Abstract
Background
Digital technologies can assess the quality of medicines in resource-challenged health facilities in a timely manner and ensure effective healthcare.
Objectives
This study assessed the quality of some non-communicable disease medicines, antimalarials, antibiotics, non-steroidal anti-inflammatory drugs and hematinics in selected healthcare facilities as part of their (potential) participation in a digital pharmaceutical supply chain management system (Med4All).
Methods
All the sampled medicines were screened using TruScan analyzer and GPHF-minilab, that is, low-medium field technologies. The quality of a representative subset of the medicines (∼11%) was determined using pharmacopoeia gold standard methods.
Results
About 75% of the medicines were manufactured in Ghana, and 16% were imported from India, with the Ghana Food and Drugs Authority registering 72% of the medicines. There was over 90% availability of medicines in health facilities from the middle and southern sectors, while the northern sector had around 64%. Approximately 87% and 88% of the medicines passed TruScan™ Raman and GPHF-minilab analysis, respectively. There was no significant difference between the two screening methods for assessing the quality of the medicines (p > 0.05). Of the 21 medicine samples, 5 (∼24%) failed, and 16 (76%) passed the pharmacopoeia test. The failed medicines were diclofenac, azithromycin and glibenclamide. Compared to the pharmacopoeia method, the performance of the TruScan™ Raman analyzer and the GPHF-minilab technologies were 71% and 63%, respectively.
Conclusion
The study provides insights into the quality of medicines in resource-limited primary healthcare facilities.
Keywords
Background
Ensuring the quality of medicine is crucial for providing safe and effective healthcare while also reducing healthcare costs. Quality assurance of medicines is vital for achieving universal health coverage (UHC) goals and is a crucial component of pharmaceutical supply systems. It ensures that the medicines reaching patients are safe, effective, affordable and of acceptable quality.1,2 Creating a thorough quality assurance program involves addressing both technical and non-technical elements. This includes establishing standards and guidelines while collaborating with stakeholders like manufacturers, wholesalers, distributors, medicine regulators, civil society organizations and consumers. 3
The Lancet Commission on Essential Medicines for UHC recommends that countries establish information systems to monitor medicine availability, pricing and affordability periodically. 4 To ensure healthy lives and access to safe, effective, quality and affordable essential medicines and vaccines, policymakers and health managers working towards achieving the Sustainable Development Goal (SDG) 3 need reliable information systems. 1 Utilizing digital technologies to improve pharmaceutical supply chains is therefore crucial to support access to quality medicines in low- and middle-income countries (LMICs) and achieve SDG 3. The Christian Health Association of Ghana (CHAG) and PharmAccess Foundation, together with the Ghana Food and Drugs Authority (FDA), have since 2017 collaborated to establish a digital pharmaceutical supply chain management system (Med4All). Med4All aims to facilitate a secure supply of quality essential medicines at competitive prices by creating smart supply chain solutions, implementing quality control mechanisms, and offering access to affordable finance. It thus seeks to increase the quality, affordability, availability and accessibility of essential medicines to patients patronizing CHAG facilities. 5
In the design architecture for Med4All, several processes guarantee the quality of medicines from production to the final consumer. One involves the post-market quality monitoring of medicines in the Med4All participating facilities. Our previous research used TruScan RM (for all samples) and GPHF-minilab (a subset of the samples) to determine the quality of some frequently used medicines in selected CHAG institutions’ healthcare facilities to be empanelled on the Med4All platform. 6 It was observed that 100% of the tested samples were within standards (at least qualitatively).
The purpose of this current study was to explore and evaluate the quality of additional selected drugs, particularly those for non-communicable diseases (NCDs). This was considered important due to the ever-increasing NCD burden in Ghana and the concomitant high demand for essential medicines for its management. 7 Also, the study assessed the quality of medicines used in selected Med4All and non-Med4All CHAG facilities using low-medium field digital technologies such as the TruScan analyzer and GPHF-Minilab (screening methods) for all samples and pharmacopoeia tests (confirmatory method) for a subset of the samples. The study also contributes to the knowledge of drug quality studies using post-market surveillance, providing insights into the quality of medicines in resource-limited primary healthcare facilities.
