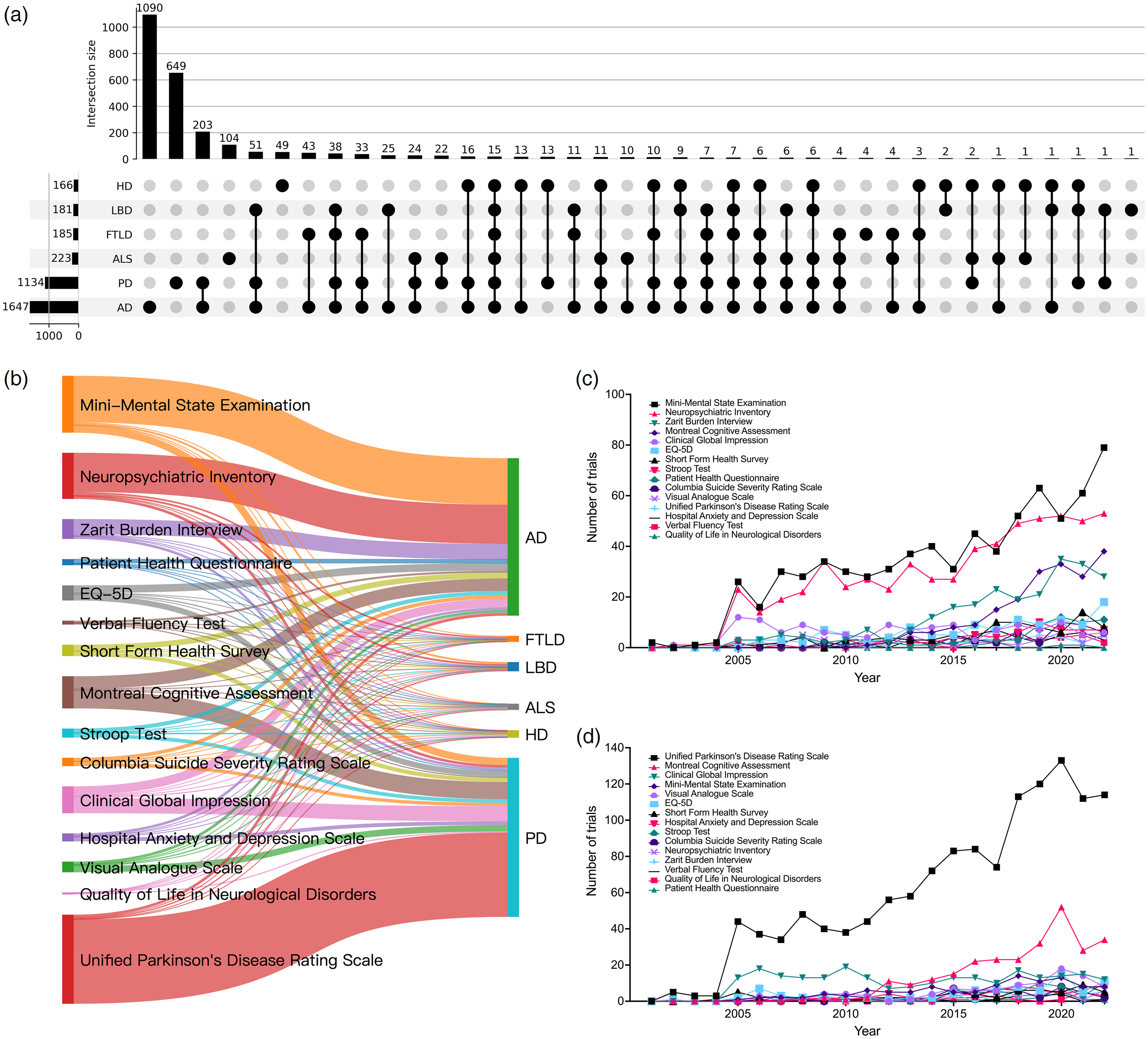
Temporal trends for 15 shared instruments among six neurodegenerative diseases. (a) UpSet diagram showing specific and common instruments among six neurodegenerative diseases. (b) Sankey diagram illustrating patterns of relationships between instruments and neurodegenerative diseases. (c) Temporal trends of the 15 instruments in Alzheimer's disease trials. (d) Temporal trends of the 15 instruments in Parkinson's disease trials. AD: Alzheimer's disease; PD: Parkinson's disease; LBD: Lewy body disease; HD: Huntington's disease; ALS: amyotrophic lateral sclerosis; FTLD: frontotemporal lobar degeneration.
