Abstract
Context
Several options are available for the care of neurological conditions including care delivered in rehabilitation centres, at home or remotely. While economic studies are available comparing centres and homes, very little economic data relates to mobile teams comparing face-to-face and remote care.
Objective
To conduct an economic study comparing face-to-face care at home and care delivered remotely (tele-rehabilitation).
Method
A randomised clinical study with two groups; a control group receiving home care and an experimental group receiving tele-rehabilitation. The primary outcome measure was the ICER (Incremental Cost Effectiveness Ratio).
Participants
Patients with severe neurological disabilities
Results
80 patients were enrolled in the study; 77 were analysed to calculate the ICER, which was positive and located in the SW quadrant. A bootstrap with 1000 replications was positioned at 72.8% in the SW quadrant.
Conclusion
Tele-rehabilitation is an acceptable alternative to the management of neurological patients at home. In the mildest cases, remote-rehabilitation may even be dominant. More extensive studies are needed to specify the indications.
Keywords
Introduction
Access to care for people with disabilities, especially those with neurological disabilities, is a national concern in France and a core part of our health policies, as highlighted in the 2014–2019 neurodegenerative diseases plan. 1 For this population, the failure to receive routine rehabilitation care has potential implications for disease progression as well as functional deterioration and psychological distress. 2 In such patients, the negative effects of social isolation may be even greater, as they require regular follow-up to minimise the impact of the disease. 3 The Saint–Hélier Physical Medicine and Rehabilitation Unit has extensive expertise in the area of neurological rehabilitation and caring for patients with traumatic brain injuries and chronic neurodegenerative conditions (MS, ALS, Parkinson's Disease, etc.). Since 2012, it has expanded its expertise to include a mobile rehabilitation and reintegration team (EMR2), which currently cares for over 200 patients with neurological conditions each year. Its primary objective is to respond to the need to provide flexible care to patients with neurological conditions in the Health Region No. 5 in Brittany, France. Following the increase in this kind of work since 2018, we have undertaken an analysis of telemedicine and its ability to meet the demand and requirement for permanent performance.4–8 Our analysis on the ease of providing access to care for people with disabilities proved to be a major challenge during the Covid-19 health crisis.9,10 Tele-rehabilitation now represents a major challenge for the healthcare system.11,12 Telemedicine was seen to offer an ideal solution for managing transmissible diseases as it reduces person-to-person contact 13 and allows therapists to (1) maintain continuity of care by educating patients through remote consultations directly in their own environment, (2) conduct a physical evaluation and plan a targeted therapeutic exercise programme, and (3) monitor the progress of patients by providing ongoing feedback and follow-up.14–16
The question of the effectiveness of tele-rehabilitation was therefore raised very early on and numerous studies have tackled this issue.17–20 In the area of neurological rehabilitation, most studies report no difference between face-to-face and tele-rehabilitation in terms of balance, functionality and quality of life. 21 A meta-analysis revealed a statistically significant but negligible effect size in six reviews in favour of tele-rehabilitation. 22
There are very few studies from a cost-effectiveness-ratio perspective. Lloréns reported no difference in the ratio between face-to-face and remote rehabilitation. 23 Other studies have shown that the use of this type of care could provide expert follow-up at lower cost, as close as possible to patients and the place where they live.24–26 In France, the medical and economic impact has yet to be assessed.
A cost effectiveness analysis (CEA) is an economic analysis method that compares the expenditure and outcomes of two or more strategies for performing the same task. In the past few years it has been commonly used in health sciences and, more recently, even more widely as a result of the importance of cost analyses in a context of seeking comparative effectiveness as well as in connection to healthcare reform. Statistical methods for CEA have been developed and a measurement called the Incremental Cost-Effectiveness Ratio (ICER) has been widely adopted by researchers and decision-makers. 27 In view of the fairly recent literature and to help guide our use of digital technology in practices within the mobile team, we have decided to conduct an economic study of the aforementioned schemes.
