Abstract
Objective
Digital twins (DTs) emerged in the wake of Industry 4.0 and the creation of cyber-physical systems, motivated by the increased availability and variability of machine and sensor data. DTs are a concept to create a digital representation of a physical entity and imitate its behavior, while feeding real-world data to the digital counterpart, thus allowing enabling digital simulations related to the real-world entity. The availability of new data sources raises the potential for developing structured approaches for prediction and analysis. Similarly, in the field of medicine and digital healthcare, the collection of patient-focused data is rising. Medical DTs, a new concept of structured, exchangeable representations of knowledge, are increasingly used for capturing personal health, targeting specific illnesses, or addressing complex healthcare scenarios in hospitals.
Methods
This article surveys the current state-of-the-art in applying DTs in healthcare, and how these twins are generated to support smart, personalized medicine. These concepts are applied to a DT for a simulated health-monitoring scenario.
Results
The DT use case is implemented using AnyLogic multi-agent simulation, monitoring the patient’s personal health indicators and their development.
Conclusion
The results indicate both possibilities and challenges and provide important insights for future DT implementations in healthcare. They have the potential to optimize healthcare in various ways, such as providing patient-centered health-monitoring.
Introduction
Digital twins (DTs) play an important role in the current developments for digital medicine and healthcare. While DTs are inherently related to Industry 4.0 and digital production processes (e.g. Björnsson et al. 1 and Kaul et al. 2 ), as well as product lifecycle management, 3 the trend towards digitizing health data facilitates the creation of various types of DTs. According to Armeni et al., 4 a DT can be defined as a multi-physical, multiscale, probabilistic simulation that uses models and sensor data to represent a real-world entity. Therefore, the concept of DTs allows to create simulations or emulations of physical processes and entities. This is beneficial in medicine and healthcare as it allows the simulation of diagnostics or therapeutic variants in general or in a personalized way. Furthermore, DTs can facilitate the development of clear and comprehensive real-time visualizations of health data. Current literature indicates, that there are application potentials for DT for different medical disorders or within the healthcare system (e.g. Sun et al. 5 ). DTs might be implemented as a service application or to support smart personalized medicine (PM), thus supporting a more individualized monitoring and treatment of patients.
However, investigating the current state of healthcare and medicine, the need for digitization is demanding. Especially in rural areas, where in comparison to metropolis states, health professionals are minorities and need to provide medical expertise for multiple areas with a huge density of patients. 6 Patients, especially elderly patients, need constant accessibility to healthcare, but often need to plan and travel long distances to see a medical expert. Here, digitization offers significant potential for the healthcare sector, from small data-driven analysis tools, like an electrocardiogram (ECG) machine, to a wholly interconnected healthcare architecture. The primary goal is to deliver personalized and data-driven healthcare tailored to the specific needs of each patient, thereby enhancing overall healthcare services. The aim of PM is to tailor medical treatments, such as diagnosis and therapy, to the individual needs and characteristics of a patient to achieve optimal therapy results. This allows the patients to be actively involved in their individual health, which provides a much deeper interconnection of health professionals and patients. 7
Interconnecting personalized DTs from industry with healthcare enables groundbreaking modern solutions for the currently needed personalized, data-driven healthcare throughout the medical infrastructure. This article provides an initial understanding of the concept of digital twins in healthcare and supplements the need of simulation in the realm of PM. The goal is to provide two simulative DT models, presenting possible infrastructure for the integration of digital twins in healthcare. This study explores the potential of DTs to simulate and monitor patients’ health data in real-time. The goal is to enable personalized and data-driven medicine with the help of a digital representation of the patient or their illness. In general, this study is differentiated into two components: (a) focus on the theoretical overview and state-of-the-art of DT in healthcare, with its possible improvements in PM; (b) implementing a DT simulation for a health-monitoring system, focusing on the demonstration of continuous monitoring and predictive analysis.
Methodology and research questions
The article is structured into two main parts: the first provides a theoretical overview of state-of-the-art DTs in healthcare and PM, while the second details the design and implementation of an exemplary use case involving two DT models developed using the AnyLogic simulation software. The theoretical part explores the role and potential of DTs and simulations in healthcare, particularly in the context of PM and simulative health-monitoring. DTs are pivotal in personalizing medical treatment strategies by aggregating heterogeneous health data from various sources, including medical diagnostics like ECG, wearables, and patient surveys. Machine learning (ML) and artificial intelligence (AI) algorithms can be used to analyze the gathered data or to train personalized medical models. The article explores DTs’ applications in various medical fields such as cancer research, network medicine, cardiology, surgery, and orthopedics to highlight different research approaches. This list of application areas and corresponding solutions is not exhaustive but serves to illustrate current implementations of DTs and their benefits for patient care, diagnostics, and treatment planning. The article also discusses the technologies, applications, challenges, research needs, and ethical and legal issues related to DTs in healthcare.
To provide a state-of-the-art overview, several research papers published from 2019 onwards were evaluated, focusing on the emergence of DTs in healthcare mainly in the last 5 years, while in the years before the focus of DT was mostly related to Industry 4.0.
The article follows these research questions:
What is the current state-of-the-art in applying DTs in the medical or healthcare domain? Which technologies are used to further the application of DTs in healthcare? Which roles play reference architectures and different data sources for the development and implementation of DTs in healthcare? How is it possible to create a practical example based on a representation model for DTs in healthcare with the help of a simulative approach?
The first three research questions are addressed in the “Theoretic background and related work” section, detailing background knowledge and related works (Figure 1) regarding digitization in healthcare, DTs in medicine and using simulation technologies in healthcare and medicine. This section also covers the requirements for a smart and PM, data acquisition for DTs, fields of applications for DTs as well as their role in emerging future medicine, before focusing on the challenges. The practical study of the article is reported in the “Design and implementation of a DT simulation for digital-based-vitals-monitoring” section and “Model description and analysis of the wearable-assisted biomarker monitoring & intervention system (BMIS)” section focusing on the design and implementation of a use case implementation of a DT for vital health-monitoring on two levels. The design will be introduced and implemented with the help of an AnyLogic simulation.
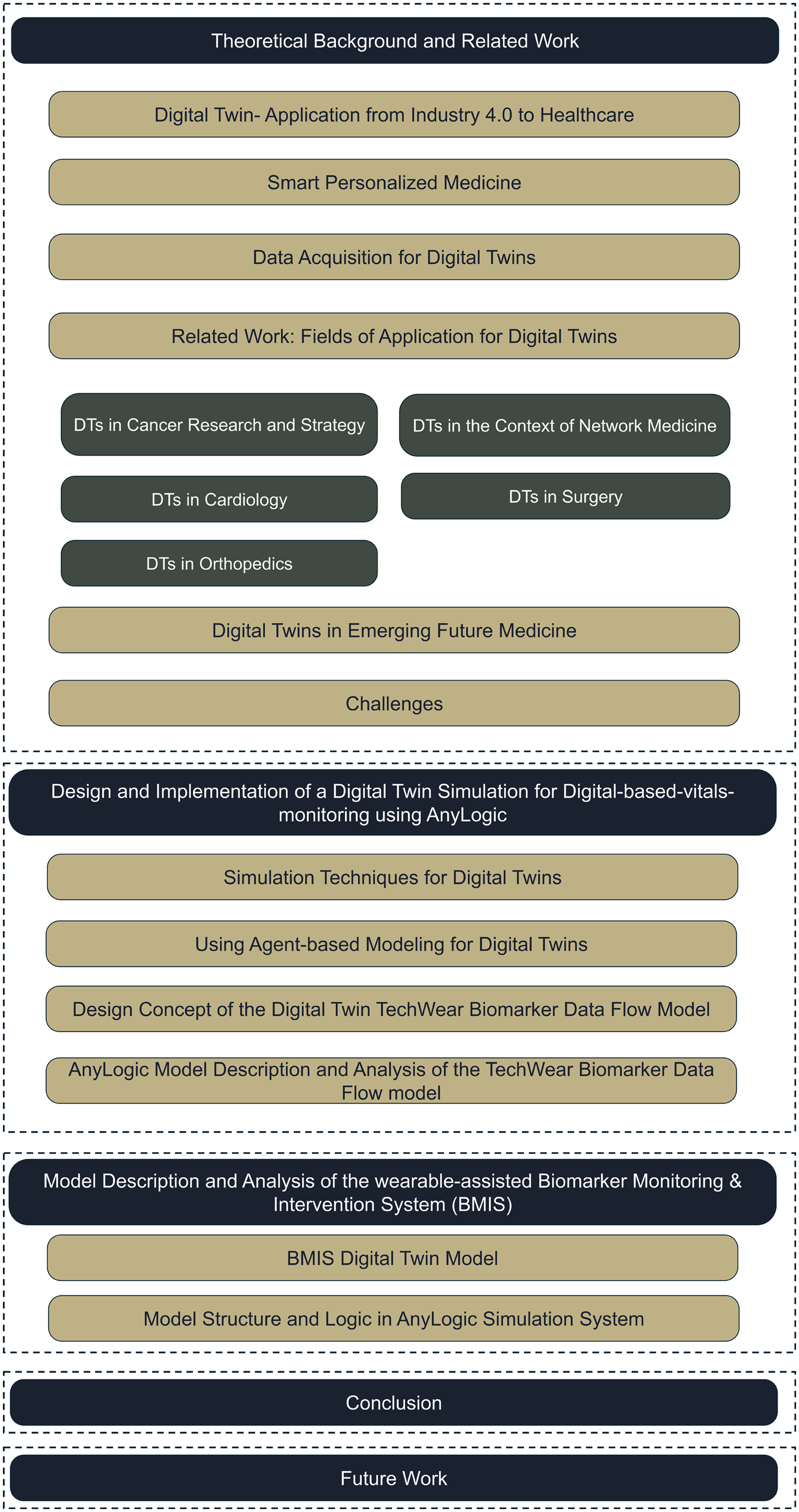
Structure of the paper.
