Abstract
Background
Personal health records (PHRs) or patient portals have been on the healthcare policy agenda for many countries as a promising mechanism to support patient-centred healthcare by making medical records accessible to patients and those assisting patients in health self-management. Studies on clinical outcome have been inconsistent. To help us to understand why, we propose to look at measures that precede clinical outcome, specifically patient engagement and activation. Patient activation describes the knowledge, skills and confidence a person has in managing their own health and healthcare.
Objective
To systematically review the current evidence on the impact of PHRs on patient activation.
Methods
A literature search was conducted for randomised controlled trials and quasi-experimental studies published up to January 2024 across EMBASE, PsycINFO, CINAHL and PubMed. Publications were included in the study if they examined any association between PHR use and activation.
Results
The search initially produced 3062 papers for review, of which 88 full-text articles were screened for eligibility. Two reviewers assessed 22 of these articles, and 8 papers were identified as meeting the selection criteria. Among these, seven studies found no statistically significant differences in activation. However, one study reported a significantly greater effect than the others. Data from seven randomised controlled trials and quasi-experimental studies examining the effects of PHRs on patient activation and similar measures were extracted for meta-analysis. Overall, the use of PHRs was associated with a 0.41 standardised mean difference increase in activation (95% confidence interval 0.31–0.51). There was a high degree of heterogeneity (I² = 98%), with one study showing a much larger effect size compared to the rest.
Conclusion
Most studies showed little impact on activation, but one study found a large effect. This study notably offered PHRs combined with health coaching and training in the use of the system to their intervention group, which may indicate an important requirement for how to get the best out of a PHR system.
Keywords
Introduction
Personal health record (PHR) implementation has been on the healthcare policy agenda for many countries. 1 The implementation of PHRs and shared digital health systems that link patient healthcare data across multiple sources has the potential to facilitate significant advancements in healthcare efficiency, quality and performance in countries where these systems are introduced. Shared information can also support clinical research, effective public health planning and the evaluation of healthcare interventions. 2
PHRs have become increasingly popular in recent years with many health systems implementing them to improve patient engagement and self-management. PHR implementation can be either implemented at a national level or at a health system level. Notable examples of health system implementation include MyChart, 3 developed by Epic Systems Corporation and implemented in several health systems across the US, including the Mayor Clinic and Cleveland Clinic; Kaiser Permanente's 4 My Health Manager system; and HealtheVet in the Veteran Health Administration in the US. 5 A number of countries have rolled out PHRs as nationwide initiatives, including Estonia, Denmark and Australia, 6 but there appear to be no published studies that demonstrate the impact of these systems. The UK's NHS has published a number of case studies demonstrating the benefits of PHRs. 7
Existing PHR systematic reviews a number of systematic reviews have been conducted to critically appraise PHR interventions with relevant patient outcomes and other benefits.1–4,15 These studies focused on evaluating PHRs’ effects on clinical outcomes, patient activation or engagement, with most finding limited impact when PHRs were used alone. Reviews that included interventions like coaching or secure messaging noted more positive effects, particularly on engagement and clinical outcomes such as glycaemic control. Studies differed in terms of the populations studied (e.g., general patients vs. chronic illness groups), the metrics evaluated (clinical outcomes vs. patient empowerment) and the functionalities of PHR systems. Overall, heterogeneity in study design and intervention methods made it challenging to draw definitive conclusions about PHR effectiveness.
Davis Giardina et al. 8 reviewed 27 studies (20 were randomised controlled trials [RCTs] and 7 were uncontrolled observational studies), and although glycated haemoglobin A1C (HbA1c) improved overall in 3 RCTs, the difference between the intervention and control groups was significant only in 1 trial. An observational study suggested an association between PHR use and improved laboratory values (glycated HbA1c and low-density lipoprotein cholesterol) and blood pressure (BP) and low-density lipoprotein cholesterol were not significantly different between intervention and control conditions in 1 of the aforementioned RCT. Two additional prospective studies examined the effect of PHR access on BP control in patients with chronic disease and found no impact.
Mold et al. 9 in 2018 reviewed 28 studies (10 using surveys, 5 RCTs, 5 focus groups and interviews, 3 longitudinal cohort studies, 1 quasi-experimental and 1 interpretive review). There was a positive association between the use of PHRs and primary clinical outcome, improved glycaemic control and general care management. A number of studies in this review included PHRs that were used together with online services, such as secure messaging and electronic health reminder letters.