The primary outcome of the study was to assess the benefit/cost ratio and the medical/economic impact of telemedicine care compared to usual follow-up by a mobile team in patients with a severe traumatic or degenerative neurological disability affecting their autonomy. Secondary outcome measures were to assess telemedicine tools, compare patient quality of life, anxiety, depression and pain in patients receiving care under the two schemes and, finally, assess carer involvement in daily life at the start and end of the care.
Material and methods
Design
This was a controlled randomised study comparing telemedicine follow-up and follow-up by a mobile rehabilitation/reinsertion mobile team in patients with a severe neurological disability.
Participants
Selection criteria were as follows: (1) patients over the age of 18 years having volunteered to participate in the study and whose primary caregiver had volunteered; (2) patients with a severe neurological disability following a neurological injury of traumatic, vascular or degenerative origin; (3) patients requiring a PRM [Physical Rehabilitation Medicine] expert (rehabilitation, technical and human means of compensation, specific treatments, etc.) and/or expert in pain management; (4) patients with reduced access to care (due to difficulties travelling, distance from expert centres, frailty, etc.) and requiring in-home professional care; (5) patients requiring coordinated care from health and medico-social professionals; (6) patients living in Health Region 5 in Brittany (France); (7) patients able to understand spoken and/or written French.
Exclusion criteria were: (1) patients with impaired understanding rendering the informed consent impossible with the refusal of the guardian or subject to a legal protection measure; (2) patients unable to use a digital tablet or absence of a person able to assist with the use of a tablet; (3) patients with no access to an internet connection (dead zone); (4) patients with a short-term vital prognosis (<3 months).
Participants have to provide written informed consent for participation in the study.
Recruitment method
Recruitment was based on referrals from general practitioners, nursing homes or hospitals. Each request was first analysed in terms of feasibility (travel distance and request compatible with the skills of the teams).
Interventions
Control group EMR2
The EMR2 operational team included PRM doctors, a psychologist, occupational therapists and a social worker. Care varied according to the request and needs identified during the first meeting. The aim of the medical intervention was to assess the person in their environment, determine with them and their family the objectives of care during the support provided by the EMR2, establish a link with medical points of contact and coordinate the actions of the EMR2. The psychologist performed an initial psychological assessment of the person and/or their carer responsible for the care of their relative, if necessary, and then proposed and coordinated a handover according to the needs identified for the patient and/or for the main carer. The social worker acted as the link between the patients, their carers and the various parties involved. The occupational therapist coordinated the testing and validation of technical aids, chair/bed positioning, and coordinated and supported the various parties intervening in the home.
TELEMED experimental group
The medical intervention and assessment at home was identical to that of the control group. However, patients included in the experimental group were followed up via teleconsultation by the usual professionals, i.e., psychologist, social worker and occupational therapist. Home visits only occurred in emergency cases requiring an in-situ intervention. A connection kit was given to the patient. The kit included a digital tablet pre-configured for the consultations. We used an approved health data connection platform provided by the Groupement de Coopération Sanitaire e-santé and mandated by the Agence Régionale de Santé Bretagne [Brittany Regional Health Agency]. As consultations were synchronous, no document was stored on the tablet. The internet configuration and network coverage were the ones habitually used by the research participants.
In the two groups, goals were set in collaboration with the patient using Goal Attainment Scaling (GAS). No duration was determined. The goals were determined with the patient and then, when they were attained, a decision to stop was taken during a multidisciplinary meeting, with the agreement of the patient.
Outcome measures
The main outcome measure was a medical-economic analysis based on the calculation of the efficiency criterion of this organisation of care. The cost-effectiveness analysis was based on international recommendations. 28 The QALY effectiveness criterion retained was quality of life as measured by SF36 at the start and end of the care. The costs included staff time, travel, carer hours and, where appropriate, teleconsultation equipment. The cost-effectiveness indicator used was the ICER (Incremental Cost-Effectiveness Ratio) representing the difference in costs and outcomes between the new intervention (TELEMED) and the control (EMR2). The numerator of the cost-effectiveness ratio was the monetary cost of the TELEMED intervention minus the monetary cost of the EMR2. Annual project costs were calculated by converting to scale the costs of the period used for the rollout of the treatment. The denominator was the QALY gained by TELEMED less the QALY gained by EMR2, based on SF36. Bootstrapping (1000 replications) was used for a pairwise comparison of costs and average effects between the TELEMED and EMR2 groups.