The “Conclusion” section concludes and closes with a consideration of future activities in the “Future work” section.
Theoretic background and related work
Digital twin-application from Industry 4.0 to healthcare
The DT, a concept originating in engineering, has taken on a transformative role across various industrial sectors.1,2 Organizations such as NASA, General Electric, Siemens, and ANSYS showcase the wide spectrum of its applications, from aerospace to manufacturing and energy production, thus positioning themselves as pioneers in the implementation and further development of the DT concept.4,8–11 DTs are a concept to create a digital representation of a physical entity and imitate its behavior while feeding real-world data to the digital counterpart. A DT is “adequate for communication, storage, interpretation, process and analysis of data pertaining to the entity in order to monitor and predict its states and behaviors within a certain context.”
12
While this definition of the Industrial Internet Consortium is tailored to physical devices instead of humans, the entity might also be a patient in the context of Smart Health solutions. In healthcare, this understanding is also applied. The DT can be a patient, but also has the possibility to represent whole systems, like hospitals or is tailored to specific illnesses. This will be reflected in the state-of-the-art applications discussed later in this work. The conceptualization of the DT includes terms such as
The DT itself creates a link between the digital model and digital shadow, enabling a comprehensive and dynamic representation of the real entity in the digital space. Through this connection, entities can be simulated, monitored, and analyzed in real-time, leading to an optimization of lifespan and performance. 3 Continuous updating with real-time data through sensors, cyber-physical-systems (CPSs), and industrial internet of things (IIoT) technologies enhances the DT’s precision and relevance, significantly supporting decision-making and process optimizations in the industry.4,13
The origins of the DT lie in product lifecycle management (PLM) systems, indicating the evolution from static product documentation to a dynamic, interactive simulation of the product lifecycle. 3 This approach of five interacting dimensions, including physical entity, virtual model/spaces, connections, DT data, and services, allows for the seamless integration of the physical and digital worlds by collecting data through sensors and IoT technologies and processing it in virtual models. The resulting DTs offer unprecedented opportunities for monitoring, analysis, and prediction, far beyond traditional methods. 15
After examining the basic structures and functions of DTs, an alternative approach opens up further dimensions of their definition and application possibilities. DTs model physical or virtual entities or human features or entities through highly precise computer-based simulations to achieve an exact bijective representation of their physical counterpart via a digital thread and using a dedicated key number.4,8,2,11 The
DTs represent a paradigm shift, revolutionizing the care landscape and enabling PM through the integration of cutting-edge technologies such as IoT devices, wearables, specialized electrodes, smart sensors, shape-memory polymers, smart electro-clothing systems (SeCSs), blockchain, and AI. A personalized DT provides a holistic virtual representation, called a “digital twin,” of an individual’s “physical twin” encompassing physical (such as sports, dietary, and sleep habits), mental (such as ideas, thoughts, and knowledge), as well as social and biological aspects, relying on multimodal and multisource databases.4,11,16
These comprehensive models offer significant value for personalized and precision medicine by visualizing individual therapy simulations, treatment outcomes, and disease progressions, enabling the prediction of disease developments through the analysis of personal history and context, thus providing tailored therapy approaches and elucidating the complex parthenogenesis of diseases.4,8,15
Pioneering models like the “AnyBody Modeling System” demonstrate the potential of DTs to simulate human body interactions and enable precise calculations such as joint contact forces and metabolism. 8 These and other DT models can promote complex body studies and computer-simulated predictions, serving as a virtual laboratory for treatment tests in medicine, complementing traditional approaches in medical research and practice.4,8 The integration of DTs with advanced image processing, algorithms, and virtual reality/augmented reality technologies enables three-dimensional (3D) representations from two-dimensional (2D) cross-sectional images 5 and supports decision-making in personalized care, risk diagnosis, and individualized treatment planning. 11
The future development of DT technology in medicine requires continuous adaptation and validation of models to ensure their precision and relevance. The use of real-time sensors, wearables, model synergy, and various data sources will enhance the clinical interpretability and integration of DTs into medical practice. 5 Interdisciplinary expert teams are essential to address the complexity of medical applications and develop precise DTs that meet the demands of PM. 17
In summary, DT technology opens new horizons in the healthcare industry in various fields (e.g. diagnosis, treatment, and prevention of diseases). Combining technological innovation, sensors, wearables, and medical expertise enables DTs to provide personalized patient care at an unprecedented level. Continuous research and development in this field promise to push the boundaries of what is possible in medicine while addressing the ethical and practical challenges associated with this advanced technology.
Smart PM
PM utilizes both individualized diagnoses and therapies, derived from experiential knowledge and results of standardized clinical trials. It is supported by exploratory data analysis and ML for modeling and analyzing diseases in DTs. Kamel Boulos and Zhang view DTs as key players
The integration and analysis of diverse types of data, from omics to clinical histories, laboratory values, EHR,
4
and mHealth data, using advanced AI algorithms, are crucial for correlating and evolving in PM.
18
These processes embed information and communication technologies deeply into the heart of
DTs are central in PM, creating comprehensive models of an individual based on structural, physical, biological, and historical features, integrating electronic patient data records (EHRs). They support patient autonomy, risk reduction through monitoring, and preventive measures against disease deterioration, and thanks to real-time health data collection, the optimization of treatment planning. 15 DTs facilitate disease prognosis by analyzing personal history and current context such as location, time, and activity. 4 Clinical decision support systems (CDSs) and DTs provide physicians with valuable tools to manage the information flow and make guideline-compliant decisions, leading to a reduction in unnecessary examinations and improved therapeutic approaches. 19
Telemedicine, including online consultations and remote therapies, minimizes distance and access barriers, supported by wearables for continuous data transmission and patient monitoring. 20 When looking into industry, companies like Atropos Health, Navina, Atomic AI, Turbine AI, and XtalPi illustrate the use of intelligent technology solutions to enhance PM and meet the requirements of this rapidly evolving field by transforming medical data into meaningful insights and optimizing medical care. These include platforms for analysis and decision-making, AI-driven health management platforms, solutions for RNA drug discovery, simulation tools for tumor behavior, and methods to accelerate drug research.22,23,21,24,25
Data acquisition for DTs
PM requires both serial data to monitor the health development of a patient as well as ad-hoc acquired data to assess the current health situation at the moment. For developing medical DTs, it is important to establish a consistent and correct as well as up-to-date database as a single source of truth. Only if the data source quality is correct and fitting it is possible to create reliable DTs and to train the used AI algorithms. 26 AI algorithms, like neural networks, can be used to calculate cardiovascular complications. 27 Treatment recommendations for rescue operations may be generated using knowledge graphs and vital signs. The results may be used for preanalysis in rescue situations, and they can increase the chance for a better treatment of the patient. 28
The medical domain provides numerous data sources, including published health metrics and research health data, public health data, development data of different illnesses and ways for treatment, data gathered from health programs or intervention measures as well as health policy data. 29
Depending on the type of illness a DT is representing, it requires different types of input data. Kaul et al. 2 describe a cancer care DT based on data such as diagnostic data to determine the type of cancer and progress, prognostic data to inform about possible progress, predictive data for possible treatment processes and outcomes as well as treatment monitoring data which might be used for training AI models and evaluation of the bodies responsiveness to treatment measures. Sub-categories of those four types of data are clinical, psychological, genomic, and demographic data used for prognosis and prediction, laboratory and scanning data as well as genomic data for diagnostics and finally, patient report data for treatment monitoring. 2 Treating a patient effectively necessitates the collection of current sensor data, using wearables as well as data describing the patients personal situation, for example, lifestyle, physical activity, or other socio-economical factors in a multi-modal approach. 18 The authors pronounce the necessary interconnection of patient-centered care data and research data for interoperability and standardization.