Han et al. 10 reviewed 24 studies (10 RCTs, 7 quasi-experimental studies, 6 cohort studies and 1 mixed method study) and found that the effects of PHRs on clinical outcomes including BP, glucose, cholesterol and weight loss were mixed. For example, of five studies in which BP was included as an outcome, only one found improved BP control. Similarly, less than half of the seven studies including glucose control as an outcome had a significant finding and this was either in a non-controlled setting with no comparison group or only for a short term (6 months). Effects of cholesterol control were also overall insignificant—only one of five studies had significant reduction in low-density lipoprotein.
de Lusignan et al. 11 in 2014 systematically reviewed 143 studies and identified several primary findings related to patients’ online access to their PHRs. Online access to health records can lead to improved patient outcomes such as better medications adherence, increased patient satisfaction and better communication with healthcare providers.
Ammenwerth et al. 12 in 2021 systematically reviewed 10 randomised control studies and cluster-randomised trails and found low quality evidence suggesting no or little effect on HbA1c level, no or little difference in systolic or diastolic BP with the intervention, little or no effect on BMI or weight and no effect on 10-year Framingham risk score.
In summary, evidence suggests that PHR interventions may have little or no effect on clinical outcomes compared with usual care. In order to understand the reasons behind the limited effects of PHRs on patient clinical outcome, it is useful to consider a purported mechanism of action, namely patient activation and engagement.
Healthcare bodies are interested in increasing patient activation. 13 Patient activation is a widely recognised concept that describes the knowledge, skills and confidence a person has in managing their own health and healthcare. The Patient Activation Measure (PAM) is the most commonly used measure of activation. 14 PAM is endorsed by NHS England as a validated measure of the concept. Evidence shows that when people are supported to become more activated, they benefit from better health outcomes, improved experiences of care and fewer unplanned care admissions. 15
According to Fumagalli et al., 16 empowerment represents the possession of conditions that make patients ‘willing and able’ to play an active role in their care. Activation and empowerment interconnect as concepts and relate to an increased ability, motivation and growing patient awareness of having an important role in the management of their own healthcare. 12 The terms ‘patient empowerment’, ‘patient activation’ and ‘patient engagement’ are frequently used in digital health and often interchangeably. Digital health practitioners 17 and researchers 16 have attempted to clarify boundaries, connections and intersection. In the Rislings’ model, 17 empowerment, activation and engagement are three stages in a process of how a user engages with a technology. Empowerment is how much control is put in the hands of the patient. Activation is a measure of how patients are getting through the early days of an intervention. If the two stages have gone well, this will result in user engagement—ongoing, demonstrated interaction with the technology that presumably will result in improved health outcomes.
Using PHRs may help patients take an active role in their healthcare 18 as a result of an improved understanding of their health, related conditions and treatment. 19 However, there is still a lack of understanding about how best to leverage PHRs to engage patients in their own healthcare and what the impact of these tools can be on patient engagement and activation. We seek to review the evidence on whether access to PHRs impacts positively on patient activation by providing better access to one's own health data. This systematic review aims to explore the evidence that supports or contradicts the hypothesis that PHRs increase patient activation.
There are diverse approaches possible to studying digital health interventions. 21 For this review, it was decided to use just RCTs and quasi-experimental studies. RCTs are considered the gold standard for testing causal relationships between an intervention and an outcome. 22 Quasi-experimental studies are useful when RCTs are not feasible or ethical, such as when the intervention is already in place or when it is not possible to randomly assign participants to groups. Despite the limitations of quasi-experimental studies, they can still provide valuable insights into the effectiveness of interventions when conducted properly. 23
Methods
Search strategy
A search was conducted in EMBASE, PsycINFO, CINAHL and PubMed (up to December 2021).
The following terms were searched in free text or key words: ((personal* health data) OR (Web-based Personal Consumer Health Record*) OR (personally controlled health record*) OR (personal health record*) OR (PCHR*) OR (PHR) OR (PHRs) OR (patient portal)) AND ((patient engagement) OR (patient activation) OR (patient empowerment)). These terms were chosen as we have noted patient portal synonyms to describe ways to give patient access to their own health data. We picked generic language for outcomes. Although PAM is commonly used, we also recognised that the terms patient activation can be used without necessarily applying a PAM measure tool.