The secondary outcome measures were: 1) Goal Attainment Scaling (GAS) and numeric scale of goal (NSG) 2) the Anxiety and Depression score of the HAD scale 3) pain assessed using the visual analogue scale and DN 4) caregiver burden measured by the Zarit Burden Interview 5) participant satisfaction measured by the visual analogue scale (VAS).
The Goal Attainment Scale is an individualised outcome measure involving the selection and scaling of goals to calculate the level of goal attainment of a patient. 29 Each patient will have his or her own outcome measure, but these measures are recorded in a standardised manner. Outcomes considered as ‘successful’ were agreed jointly before the intervention. Each outcome was graded using a 5-point scale:
+2 = much more than expected
+1 = a little more than expected
0 = the patient reached the expected level
−1 = a little less than expected
−2 = much less than expected
The overall score was calculated by incorporating the scores of the objective outcomes into a single aggregate score.
Collection of cost data
Costs are broken down into three parts. The first part is contact with the patient, whether face-to-face, remotely via the telemedicine platform or by telephone. Each time item collected is assessed by cost based on the hourly wage of the intervening professional: doctor, physiotherapist, occupational therapist, psychologist and social worker. The second part includes the travel costs which include the mileage adjusted to the distance travelled, plus an hourly cost of the travelling professional adjusted to their travel time. The third part includes the time spent reporting and drafting records adjusted to the hourly rate of the professional preparing them. Costs are expressed in euros. No adjustment for inflation was made, as wages remained stable over the study period and inflation rates were negligible: 2018 1.85%, 2019 1.11%, 2020 0.45% (source https://www.donneesmondiales.com/europe/france/inflation.php).
Sample size
The number of subjects to be included could not be calculated and was based on the EMR2 activity reports of the last four years with an annual recruitment of 281 requests in 2016, a number that was on the rise. The plan was to include 40 patients in each arm to obtain a total of 80 patients. For 40 patients per group, a sigma level of 0.05 and a power of 0.8, a detectable effect size (Cohen's d) is calculated at 0.64 (r statistic, package pwr v 1.3-0).
Randomisation
Randomisation was carried out using online software (https://www.random.org/lists/) by the research unit of the St Hélier centre, which was external to the investigators.
Allocation
A series of sealed envelopes were used for the allocation according to the order of inclusion. the physician recruits the patients and calls a clinical research associate who indicates the allocation.
Blinding
This was an open-label study and patient assessors were the health professionals involved in the study, who were not blinded. However, the assessment criteria were mainly produced using self-questionnaires. In addition, the assessment of the economic costs was compiled per patient.
Statistical methods
Data were analysed via R. studio V 3.6.3 packages Dampak for the ICER criterion. In the case of missing data, an attribution of statistical data using R package missMDA was envisaged. 30 The calculation was based on Principal Component Analysis (PCA) taking into account both the similarities between individuals and the links between variables. The attributed values will have no effect (i.e., no weight) in the calculation of the dimensions of the factor analysis.
Results
Recruitment period
The recruitment period ran from 11/10/2018 to 22/12/2020. The last assessment was carried out on 24/01/2022.
Flow of participants
Eighty participants were recruited; 40 of whom were allocated to the EMR2 group and 40 to the TELEMED group (Figure 1). Two participants were lost to follow-up and one participant withdrew their consent. Ultimately, 38 participants were analysed in the control group (EMR2) and 39 in the experimental group (TELEMED).

Flow chart.
Initial data
The mean age of participants was 59.37 (SD 16.22) in the EMR2 group and 61.92 (SD 13.10) in the TELEMED group. The gender distribution in the two groups was similar, as was the disease length of time (Table 1).
Baseline demographic and clinical characteristics.

Fcg: family caregiver; Pcg: professionnals caregiver; Amyo lat scle: Amyotrophic lateral sclerosis; PF: physical functioning; RP: role physical; BP: bodily pain; GH: general health; VT: vitality; SF: social functioning; RE: role emotional; MH: mental health; HT: Change in Health. *p value < 0.05.