One type of sensor data is biomedical signals, which are generated from the electrical, chemical, or mechanical activities of a patient. Electrodes may be placed invasive or non-invasive with, for example, smart patches or integrated into smart textiles, such as SeCSs. 16
Sensor units are able to detect, for example, movement, gestures, positions, temperatures, vital parameters, interactions, environmental factors or objects.
11
In addition to the data gathered from a patient, other medical disciplines, such as oncology or radiology, require additional data. The generation of cancer diagnosis and treatment plans requires the consideration of different risk indicators and results from existing clinical studies or related data sets next to the patients own medical data. In this way, representative models, diagnoses, treatment plans, and prognoses may be generated with the help of AI. 2
State-of-the-art: Fields of application for DTs
In medicine and eHealth, DTs are of central importance, as they enable a virtual representation of real patients, fostering PM. They serve as virtual counterparts for tests and simulations, including predicting medication efficacy. AI-based models analyze patient data provided by DTs, simulating real-time scenarios for optimized treatment approaches. This supports the development of tailored therapeutic strategies and medications for both healthcare professionals and patients.4,2,11 The combination of AI and DTs, assisted by technologies like IoT devices and sensors, enhances data evaluation and management across diagnosis, prognosis, treatment selection, decision-making processes, quality of care, and patient satisfaction.30,2,11,15 The application of DTs in healthcare is considered an innovative method, utilizing technology in conjunction with multidisciplinary, multiphysical, and multiscale models, as well as diverse databases. This approach aims to deliver robust, precise, and effective medical services.11,15 DTs significantly contribute to understanding complex disease mechanisms by efficiently combining various datasets, from wearable digital devices and omics to hybrid electronics, imaging, and electronic medical records. This enables in-depth insights and improved diagnostic and therapeutic approaches.
1
DT technology finds diverse applications in various research centers and projects aimed at enhancing personalized diagnosis and treatment. Sahal et al.
11
summarized examples such as the Swedish DTConsortium (SDTC),
31
Human Digital Twin OnePlanet research center
32
or the Empa research center
33
focusing on specific aspects of DT-based health research. DigiTwin Consortium,
34
a global network of academics, clinicians, and industry partners, utilizes DTs to optimize various diagnostic and treatment approaches.
1
The work of the aforementioned research centers focuses on creating DTs that depict all relevant molecular, phenotypic, and environmental factors of an individual patient. These DTs are used to test thousands of drugs and identify the most effective medication for the patient.1,35,11 While there are many advantages mentioned in the publications above, Moztarzadeh et al.
36
also view a progress in applying DT in healthcare, but miss a systematic methodology to implement DTs when facing the challenge of
In the following various applications of DTs in the field of healthcare will be presented:
DTs in cancer research and therapy
In oncology, interdisciplinary collaboration, for example, with other clinical disciplines
37
and informed decision-making through tumor boards enable personalized therapeutic approaches. Molecular pathology identifies individual tumor vulnerabilities, and AI could optimize these processes in the future.38,37,40–42,39 Advances in cancer research and precision medicine provide a wealth of data enabling tumor classifications and more personalized treatments based on the genetic profiles, supported by DT and computational oncology. Epigenetics, which involves the analysis of DNA methylation and gene expression patterns, plays a crucial role in treatment optimization.
43
Batch et al.
44
introduced a DT for predicting the
Thiongó and Rutka discussed the possibilities of using DTs as a future technology to improve the prediction of neurological complications of pediatric cancer patients, focusing on the factors of precision medicine, predictive analytics, possibilities to model cancer care and to level out different opinions of clinicians, or extend clinical trials with the help of DTs. 45
Kim et al. 46 developed a DT model for prostate cancer patients. It incorporates clinical patient data, prostate cancer process knowledge, expert knowledge and uses ML for training a prediction model for pathology and biochemical recurrence. The DT supports clinical decisions regarding the illness. The DT solution by Moztarzadeh et al. 36 incorporates a metaverse, a layer of the DT that receives new data on breast cancer patients and updates the DT based on different ML models, thus providing a new basis for decision support. They recommend using individual DTs for each patient to enhance monitoring within the metaverse. 36 In the field of tumor-agnostic cancer therapy, eHealth and PM targeted therapies are developed in DT utilizing antibodies and inhibitors, often combined with chemotherapy. Advanced molecular diagnostics and AI methods aid in identifying personalized therapies, especially in rare cancer cases or when standard treatment options are lacking. Tumor-agnostic cancer therapy, focusing on specific cell characteristics, is becoming increasingly relevant. 47 A concrete example of this targeted therapy in PM is the treatment of melanoma using DNA analysis. Medications that target specific mutations can temporarily slow tumor growth; however, they offer no benefits to patients lacking these mutations.38,7
DTs in the context of network medicine
Network medicine utilizes tools from network science, encompassing networks of biological, technological, or social systems, to investigate the molecular complexity of diseases and the relationships between different phenotype. 48 This leads to the identification of disease genes, modules, and pathways or the discovery of drug targets and biomarkers. A better understanding of cellular networks can improve clinical practices through more accurate biomarkers, refined disease classification, and personalized therapeutic approaches. 48
Medical DTs should integrate all relevant data sources and variables to form multi-layered modules encompassing biological molecules such as mRNAs, genes, and proteins.48,1 These modules enable computer-simulated procedures and treatment. Experiments are conducted virtually to support hypothesis development, testing, diagnostics, and therapy. Diagnostic and therapeutic decisions should also consider other data types, including symptoms, psychopathology, phenotypic modules, and environmental factors.48,1 Hussain et al. 49 presented a digital twin in the field of network medicine by using the DT concept to create a diagnostic method for strokes, which may support clinical decision-making for patient treatment.
DTs in cardiology
Research about heart digital twins (heart DTs) offers novel diagnostic possibilities and improved therapeutic approaches for cardiovascular diseases.4,8,50,51,11,52 Rudnicka et al.
53
conducted a literature study that concluded improvements in data quality, image processing, and 3D imaging, combined with technologies like extended reality and AI, may contribute to developing effective heart diagnostics and personalized cardiology. The aforementioned technologies may be the basis for the construction of a heart DT, which again may support applications related to diagnosis, prognosis and therapy optimization. While these are positive aspects, greater importance should be placed on the ethical dimensions of DTs, such as the responsible integration of AI.
53
Corral-Acero et al.
54
discussed a vision of using DTs for precision cardiology with the help of using computational models. Like Rudnicka et al.,
53
they also envision an application of DTs for diagnosis, prognosis, and treatment plans.
54
In precision cardiology, DTs should incorporate statistical and mechanistic models to create synergies and support the specified applications.
54
In addition to researchers, technology companies such as Siemens are also working on creating medical DTs.4,8 Technologies like Philips’
Heart DTs provide more precise diagnostic and treatment options for cardiovascular diseases. These models can non-invasively identify biomarkers and create personalized DT models, offering applications such as predicting pressure drops in flow obstructions and optimizing medication.11,5 Electrocardiographic imaging (ECGI) combines imaging and electrophysiological data non-invasively, supports heart function monitoring at home, and transmits results to doctors via a mobile app. It involves advanced techniques like ECG classification learning algorithms, nonlinear MRI registration, heart surface mesh extraction, and high-density sensors. 5
DTs can be used in medicine to simulate dosage effects and device reactions before treatments, assessing the suitability of therapy or devices for patients. FEops’ HEARTguide, for example, provides personalized heart DTs to optimize the care of patients with specific structural heart diseases after transcatheter aortic valve implantation.