In Figure 2(b), we presented the usage patterns between the 15 instruments and 6 NDDs. The thickness of lines reflects the frequency of usage of each instrument in each disease. We observed a high frequency of usage of the UPDRS for PD. Similarly, MMSE, NPI and Zarit Burden Interview (ZBI) have high frequency usage for AD. Other instruments such as EuroQol 5 Dimensions (EQ-5D), MoCA and Clinical Global Impression (CGI) are often used in both AD and PD, and no significant difference in frequency. Figure 2(c) and (d) illustrated the temporal trends of the 15 instruments in AD and PD trials, respectively. The results indicated that a majority of the instruments are utilized sparingly, whereas certain instruments are predominant and exhibit increasing usage over time. Specifically, the MMSE in AD trials, while UPDRS in PD trials.
Association patterns between different instruments
Over 76.75% of trials employed more than one instruments (Figure S3). However, 63.27% (1578) of the instruments were employed only once in clinical trials of NDDs (Figure S4), indicating that most instruments were employed extremely infrequently. To illustrate the association usage patterns among these instruments, we present a network diagram in Figure 3.
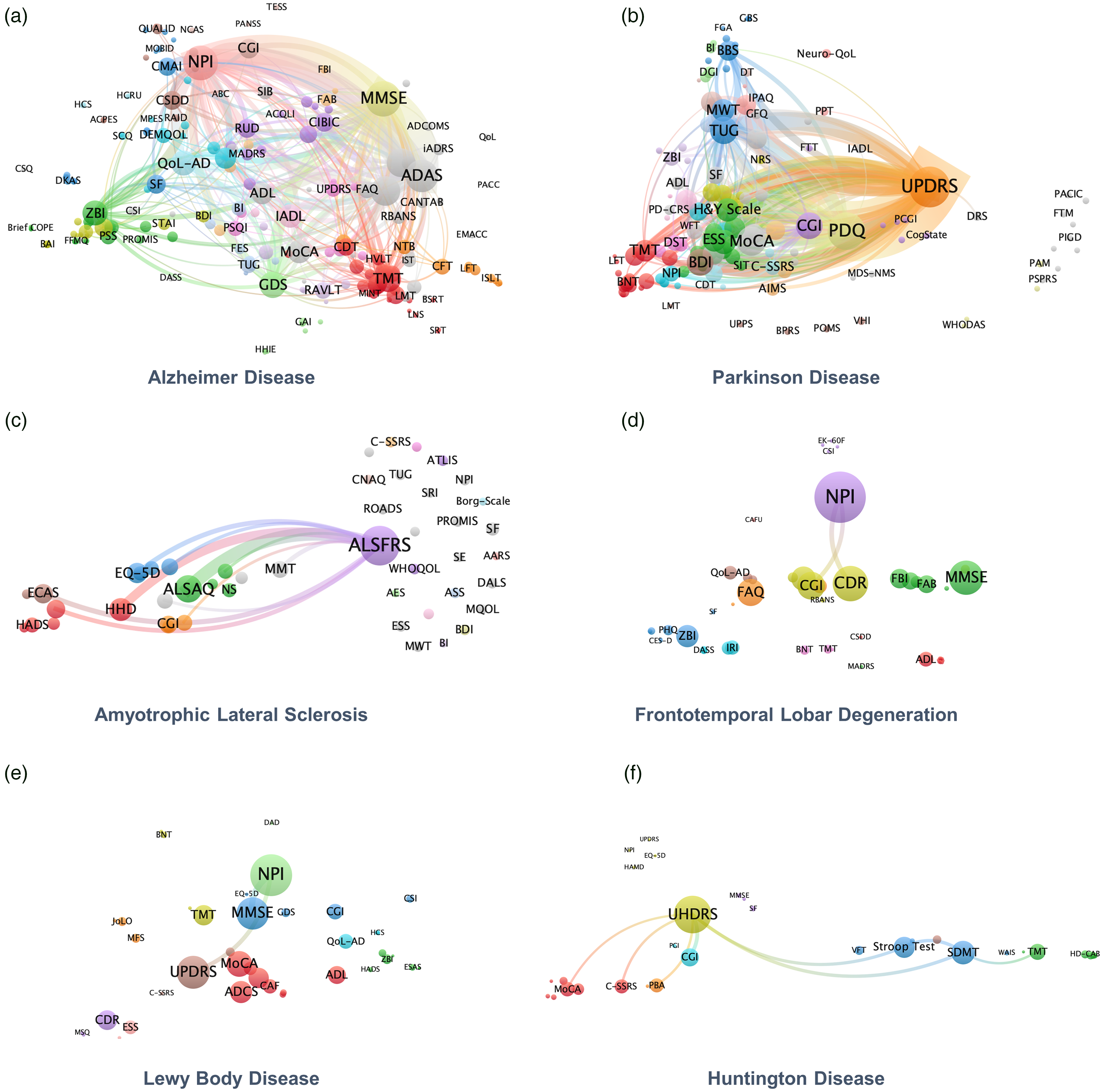