Methods
Study design
This cross-sectional study was conducted between July 3 and July 15, 2023, and adopted a two-prone testing approach to assess the quality of the drug samples. This included (1) two field screening technologies and (2) pharmacopoeia quality assessment (confirmatory method). Firstly, the screening methods, that is, TruScan Raman (RM) analyzer and GPHF-minilab, were used to assess the quality of all the drug samples. Next, a subset of the drug samples was tested using pharmacopoeia methods. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline was adopted for this study (Table S1). 8
Study site selection
The study was conducted in 24 CHAG health facilities in Ghana, the list of which is indicated in Table S2. The inclusion criteria for health facilities sampling were (1) registered CHAG facilities, (2) facilities enrolled in the digital pharmaceutical supply chain management system (Med4All), or facilities potentially earmarked for participation.
A multistage sampling approach was used to select 12 Med4All and 12 non-Med4All facilities. The facilities have been enrolled on the Med4All platform for a few weeks to less than three years. The first sampling stage reflects representation from Ghana's three main ecological zones, that is, northern, middle and southern belts. Two regions were selected from each zone based on the availability of Med4All health facilities. For the northern zone, Savanna and Northern regions were selected. Ashanti and Bono regions were selected for the middle belt, while Western and Greater Accra regions were selected from the southern zone. In each region, two Med4All facilities and two non-Med4All facilities were selected.
To enable study interpretation to be informed by other previous and ongoing research and intervention activities being jointly carried out by CHAG and PharmAccess Ghana (PAG), the list of facilities was also drawn based on the additional criteria that these facilities were part of the PAG Ghana-CHAG, 2017 supply chain survey.
In addition, medicine suppliers who provide services on the Med4All platform were selected to obtain drug samples as references for the TruScan Raman (RM) analyzer.
Medicines selection
A maximum of ten medicines were sampled from the selected Med4All and non-Med4All CHAG facilities and medicine distributors for the Med4All programme. The inclusion criteria for medicines sampling were (1) medicines that were part of a previous study (lisinopril, metformin, artemether/lumefantrine, ciprofloxacin, diclofenac), 6 (2) medicines commonly used in CHAG facilities for NCDs (amlodipine, atenolol, glibenclamide, lisinopril, metformin), antimalarials (artemether/lumefantrine), antibiotics (azithromycin, ciprofloxacin), non-steroidal anti-inflammatory drugs (NSAIDs) (diclofenac) and a hematinic (folic acid).
The medicines were collected randomly from the facilities by a field team. For each drug product, 60 dosage units (tablets, capsules, caplets) were sampled, adequate for the screening and confirmatory analysis. The samples were transported in the original package, including the package leaflet, and kept for analysis using the GPHF-minilab and/or high-performance liquid chromatography (HPLC). The TruScan Raman (RM) analysis was performed on-site during sampling. The samples were obtained using an appropriate packaging material where the original package was unavailable. A unique code number is used to identify each sample product and show the strength and dosage form to avoid mix-ups. Adequate information on the sample product was gathered during sampling before testing. Information includes the product type, sampling level, sampling date, sample sequential number, ecological zone, region and facility type from which products were sampled (Table S2).
Health facilities and medicines inspection
The availability of thermohygrometers was checked in the health facilities. Also, their calibration by the Ghana Standards Authority (National Standards Authority in Ghana) was checked to ensure accurate temperature and relative humidity measurement.
The country of manufacture, the dosage form, batch numbers, expiry dates and Ghana FDA product registration numbers were checked for each drug product packaging container.
Truscan Raman (RM) analysis
The TruScan Raman analyzer was used to analyze the drug product samples per the manufacturer's instructions, as previously described. 9 The spectra were generated for the drug products using the instrument's built-in method without modification. Each drug product (tablet, capsule, caplet) was placed in the sample compartment, from which a spectrum is generated by emitting a laser beam from the device, which is placed in contact with the sample compartment. A reference spectrum was generated initially for each drug product and used for comparison with the spectra of the corresponding drug product samples. The results of each drug product test indicated either a weak pass, strong pass, weak fail or strong fail.
GPHF-minilab analysis
The GPHF-minilab Kit was used to analyze the drug product samples per the manufacturer's instructions10,11 and as previously described.12,13 The GPHF-minilab kit contains the equipment, reference standards and a manual of procedures for basic tests such as thin-layer chromatography (TLC). The GPHF-minilab analysis includes visually inspecting the dosage form, disintegration test and TLC. Folic acid drug samples were not analyzed since a GPHF-minilab method had yet to be developed.