DTs play a growing role in treating cardiovascular diseases and aneurysms, enhancing ablation guidance for infarct-associated ventricular tachycardia (higher heart frequency based on an infarct), and generating synthetic physiological data for comprehensive information on blood pressure and flow changes. Patient-specific aneurysm DTs by the French startup Sim&Cure support surgeons in identifying optimal implants through the creation of a 3D aneurysm model for minimal invasive endovascular repairs, with promising initial results. Further research is necessary to explore these applications fully.5,59,61
DTs in surgery
DT technology enables more accurate surgical planning and minimizes unintended anatomical damage during neurosurgery, vascular surgery, and interventional surgery. In vascular surgery, it contributes to the development of diagnostic tools and the simulation of surgical procedures. The combination of in-vivo vibration measurements and virtual data increases the accuracy of DT computer models and expands diagnostic and treatment options, taking into account clinical, morphological and hemodynamic data.
5
Bjelland et al.
62
conducted a study to determine the applicability of DTs for arthroscopic surgery, in particular a knee surgery. Their motivation is the trend of using simulators for training surgeons, which could be enhanced with DTs incorporating patient-specific and intraoperative data. The authors view advantages in using DTs over existing simulators in their adaptability and flexibility and not a pre-fixed training sequence. They determined key aspects for DTs in the form of
DT technology also enhances the training of surgeons and teaches technical skills in various disciplines.
DTs in orthopedics
DTs in orthopedics enable real-time monitoring and analysis of the lumbar spine, as well as the prediction of biomechanical properties. This requires combining physics-based experimental models, data-driven numerical models, and multi-agent system technologies.15,5 A research team developed a DT of the lumbar spine using human motion capture technology, wearable VR devices and sensor data. 64 VR devices and sensor data capture body position and posture, while inverse kinematics, finite element methods, and Gaussian process regression are utilized for analysis. They developed a 3D virtual reality system, which enables orthopedic treatments and effective warnings, especially in spinal rehabilitation.15,5 Furthermore, DTs have been used in orthopedic surgical models to provide clinical decision insights. DT models evaluate factors like mechanical stability and interfragmentary stress, which help assess the risk of recurrent fractures. A patient-centered model combining CT, AI, and DT technologies has been developed to improve the accuracy of alignment and adjustment examinations of the subtalar joint axis. 5
Digital twins in emerging future medicine
In recent years, the number and types of biomedical sensors, wearables, or smart devices to monitor a person’s fitness and health have been rising steadily. 65 Fitness trackers or sensors connected to smartphones often aim to “optimize” individual’s use. The gathered data directly relates to the person and may have limited comprehensibility. 18
Wearables or trackers use IoT-technologies for gathering the related data. Modern hospital monitoring of chronically ill patients using their physiological parameters and real range of capabilities to gather and process personalized data for precision medicine. 66 Other trends include combining medical technology, robotics, and AI to create exoskeletons, for instance. 67 Future trends in healthcare may focus on health-monitoring, precise diagnostics and personalized treatment plans. One example would be the monitoring of chronically ill patients using their physiological parameters and real-time health data to detect anomalies or risks, thereby adapting treatment plans. 5 On a broader scale, it might be possible to create lifelong DTs that access a person’s health records and generic epidemiological data. 5
According to Mukherjee et al.,
68
there are three trending areas of precision medicine for the upcoming years which also offer a basis for a DT or the needed data to create it:
complex AI algorithms, digital health applications, “omics”-based tests or biomarker.
A DT for a precise diagnosis would require multi-layer, multi-modal, and multi-source data (e.g. health exams, wearables, and simulations) in an integrated DT model.69,11,15,5 A DT for precise treatment plans requires multi-modal and multi-source data as well, which might be data from medical studies, risk factors, correlations between factors, data of virtual experiments, new research data, therapeutic outcomes, etc. AI might be used for learning treatment plans and outcomes.
5
Cancer patients might benefit from a personalized DT for follow-up examinations after the treatment process is over, to provide an individualized healthcare and to detect new risk factors.
11
On a broader scale, DTs might not only be used for personalized or precision medicine, but for optimizing the resources and efficiency of a hospital. The necessary input data, in this case, includes the number of certain treatments, patients, waiting times, etc. Simulations might be used to optimize internal processes or for learning purposes.4,5 To provide more processing power for DT applications based on AI, quantum computing may be employed in AI in the future.70–72 Other trends discuss the application of DT for clinical decision support apps for mobile devices. 19
Challenges
The integration and use of DTs and other technological advancements in the field of medicine present formidable challenges across technical, ethical, and regulatory dimensions. Addressing these challenges is crucial for fostering responsible and effective integration of such technologies into healthcare:
Technical challenges for implementing DTs in healthcare
In the application of DTs in healthcare, such as patient care and hospital management, there are challenges ranging from the precision of the simulation, validation approaches, technical limitations and data collection and integration.73,72,74,5,75,4 The lack of a standardized architecture and individual solutions as well as the limited use of the potential of DTs over its entire life cycle pose further problems. Effective use of DTs requires a data flow between different companies 76 and a sufficient data quality. 4 Creating complex high-fidelity models for DTs is a technical challenge. This is particularly true when considering multiphysical processes (mechanical, thermal, electrical, magnetic, chemical, or biological) and the integration of information across different scales or levels, coupled with a lack of experimental data. Such models require advanced mathematical methods, algorithms, and powerful computing resources, including 5G, sensor technology, and quantum computing.73,72,74,5,75 There are also technical limitations between the interaction of DT objects and DT people, such as the real-time connection and the need for standardization. 8 Times and latency for data gathering and transfer between sensors and data analysis components need to be considered. Additionally, bridging the gap between DT-calculated results and real-world scenarios remains a challenge. For example, unforeseen or underestimated risks may arise during the planning of a surgical procedure and become apparent during the actual surgery. 5
Security, data protection, and ethics of DTs
PM poses challenges at the intersection of business and data protection, particularly in the fields of pharmacy and medical technology, including standards for data trading and patient information. Medical innovations and data transfer are facilitated by manufacturers, with state repositories serving as data storage. This also involves far-reaching adjustments in infrastructure, organization, financing and compliance with regulatory and data protection aspects. 66
The comprehensive application of DTs and AI in the healthcare sector requires solutions for current regulatory and data protection challenges. While medical environments are subject to strict (country specific) regulations such as HIPAA (US Health Insurance Portability and Accountability Act), FDA (US Food and Drug Administration), or the EU GDPR (EU General Data Protection Regulation), non-medical areas are less regulated.4,77,5 The exploitation of these regulatory loopholes by technology companies such as Facebook and Google collects and monetizes personal health data, which requires a review of the role of medical devices and data. 77
There is also mistrust of AI-supported decisions, for example, with regards to reliability of the proposed recommendations or potential bias against patient groups due to used training data or detected patterns. Armeni et al., 4 for example, mentioned that currently there is oftentimes a bias in health data due to data of white men being overrepresented, 4 which might pose the question if the same recommendation may be applied for women. Implementing validation and approval regulations for predictive biomedical computer models could reassure doctors and patients, who are often skeptical about AI-supported decisions.4,8 The blending of medical and health-related areas through digital biomarkers increases the difficulty of integrating wearables into medical applications, as current regulatory frameworks do not sufficiently consider data outside clinical environments. Interdisciplinary collaboration is needed to investigate these challenges and discuss international data processing standards, while analyzing the underlying technologies and their impact on medical and health-related areas.78,77 Wearables in medical applications of wearables require adjustments to the regulatory framework to ensure security and data protection. The distinction between end-user and medical technology, as well as meeting regulatory requirements, are key challenges. There is a need for a technical platform for recording vital parameters and a clinical integration, taking security and encryption techniques into account. The implementation of DTs and AI in healthcare requires an examination of technical, regulatory, ethical and responsibility issues. This necessitates intensive interdisciplinary scientific exchange.
Limitations
A major challenge of integrating DTs in healthcare is the integration of real-time data from multiple sources. A digital twin is a complex structure representing a physical entity, such as a patient or an illness. The real-time integration of data from various sources and its following analysis remains highly complex. For that advanced architectures are needed, which are highly secure, patient centric and provide a high standard of data protection. The healthcare infrastructure needs to be adjusted to the idea of digital twin applications, which may be challenging, but future-oriented. In addition, the concerns regarding to the aforementioned bias in medical training data needs to be addressed to prevent non-fitting recommendations or decisions based on the DT.