The association patterns among different instruments (a) Alzheimer’s disease. (b) Parkinson’s disease. (c) Amyotrophic lateral sclerosis. (d) Frontotemporal lobar degeneration. (e) Lewy body disease. (f) Huntington’s disease. Each node on the network graph corresponds to a specific instrument, and the edges between them represent the number of trials where both instruments were utilized. The size of each node is proportional to its frequency of usage, and the thickness of the edges reflects the number of trials. We sorted the instrument pairs in descending order of usage frequency and displayed only the top 10%. Additionally, we have limited the display of edges with 10 or more trials. MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; UPDRS: Unified Parkinson's Disease Rating Scale; ADAS-Cog: Alzheimer's Disease Assessment Scale-Cognitive Subscale; NPI: Neuropsychiatric Inventory; ZBI: Zarit Burden Interview; CGI: Clinical Global Impression; ADAS: Alzheimer's Disease Assessment Scale; PDQ: Parkinson's Disease Questionnaire; ALSFRS: Amyotrophic Lateral Sclerosis Functional Rating Scale; UHDRS: Unified Huntington's Disease Rating Scale; TUG: Time Up and Go Test; ALSAQ: Amyotrophic Lateral Sclerosis Assessment Questionnaire; HDD: Handheld Dynamometry; CDR: Clinical Dementia Rating Scale; EQ-5D: EuroQol 5 Dimensions; CGI: Clinical Global Impression; ADCS: Alzheimer's Disease Cooperative Study; MWT: Meter Walk Test; PGI: Patient Global Impression; PHQ: Patient Health Questionnaire; NLP: Natural Language Processing; MDS: Movement Disorder Society; UHDRS-TMS: Unified Huntington's Disease Rating Scale-Total Motor Score.
