Abstract
Background
Prehabilitation optimizes colorectal cancer patients’ health during the preoperative waiting period, by increasing functional capacity, reducing postoperative complications, and speeding recovery. However, challenges in implementation include patients’ willingness to attend multiple hospital visits, and hospitals needing trained personnel, facilities, and financial resources. An app-based prehabilitation program could address these issues by allowing patients to participate from home with remote support from healthcare professionals.
Objective
To develop and evaluate the feasibility of a digital application to offer multimodal home-based prehabilitation for older patients undergoing surgery for colorectal cancer
Materials and Methods
This single-center prospective cohort study at Northwest Clinics (Alkmaar and Den Helder, The Netherlands) included patients scheduled for elective surgery for the curative treatment of colorectal cancer. The Patient Journey App was used to deliver prehabilitation. The primary outcome was the number of patients who downloaded and activated the app. Secondary outcomes included usage information, videos viewed, questionnaires answered, and signals triggered.
Results
Ninety-seven patients were included in the study (age 72.1 [SD 0.8], 62.9% male). All patients used the app daily for a median of 29 days (IQR 23–28). Exercise videos were viewed most. Ninety-five patients activated daily exercise and protein shake reminders. Patients provided 1367 answers, triggering 79 signals related to smoking cessation, nutrition, and exercise. Response rates to in-app questionnaires were high, up to 90.5%.
Conclusions
Home-based prehabilitation via an app for older colorectal cancer surgery patients is feasible. Given the effectiveness of prehabilitation programs, the scarcity of healthcare professionals, and patients’ reluctance for frequent in-person visits, home-based prehabilitation programs via an app could become a valuable added modality for offering these programs.
Trial Registration
2020.0600 (VU University Medical Center).
Introduction
Prehabilitation is a concept in which the preoperative waiting time is used to optimize patients’ health. 1 Originating in 1946, it regained interest over the last decades, evolving into a four-pillar multimodal approach that improves a patient's physical, nutritional, and mental status, and supports intoxication cessation (e.g. smoking, alcohol). 2 In recent years, prehabilitation programs have demonstrated increased functional capacity, and a trend towards fewer postoperative complications, and early recovery in patients undergoing colorectal cancer (CRC) surgery.3–10 Standard multimodal prehabilitation includes multiple visits to an (oncological) physiotherapist in the hospital or physiotherapist practice, where patients receive supervised (high intensity) physiotherapy training, dietary advice, smoking, and/or drinking cessation support, and mental support.
Despite the positive results, the implementation of prehabilitation comes with multiple challenges for both patients and hospitals. On the patient side, there exists a dual imperative: the logistical feasibility and the willingness to undergo multiple visits per week, sustained over several consecutive weeks. This is especially true for the most vulnerable patients (e.g. frail, comorbidity, older age), who could benefit the most from such a program.3,11,12 On the hospital side, trained personnel, and adequate facilities need to be available to support these time-intensive programs. Moreover, a financial hurdle looms, as supervised prehabilitation is costly, and reimbursement is often not (fully) regulated. These thresholds might lead to postponing or even aborting the implementation of prehabilitation.

An overview of the elements of home-based prehabilitation.
Given the current state of digital care initiatives to remotely educate and monitor various patient populations,13–18 app-based prehabilitation could be a promising solution in addition to regular prehabilitation programs. An app would allow patients to participate in prehabilitation from the comfort of their own home, while providing them with all the necessary information and activities and being remotely monitored and supported by a healthcare professional (Figure 1).
Objectives
The aim of the study was to develop and evaluate feasibility of a digital application to offer multimodal home-based prehabilitation for older patients undergoing surgery for CRC.