Eligibility and inclusion
Inclusion criteria were studies with an element of primary data collection; using RCT and quasi-experimental study designs; and an outcome of patient activation, including PAM or similar.
Full-text articles were screened for eligibility and rejected papers were randomly crossed-checked. Possible papers for inclusion were reviewed by two reviewers to identify those fully meeting selection criteria. Any discrepancies were resolved through discussion.
Data extracted included author, year, country of study, study design, study duration, population, selection criteria, sample size, age in years, intervention type, description of intervention, outcome measures, control group treatment and effect size/impact/findings.
Quality appraisal
Data quality was appraised using the Joanna Briggs Institute (JBI) appraisal checklists for RCTs 24 and quasi-experimental studies. 25 The JBI checklists composed of 13 questions for RCTs and 9 for quasi-experimental studies. Studies were rated 1 if they included a component of the quality rating and 0 if they did not identify and each study total score is calculated (Tables 1 and 2).
RCTs: JBI checklist and scores of included studies.
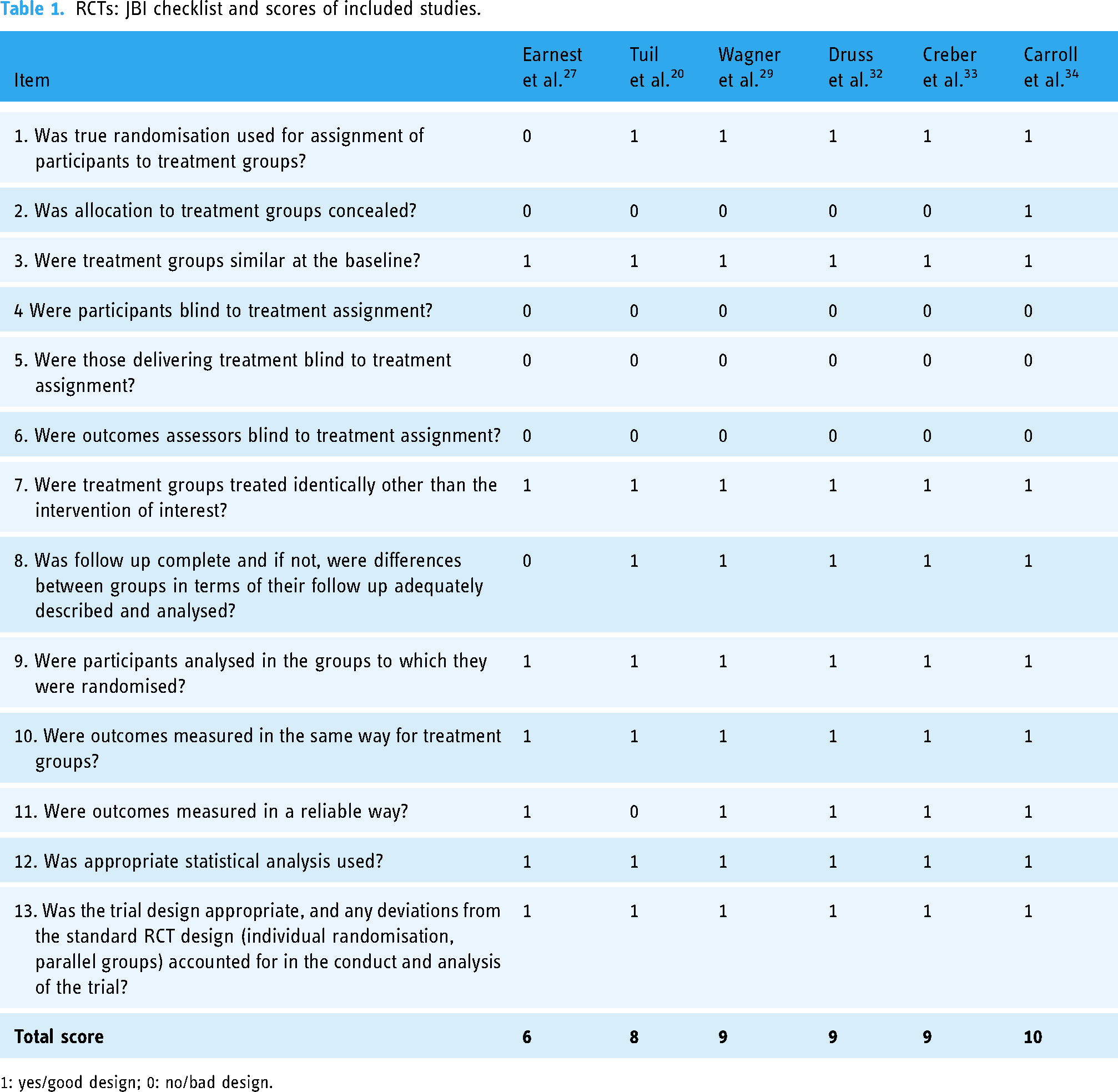
1: yes/good design; 0: no/bad design.
