Abstract
Introduction
The objective of Virtually Assisted home rehabilitation after acute STroke was to offer a fully remote telerehabilitation intervention to stroke survivors during the COVID-19 pandemic.
Methods
Participants were recruited from acute care, inpatient rehab, or provider/self-referral if they had a stroke within the previous year, prestroke modified Rankin Scale < 3, were recommended to participate in rehab, and had internet access. Exclusions were: prior injury/diagnoses that impacted functional level, life expectancy of less than six months, or safety concerns. Outcomes were within one week prior to initiation and within one week after completion of telerehabilitation sessions. Video call sessions were completed 1–2 times a week for 12 weeks. Occupational therapy, physical therapy, and/or speech therapy were provided. Analyses involved descriptive statistics; qualitative comments were aggregated and analyzed for broader themes. Twenty participants were consented.
Results
Of the 20 participants (39–71 years old, 35% White, 50% female), 12 completed study activities, five completed exit interview; 10 demonstrated improvement on National Institute of Health Stroke Scale from a mean of 5.6 at baseline to 2.7 at end of study, six demonstrated improvement in modified Rankin Score and EuroQol (EQ-5D-5L) scores from a mean of 2.8 to 1.9 and 13.3 to 9.3, respectively, and five demonstrated an increase above the minimally clinically important difference (mean change = 5) on Montreal Cognitive Assessment. On the Stroke Impact Scale rating of total recovery, six of the 12 scored themselves with improvement (mean change = 18.2). Exit interview data revealed an emerging theme: while in-person therapy was preferred, telerehabilitation was an important resource for those without access to in-person therapy.
Conclusion
Our study adds to the growing data on the practice of telerehabilitation for survivors of stroke. Future studies are underway to address telerehabilitation serving the uninsured, underinsured, and populations in rural areas.
Clinicaltrials.gov ID
NCT05659784.
Introduction
Rehabilitation is a critical component of healthcare for stroke survivors. However, access to rehabilitation is limited in rural and uninsured populations.1,2 Persons of low socioeconomic status are at a much greater risk of stroke due to lower health literacy, reduced management of risk factors, and higher rates of tobacco use, but are much less likely to have access to rehabilitation due to insurance or language barriers. 1 Rural populations may not have access to a rehabilitation facility at all. In Texas, 71% of rural counties have zero outpatient rehabilitation clinics and within nonrural counties, 19% have zero outpatient rehabilitation clinics. 2
To address these barriers to accessing rehabilitation in disadvantaged populations, telerehabilitation is being explored as an effective alternative to in-clinic rehabilitation.3–12 Not only does telerehabilitation remove transportation barriers, it also allows patients to practice exercises and functional tasks in their own homes instead of artificial or simulated environments. Instead of practicing on a stair machine, sit to stand from a standardized chair, or reading from random magazines, patients are able to practice on their own stairs, from their own chairs, and with their own favorite books. 4 Therapists can make personalized exercise recommendations based on the patient's home and what they have access to, which may further facilitate patient engagement and motivation. 4 Previous research on telerehabilitation in poststroke recovery has shown that outcomes from telerehabilitation are comparable or exceed outcomes of traditional in-person rehabilitation. 11 While there are many technologies, such as video games, virtual reality, web-based platforms, and mobile applications, 12 being trialed in telerehabilitation, more studies are needed on the safety, feasibility, and outcomes of delivering telerehabilitation to underserved, socially vulnerable populations. In particular, studies are needed on telerehabilitation that does not require specialized equipment or technologies.
We studied the delivery of telerehabilitation in Harris County, Texas, where the population has a high social vulnerability index (SVI = 0.9201; range 0–1). 13 This study was undertaken to address the problem that was occurring in stroke rehabilitation due to the COVID-19 pandemic: a lack of postacute rehabilitation for many stroke survivors across Texas due to clinic closures and social distancing, but continued throughout the endemic addressing rehabilitation utilization for the underserved. Unlike previous telerehabilitation studies, Virtually Assisted home rehabilitation after acute STroke (VAST-rehab) allowed all study aspects to be completed remotely, including the consent process and outcome measures, to improve accessibility and high-quality neuro-rehabilitative care to an already high-risk population. The purpose of this study was to examine the feasibility of a fully remote telerehabilitation program and its potential to improve outcomes in stroke survivors.