In AD trials, ADAS-MMSE and MMSE-NPI emerged as the most frequently employed instruments combinations. In PD trials, UPDRS was the most commonly used instrument, which was frequently used with PDQ, MoCA, CGI, and Time Up and Go Test (TUG). In ALS trials, researchers frequently utilized ALSFRS in conjunction with Amyotrophic Lateral Sclerosis Assessment Questionnaire (ALSAQ) and Handheld Dynamometry (HHD). In FTLD trials, the most commonly used instruments combinations were NPI and Clinical Dementia Rating Scale, as well as NPI and Clinical Global Impression (CGI). In LBD trials, the NPI-MMSE combination was frequently employed, while in HD trials, the UHDRS-CGI combination was the most commonly used.
Usage of different versions
OMIs typically contain multiple modules or various versions to address cultural differences, accommodate diverse populations, and enhance reliability and validity. Figure 4 shown the usage of different modules and versions for 8 commonly employed instruments, including ADAS, UPDRS, Alzheimer's Disease Cooperative Study (ADCS), Meter Walk Test (MWT), CGI, Patient Global Impression (PGI), Patient Health Questionnaire (PHQ), and EQ-5D. The results indicated that some certain subscales of instruments, such as ADAS-Cog Subscale and ADCS-Activities of Daily Living (ADCS-ADL), were utilized particularly frequently. For instruments such as CGI and PGI, which are instruments for assessing the overall severity of illness or improvement in clinical trials, and multiple versions for change (CGI-C, PGI-C), severity (CGI-S, PGI-S) and improvement (CGI-I, PGI-I) are available. The UPDRS was often used in its entirety in some trials, while other trials utilized only a module of it. When employing the MWT, the 10 MWT was the most commonly used, although other versions such as the 4 MWT, 6 MWT, 30 MWT, 4 × 3 MWT, and 4 × 10 MWT were also used. Similarly, for PHQ, the PHQ-9 was the most frequently utilized version, while the PHQ-2, PHQ-4, PHQ-8, PHQ-15 were also used.

The usage patterns of different modules and versions for eight commonly employed outcome measurement instruments, including Alzheimer's disease cooperative study (ADAS), Unified Parkinson's Disease Rating Scale (UPDRS), Alzheimer's disease cooperative study (ADCS), Meter Walk Test (MWT), clinical global impression (CGI), patient global impression (PGI), Patient Health Questionnaire (PHQ), and EuroQol 5 dimensions (EQ-5D).
Platform overview
The NDDOMI (Figure 5), a pioneering knowledge-guided platform dedicated to OMIs in NDDs, including Alzheimer's disease (AD), Parkinson's Disease (PD), Lewy body disease (LBD), Huntington's disease (HD), Amyotrophic lateral sclerosis (ALS), and Frontotemporal lobar degeneration (FTLD). We have developed an online information retrieval system specifically designed for intelligent terminals, such as smartphones or tablets. The NDDOMI is constructed and available at http://bioinf.org.cn:3134/index.

A schematic overview of the NDDOMI platform, including homepage, search page, explore page, statistics page, about page, and contact page. Homepage: Introduces the significance of outcome measurement instruments in NDDs and platform features. Search page: Allows browsing, searching, analyzing, and visualizing instruments, including information retrieval, prevalence analysis, and usage patterns. Explore page: Enables detailed exploration of clinical trial details by NCT number, covering basic information, used instruments, and primary/secondary outcomes. Statistics page: Displays key study results, figures, and tables. About Page: Provides an overview of the platform content. NDDs: neurodegenerative diseases; NCT: national clinical trial; NDDOMI: outcome measurement instruments in neurodegenerative diseases.
The homepage of NDDOMI serves as an introductory gateway, outlining the significance of OMIs in NDDs and highlighting the key features of the platform. The search and explore page of NDDOMI is designed to cater to the diverse needs of users. The search page is designed to provide user-friendly access to browse, search, analyze, and visualize. The search page provides three key functionalities: information retrieval, temporal prevalence analysis, and usage pattern examination of different instruments or versions. By clicking on individual OMIs, users can access OMIs basic information, temporal prevalence, distribution in NDDs, distribution in versions, distribution in combinations, and detailed clinical trials information. The explore page enables users to delve into comprehensive trial details by searching NCT number. The explore page presents relevant trial information, including basic information, primary outcome, secondary outcome, and other outcomes. The statistics page displays the main results, figures, and tables presented in our study. The about page details the content of the knowledge base, providing users with a clear understanding of the data contained within.