Materials and methods
Study design
This single-center prospective cohort study was performed in the surgical department of the non-academic teaching hospital Northwest Clinics (Alkmaar and Den Helder, The Netherlands). After the development of the app was completed, patients scheduled for elective, curative CRC surgery were invited to participate. Usage statistics were collected during the 4-week preoperative time window in which prehabilitation was offered. One-year follow-up by questionnaires was collected in the app. No changes were made to the design after the study was commenced. We followed the STROBE guidelines for reporting data in a cohort study. 19
Informed consent and ethical considerations
This study was evaluated by the Medical Ethics Committee of VU University Medical Center, who established that the Medical Research Involving Human Subjects Act (WMO) was not applicable (reference number: 2020.0600). Consent was gained at the outpatient clinic via the colorectal surgeon who invited patients to participate in the study. Patients willing to participate received all the necessary study information. Patients were offered several days to reflect on the information. In case of any questions, patients could contact the surgery outpatient clinic (surgeon, resident, case manager). When willing to participate, patients gave written consent, after which download instructions were provided via email. In addition, patients were offered a 15-min session with the case manager to install and activate the app. Clinic staff received a 30-min online training session prior to the study. In addition, they received short videos on how to include new patients and look up data and signals from existing patients.
Participants
Patients scheduled for elective, curative CRC surgery, aged 60 years and above were eligible for inclusion. Additionally, participants were required to be fluent in Dutch and possession of an email address. A smartphone or tablet was required to use the app. Patients who did not have such a smart device were offered one by the hospital. Exclusion criteria consisted of a cognitive disorder (e.g. severe dementia, Alzheimer's), CRC treated via trans-anal endoscopic microsurgery (TEM), paraplegia (inability to voluntarily move the lower parts of the body), or chronic renal failure stage 3 or higher. Patients had to be (medically) able to participate for at least 3 weeks preoperatively.
Home-based prehabilitation
The app was based on the initial Fit4SurgeryTV pilot study, which demonstrated that the use of technology to support elderly patients undergoing CRC surgery is feasible. 20 In this study, the app was further improved to meet multimodal prehabilitation standards, including exercise (video) instructions, nutrition advice, mental support, and intoxication cessation. 21 All content in the app was developed specifically for this study and was derived from the existing, supervised prehabilitation program that was already offered by the hospital. Furthermore, data collection was added to perform remote patient monitoring, assess therapy compliance, and for research questionnaires. Information about polypharmacy (the chronic use of five or more medications) and laboratory findings (iron deficiency) was not available in the app. In addition to information about prehabilitation's main pillars, the app provided general education about the disease and the preparations for the surgery as well (e.g. fasting, what to bring, when to contact the hospital).
Digital care platform
The Patient Journey App (Interactive Studios, ‘s-Hertogenbosch, The Netherlands) platform was selected for this study due to its established presence and proven reliability in the Dutch healthcare market since 2015. The platform has undergone rigorous scientific validation, demonstrating its effectiveness in delivering tailored healthcare interventions. Additionally, the PJA adheres to all necessary privacy and security controls, ensuring compliance with stringent healthcare regulations and safeguarding patient data. After downloading the app and accessing it via a personal code and date of birth, patients were directed to their personal prehabilitation timeline. The app's timeline provided patients with timely information during the program. In addition to the timeline, the app offered an information library, a page with contact details, and a profile section (Figure 2). The information in the app was generic to all patients, but the availability and timing of the information were automatically adjusted to the start date of the app and the patient's date of surgery. Depending on the patient's physical fitness at baseline, defined by a physical exam, an exercise program was prescribed and adjusted during the consecutive weeks based on exertion levels measured by the Borg CR10 scale in the app. 21 Two main exercise programs were developed, consisting of a bodyweight program and a resistance band program. The latter could be adjusted by the use of heavier bands (Bemaxx, Wien, Austria) if the target exertion was not reached.