Methods
VAST-rehab is an exploratory pilot study that began in June of 2020 following approval by the Committee for Protection of Human Subjects at The University of Texas Health Science Center at Houston and Clinical Innovation & Research Institute at Memorial Hermann Hospital. This study was registered on clinicaltrials.gov (Clinicaltrials.gov ID: NCT05659784). A pilot exploratory design was chosen to gather preliminary data on our fully remote telerehabilitation approach. Written consent was obtained from eligible and agreeable participants either in person while hospitalized for acute stroke or electronically following discharge using an informed consent form approved by the Committee for Protection of Human Subjects at The University of Texas Health Science Center at Houston. If consented electronically, participants engaged in consent process over phone or video call and signed the consent form electronically via RedCAP. All assessments and telerehabilitation sessions were completed over video call through the Health Insurance Portability and Accountability Act compliant Webex platform. All study activities were completed by May 2023.
Participants
Participants were approached while in acute care or inpatient rehabilitation at Memorial Hermann—Texas Medical Center in Houston, Texas. Participants were also welcomed through referrals from a provider or self-referral within a year poststroke, as telerehabilitation could benefit in each stage of recovery. Patients met inclusion criteria if they had a hemorrhagic or ischemic stroke, were at least 18 years old, had a prestroke Modified Rankin Scale (mRS) less than 3, were recommended to participate in rehabilitation therapy by a physician or rehabilitation therapist, had sufficient cognitive and language ability to understand commands and complete study assessments as deemed by medical team or had availability of LAR to assist, were able to access the internet via a personal device, and able to understand and speak English or Spanish. Bilingual study staff were involved in sessions with Spanish-speaking participants. Patients were excluded if they had a history of a neurological diagnosis that resulted in a significant functional impairment or severe comorbid diagnosis with an expected life expectancy of less than six months. Participants were excluded at the opinion of the investigators if telerehabilitation was deemed unsafe due to severity of deficits or medical instability following a medical history review.
Telerehabilitation program
Participants were scheduled for baseline assessments within 1–2 weeks of consent. Outcome measures included National Institute of Health Stroke Scale (NIHSS), mRS, Barthel Index, EuroQol-5D-5L (EQ-5D-5L), Montreal Cognitive Assessment (MoCA), and Stroke Impact Scale (SIS). Outcomes were collected within one week prior to initiation of telerehabilitation sessions and again within one week after completion of telerehabilitation sessions. An exit interview was developed based on investigator's previous clinical experiences and was completed by participants at the end of study. Outcomes were completed by a study coordinator or treating therapist. A physical medicine and rehabilitation physician reviewed medical concerns with the patients as needed.
Participants were scheduled for a 1-h rehabilitation session, per discipline, once a week over video call for 12 weeks at a time that was convenient for them, their caregiver (if appropriate/needed), and the rehabilitation therapist. Participants were scheduled with an occupational therapist, physical therapist, and/or speech language pathologist, depending on their individual needs. Therapy disciplines were selected based on individual participant goals/needs and therapist availability. If needed and available, participants received two disciplines each week. In this case, therapy sessions were scheduled successively when available. The telerehabilitation sessions consisted of individualized home exercise, functional training, and stroke education. Participants identified goals and the therapists targeted those goals through the education-based intervention. Sessions were guided by participant goals, included rehabilitative and compensatory techniques, and addressed motor, sensation, vision, cognition, language impairments, and activity/functional limitations. Therapy sessions were individualized to participants needs and included stroke specific education, such as importance and logistics of blood pressure monitoring, medication compliance, and secondary stroke prevention, as well as rehabilitative therapy intervention. The rehabilitative therapy intervention focused on home exercise prescription, safety, and practice, and was updated each week as needed. Treatment techniques were selected at the discretion of the therapists based upon being able to maintain safety during performance of the activities. Intervention was provided at no cost to the participants by rehabilitation therapists licensed in the state of Texas.