Design and implementation of a DT simulation for digital-based-vitals-monitoring
Simulation techniques for DTs
Advantages of simulation techniques for DTs in healthcare
Simulation techniques offer numerous advantages in the development and optimization of DTs. They enable the creation of simulation systems of DTs to create a realistic representation and analysis of the physical twin. Through simulation, the behavior and responses of these systems under various conditions and scenarios can be examined without directly affecting real systems or processes.81,80
Advantages of simulation techniques in relation to DTs:
Simulation techniques can be divided into various categories, including stochastic simulations, dynamic simulations, and agent-based simulations.
83
Each of these techniques has its specific application areas and advantages. Choosing the appropriate simulation platform depends on the specific project requirements.
Simulation software solutions
There are various simulation software solutions available on the market. Abar et al. 79 conducted a comprehensive survey of 85 ABMS tools. This survey was based on criteria such as the availability of source code, agent interaction, the availability of the programming language used, APIs or graphical programming interfaces, the necessary compiler, operating system, the effort required to implement a use case, application areas, and scalability. 79 Although healthcare applications are mentioned and various tools classified as applicable, the study does not specifically discuss DTs implemented with ABMS, likely due to its 2017 publication date. Nonetheless, the study also evaluates Anylogic which was chosen for the prototypical implementation of the DT in this article due to its extensive functional scope. AnyLogic is evaluated as applicable in healthcare as a high- or large-scale ABMS tool and a “user-friendly graphical environment for visual model development.” 79
AnyLogic is a widely used platform for creating various types of simulations, including production processes, traffic flows, supply chain management, and personalized medical applications. It provides a versatile environment suitable for modeling various systems and scenarios in PM.84,90 Other solutions on the market include Simio, 91 Arena by Rockwell Automation, 92 and others.
Compared to the other aforementioned solutions, AnyLogic stands out for its flexibility, versatility, and support for a broad range of simulation methods, which might be used in DT development. AnyLogic allows for integrating system dynamics, discrete events, and agent-based models within a single environment, significantly enhancing model efficiency and optimizing solutions in PM. This allows users to select the simulation approach that best meets their specific needs. This feature is particularly useful in interdisciplinary applications, such as PM, where complex and dynamic systems need to be accurately modeled. Furthermore, AnyLogic provides a free version that already offers a wide range of features, and special licenses are available for universities, making it accessible for educational and research purposes.84,86,87,85,88,89
Short comparison between simulation software solutions:
Using Agent-based modeling for DTs
Introduction to agent-based modeling with AnyLogic
Agent-based modeling is an advanced simulation method used to mimicking complex systems and multi-agent systems through autonomous agents, which range from individuals to organizations. These agents interact with one another, resulting in emergent behaviors that contribute to the system’s overall dynamics. One software platform for different types of multi-agent simulations is AnyLogic. AnyLogic is a powerful software platform that integrates agent-based modeling with other simulation methods, enabling detailed analyses of complex systems. It supports the representation of DTs, increasingly significant in the era of Industry 4.0 and PM. 85 AnyLogic facilitates the creation of realistic models through the integration of high-resolution real data. Its multi-method approach allows for individual and autonomous agent modeling, provides a platform for training AI and ML algorithms, and supports synthetic data generation. This approach bridges the gap between theory and practice, assisting businesses in making informed, evidence-based decisions. It supports the modeling of complex scenarios, such as pharmaceutical launches or the use of wearables, by simulating the dynamic interactions between various actors.84,86–88 In the next section, a design concept will be introduced, which will later be implemented in the form of an agent-based simulation with AnyLogic.
Design concept of the DT TechWear Biomarker Data Flow model
The emergence of medical DTs—an industrial legacy
The transition of DTs from industrial applications to medical uses marks a significant advance in simulation technologies. Initiated in the manufacturing industry to optimize product life cycles and maintenance, the technology is undergoing a transformative adoption in the healthcare sector. The transition is facilitated by rapid digitization and access to extensive datasets, which supports the creation of precise, patient-specific models. In medicine, DTs utilize advanced industrial simulation techniques that have been adapted to address the unique challenges of medical data analysis and diagnostics. Integrating this technology to medical practice promises a revolution in PM by enabling individual treatment plans and preventive measures based on the specific patient data. Furthermore, integrating DTs into medical workflows facilitates research and development of new therapies. This is achieved by offering a deeper understanding of disease mechanisms, pathogenesis, and drug effects on the human body.97,15
PM with DTs
At the intersection of health and technology, DTs offer a visionary perspective for medicine. This DT model, called
DT model and application use case
The core component and idea of the DT-TechWear Biomarker Data Flow model is a simulation of a holistic data processing process from patient to application in diagnosis, analysis, or treatment decisions. The DT model serves as a crucial link between the real and digital worlds, with a specific focus on the cardiovascular system.
The concept of the model allows the capturing of different kinds of data related to the patient and the treatment process. Therefore, it could be simulated cyclically to capture biomarker data from, for example, wearables or smart patches, which are then processed in a dynamic health monitoring system. Here, synthetic data was used to represent the process. However, real-time data can also be used to run the model. The process, as shown in Figure 2, therefore, encompasses a continuous data collection state called
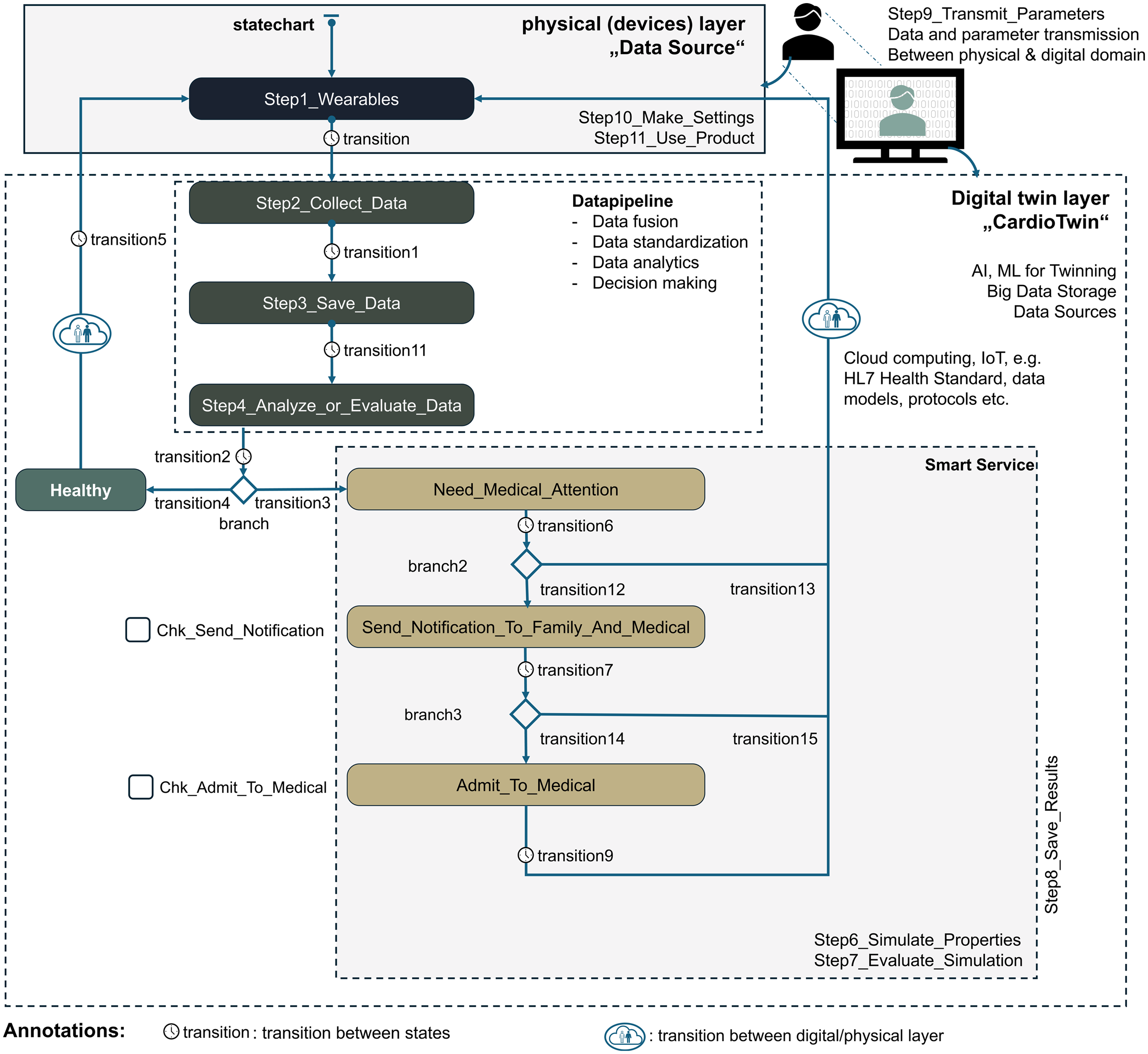
The digital twin TechWear Biomarker Data Flow model, implemented with AnyLogic, represents a monitoring system that captures and analyzes multiple biomarkers for patient health monitoring.