The majority of participants in both groups were accompanied by a family carer who was present for a similar number of hours each week.
In the EMR2 group, most participants were accompanied by a professional carer. In the TELEMED group, most of the participants were accompanied by a professional carer.
Three dimensions of the SF36 seemed to be higher in the TELEMED group, bodily pain (BP) (p value = 0.017), social functioning (SF) (p value = 0.018) and change in health (HT) (p value = 0.012).
Primary outcome: incremental cost-effectiveness ratio
Management of missing data
The calculation of effect differences was affected by missing data in the SF36 measurement survey. A single missing data item prevented the calculation of a dimension score and therefore a dimension effect difference and then overall effect difference. Data was allocated successfully. The description and allocation results are available in the supplemental material.
ICER calculation
In terms of quality of life (Table 2 ICER), there were variations in gains and decreases across the different dimensions of the SF36. None of the dimension scores passed over the minimum detectable change from baseline to end of treatment in either the control or experimental group (MDC = 12 points for PF, 23 points for RP, 15 points for BP, 18 points for GH, 16 points for VT, 26 points for SF, 28 points for RE, and 24 points for MH). 31 . The highest effect sizes related to the 1) physical role (PR) which worsened in both the EMR2 and TELEMED groups (respectively mean (SD) = −13.6 (28.7); −12.4 (20.7)) 2) social functioning (SF) which improved in both groups (respectively mean (SD) = 13.2 (28.6); 4.86 (20.3)). 3) A significant difference was observed between the two groups on the perception of change in health (HT) in favour of the EMR2 control group compared to TELEMED (respectively mean (SD) = 11.1 (27.8); −8.32 (19.6); p value = 0.0008***).
ICER calculation.