Outcome measures
The NIHSS is a measure of deficit poststroke, with scores ranging from 0 to 42 (very severe: > 21, moderate to severe: 16–20, moderate: 5–15, minor: 1–4). 14 The mRS assesses global disability on a six-point scale: 0 = no symptoms, 1 = no significant disability despite symptoms, 2 = slight disability, 3 = moderate disability, 4 = moderately severe disability, and 5 = severe disability. 15
The Barthel Index is a patient report measure of participation level in activities of daily living (ADLs) and mobility with a score of up to 100. It has excellent criterion validity with the Functional Independence Measure (FIM) motor. 16 The Barthel Index does, however, have a ceiling effect, which is seen within the results of this study. 17
The EQ-5D-5L is a self-report measure of perceived quality of life in mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. 18 For the purposes of this study, the EQ-5D-5L raw score is utilized to determine pre- and postintervention change in overall quality of life.
The MoCA is a screen for mild cognitive impairment that assesses executive functioning, naming, memory, attention, language, abstraction, and orientation with a maximum score of 30. It has excellent internal consistency (0.78) and criterion validity with the Mini-Mental Status Examination (r = 0.79) and cognitive FIM scores (r = 0.67). 19
The SIS is a patient report outcome measure that assesses eight domains: strength, hand function, ADLs, mobility, communication, emotion, memory and thinking, and participation. 20 Each question is scored on a five-point Likert scale; summative scores for each domain range from 0 to 100. The SIS has excellent internal consistency (>0.8121,22) and is sensitive to change from one to three months for mild and moderate strokes, three to six months for moderate strokes. 22 The domains have excellent criterion validity with the Fugl Meyer Assessment, FIM, Barthel Index, and NIHSS. 22
Outcomes were completed utilizing standardized instructions, with the exception that they were completed over video call instead of in person. Study staff shared their screen when parts of the outcomes required the participant view items, such as in the MoCA, or when answer choices needed to be viewed, such as with the SIS. In certain situations, the family member or caregiver was utilized as a proxy for the outcome measures, such as in the motor items for the NIHSS.
Standardized functional outcomes measures were utilized at the discretion of the therapists, however were not completed with all participants. These functional outcome measures were not designed as secondary outcomes for the study, as it was not anticipated that all participants would be appropriate to complete them due to functional level, lack of caregivers, or safety concerns.
Analyses
Descriptive statistics were used to analyze quantitative change in outcome measures. Where appropriate, minimally important clinical differences (MCIDs) were compared to individual change on outcome measures. Qualitative comments were aggregated and content was analyzed for broader themes.
Results
A total of 20 participants were consented and enrolled into the program. Demographics are detailed in Table 1. Nearly all the participants, except two, had an ischemic stroke. Of the 20 participants, the average age was 55 years old, with a range of 39–71. Participants were 50% men and 50% African American. Nearly half began study activities within 30 days of their stroke in the period most receptive to behavioral induced neuroplasticity (Table 1). Details of participation in study activities are displayed in Figure 1. Eight participants received occupational therapy only, three participants received physical therapy only, two participants received speech therapy only, three participants received occupational and physical therapies, and one participant received occupational and speech therapies (Figure 1).

Participant enrollment and retention.
Participant demographics, stroke characteristics, and baseline measures.