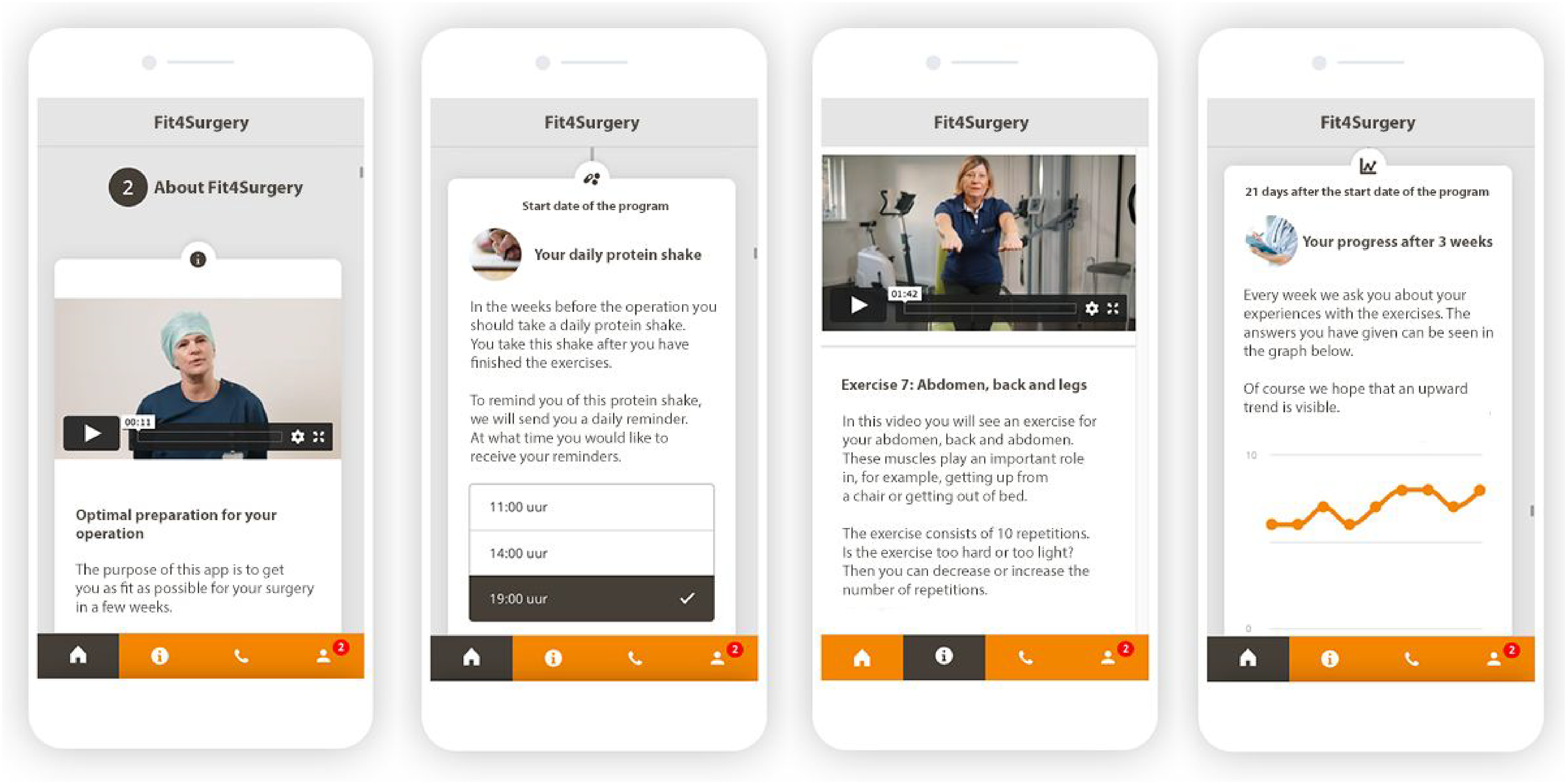
Examples of the interactive app used as an intervention, translated from Dutch (language used in the study) to English. From left to right: video with general information on prehabilitation, setting daily reminders for protein shakes, video with bodyweight exercises, personal progress tracker.
Push notifications were used to actively alert patients about information being available. The timing of the push notifications was configured per information item (e.g. information about protein-rich food on day 2 at 11:00, a motivational speech for the start of week 2 on day 8 at 10:00, a recipe for a protein-rich meal on day 12 at 10:00). In addition, patients could set their own reminder schedule for their daily exercise program and protein shake. The app's content (text, photos, and video) was developed specifically for this study, in close collaboration with surgeons (specialized) nurses, a specialized oncologic physiotherapist, and dieticians (Table 1). Patients and communication professionals were involved to assess the readability and understandability of the app. All information in the app was presented in Dutch.
Overview of app-subjects and health care professionals involved.