Quasi-experimental studies: JBI checklist and scores of included studies.

1: yes/good design; 0: no/bad design.
Each article was reviewed by one reviewer (IO) and checked by a second reviewer (HP). No articles were excluded on the basis of their review and score.
Screening
Studies were selected in line with the process shown in Figure 1.

Flow diagram showing article selection strategy in PRISMA format. 26
Results
Description of included studies
The search produced 3062 papers for initial review. 88 full texts were read. Eight studies met the inclusion criteria: Earnest et al. 27 ; Tuil et al. 28 ; Wagner et al. 29 ; Riippa et al. 30 ; O’Leary et al. 31 ; Druss et al. 32 ; Creber et al. 33 ; and Carroll et al.. 34 These are summarised below and in Table 3.
Characteristics of included studies.

A final total of 8 papers were subject to full data extraction. Through quality appraisal, we identified strengths and weaknesses in studies’ methodology, but the overall analysis and conclusions have not been affected.
Six of the eight included studies were based in the US and remaining two in Europe (the Netherlands and Finland).
Of the US studies, only two were based in hospitals and the remainder in primary care and specialist clinics. Patients across eight studies had long-term health conditions ranging from serious mental health disorder to hypertension, long-term heart condition, HIV and undergoing IVF. Only one study 31 had a mixed range of patient conditions from acute, such as injury and poisoning, to a wide variety of chronic conditions.
Carroll et al. found a large, significant effect that stands out compared to the other studies. Six of the other seven studies did not find any statistically significant differences in activation and patient empowerment measures. Ernst et al., Druss et al. and Creber et al. conclude PHRs are beneficial, but based on other outcome measures in their studies. Riipa et al. and Tuil et al. focused on activation or empowerment and report non-significant results but still offered optimistic conclusions in favour of PHRs. In contrast, O’Leary et al. conclude that PHRs do not make a difference to activation. Wagner et al., with a non-significant result on patient activation, are equally pessimistic, although they do report a significant improvement on a separate patient empowerment score.
Earnest et al. 27 evaluated the experiences of patients and physicians using System Providing Patients Access to Records Online (SPPARO) in the US. No statistically significant differences were found between groups, but the Patient Empowerment Score declined for patients as a whole.
Tuil et al. 28 evaluated an Internet-based PHR for patients undergoing IVF treatment in the Netherlands. The PHR users were relatively young and well-motivated patients, who expressed the explicit need for Internet-accessible medical records. The authors viewed the concept of patient empowerment as having conflicting definitions and connecting concepts such as self-efficacy, knowledge about disease and treatment and patients’ involvement in decision-making process. They thus used as the main outcome measure a questionnaire that consisted of items about self-efficacy, actual and perceived knowledge, and involvement in the decision process. The study found no statistically significant differences between an experimental and a control group on their various measures of empowerment (ps > 0.4).
Wagner et al. 29 conducted a cluster randomised effectiveness trial with 24 primary care physicians recruited from one Family Medicine and one from Internal Medicine ambulatory clinics at a tertiary academic medical centre in the US. Physicians were randomised into two groups: intervention (PHR) or control (care as usual). This study employed the Centre Corporation proprietary PHR system (IQHealth) deployed under the brand name MyHealthLink. PAM scores were not significantly different between the experimental and control groups (p = 0.23). However, Wagner et al. did show a significant difference on a related outcome, the Patient Empowerment Scale (PES; p = 0.019). The PES assesses patient's perceived risks and benefits of access to their own health information and, similar to PAM, assesses patients’ knowledge about their condition and better understand their doctor's instructions. Few patients provided with a PHR actually used the PHR with any frequency. The authors concluded simply providing a PHR may have limited impact on empowerment and other outcomes without additional education or clinical intervention designed to increase PHR use.