Effect: difference assessment end of treatment_baseline; PF: physical functioning; RP: role physical; BP: bodily pain; GH: general health; VT: vitality; SF: social functioning; RE: role emotional; MH: mental health. HT: Change in Health; Effet Diff: mean of difference between groups; Cost Diff: difference of cost between group; ICER: incremental cost effectiveness ratio; SE: sud est, SW: sud west; NE: nord est; NW: nord west. ***p value < 0.001.
Costs calibrated over one year were almost identical for both types of interventions (p = 0.99) with a high variability in both groups (EMR2 mean = 581.40 SD = 296.13, TELEMED = 583.55, SD = 388.04).
The ICER calculated was positive and close to 0 (ICER observed = 0.0299, ICER bootstrap = 0.0285). The % probability of ICER in the quadrants was 1.0% for the SE quadrant, 1.4% for the NE, 72.8% for the SW and 24.8% for the NW 1.0. 5 (Figure 2).
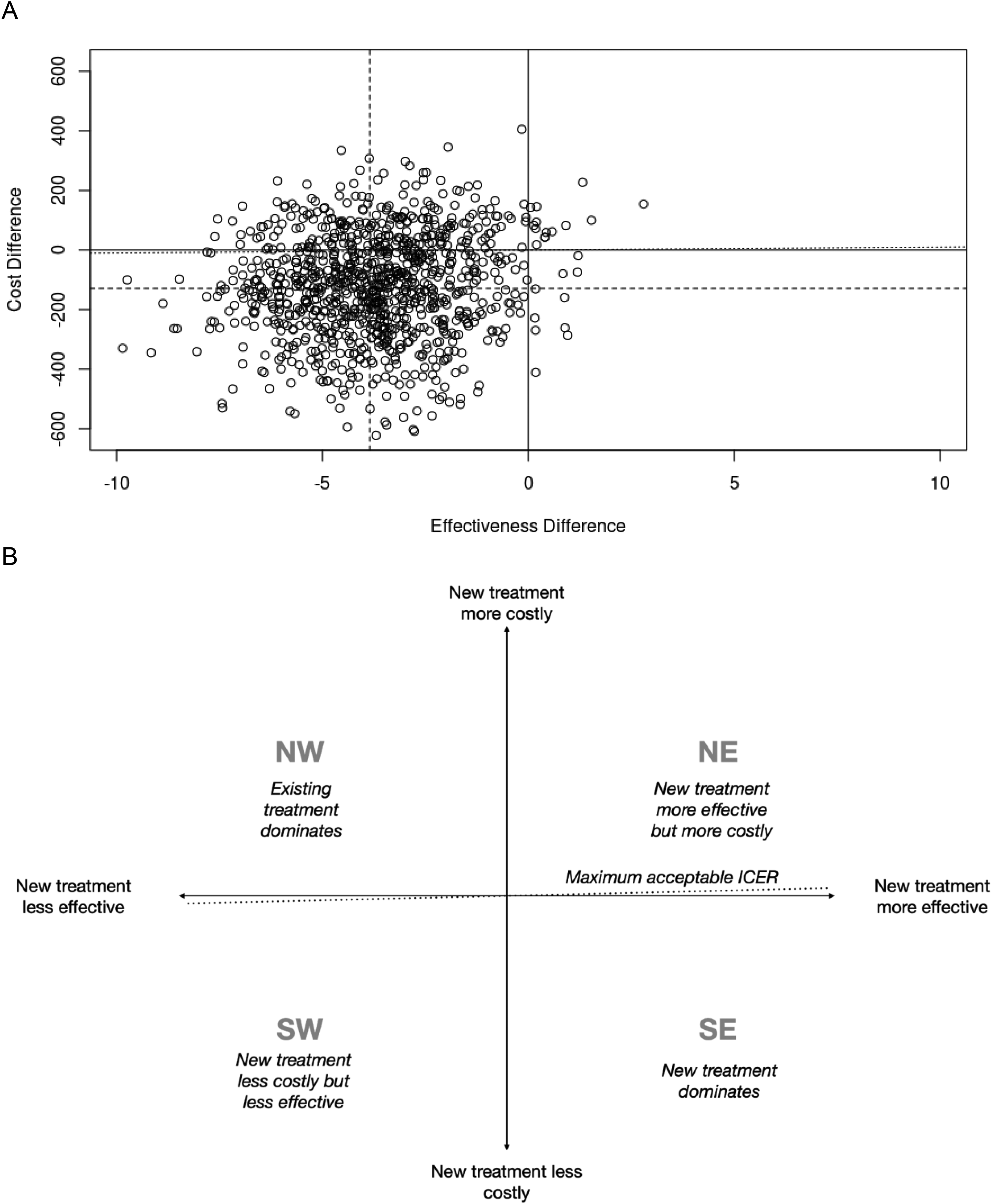
ICER, incremental cost effectiveness ratio; A = Bootstrap 1000 replication of ICER ; B = Key to interpretation of incremental cost effectiveness ratio plots. NW = North West; NE = North East; SW = South West; SE = South East.
Secondary outcomes
Goal attainment scalling (GAS), numeric scale of goal (NSG)
The results show a positive score which reflects the goals attained in both groups with an advantage for the EMR2 group over the TELEMED group, although with a non-statistically significant difference, respectively mean (SD) = 0.53 (1.06); 0.06 (1.07); p value = 0.07 (Table 3).
Secondary outcomes.

GAS: Goal Attainment Scaling; NSG: Numeric Scale of Goal; Hospital Anxiety and Depression scale; ZBI: Zarit Burden Interview; 1.2.3: time of assessment.
No statistically significant difference was observed in the NSG score at the end of the intervention between the EMR2 and TELEMED groups, respectively mean (SD) = 6.23 (2.68); 5.36 (2.92); p value = 0.118.
Satisfaction
The satisfaction scores revealed no difference at any time in terms of the care pathway. A high rate of satisfaction was reported in the two groups at the start of treatment that weakened at the end of treatment, respectively mean (SD) = 5.97 (3.17); 5.59 (2.45), p (value = 0.441.