Pharmacopoeia analysis
The pharmacopoeia analysis of the drug samples was performed at the Ghana FDA Centre for Laboratory Services and Research (CLSR). The CLSR is accredited to the ISO/IEC 17025:2017 by the ANSI (American National Standards Institute) National Accreditation Board (ANAB) and prequalified by the World Health Organization (WHO) as a Good Practices for Pharmaceutical Quality Control Laboratory (GPPQCL).
Pharmacopoeia test methods from the United States Pharmacopoeia (USP), and British Pharmacopoeia (BP) monographs were used.14,15 When an in-house specification was used for a drug product, a validated analytical method from the manufacturer already transferred to the CSLR was used for the quality assessment of the drug products. The pharmacopoeia testing includes identity, uniformity of weight/uniformity of dosage units, the content of each active ingredient and dissolution.
The identity of the drug products was determined using either infrared (IR) spectroscopy, ultraviolet-visible (UV) spectroscopy, TLC, or HPLC as described in the respective pharmacopoeia monograph or manufacturer's method. The uniformity of weight or uniformity of dosage units was determined using the BP or USP. However, the uniformity of weight/uniformity of dosage unit test was not applicable for ciprofloxacin and lisinopril tablets. The assay of each drug sample was performed using HPLC. The dissolution was performed using either the basket or the paddle method, as well as UV or HPLC, to determine the amount of drug release.
Materials
The following materials were used for the TruScan, Minilab and pharmacopoeia analysis of the medicines: TruScan RM (Model #: TruScan RM, SN #: TM6512, Thermo Fisher Scientific, USA), Global Pharma Health Fund (GPHF) Minilab Kit (Merck, Germany), HPLC (1260 Infinity Series, Agilent Technologies, Germany), Erweka DT 800 dissolution tester (Erweka, Germany), Amlodipine besylate RS (Lot #: R09060, USP), Artemether RS (Lot #: 3.0, EDQM), Atenolol (Lot #: R101E0, USP), Azithromycin (Lot #: R103C0, USP), Ciprofloxacin hydrochloride (Lot #: R05170, USP), Diclofenac sodium (Lot #: R038E0, USP), Folic acid (Lot #: R056H0, USP), Glyburide (Lot #: R157E0, USP), Lisinopril (Lot #: J1F043, USP), Metformin hydrochloride (Lot #: R069H0, USP), Lumefantrine (Lot #: 2.0, EDQM), Methanol (Fischer, UK), Acetronitrile (MERCK, Germany), Potassium Dihydrogen Phosphate (VWR, Spain), Hydrochloric acid (VWR, France), Triethylamine (VWR, USA), Phosphoric acid (Park Scientific, UK), Sodium hexanesulphonate (VWR, UK), Sodium hydroxide (Merck, Germany), Ammonia (VWR, France), Propan-1-ol (Fischer, UK), Ethanol (Merck, Germany), Disodium hydrogen phosphate (VWR, Germany), Glacial Acetic Acid (Fisher, UK), Ethyl Acetate (Merck, Sweden), Ninhydrin (Fluka, Czech Republic), Tribasic Sodium Phosphate (SCP, UK) and Sodium dihydrogen Phosphate (Merck, Germany).
Ethics
The Institutional Review Board of CHAG approved the study with approval number CHAG-IRB01052023.
Results
Health facilities and drug product characteristics
Among the 24 health facilities, 11 (46%) were health centres/clinics and 13 (54%) were hospitals (Table 1).
Health facilities and drug product characteristics.

FDA registration number not indicated on the product packaging material.
Of the 24 health facilities, only five (21%) had thermohygrometers for monitoring temperature and relative humidity. Among the five facilities with thermohygrometers, two had their thermohygrometers calibrated. All five facilities with thermohygrometers recorded temperature and relative humidity regularly except one, which had no recordings of relative humidity (Table 1 and Table S2).
The drug products obtained from the health facilities were manufactured mostly in Ghana (150/199, 75%) and India (32, 16%) (Table 1). The rest were manufactured in the United Kingdom (UK) (8, 4%), Poland (3, 2%), Slovenia (3, 2%), and one each from China, Germany, and the Republic of Macedonia (Table 1). The dosage form of the drug products was predominantly tablets (190, 95%) with a few capsules (5, 3%) and caplets (4, 2%) (Table 1). All the drug products had batch numbers and expiry dates. The far majority, 191 (96%) drug products had manufacturing dates. It was observed that 143 (72%) products were registered with the Ghana FDA, while 56 (28%) were not (Table 1).