Riippa et al. 30 conducted a study in a Finnish public primary care setting. Study participants were at least 18 years old, with at least two treatable health conditions assessed by a health professional, bank identifiers (i.e., electronic credentials for online authentication provided by their bank), access to the Internet, and were willing and able, both according to themselves and to a healthcare professional, to engage in using the portal. The intervention group ended higher on the 13-item Short Form PAM (PAM-13) than the control group, but the difference was not statistically significant on raw figures (p = 0.9) or when using propensity score matching.
O’Leary et al. 31 conducted a study in a large academic hospital in Chicago, Illinois and found that hospital in-patients given a PHR system (n = 100) scored higher on the PAM-13 compared to a control group (n = 102), but this was not statistically significant on a univariate analysis (p = 0.5), nor when adjusting for age (p = 0.4). This study did not explicitly describe the control group intervention.
Druss et al. 32 evaluated the effect of a PHR on the quality of medical care in a community mental health setting in the US. Although having a PHR resulted in significantly improved quality of medical care, increased use of medical services among patients, there was not significant difference between experimental and control groups.
Creber et al. 35 conducted a three-arm RCT study (usual care; iPad with general internet access; or iPad with access to the personalised inpatient portal) in two cardiac medical–surgical units at an academic medical centre in New York City in 2014–2017. The patient portal contained clinical information from the hospital's EHR and useful information about the patient's care team, a list of administered medications and educational content and functionality to communicate comments and questions to the care team. There were no significant differences in PAM scores among the three groups in unadjusted (p = 0.39) or adjusted (p = 0.42) analyses. Participants in the portal group who used the portal more frequently (two or more total logins) were not found to have higher activation (p = 0.11).
Carroll et al. 34 conducted a pragmatic RCT in eight practices in New York and two in New Jersey serving patients living with HIV (PLWH). The objective of the study was to evaluate the effect of a multimodal self-management program, consisting of access to the URHealth app (electronic PHR [ePHR]) loaded onto an iPod with internet connection, combined with a targeted, peer-led and group-based intervention for PLWH. The intervention consisted of six 90-min group training sessions and a single 20- to 30-min individual pre-visit coaching session. The study primary measure was change in PAM. The study participants were at least 18 years old (mean age 52 in the intervention group and 51 in the control group) and with confirmed HIV status. Control group participants also received the iPod device after their follow-up evaluation was complete. The intervention group (n = 180) ended statistically significantly higher on the PAM-13 than the control group (n = 179), with a 2.8-unit improvement in patient activation. Patient activation improved at 12 months to 73.4 ± 1.13 and 70.5 ± 1.14 in the intervention and usual care groups, respectively (p = 0.027). The intervention was found to have a moderating effect on PAM level. The interaction between the intervention and PAM level was statistically significant among those in the lowest PAM quartile at baseline (p < 0.05). Notably, the intervention was associated with double the odds of improving one PAM level compared with control (odds ratio 1.96; 95% confidence interval 1.16–3.31).
Meta-analysis
A random effects meta-analysis was performed on seven of the included trials (Figure 2). Due to a lack of detail in the Earnest et al. study, data from this paper could not be extracted for the meta-analysis.

Forest plot showing effects of PHRs on patient activation.
The quality assessment found all the studies to be of a reasonable and similar quality. The lowest scoring study was Earnest et al., which already had to be excluded from the meta-analysis. No analyses by quality level were thus conducted.
Wagner et al. 29 give a mean in both groups at the end of study, but not a standard deviation. However, a baseline standard deviation is provided and we used this. The paper also gives a p-value for a comparison at end of study adjusting for background variables, including gender and literacy. This shows less difference than the raw numbers that we used in the meta-analysis. Wagner et al. did show a significant difference on another related outcome, PES.
The overall effect in a random effects meta-analysis is statistically significant (z = 8.12, p < 0.001). Heterogeneity is apparent: I2 = 98%, χ2(6) = 253.9, p < 0.0001 (Table 4). The effect seen in Carroll et al. is not consistent with the other studies, as is apparent from the forest plot (Figure 2).
Meta-analysis of seven studies.