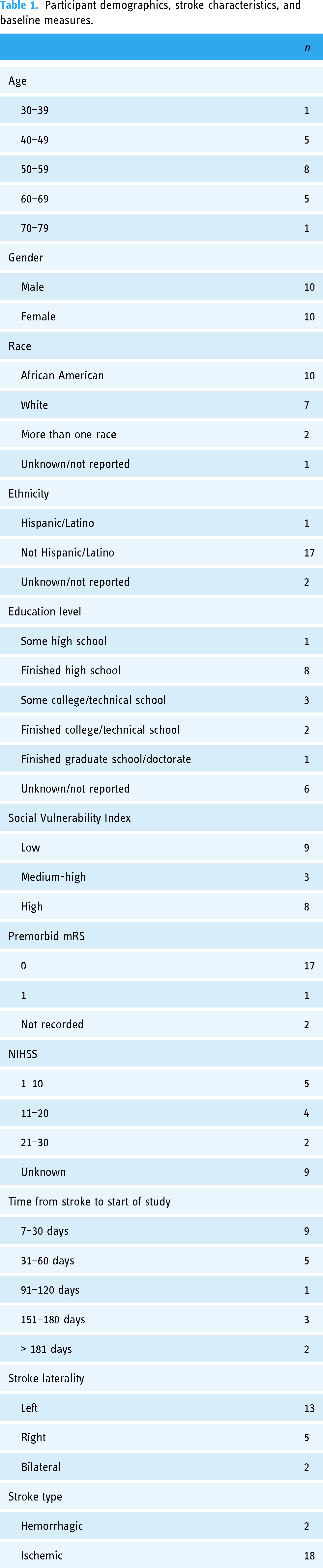
Demographics were reviewed in relation to amount of study activities completed, as seen in Table 2. The majority of participants who completed all study activities were women (n = 8), at least high-school educated (n = 14), and were within 60 days from stroke onset (n = 8). Race and social vulnerability index within this group was equally distributed. Of the eight participants who were consented, but did not complete the study, the majority were African American (n = 5), men (n = 7), and lived in an area with a low social vulnerability index (n = 5). Participants who did not complete the study activities had a higher NIHSS (mean = 15, SD = 8.43) at hospital admission for the stroke than those who completed the study in its entirety. At baseline assessment, participants demonstrated moderate residual stroke severity on the NIHSS (mean = 5.39, SD = 4.02, range 0–14) and self-reported recovering halfway from their stroke on the SIS (mean = 53.4, SD = 28.1, range 0–90). Participants averaged slight to moderate disability on the mRS (mean = 2.8, SD = 1.2, range 1–5) (Table 2).
Demographics, study progress, and attrition.

Ed.: education level; SVI: Social Vulnerability Index; TS: type of stroke; LS: laterality of stroke; NIHSS: National Institute of Health Stroke Scale; TtS: time poststroke to start study activities; M: male; F: female; AA: African American; W: White; UK: unknown; SVI L: low; SVI MH: medium high; SVI H: high; TS I: ischemic; TS H: hemorrhage; LS L: left; LS R: right.
Due to logistical and scheduling issues, five of the 12 participants completed the exit interview, as detailed in Table 3. The five participants reported no prior knowledge of telerehabilitation before initiating study participation, with two reporting they had never used their device's camera prior to study activities. All of the participants reported confidence that their privacy was protected through the telerehabilitation sessions and reported their needs were understood by the therapists. One participant reported that they were not able to understand all of the instructions provided by the therapists during the calls, stating that “(it is) hard to pay attention over video call.” Therapists went back and forth between the computer and the video call; the therapists were not always looking at the camera due to using multiple screens or camera position. The majority of participants did report some issues with technology during their video call (sound/video would not come on, internet went out, issues with video call links, call dropped). Participants were asked to rate their satisfaction out of 10 (extremely satisfied) with the visits; four of the five rated it a 7 or above. One participant did rate their satisfaction at a 2 due to “wish(ing) it would have been in person.” Despite these concerns, all of the participants reported they would recommend telerehabilitation to others, with two saying they would prefer telerehabilitation over in-clinic sessions (Table 3).
Exit interview.

The five exit interviews revealed a preference for in-person therapy, but that telerehabilitation was an important tool for those without access to in-person therapy. One of the participants reported in the exit interview, “if you don't have anything, video is something to do” and rated the telerehabilitation program as “9/10.” Other participants had similar thoughts, with one stating, “…I wish there had been more time.” Another participant stated, “In person would be better, but if it is the only option it is very good.”
Multiple participants reported perceiving that rehabilitation was better in person versus telerehabilitation. These participants tended to be those with higher levels of disability on the mRS, as well as higher reported levels of pain throughout the sessions. The authors agree that those with more impairment and pain may require physical handling interventions that have limited ability to be addressed over video call, requiring the caregiver to be present. Still, those participants that reported strong preference for in-person therapies noted that the telerehabilitation did help them, with one participant demonstrating improvements on the Barthel Index from a 75 to 85, and another a reduction in NIHSS score from 6 to 2. Further details are provided in Table 4.