Patients were enabled to use their personal app code on multiple devices, allowing them to use the app on their smartphone and/or tablet, or to involve their partner and/or informal caregiver(s) in the program. Each personal app code could be used on a maximum of five devices.
In addition to the timely delivery of information, the app was used to collect data, ranging from Patient Reported Outcome Measures (PROMS) to daily monitoring of patients’ well-being and therapy compliance, such as participating in exercises and protein shake intake. For some of the questions on the timeline, an alert was configured when a specific answer was provided (e.g. when a patient reported suffering from nausea or diarrhea). This alert was automatically sent to the case managers at the hospital, who could then process the alert or forward it to another healthcare professional (e.g. surgeon, physiotherapist, dietician).
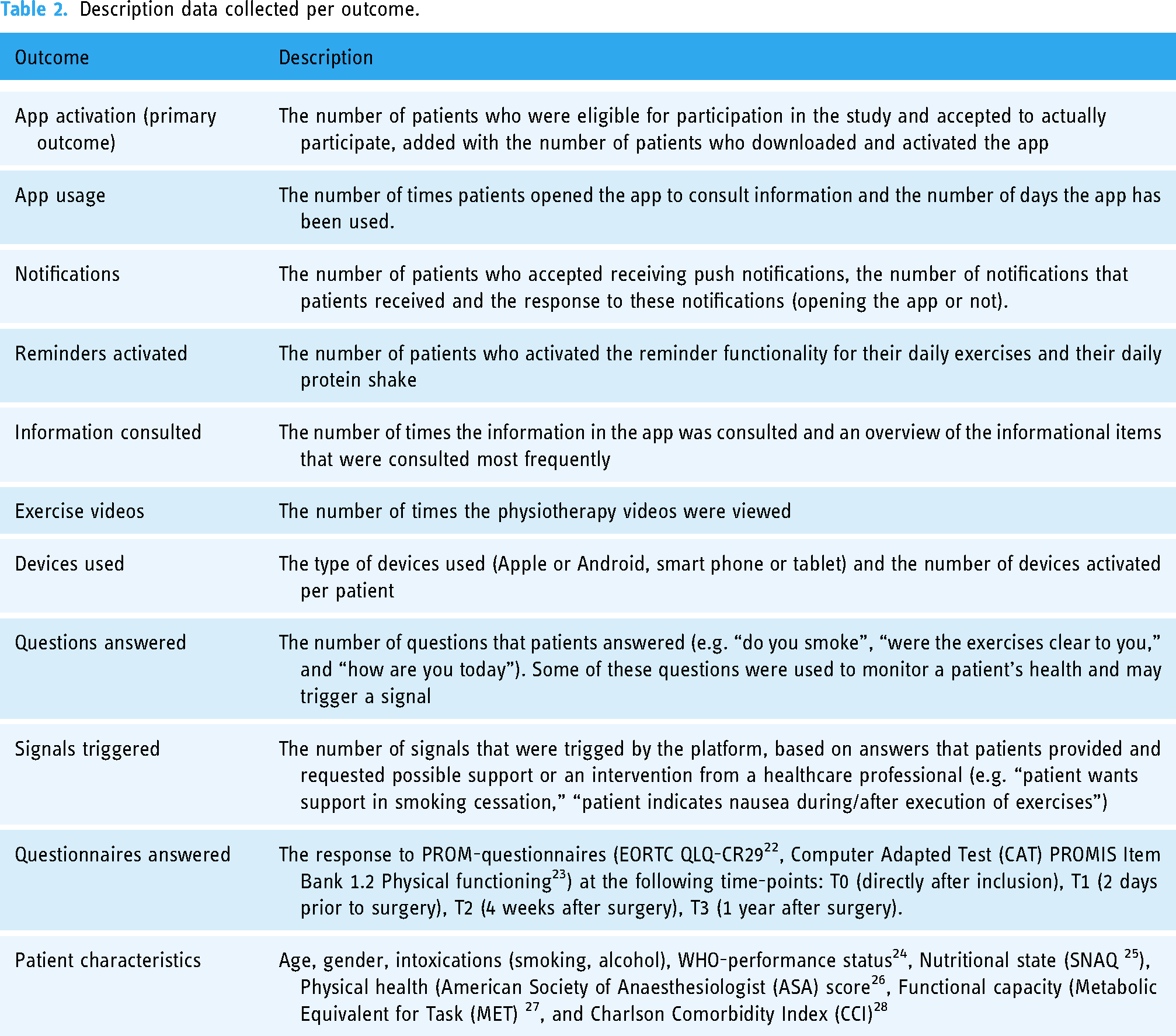
Outcomes and measures
The primary outcome was the number of patients who were eligible for participation in the study and accepted to actually participate, added with the number of patients who downloaded and activated the app. The secondary outcomes were information about usage, notifications, reminders, information consulted, videos watched, devices used, questions answered, questionnaires answered, and signals triggered (Table 2). Validated questionnaires were used to collect outcome data at baseline, 2 days prior to surgery, 4 weeks after surgery, and 1 year after surgery (references in Table 2). Self-developed questionnaires were used to assess patient characteristics such as smoking, alcohol usage, and “how are you today” (see Supplementary file A). Self-developed questions were tested for clarity and interpretation in 5 patients. No changes came from this session. All data was collected in real-time for the duration of the study via the Patient Journey App platform, except for the data about the videos watched, which came from Vimeo (Vimeo Corp.).