In summary, the NDDOMI platform provides a comprehensive resource for researchers and clinicians interested in exploring the utilization of OMIs in NDDs. With its robust functionalities and user-friendly interface, NDDOMI aims to facilitate the advancement of clinical research in this critical field.
Platform usability evaluation
The average SUS score was 88.19 ± 11.78. According to the SUS grading standard, NDDOMI was rated as excellent (Figure S5A), indicating high user satisfaction. The proportions of users graded as A and B were 61.11% and 22.22%, respectively (Figure S5B). The NPS score ranged from 0 to 10, with an average score of 9.56 ± 1.15. 88.89% (16/18) of users were promoters (scores 9–10), 5.56% (1/18) as passives (scores 7–8), and 5.56% (1/18) as detractors (scores 0–6) (Figure S5C). The final NPS value was 83.33, indicating a high recommendation level for the NDDOMI platform (Figure S5D).
Discussion
In this study, we identified 2494 OMIs from 4619 clinical trials for 6 NDDs, and comprehensively investigate the utilization patterns and temporal trends. In the process of instrument extraction and normalization, a total of 10,679 instrument name synonyms were recorded. Considering research purpose and data, we chose manually annotation instead of natural language processing technology. The primary objective of this study is to explore the utilization patterns and temporal trends of instruments. To ensure the reliability of the results, accurate instrument identification is deemed crucial. Therefore, we engaged neurology clinician and neuroscience researcher in the field of NDDs to perform manual instrument annotation. The data used in this study has been sourced from the outcome measurement section of clinical trial registration files. The information is presented in a highly readable textual format, which enable effortless extraction and normalization of instrument names.
The comparison of different NDDs revealed heterogeneity in instruments utilization patterns. One potential explanation is the difference of research attention for the diseases. For instance, AD and PD are two of the most common NDDs and attracted significant attention from researchers and clinicians, which leading to extensive research efforts.37,38 These instruments, such as MMSE and ADAS for measuring cognitive impairment and neuropsychiatric symptoms, have gained popularity over time and continue to be widely utilized owing to their recognition as established measures of cognitive decline, dementia severity, and treatment response.28,30 Consequently, a significant number of diagnostic and assessment instruments have been developed, validated and employed. 11 On the other hand, ALS, FTLD, LBD and HD are relatively less common than AD and PD. As a result, there are relatively fewer instruments have been developed for them. However, our findings shown the emergence of a few authoritative instruments, such as ALSFRS for measuring physical function and disease progression in ALS, and UPDRS for assessing motor and non-motor symptoms in PD, which have gained widespread acceptance and employment in clinical trials for outcome measurement. Additionally, the guideline recommendations may also contribute to this phenomenon. Our analysis revealed that the most frequently employed instruments in each NDDs align closely with those recommended by key organizations and clinical guidelines. For example, the MMSE and ADAS, the most commonly used instruments in AD trials, are widely endorsed for assessing cognitive function. The Movement Disorder Society (MDS) version of UPDRS is the most commonly used rating scale in PD research and is recommended to measure PD related disability and disease progression.39,40 The UHDRS-Total Motor Score (UHDRS-TMS) is a widely used clinician-rated scale that was developed by the Huntington Study Group, and is recommended for the assessment of motor dysfunction and disease severity in HD. 41 As the field moves toward greater standardization, the data presented in this study can help inform updates to existing recommendations, ensuring they reflect both established best practices and emerging trends in outcome measurement.