Hospital anxiety and depression scale
No significant difference in scores was noted, either in terms of the change over time or between the two groups. Anxiety values were in line with the reference values measured in the groups of patients with chronic neurological disease, whereas depression values (mean = 5.7 →7.97) were above the reference values (mean = 5.5).32,33
Zarit Burden Interview
No statistically significant difference was observed, either in terms of change over time or between the EMR2 and TELEMED groups, respectively mean (SD) = 21.82 (22.15); 18.44 (21.15), p value = 0.568. Scores remained below the cut-off score determining a high burden level: cut-off score >25. 34
Analysis and discussion
Interpretation of the ICER
The NW and SE quadrants reported negative ICER values (Figure 2).
These two quadrants are relatively easy to interpret. 35 In the SE quadrant, the new treatment was more effective and saved money compared to the old treatment. When this was the case, the new treatment dominated overs the old treatment. In the NW quadrant, the opposite is observed, and the new treatment was less effective and also increased costs. Here again, it is also relatively easy to take a decision, as the old treatment dominates over the new one, and there is no added value in adopting the new treatment regimen. ICER values in the north-east (NE) and south-west (SW) quadrants were positive. As most of the new treatments are expected to be more effective and more expensive, the literature focuses mainly on the NE quadrant. The bootstrap results show that the probability of ICER in the SW is 72.8% and 24.8% in the NW but with an ICER close to 0 (Table 2). Thus, in this context it is impossible to accept or reject tele-rehabilitation technology on the basis of cost effectiveness. 35 The acceptability of telemedicine for an identical cost is therefore 72.8%.
We confirmed that tele-rehabilitation fulfils the same goals as face-to-face in-home rehabilitation without economic gains. This finding is consistent with the conclusions of other economic studies on rehabilitation. This finding is consistent with recent studies showing the benefits of telerehabilitation for neurological diseases 36 and other economic cost studies in rehabilitation. The study by Housley et al. 37 was the only neurological study that reported significant differences in the cost of tele-rehabilitation and savings per person compared to traditional in-centre rehabilitation. Lloréns et al. 23 also observed that tele-rehabilitation programmes involved lower costs, but did not specify whether the differences were statistically significant. Bendixen et al. 38 examined healthcare costs after 12 months of the tele-rehabilitation intervention and found no significant difference in cost. The study by Rennie 39 showed that if the rehabilitation objectives are correctly met in both groups, the difference in cost is minimal and mainly related to the reduction in travel costs.
It may be that in interventions where physical activities are simpler to implement, such as in pulmonary problems or chronic low back pain, the economic benefit of tele-rehabilitation is more perceptible. The 2021 study by Liu et al. 40 reported that home rehabilitation in obstructive pulmonary disease was dominant over in-patient rehabilitation and tele-rehabilitation was dominant over home rehabilitation. In another economic assessment of an intervention for the treatment of patients with coronary artery disease, Brouwers et al. 41 showed that tele-rehabilitation is likely to be cost-effective compared to a centre-based intervention.
We compared an in-home care system with a system based on tele-rehabilitation. However, it was shown that there is a real economic gain between rehabilitation in a centre and rehabilitation at home. The 2022 study by Candio 42 on a European level showed that the cost effectiveness was largely in favour of in-home rehabilitation in 26 of the 32 countries studied. The EMR2 group is therefore probably already economically viable compared to on-site care. Tele-rehabilitation therefore appears as a possible alternative at no extra costs. It is feasible that in less complex cases, as has been shown in other conditions, that it could become the predominant strategy. Further studies are required to clarify this point.
Interdependence of social factors with anxiety, depression and caregiver burden
Data is telling us something else. The correlation study shows the inverse relationship between social functioning (SF) and anxiety and depression, (respectively r = −0.69; −0.86; p value <0.001). The origin of the disease also has a significant impact on depression, with an increased score in conditions of degenerative origin compared to traumatic or vascular origins (r = 0.25, p value = 0.04).
We fitted a linear model to predict the burden of family caregivers. This model showed that only the effect of depression has a statistically significant and negative effect (p = 0.008). These findings are consistent with other studies that have shown that the cognitive/emotional category is a better predictor of caregiver burden than the motor/functional category.43,44 Similarly, a positive effect of social functioning (SF) was observed in both groups (effect size EMR2 and TELEMED respectively mean (SD) = 13.2 (28.6); 4.86 (20.3)), while physical role (PR) deteriorated (respectively mean (SD) = −13.6 (28.7); −12.4 (20.7)).