Description data collected per outcome.
Sample size
In this study, a consecutive, convenience-based sample of 100 eligible patients was included.
Statistical analyses
Categorical variables are presented as numbers and percentages. Continuous variables are presented as means (with SD). Non-normally distributed data are presented as median values and interquartile ranges. All data were analyzed using IBM SPSS Statistics for Macintosh, version 25.0 (IBM Corp).
Results
Between October 2021 and December 2022, a total of 182 patients underwent elective colorectal surgery. Of these patients, 114 met the study's inclusion criteria (Figure 3). After informing patients about the study, 14 patients were not willing to participate. A total of 100 patients were included in the study. During the study, three patients were excluded because of acute surgery due to gastric bleeding (n = 1), conversion to TEM surgery (n = 1), and ongoing blood loss and pain (n = 1). Finally, 97 patients were analyzed in the study.

Patient flow diagram.
Patient characteristics
The mean age was 71.2 years (SD: 0.8) and 62% of participants were male (n = 61). Of patients aged 70 years and older, 11 of 53 patients (20.8%) were screened to be frail. Smoking was reported by 5.2% of participants (n = 5) and drinking alcohol by 61.8% (n = 60). The majority of patients (75.3%, n = 73) were able to perform normal activities (WHO-performance status 0), and 85.6% (n = 83) were able to perform moderate to vigorous activities (METs > 4). Screening of nutritional state revealed normal nutritional status in 79 patients (81.4%) SNAQ. Most patients had severe systemic disease (ASA 3) and a mean Charlson comorbidity index (CCI) of 5.3 (SD 0.23). Predominantly, a colon carcinoma was diagnosed (79.4%, n = 77), and a laparoscopic right colectomy was performed in 45 patients (46.4%). For more data, see Table 3.
Patient characteristics.

Values are n (%) unless otherwise indicated.
ASA: American Society of Anesthesiologists physical classification score; METs: metabolic equivalent for task units.
Primary outcome: app activation
In total, 115 patients were eligible for inclusion, of which 100 patients accepted participation in the study (87%). Finally, 3 patients were excluded due to medical reasons, leaving 97 patients being invited to download the app and activate it via their personal code. Out of these 97 patients, 100% (n = 97) downloaded and activated the app.
Secondary outcomes
App opens, devices used, notifications, reminders
In total, the app was opened 7066 times (M: 73, IQR 25–112 per patient). Patients used the app for approximately 4 weeks (M: 29 days, IQR 23–38). Almost all patients linked a second device to their account (M: 2, IQR 1–3). Ninety-five patients (98%) accepted to receive push notifications, which triggered them to open the app 69% of the time. Reminders for daily exercises were activated by 95 (98%) of the patients. Based on a paper-based 3-day pre-consultation nutritional diary, all patients were advised to take additional protein shakes, leading to 95 (98%) of patients activating the protein shake reminders (Table 4). All patients had a smartphone or tablet, though seven patients used the hospital-provided tablet in addition to their smartphone due to the convenience of the app being pre-installed and the bigger screen size.