Another notable observation is the prevalence of instrument submodules, subscales and various versions. For example, the UPDRS is a validated tool that specifically designed to measuring the severity of symptoms in PD, which consists of four submodules that can be utilized as a whole or in parts, depending on the research objective and trial design. Version issues primarily encompass two types. The first is different iteration versions. As scientific research develop, older versions of instruments may not meet new clinical needs, prompting researchers to continuously improve, design, and validate new instruments to adapt to clinical practice. For example, the PHQ 42 includes PHQ2, PHQ4, PHQ8, PHQ9, and PHQ15. Additionally, the selection of a particular version depends on the specific research question and study design. For example, the CGI has multiple versions CGI-C, CGI-S, and CGI-I. If a trial aims to compare the efficacy of two treatments, the CGI-I may be appropriate to assess overall improvement. If the trial aims to assess the overall changes in clinical status, the CGI-C may be more suitable. The second is different language versions. This is mainly to adapt to the various customs, cultural differences, and language barriers of different countries or regions. The most common ADAS-Cog language versions include Korean, Japanese, Cantonese, Mandarin, Swedish, Norwegian, among others. This diversity suggests ongoing efforts to refine measurement tools for different research contexts and patient populations. These findings demonstrate the importance of selection of appropriate modules or versions of instruments in clinical research. By selecting the appropriate modules or versions of an instrument, researchers can improve the reliability and validity of their findings.
The considerable variation in OMIs across clinical trials highlights the ongoing challenge of standardizing outcome assessments in NDD research. Standardization is crucial for enabling meta-analyses, facilitating cross-study comparisons, and improving the reproducibility of findings. Without a consistent set of instruments, synthesizing evidence across trials becomes difficult, limiting the generalizability of research outcomes and slowing the development of evidence-based guidelines. However, we also observed emerging trends toward the adoption of certain instruments over time. These trends indicate that the field may be gradually moving toward standardization, driven by factors such as guideline recommendations, regulatory requirements, and the widespread use of validated instruments in clinical practice.
More than three-quarters of trials employed more than one instruments, which means the instruments were often used in combination. In clinical research, a comprehensive measurement of participants’ physical and cognitive status is often necessary to determine the effectiveness of treatment interventions. Different instruments typically cover different domains and dimensions, such as cognition, emotion, behavior, physiology, and so on. 43 The combinations of multiple instruments can compensate for the limitations of a single instrument by providing greater comprehensiveness and accuracy in evaluation. Moreover, the development and validation of a new instruments often require comparative analyses with existing instruments, 44 thus increasing the prevalence of combined instrument usage. In this study, we identified the most common combinations of instruments, and these results can provide valuable reference for the clinical trial design and novel instrument development.
Methodologically, this study demonstrated how clinical trials registration data can be used to explore the utilization patterns and temporal trends of instruments. We have identified the top commonly employed instruments and its combinations, revealed the utilization patterns and temporal trends in past two decades, and also analyzed usage for submodules or different versions.
The NDDOMI was built for clinical researches and it integrates vast amounts of clinical trial data, offering empirical evidence for the selection of OMIs. The NDDOMI collects and organizes 2494 OMIs used in 4619 clinical trials of six common NDDs over the past two decades, providing clinicians and researchers with a comprehensive and easily accessible knowledge base.
The NDDOMI's information includes the basic information, such as name, abbreviation, synonyms, related disease, definitions, and available download link, and prevalence information, such as number, frequency, versions, combinations of OMIs used in clinical trials of different NDDs. The NDDOMI offers functions such as information retrieval, temporal prevalence analysis, and usage pattern examination. For instance, the information retrieval function allows users to quickly identify commonly used OMIs for specific NDDs; the temporal prevalence analysis function enables users to understand the trends in OMI usage over different years; and the usage pattern examination function provides insights into how OMIs are utilized, and their versions and combinations in various trials. This enables clinicians to gain a more comprehensive understanding of OMIs and make more informed choices. In summary, the NDDOMI platform serves as a comprehensive, convenient, and efficient knowledge base for the selection and application of OMIs in clinical research for NDDs. NDDOMI promotes consistency and standardization by providing information retrieval, temporal prevalence analysis, and usage pattern examination of different instruments or versions. Furthermore, in NDDOMI users are allowed to explore the detailed trials information by surveying NCT number, composing of 4619 trials. NDDOMI can assist users in making clinical OMIs selection through organized, structured, and evidence-based knowledge. We believe NDDOMI will become an invaluable resource for the clinical research of neurological disorders.