Comparison of exit interview comments with pre/postquantitative outcomes.

Of the 12 participants that completed the study, nine began at a BI score of > 80 (mean score = 79.16). Sixty-seven percent of the participants (n = 8) demonstrated improvement in Barthel Index score with averages increasing from 73.33 at baseline to 84.38 at end of study (minimal detectable change (MDC) = 4–7 points23,24). The majority of participants (n = 10) demonstrated a reduction in NIHSS scores, averaging 5.6 at baseline to 2.67 at end of study. Half of the participants (n = 6) demonstrated improvements (reduction) in mRS and EQ-5D-5L scores; these six participants improved from a mean of 2.8 to 1.9 and 13.3 to 9.3, respectively. However, four of the 12 participants had worse scores on the EQ-5D-5L quality of life score, with an average change of 3.5 points. Five of the 12 participants demonstrated an increase above the MCID (mean change = 5) on the MoCA (MCID = 1.22–2.15 25 ); three of the 12 participants had a decrease in score that falls within the standard error of measurement (mean change = 1.33; SEM 1.5, MDC ≥ 4 26 ).
On the SIS subjective rating of total recovery, six participants scored themselves with overall improvement in recovery (mean change = 18.2; MCID not reported), three participants scored themselves lower than at baseline (mean change = 5), and one participant scored themselves the same. On the SIS subscales, four participants reported improvement in strength (mean change = 26.6; MCID = 9.2 27 ), five in memory (mean change = 13.9; MCID not reported), seven in emotion (mean change = 18.2; MCID not reported), seven in mobility (mean change = 17.6; MCID = 4.5 29 ) six in communication (mean change = 12.5; MCID not reported), seven in ADL (mean change = 17.9; MCID = 5.9 27 ), eight in social, (mean change = 27.9; MCID not reported), and seven in hand (mean change = 32; MCID = 17.8 27 ).
No serious adverse events occurred related to the study intervention. One participant reported an increase in subjective facial weakness and sensory changes; however, they were medically evaluated at an emergency care facility with no change in medical condition reported. Several minor nonstudy-related adverse events were reported during the intervention, in order of frequency reported: elevated blood pressure, pain, noninjurious falls, spasticity, COVID + (asymptomatic), urinary tract infection, ear infection. Those participants who reported elevated blood pressure had been attempting to manage elevated blood pressure prior to engagement in the study and were encouraged to return to or call their physician. The fall that was reported occurred prior to the initiation of the study intervention. Spasticity and pain reported were present prior to initiation of study intervention and are known to be common symptoms during post stroke recovery.
Discussion
This exploratory pilot study aimed to determine feasibility of a fully online telerehabilitation program that provided stroke specific and home exercise education. Our sample tended to be young, high school educated, from underrepresented groups, and lived in areas with medium to high social vulnerability. Sixty percent of the enrolled participants completed all study activities, which exceeds typical averages of outpatient rehabilitation utilization of 30–40%. 28 Of those that did not complete all study activities, two chose to return to in-person outpatient therapies once clinics opened. Three participants stopped attending study activities and, despite three attempts to contact them via phone and email, no longer returned communications. While we do not know the specific reasons why participants did not return our calls, we speculate that motivation, technological barriers, and disabling symptoms may have contributed. Previous telemedicine and telehealth studies report similar attrition. In a study on telemedicine for diabetes management, Myers et al. report 47% of participants declined participation or stopped participation in telehealth activities early. 29 Further, in a study offering parent training in pediatric primary care, Craig et al. reported 52% attrition by the end of the offered intervention sessions, with 56% completing an end of study survey and 33% completing an end of study interview. 30 Based on the percentage that completed all study activities in VAST-rehab and the lack of study related adverse events, one may conclude that this type of intervention is feasible but may not be considered a priority. It is important to note that those with minimal experience with technology demonstrated greater challenges with access to the video call platform.