Results for app opens, devices, notifications, and reminders.
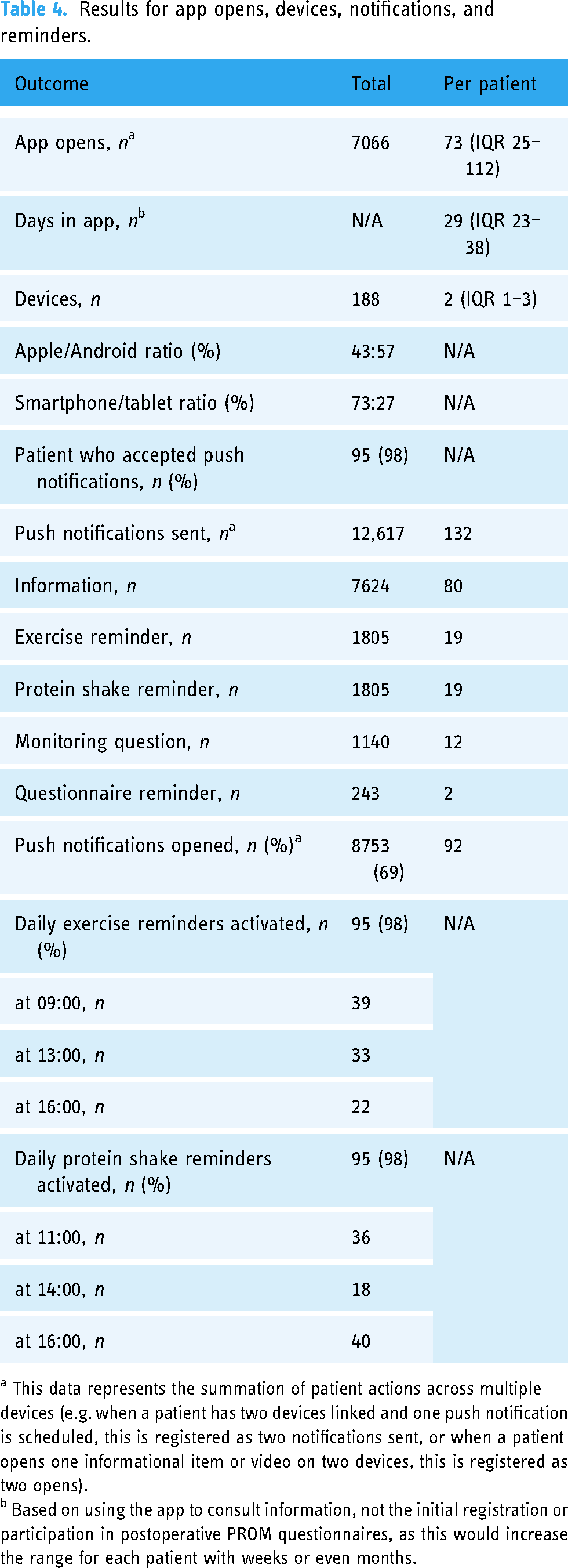
a This data represents the summation of patient actions across multiple devices (e.g. when a patient has two devices linked and one push notification is scheduled, this is registered as two notifications sent, or when a patient opens one informational item or video on two devices, this is registered as two opens).
b Based on using the app to consult information, not the initial registration or participation in postoperative PROM questionnaires, as this would increase the range for each patient with weeks or even months.
Information consulted
Information in the app was consulted 18,484 times (190 times per patient). Exercise videos were viewed 8217 times. The videos were divided into two groups: bodyweight exercises (2539 views) and resistance band exercises (5678 views) (Table 5). Per group, separate videos were available for each exercise. Information about the team, nutritional advice, and recipes for protein-rich meals were consulted frequently as well (Table 6).
Views per physiotherapy exercise video.

Information consulted (top 10).