Emulation of human health data
A key feature or requirement of the DT TechWear Biomarker Data Flow model is its ability to realistically emulate human health data. It uses
In the AnyLogic models, real-time data is generated by producing random numbers that follow known distributions. For instance, values for bio-vital parameters, such as “heart rate,” are randomly selected from a defined healthy range at the start of the simulation, for example,
AnyLogic model description and analysis of the TechWear Biomarker Data Flow model
The digital twin
The model uses
Specific and important functions for the model are the ones for initializing the biomarkers
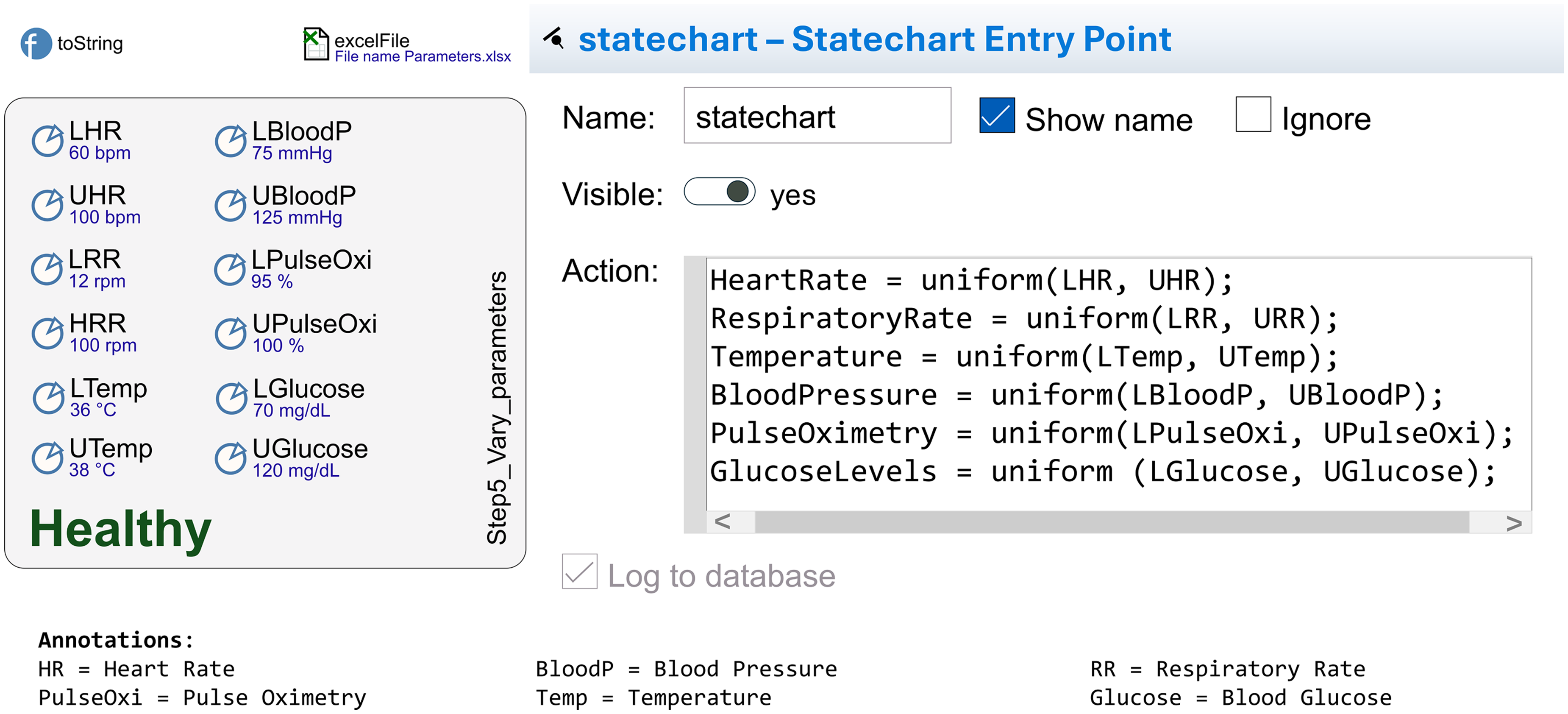
Step 5: Vary parameters and initialization actions. This figure presents a detailed view of the fifth step to detail the used health indicators (based on AnyLogic).
The key aim of the AnyLogic implementation of the DT model is the simulation variability. The model simulation captures the inherent variability of health indicators by representing a range of health states based on random variables within defined boundaries. State transitions are guided by these initialized variability values, facilitating dynamic and realistic health monitoring.
States & Transitions
State: Step1_Wearables—integration, initialization, and monitoring
The
Step2_Collect_Data—Synthesis and data modeling

Detailed view of “Step2_Collect_Data”: Generation of synthetic data with triangular distribution and variables (based on AnyLogic).
Step3_Save_Data—Data processing and storage
In the data processing cycle,
Step4_Analyze_or_Evaluate_Data—Analysis and evaluation
The
State transitions and management in the context of the DT TechWear Biomarker Data Flow model
After explaining the different states, this paragraph focuses on the state transitions within the DT model:

Conditions and parameter: This figure presents a detailed view of the conditions and parameters of transition12 (based on AnyLogic).
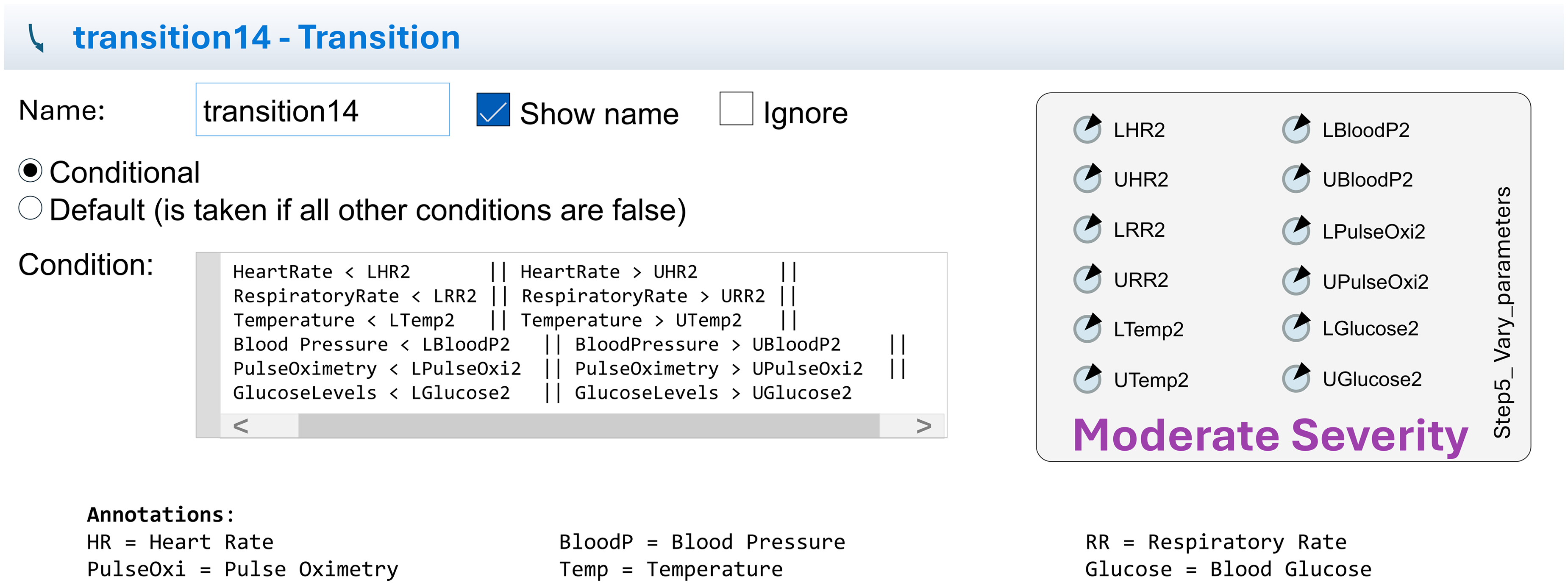
Conditions and parameter: This figure presents a detailed view of the conditions and parameters of transition14 (based on AnyLogic).