This study has several limitations. First, we utilized clinical trial registration data, which although closely aligned with real clinical practice, may not adequately represent the breadth of researches on NDDs. In the future, it may be beneficial to include a larger volume of published literature data. Second, the process of extracting and categorizing OMIs relied on manual annotation and normalization, which, despite rigorous cross-checking, may introduce subjectivity and potential oversight. Third, while we used the SUS and NPS to evaluate the usability and overall satisfaction of the NDDOMI platform, these assessments did not directly measure whether the platform's content sufficiently meets users’ needs in selecting appropriate OMIs. Future iterations of the platform will aim to incorporate more comprehensive content evaluation, guided by further user feedback, to better support the selection of OMIs in clinical and research settings. Finally, the platform currently focuses on providing basic instrument information, usage frequency, and temporal trends, without detailing the specific domains or constructs assessed by each OMI, their psychometric properties, or endorsements from authoritative guidelines like the NIH Toolbox or Clinical Practice Guidelines. While integrating these elements would enhance the platform's utility, the process requires extensive data collection and validation. A notable improvement would be the inclusion of personalized guidance in instrument selection. A guided workflow could help users navigate the selection process, from forming an initial research question to identifying the most relevant OMIs. Future iterations will prioritize incorporating these aspects to better support clinicians and researchers in selecting appropriate instruments.
Conclusion
NDDOMI serves as the first knowledge base for OMI navigator. As a medical knowledge resource, NDDOMI has the potential to assist clinicians in selecting OMIs. NDDOMI presented a comprehensively investigate of the utilization patterns and temporal trends of OMIs in NDDs by analyzing clinical trial registration data spanning two decades. A total of 2494 unique instruments were identified. The utilization patterns indicated that most instruments were employed extremely infrequently. More than three-quarters of trials employed instruments in combination. The top 10 frequently employed instruments for each disease were determined, and their temporal trends were analyzed. Additionally, the submodules and versions of instruments were thoroughly explored. These findings have highlighted complexity of instruments employment and provided valuable insights for the diagnosis and management of NDDs.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251335544 - Supplemental material for Outcome measurement instruments in neurodegenerative diseases: Comprehensive analysis and platform construction
Supplemental material, sj-docx-1-dhj-10.1177_20552076251335544 for Outcome measurement instruments in neurodegenerative diseases: Comprehensive analysis and platform construction by Hui Zong, Jiaxue Cha, Liang Tao, Muyun Shi, Yan Zhao, Ru Zhang and Bairong Shen in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors acknowledge the use of generative AI for language polishing in the preparation of this article. The authors take responsibility for the content and intended meaning of this article.
Author contributions/CRediT
H.Z., J.C., L.T., M.S., Y.Z., R.Z., and B.S. involved in the study concept and design. HZ collected and preprocessed the data. H.Z. and J.C. participated in the data annotation. H.Z. and J.C. conducted data analysis, results interpretation, and manuscript preparation. H.Z., J.C., L.T., M.S., Y.Z., R.Z., and B.S. contributed to the editing of the manuscript. B.S. supervised the research. All authors read, revised, and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (Grant Nos. 32270690 and 32070671) and Sichuan Science and Technology Program (Grant No. 2024YFHZ0205).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The codes are available at https://github.com/zonghui0228/NDDOMI. The data analyzed and reported in this study are available at ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