Questions answered and signals triggered
In total, patients provided 1367 answers to 8 different questions (Table 7). Some questions were asked only once (smoking and drinking status and support, explanation of exercises). Other questions were asked weekly (general well-being) or even multiple times per week (performing the exercises). Based on predefined rules, these answers triggered a total of 79 signals. Signals ranged from patients indicating they were considering smoking or drinking cessation, to trouble performing the exercises, or a low score on general well-being. Hospital staff was notified about a new signal by email and could then determine the appropriate follow-up action (e.g. send an in-app answer to a question, call the patient, ask a physiotherapist or dietician to reach out to the patient).
Questions answered and signals triggered.

a These questions were asked once, at baseline.
b These questions were asked weekly.
c This question was asked 3 times per week, 0–10 score (higher score is better).
Patient-reported outcome measures and adherence data collected
In total, patients were invited to participate in three PROM measurements: baseline (74% response, n = 70), 4 weeks after the surgery (89% response, n = 88), and 1 year after surgery (90.5% response, n = 86). Baseline response was relatively low due to a misconfiguration in the data management, which was adjusted later in the study. Data that were not collected via the app were collected manually by hospital staff. In addition to the PROM measurements, patients were invited to maintain a daily diary, which was completed by 78.4% (n = 76) of patients. Data showed that adherence to taking the daily protein shakes ranged between 98.6% (n = 96) and 100% (n = 97) during the program.
Discussion
The results of our study demonstrate the feasibility of implementing an app to support older patients with a home-based prehabilitation program prior to elective surgery for CRC. Regarding the primary outcome, 100 patients out of 115 eligible patients (87%) were willing to participate in the study, from which 3 were excluded due to acute medical reasons. Out of the 97 patients left, 100% downloaded and activated the app—in a population that is often deemed unsuitable for these kinds of interventions. The group characteristics align closely with those reported in previous studies (refs), with our cohort being, on average, slightly older and comprising a higher proportion of ASA three patients. 7 The information in the app was delivered in a timely manner and consulted daily. Protein shake reminders were used by all patients, and exercise videos were viewed over 8000 times. In addition, the platform demonstrated its usability for remote monitoring and data collection (PROMS and adherence diary) and reported high response rates even at 1-year follow-up.
Providing patients with digitally supported home-based prehabilitation programs allows them to actively participate in the preparation for their surgery, from the comfort of their homes—removing the barriers of having to travel to the hospital or local physiotherapist and/or the related costs. 3 In addition, the hospital staff remains in control of the program's content, is enabled to personalize the content per patient, and collects continuous data for remote monitoring and scientific research. Given the costs of current prehabilitation programs (969–1134 euros per patient 6 ) and the scarcely available medical staff and other resources, the results of our study could facilitate a cost-effective and scalable modality to implement prehabilitation programs further and more rapidly for CRC patients as well as other diagnoses.
A major strength of our study was the holistic approach to the app's content, combining insights from surgeons, physiotherapists (specialized) nurses, dieticians, communication professionals, e-health experts, and, of course, patients. Even more so, the care professionals were all from their treating hospital, which aids in gaining trust in the care team and gives the app a more personal tone. Providing patients with timely information and push notifications to actively alert patients about newly available information has previously demonstrated its effectiveness 18 and resulted in an average use of the app of 73 times per patient over a 29-day course. Videos were viewed over 10,000 times, with about 8000 views coming from the exercise-related videos. A 2023 review demonstrated barriers and facilitators that move patients to participate in a prehabilitation program, including lack of knowledge, lack of physical and exercise resources, and transport and parking limitations. 3 In addition, autonomy was mentioned as one of the key factors to participate in prehabilitation programs. 29 The fact that patients did not have to consume time and financial resources to attend the hospital for their prehabilitation program increases the potential participant rate in future implementation. Furthermore, older patients often have other societal tasks (e.g. caretaker for a partner, watching the grandchildren) that could be continued since the app was more adaptable to a patient's own schedule.