The highest and lowest drug products obtained were 10 and 3, respectively (Table S3). The number of drug products obtained from each health facility in the middle and southern sectors averaged 9.4 ± 0.5 and 9.1 ± 0.8, respectively (Table S3). The northern sector had the lowest number of drug products per health facility, averaging 6.4 ± 2.2. The Savannah region had the least drug products per health facility, averaging 5.8 ± 3.1 (Table S3).
Quality assessment of drug products using screening methods
A total of 183/199 drug samples were analysed using TruScan™ Raman (RM) analyzer, while 16/199 were not. Of the 183 samples, 159 (87%) passed, while 24 (13%) failed. All samples from artemether/lumefantrine, azithromycin, ciprofloxacin and metformin passed (Table 2 and Table S4).
Truscan test results per drug product type.

Of the 99 samples obtained from 12 CHAG facilities using the Med4All logistic platform, 87 (87.9%) passed, while 12 (12.1%) failed. On the other hand, 72/84 (85.7%) samples from 12 facilities not using the Med4All logistic platform (Non Med4All) passed, while 12/84 (14.3%) failed (Table 2).
The quality of 177/199 drug samples was analyzed using GPHF-minilab. From the GPHF-minilab results, 156/177 (88%) passed all test parameters, such as visual inspection, TLC and disintegration, while 21/177 (12%) failed one or more of the test parameters (Table 3 and Table S4). Also, all samples from amlodipine, artemether/lumefantrine, ciprofloxacin and glibenclamide passed the GPHF-minilab test parameters (Table 3 and Table S5).
Minilab test results per drug product type.

na: not applicable.
The GPHF-minilab results showed that 79/91 (87%) samples from facilities using the Med4All logistic platform passed, while 12/91 (13%) failed. For samples from facilities not using the Med4All logistic platform, 77/86 (90%) passed, while 9/86 (11%) failed (Table 3).
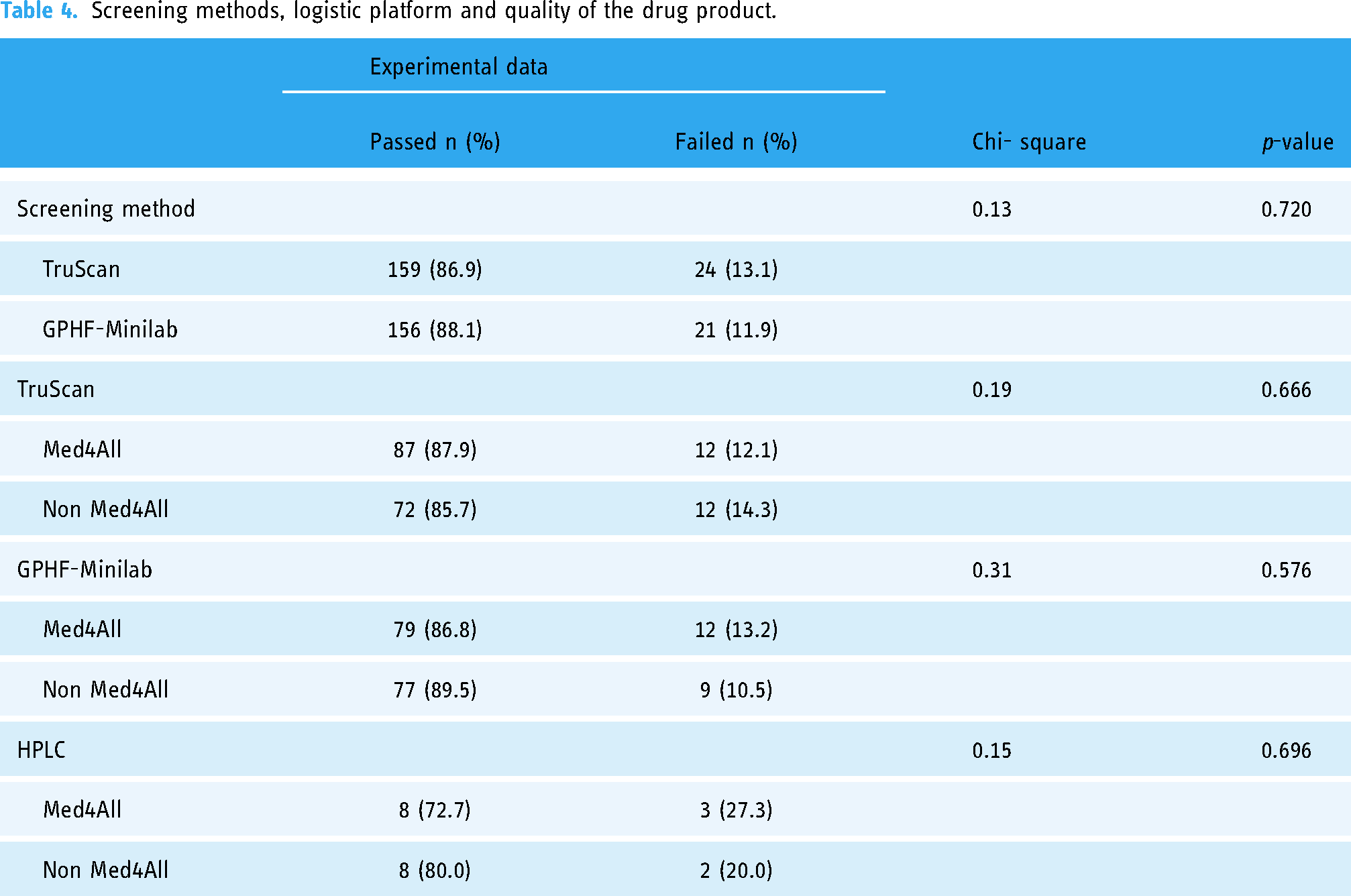
The quality of the drug products determined using the TruScan™ Raman (RM) analyzer was compared with the GPHF-minilab. There was a marginal variation between the pass rate of the drug products using the TruScan™ Raman (RM) analyzer (87%) and GPHF-minilab (88%) (Table 4). However, there is no significant difference between the two field screening technologies for assessing the quality of the drug products (p > 0.05) (Table 4).
Screening methods, logistic platform and quality of the drug product.
Pharmacopoeia quality assessment of the drug products