Transition conditions
Transitions are based on algorithmic checks of biomarker data. For example,
Data storage and visualization
In the model, real-time visualization of biometric data occurs under the agent “Patient.” The
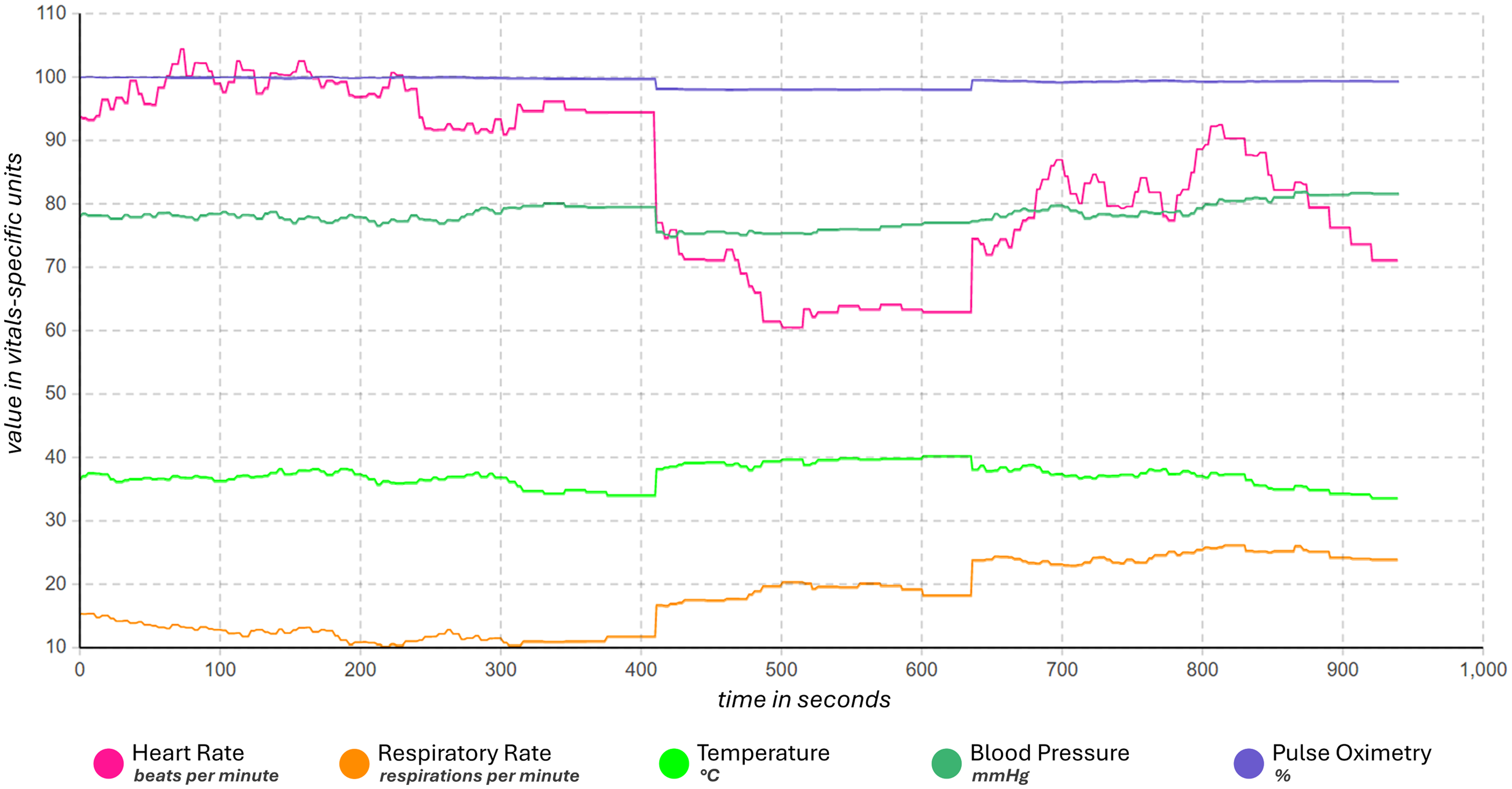
Time plot for various physiological parameters: Exemplary visualization of various vital sign parameters of a patient (heart rate, respiratory rate, temperature, etc.) over time (here in seconds) (based on AnyLogic).
In the future, the DT TechWear Biomarker Data Flow model can be enhanced by testing the model with real-time data, and evaluating its response to the various data and data types of medically certified devices. Such real-time real data testing, can increase the effectiveness of the model.
Model description and analysis of the wearable-assisted Biomarker Monitoring & Intervention System (BMIS)
BMIS digital twin model
The Biomarker Monitoring & Intervention System (BMIS) is a more complex DT model (BMIS-DT model) that enables dynamic and continuous health monitoring through wearables for capturing various biomarker data in individuals or populations. Figure 8 depicts the setup and state transitions. The model can be used to compare a monitored or non-monitored patient or a larger population of patients with each other. Variations resulting from factors such as data transmission delays or physical activities affect system accuracy and response time, leading to false alarms or delayed interventions. These aspects are addressed in the wearable-assisted BMIS model through the use of specific algorithms and thresholds.
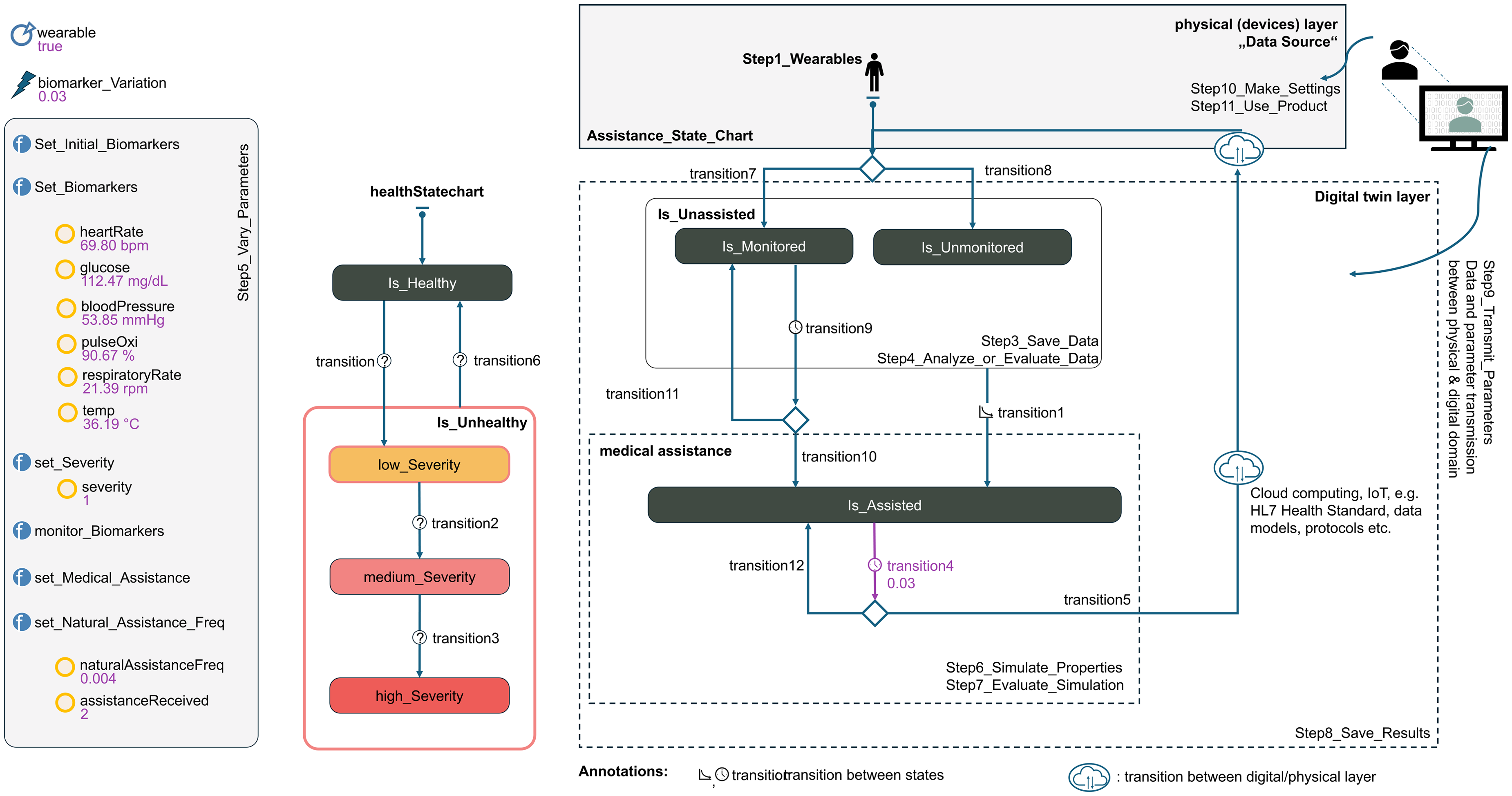
Wearable-assisted Biomarker Monitoring and Intervention System (BMIS) implemented with AnyLogic.