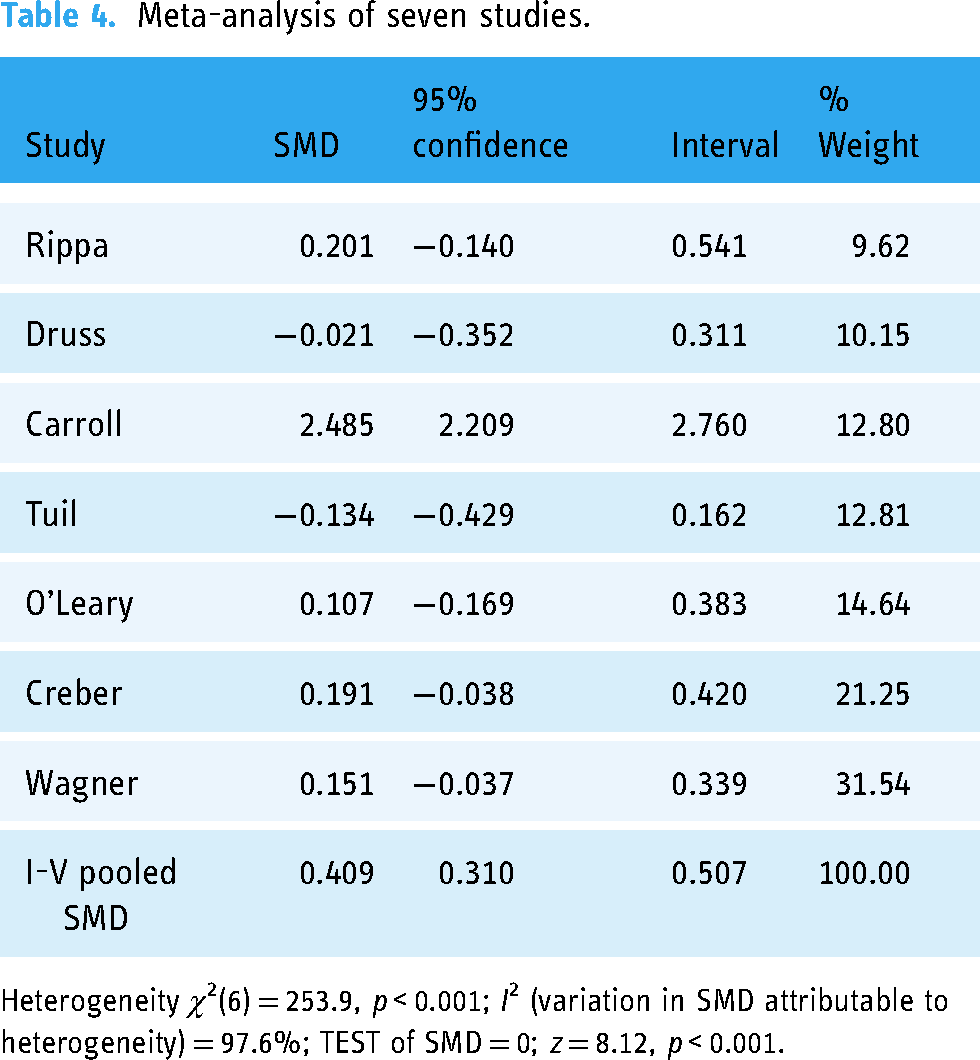
Heterogeneity χ2(6) = 253.9, p < 0.001; I2 (variation in SMD attributable to heterogeneity) = 97.6%; TEST of SMD = 0; z = 8.12, p < 0.001.
In a meta-analysis of six studies excluding Carroll et al., there is no statistically significant difference between intervention groups using a PHR and control groups: z = 1.93, p = 0.054. However, the result does approach statistical significance. There is now no heterogeneity apparent: I2 = 0%, χ2(5) = 4.1, p = 0.5.
Discussion
Principal findings
We found eight studies, six RCTs and two quasi-experimental, directly assessing the effects of PHRs on patient activation and similar measures. The effect seen in the Carroll et al. study was markedly greater than in the others. Across the other seven studies, we failed to find a statistically significant difference in the activation and patient empowerment measures, either individually or when pooled in a meta-analysis, although the pooled result was close to the 5% cut-off. The study by Wagner et al. has the greatest weight (28%) in the meta-analysis. The paper itself concludes a slight positive effect, although our version of the analysis to allow a consistent meta-analysis does not reach statistical significance.
We note that there are substantial differences across all the studies included in the meta-analysis. Studies ranged from publication in 2004 (Tuil et al. and Earnest et al.) to 2019 (Creber et al. and Carroll et al.) and are on different patient populations. There are functional differences in the PHR systems in these studies and in the approach to how the health systems introduced PHRs to patients and engaged them in using PHRs. Although limited information is available on the functional differences between the systems, we note that PHRs in five studies (Riippa et al., Tuil et al., Wagner et al., Carroll et al. and Creber et al.) had access to health data and information as well as the ability to interact with clinicians via some form of messaging system. PHRs in the other two studies (O’Leary et al. and Druss et al.) only contained access to various forms of health data and health-related information.