While studies indicate equivalence between in-person rehabilitation and telerehabilitation,5–7,31 several participants would have preferred in-person if such services had been available to them. However, in the absence of in-person services, our study adds to the growing literature on the benefits of telerehabilitation, particularly for those who may not have the option due to rural geographical location, transportation, or lack of insurance coverage. In our study, many of the participants were uninsured or underinsured and had no access to outpatient or home health therapies outside of the telerehabilitation offered within the study. Another benefit is access to clinicians to assist with other medical problems. Participants that had unstable hypertension during study enrollment were able to receive support of the therapist to navigate the process of locating accessible medical follow-up as well as education on blood pressure management in regard to secondary stroke prevention.
The VAST-rehab study offered free postacute support and stroke specific education to those who may not have had access to these services otherwise. Participants experienced improvements in functional scores on the Barthel Index, mRS, and SIS subscales of ADL, mobility, and hand function. A reduction in impairment was also noted in the majority of participants, measured by improvements on the NIHSS. In addition, over half of the completing participants demonstrated an improvement in quality of life (50% on EQ-5D-5L and 60% on overall recovery on SIS), thereby aligning with the improvements seen in the emotion and social subscales of the SIS. While less than 50% of the participants improved on the MoCA, of the seven participants who scored below normal limits at baseline (< 26), five of them (71%) demonstrated improvements at the end of the study. These results seem to mirror the self-reported improvements that were seen on the communication and memory subscales of the SIS. The combination of these results supports the hypothesis that individualized telerehabilitation services can be beneficial for stroke survivors.
Upon reviewing the demographics of the participants who scored themselves lower on total recovery on the SIS at end of study than at baseline, three of four of these participants had higher levels of arm impairment, poststroke reports of pain, and subjective reports of hypertonicity at baseline, which continued throughout the study. The consistency and severity of the impairments are likely contributors to their related SIS scores. Ideally, these patients might benefit more from in-person rehabilitation.
Telerehabilitation has a definite role in stroke rehabilitation and recovery.3,5,6,8–10 Based on the results of the VAST-rehab trial, telerehabilitation serves two main purposes: (1) to serve as a bridge for those who may receive outpatient therapy later poststroke to prevent loss of critical recovery time and (2) to be a low-cost option for stroke survivors who never receive any postacute rehabilitation due to lack of funding or access, related to geography, finances, transportation, or other factors.
VAST-rehab is novel in that it offered a fully remote telerehabilitation program, inclusive of outcome measures, that is closer to personalized standard of care than seen in previous telerehabilitation trials.3,8–10 The intervention was individualized to each participant's goals, impairments, activity limitations, and participation limitations. VAST-rehab study activities were completely remote, allowing the participants to complete all aspects of the telerehabilitation service without leaving their homes. The in-home program increases accessibility when compared to previous telerehabilitation trials that involved some component of in-person assessment or equipment set-up.8–10 VAST-rehab outcomes support the idea that there is benefit of a fully remote rehabilitation program in improving functional outcomes of stroke survivors, in turn facilitating accessibility to services for those unable to attend any in-person visits due to transportation, rural location, or other barriers.
Fifty percent of the participants lived within Harris County, where the researchers and primary referral sources are located. Almost 25% of Harris County residents are below 150% of the poverty line and almost 25% of residents are uninsured.13,32 Harris County ranks in the 64th percentile for transportation. 13 These are all limitations to access of medical care resources within this county. 13 Outside of a pandemic, these factors alone can prevent adequate stroke follow-up care. Telerehabilitation provides an opportunity to lessen the gap in follow-up care for this population.
Limitations and next steps
One of the main limitations of the study is the lack of transferability, due to the small sample size, attrition, and heterogeneous nature of the participants’ impairments and time since stroke/phase of recovery. In addition, less than 50% of the completing participants completed the exit interview that provided more information on feasibility of this type of rehabilitation therapy from the participant perspective. These limitations attest to real world challenges in the implementation of telerehabilitation. Future studies within telerehabilitation could include reflective practice within each session to allow for increased opportunities for feedback from participants on barriers and supports for telerehabilitation.