Strengths and limitations
Regarding the strengths,
This study represents an early and pioneering investigation into the impact of tele-rehabilitation on a population characterised by individuals with severe neurological disabilities, who often face geographical barriers to accessing healthcare services. Notably, this research predates the onset of the COVID pandemic, underscoring its significance in laying the foundation for subsequent studies in this area.
Importantly, this study stands alone in its focus on mobile team activities that incorporate the utilisation of telemedicine, rendering it distinct in its scope and contribution to the field.
Furthermore, the inclusion of a medical-economic evaluation in this study provides a valuable avenue for effectively targeting and allocating appropriate resources for the rehabilitation and ongoing monitoring of individuals with neurological disabilities. This assessment not only encompasses the clinical aspects but also takes into account the economic considerations, enhancing the efficiency and effectiveness of the interventions. Consequently, the findings of this evaluation have the potential to instigate paradigm shifts in clinical practices, specifically by directing attention to the technological and human resources deployed in the context of mobile teams serving this population.
Regarding the limitations.
The collection of time data is probably incomplete. The professionals involved probably failed to report the exact total amount of the time spent. It is very difficult to monitor the accuracy of time data retrospectively.
The difference in costs can only be established for professional travel when details are supplied of both the cost of the mode of travel and the hourly cost of the professional. It should also be noted that patient interventions are not always compatible with telemedicine, in terms of spatial layout or wheelchair adjustment. Such interventions also require considerable travel, which was accounted for in both groups, thus reducing the likelihood of economic gain in the TELEMED group.
Conclusion
Our study showed that for the same cost, telecare is an acceptable alternative in the care of chronic neurological diseases compared to face-to-face care at home. However, the strong relationship measured between the psychological dimensions (HAD), caregiver burden (ZBI) and social functioning (SF) and quality of life suggests that in complex psychological situations, face-to-face care appears to be a more appropriate approach than telecare. Other studies are required to further explore the choice between in-situ and remote interventions.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231191001 - Supplemental material for Cost effectiveness of a telerehabilitation intervention vs home based care for adults with severe neurologic disability: A randomized clinical trial
Supplemental material, sj-docx-1-dhj-10.1177_20552076231191001 for Cost effectiveness of a telerehabilitation intervention vs home based care for adults with severe neurologic disability: A randomized clinical trial by Aurélie Duruflé, Claire Le Meur, Patrice Piette, Bastien Fraudet, Emilie Leblong and Philippe Gallien in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076231191001 - Supplemental material for Cost effectiveness of a telerehabilitation intervention vs home based care for adults with severe neurologic disability: A randomized clinical trial
Supplemental material, sj-docx-2-dhj-10.1177_20552076231191001 for Cost effectiveness of a telerehabilitation intervention vs home based care for adults with severe neurologic disability: A randomized clinical trial by Aurélie Duruflé, Claire Le Meur, Patrice Piette, Bastien Fraudet, Emilie Leblong and Philippe Gallien in DIGITAL HEALTH
Footnotes
Acknowledgements
Thanks to the people involved in the research, occupational therapists, psychologists, social workers for their application to collect data with rigorousness and patience.
Contributionship
AD, ELB and PG designed the study; AD performed the experiments, supervised the work ; CLM and ELB was involved in protocol development and patient recruitment ; BF . helped supervise the project ; PP analysed the data and wrote the paper with input from all authors ; All authors provided critical feedback and helped shape the research, analysis and manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The Ethics Committee (EC) Ouest II Angers, France gave its approval on 18 May 2018.
Funding
This research received funding from Paul Bennetot Foundation under direction of future for applied medical research, project number : AP-FPB-17-006 and Fondation de l'Avenir pour la Recherche Médicale Appliquée.
Guarantor
AD
Protocol and registration
The study was registered in the ClinicalTrials National Library of Medicine on 24 October 2018; registration number NCT03717974.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