The Carroll et al. study stands out. Without it, the evidence for an effect by PHRs on activation is very weak. Carroll et al. evaluated the effect of a PHR for people living with HIV. The intervention consisted of an ePHR loaded on an iPod as well as coaching and peer support to increase knowledge and confidence in managing their health and healthcare as well as skills and tools to facilitate self-management. The population in Carroll et al. study also had relatively high levels of baseline PAM and a high level of self-management, which is typical for HIV patients. 36 This study showed a greater effect on patient activation among those with the lowest levels of baseline patient activation, which might partly reflect a ceiling effect.
Limitations
During this systematic review, we found only a fairly small number of studies that met our search criteria that evaluated the impact of PHRs on patient activation or empowerment measures. Apart from Wagner et al., most studies were relatively small in size; a larger number of studies and studies on bigger samples would provide greater statistical power. While Carroll et al. found a large effect, more studies are needed to replicate such findings among non-HIV patients with low income and with low health technology used to assess impact and the cost-effectiveness of multimodal approaches to patient activation. There is also a question on how self-management can be better integrated with standard care such as interactions with clinicians.
When we began this review, our hypothesis was that the use of PHRs leads to higher activation and therefore to better clinical outcome. As the systematic review concluded that the evidence to support this hypothesis was not significant, perhaps future research needs to look at preceding factors to patient activation and empowerment such as patient's engagement, ability and willingness to use PHRS. Below is a proposed logical model that breaks down the process and impact into a smaller stages and maybe helpful to visualise the journey of using PHRs for better health outcomes.

Conclusion
Digitally enabling patients is a primary theme to many healthcare organisations and governmental initiatives. Central to these are patient access and interaction with their health records giving patients control over viewing, modifying and sharing their clinical records, and sharing the information between healthcare professionals, patients and carers.
The Carroll et al. study shows a notable shift in patient activation, and this is the study that pushed hard on patient engagement with the PHR. Literature review has demonstrated that patients’ interest to access and update their own health data is growing but the ability to use patient portals is often influenced by personal factors including age, education level, health literacy and health status. 2 National PHR adoption strategy and healthcare providers’ endorsement appear to be some of the fundamental drivers in PHR adoption. 37 As the patient-facing features of PHR portals are still evolving, redesigned care pathways to incorporate the use of PHR technology, where appropriate, will be fundamental in creating a sustainable environment for patient portals use. Adoption by patients and endorsement by providers will come when existing patient portal features align with patients’ and providers’ information needs and functionality. 38
If patient health records are to become the norm, then there needs to be a better understanding of the factors that contribute to creating value and practical use.30,31 PHRs have the potential to impact on patient activation, but the effect does not appear to be consistent. How the PHR is introduced may be important to realising that effect. In Carroll et al. study, we observed that the PHR combined with coaching and training created a more significant impact on PAM but there may be also an opportunity to personalise the patient experience through better design of a PHR system.
Future research in these areas should focus on interventions that target and measure actual PHR usage, engagement in care and what modifiable factors contribute to meaningful PHR use by patients. This can give insights to how best improve PHR system design, better introduce systems to patients and support them in making the best use of PHRs for their healthcare. Further investigation is needed to see how certain approaches in personalisation could have a positive effect.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251315295 - Supplemental material for A systematic review of the effect of personal health records on patient activation
Supplemental material, sj-docx-1-dhj-10.1177_20552076251315295 for A systematic review of the effect of personal health records on patient activation by Irina Osovskaya, Ann Blandford and Henry WW Potts in DIGITAL HEALTH
Footnotes
Acknowledgements
Only authors of this study contributed to this research.
Contributorship
IO designed the study, led the review and wrote the first draft of the paper. HP is IO's primary PhD supervisor and helped to build the search strategy, review selected articles, direct analysis, review and approve for publication. AB is IO's secondary PhD supervisor and reviewed and approved this paper for publication. All authors contributed to writing the paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This is a systematic review of published studies and therefore requires no ethical approval from UCL.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Irina Osovskaya is currently self-funded PhD student and has had partial PhD fees paid by AstraZeneca.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