A limitation of our study is the fact that it was performed as a single-center study, which could limit the generalizability. It is noteworthy that the patient demographics observed in our study closely mirror those reported in prior investigations.4,5,7 The consistency in patient characteristics across studies lends support to the generalizability of our findings beyond the confines of a single institution. However, true adherence (viewing an exercise versus executing it (in the right way)) remains challenging and might never compare to a physiotherapist standing next to the patient. In future studies, video consultations or movement sensors could be used to gain more insightful knowledge. Another limitation is the fact that home-based prehabilitation was new to the clinical staff and presented them with a learning curve. The additional time investment, however, was limited. In addition, clinical staff was now facilitated to remotely monitor their patients, which was previously impossible and might now even facilitate more patients to safely participate in home-based prehabilitation programs. Finally, it needs to be taken into account that the high level of activation of the app was because this group of patients actively agreed to participate in the study after being assessed for eligibility criteria. Future research should demonstrate adherence rates when provided to the entire population of CRC surgery patients. In addition, future research should demonstrate the clinical benefits of an app-led, home-based prehabilitation program, as well as its cost-effectiveness for CRC patients and other patient populations. Moreover, additional research is required to further optimize and personalize the content in the app, e.g. a more diverse exercise program, data-driven increase/decrease of exercises, personalized recipes, multi-lingual and/or multi-cultural support, and additional attention for psychosocial wellbeing.
Conclusion
Supporting patients undergoing surgery for CRC with home-based multimodal prehabilitation via an app is feasible from an implementation and usability perspective. 97 patients (85%) used the app, consulted it daily, watched videos with information and exercises, and activated the protein-intake reminders. In addition, the data collected enabled healthcare professionals to remotely monitor their patients’ progress and respond to their questions. Given the growing evidence of prehabilitation programs’ effectiveness, the scarcity of healthcare professionals to supervise these programs in-person, and the unwillingness of some patients to undergo multiple in-person visits per week, home-based prehabilitation programs via an app could become a valuable added modality for offering these programs. Further research is required to evaluate its clinical benefits and cost-effectiveness.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251317760 - Supplemental material for Development and implementation of a home-based prehabilitation app for older patients undergoing elective colorectal cancer surgery. A Prospective Cohort Study
Supplemental material, sj-docx-1-dhj-10.1177_20552076251317760 for Development and implementation of a home-based prehabilitation app for older patients undergoing elective colorectal cancer surgery. A Prospective Cohort Study by Thomas GC Timmers, Lennaert CB Groen, Hermien Schreurs and Emma RJ Bruns in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076251317760 - Supplemental material for Development and implementation of a home-based prehabilitation app for older patients undergoing elective colorectal cancer surgery. A Prospective Cohort Study
Supplemental material, sj-docx-2-dhj-10.1177_20552076251317760 for Development and implementation of a home-based prehabilitation app for older patients undergoing elective colorectal cancer surgery. A Prospective Cohort Study by Thomas GC Timmers, Lennaert CB Groen, Hermien Schreurs and Emma RJ Bruns in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to thank all the healthcare professionals from the Northwest Clinics in Alkmaar for their unlimited time and energy to turn this project from an idea into this manuscript. Furthermore, they would like to thank the team of Interactive Studios for creating the app. Finally, they would like to thank all patients who were willing to participate in the study.
Contributorship
Conceptualization: TGCT, ERJB, and HS. Methodology: TGCT, ERJB, and HS. Formal analysis: TGCT and LCBG. Investigation: LCBG and ERJB. Data curation: LCBG. Resources: HS, LCBB, and ERJB. Visualization: TGCT and LCBG. Writing—original draft preparation: TGCT and LCBG. Writing—review and editing: TGCT, LCBG, HS, and ERJB.
Data availability
The data that support the findings of this study are available from the corresponding author, [TGCT], upon reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The principal investigator, Thomas Timmers, is one of the cofounders of Interactive Studios. Interactive Studios is the company that developed the app used in this study. Interactive Studios offered the app used in this study free of charge. The coauthors declare that the research was conducted in the absence of any other commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This study was evaluated by the Medical Ethics Committee of VU University Medical Center, who established that the Medical Research Involving Human Subjects Act (WMO) was not applicable (reference number: 2020.0600).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Consent was gained at the outpatient clinic via the colorectal surgeon who invited patients to participate in the study. Patients willing to participate received all the necessary study information. Patients were offered several days to reflect on the information. In case of any questions, patients could contact the surgery outpatient clinic (surgeon, resident, case manager). When willing to participate, patients gave written consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