The aim of the model is to simulate daily fluctuations of defined biomarker data and assess health conditions to initiate precise medical interventions upon exceeding certain thresholds or pathological deviations, allowing for a timely response to critical health developments. The BMIS-DT model is used for predictive analysis in healthcare to detect potential health deteriorations early. It may be applied in telemedical monitoring and clinical research, aiming to optimize treatment efficiency and steer patient-specific interventions.
Example scenarios for model application
The following scenarios illustrate the application of the “BMIS-DT model”:
State machines are used for dynamic management of health states and to define possible state transitions, contributing to real medical scenarios by investigating factors influencing individual health and evaluating intervention strategies. The BMIS-DT model addresses the need for precise and proactive healthcare through the continuous capture and analysis of biomarker data.
Wearables play a central role in the BMIS-DT model by enabling continuous monitoring. Individuals are divided into two main groups, monitored
Various biomarkers such as heart rate, blood pressure, and oxygen saturation (see Figure 8) are captured precisely and continuously. Exceeding the set thresholds of a biomarker is considered an indicator of a potentially critical health condition, signaling the need for medical interventions and enabling the use of predictive models. This includes measures such as medication dosing, notifications to medical personnel, and automated emergency calls. Exceeding these thresholds, such as a tachycardia with a heart rate over 100 beats per minute, triggers an escalation in the treatment process.
Model structure and logic in AnyLogic simulation system
The AnyLogic simulation of the BMIS-DT model consists of the following core components:
This model architecture facilitates precise and realistic simulation of health monitoring and intervention mechanisms, which are supported by the analysis of synthetic biomarker data. The
Figure 9 presents the patient simulation area, whose goal is to simulate modern health monitoring and to compare between patients wearing a wearable device for health monitoring (indicated by colorful dots with a blue border) and patients without a wearable (colorful dots without border). Three colors are used to differentiate the patients’ current health status: Green describes healthy patients who do not need active treatment. Light yellow describes the patient, where a minor abnormality within the biomarkers has been identified, which leads to health monitoring but not necessarily an intervention. Red describes an acute patient, who needs immediate treatment or intervention. Of course, the given figure is static and only shows the current situation of the comparison between patients with- or without a wearable and, therefore, only indicates the number of healthy, slightly abnormal, or critical patients at this point of time. The graphs in Figure 9 present the simulation of an average number of medical assistance visits in hours and the average severity during the simulation, over time (also in hours). It shows clearly that with the help of a wearable the severity level maintains to a specific level, in comparison to the non-wearable patients, where it increases by time.

“Main”—patient simulation area (based on AnyLogic).
Functions
Key functions such as
BMIS-DT model functions.

State transitions
State transitions are based on well-thought-out decision trees fed by multidimensional data analyses. They are essential for the dynamic adaptability of the model to changing health data.
Evaluation and enhancements
Analysis of the in-house development of the BMIS-DT model
The BMIS-DT model underscores the transformative role of DTs and wearable technologies in health monitoring and intervention. It demonstrates how continuous and differentiated monitoring through wearables enables individual and precise medical actions based on the simulation and analysis of biomarker data. By doing so, it tackles the challenges associated with dynamic health-state modeling and the necessity for adaptive intervention strategies. The development and analysis of the BMIS-DT model highlight its potential to enhance patient care through advanced technologies and emphasize the importance of proactive health monitoring for early detection and treatment of critical conditions. Future improvements may include the integration of advanced biomarkers and the application of ML to increase the accuracy of predictions, further promoting PM.
Extension and optimization of the model for wearable-supported biomarker monitoring
In the ongoing development and enhancement of systems like the BMIS-DT model, it is prudent to expand the discussion to the optimization of underlying models, especially in terms of patient data simulation. Using such data is not only a valuable resource to demonstrate the model’s functionality and efficacy, but it also opens the possibility of significantly broadening its application scope. Through this methodological expansion allows for comprehensive validation of the system’s functionality, enabling more precise and wide-ranging health monitoring and interventions by integrating additional dimensions and parameters. These approaches promise improved diagnostics and therapy support by enabling more detailed and individually tailored data collection and analysis.
The following paragraphs summarize further model enhancement opportunities:
Conclusion
The article provides an overview of the current state-of-the-art DTs in healthcare and their utilization for smart PM. The second part of the article focuses on the design and implementation of a DT simulation for digital-based vitals monitoring and a wearable-assisted biomarker monitoring and intervention system. For implementation purposes, AnyLogic has been used, combining the idea of a DT and simulation techniques.
Concerning the posed research questions, the literature overview in section two reveals the current trends and technologies used for implementing DTs in healthcare, as well as using DTs for smart, PM. The availability of high quality data sources for implementing a DT, tailored to a certain disorder, is a deciding factor as well as supporting to involve the patient for gathering individual data to personalize a DT-based application. Similarly, healthcare and medicine have high standards for data security and privacy, as well as ethical aspects to consider, which pose challenges for implementing DTs in this sector. Unlike machines, patients always provide individual data and require an application that is easy to understand and transparent concerning their current health situation or parameters. The two presented examples of a practical simulation of DTs in healthcare underline the immense potential.
Future work
In future, DTs can be a groundbreaking technique in the field of healthcare and medicine. Its advancements can lead to a globally available dynamic infrastructure, leading to a great interconnected digital healthcare. In the following some concepts for future work are presented:
Simulation to reality
The presented implementation of two DTs is a demonstration prototype utilizing artificially generated data input. One aspect for future extension is to conduct a patient study using different sensors and wearables to provide real data. This will help further tailor and detail the personalization aspect of the implementation and to prove the concept behind the vital monitoring.
Integration of n-many data sources
Another aspect is integrating other data sources, such as bio-bank data and diagnostic or therapeutic data, to provide better reference data for ML integration. Real-time analysis and feedback will benefit the patient and can indicate emergency situations. Here the field of knowledge fusion can enhance the concept with great additions.
AI algorithms
Furthermore, an extension of the current model could focus on integrating different AI algorithms, contributing to ad-hoc or long-term patient monitoring and analysis. Medical services, like disease detections, treatment recommendations can be integrated as a service for DTs, which allows pre-analysis based on the patient’s own data. From an ethical perspective, it is essential to prevent potential bias due to patient backgrouds, age, gender, etc. when generating treatment recommendations.
Security and privacy
Moreover, further consideration is needed for data security and privacy, interoperability with other systems and platforms, and patient interaction. From a research point of view, the empirical validation of the model, patient acceptance, and integration of the model Integration into existing healthcare processes needs to be evaluated.
Communication and interoperability
Interesting would be the communication and interoperability schemes, developed to prepare multiple DTs or its services, to interconnect and communicate. Especially, the development of standardizations between models and sources can be further investigated. If further tests show promising results, the inclusion of additional health parameters and disorders in the DT model and monitoring system is aspired.
Footnotes
Acknowledgements
Not applicable.
Contributorship
MN, SK, and MD: substantial contributions, study objective, study design, research questions, search strategy, identification of relevant studies, draft writing, and visualization. MN, MD, and CW: supervision, proof-reading, and writing extension. MN and SK: data retrieval from literature. SK: AnyLogic design and demonstrator implementation. MN, MD, and MF: project administration. All authors contributed to the article and approved the submitted version.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This article does not include any studies involving human or animal participants.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Guarantor
CW.