This study was developed during the COVID-19 pandemic as a way to trial a fully remote telerehabilitation option for postacute therapy, due to closures of many outpatient clinics during that time and concern if the risk of exposure was worth having therapy services. As this study continued through the endemic, there was a noted impact in availability of caregivers to assist with video calls due to returning to work and patient interest in telerehabilitation due to returned option of in person therapies. Future studies could explore inclusion criteria of subpopulations that would not require the presence of caregivers. Alternatively, incorporation of community health workers as an extension of the therapist within the home to assist with technology setup could mitigate this barrier to telerehabilitation.
The therapies themselves were not standardized, but were instead tailored to each individual participant. While this can be seen as a limitation, especially when considering reproducibility, it is realistic when compared to the individualized nature of traditional postacute outpatient and home health therapies. Future studies could trial assorted frequency and duration of telerehabilitation to determine if this plays a role in attrition.
To be completely remote, outcome measures were primarily patient report measures. There was limited availability to complete traditional clinical stroke outcome measures due to lack of validation over video call, safety concerns, or needs for equipment. However, standardized functional outcome measures were used as able, if patients were deemed safe to perform, a caregiver appeared able to adequately guard the participant during performance, and/or adequate camera angles allowed sufficient therapist viewing. As discussed in the outcome measures section, the psychometric properties of the self-report assessments demonstrate excellent criterion and internal validity. A vital next step in telerehabilitation research is identification and validation of traditional outcome measures over video call to provide a more thorough assessment of change in impairment and function.
Reported benefits of telerehabilitation have been the reduction of such barriers as lack of access to clinics for services, lack of transportation, funding, and time and energy expenditure of the patient and caregiver. Although VAST-rehab attempted to reduce these barriers (including but not limited to providing occupational, physical, and/or speech therapies for free), attendance was limited, with cancelations due to other appointments, work or other demands of caregiver, stroke survivor “not feeling well,” as well as uncontrolled blood pressure or other symptoms requiring immediate consultation with study physician, or “no shows” to appointments. Study coordinators and therapists maintained frequent contact with participants and caregivers via preferred communication methods throughout the study to mitigate attrition. However, some situations, such as caregivers returning to work or a general feeling of being overwhelmed, were unavoidable within the scope of this study. Future research should include caregivers and/or family members within feasibility and usability assessment. A comparison of attrition within telerehabilitation and home-based therapies is warranted. Further analysis of cost-benefit should occur.
Conclusions
VAST-rehab showed that telerehabilitation is a safe and viable option for stroke survivors with mild to moderate impairments, caregiver support, and access to a device with a camera who may not have the option to complete in-person postacute therapies. The majority of participants in this study demonstrated improvements in functional level on the Barthel Index and a reduction in impairment on the NIHSS, while half of the participants demonstrated reduced impairment on the mRS and an improvement in quality of life on the EQ-5D-5L. Almost half of the participants demonstrated an improvement in cognition on the MoCA. Future studies are planned to address higher intensity telerehabilitation serving the uninsured, underinsured, and populations in rural areas without convenient access to neuro-specialty rehabilitation services to determine best practice to better serve this subset of stroke survivors.
Footnotes
Acknowledgments
The authors would like to acknowledge the hard work of the research team to manage the logistics and implementation of this study, as well as all of the participants for their engagement during the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
The corresponding author (E.A.S.) made significant contributions to data curation, analysis, investigation, project administration, writing, and editing of the manuscript. N.M. contributed to writing of the original manuscript draft. C.P.D. contributed to data curation, resources, writing, and editing of the manuscript. L.R. assisted with reviewing and editing the manuscript. A.S. and A.S.R. contributed to data curation, reviewing and editing the manuscript. H.S. completed data curation, project administration, and assisted with resources during the study. S.M. assisted with data curation. M.E.R. contributed with idea conceptualization, investigation, data curation, reviewing, and editing the manuscript. S.I.S. contributed to conceptualization, investigation, methodology, reviewing, and editing the manuscript. All authors were agreeable to submission of this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