The drug samples were assayed using pharmacopoeia HPLC methods. Of the 21 drug samples, 5 (24%) failed, and 16 (76%) passed the pharmacopoeia test (Table S6). All the samples passed the identification and uniformity of weight/uniformity of dosage unit tests (Table S5). The five samples failed either the dissolution (diclofenac (1/3), azithromycin (1/2), glibenclamide (1/2)) or the assay and dissolution tests (diclofenac sodium (1/3), glibenclamide (1/2)) (Table S6).
Compared to the HPLC method (gold standard), the performance of the TruScan™ Raman (RM) analyzer and the GPHF-minilab technologies was 15/21 (71%) and 12/19 (63%), respectively (Figure 1). Four out of the five NCD drugs tested passed the pharmacopoeia analysis (Table S6).

Summary of quality assessment of medicines. Venn diagram representation of TruScan™ Raman (RM) analyzer, GPHF-minilab and HPLC methods. The numbers in the bubbles of the Venn diagram represent test results from the screening and confirmatory methods.
Effect of the logistic platform on the quality of the drug products
There was no association between the logistic platform (Med4ALL or Non Med4ALL) and the quality of medicines (p > 0.05) using either TruScan™ Raman (RM) analyzer, GPHF-minilab or HPLC method (Table 4).
Discussion
Regional distribution of CHAG facilities and availability of tracer medicines
CHAG is Ghana's second largest health service provider, delivering ∼40% of national health service needs to the most vulnerable and underprivileged population groups in mostly remote areas with no public health facilities and with the urban poor.16–18 Due to the multistage sampling level for this study, the percentage of health centres/clinics and hospitals selected from the CHAG facilities did not reflect the current distribution of ∼67% and 33% for health centres/clinics and hospitals, respectively. 18 An average of 10 drug products were expected to be obtained from each health facility. Since the availability of medicines is relatively higher in hospitals than in health centers,19,20 this may account for the lowest number of drug products per health facility obtained from the northern sector, mainly health centres/clinics.
Most drug products (75%) from the health facilities were manufactured locally, reflecting the inclusion of 4/10 of the drug products on the list of drugs reserved for local production. 21
CHAG facilities in the southern and middle sectors had above 90% of the expected ten tracer medicines compared to around 64% for facilities in the northern sector. This agrees with the availability of tracer medicines in Ghana Health Service facilities in which the regions from the northern sector were within 63–69%, and 75–93% for regions within the middle and southern sectors. 22 The registration status of ∼72% of the drug products is along the lines of the WHO-reported 86% in six countries of sub-Saharan Africa. 23
Medicines quality in the CHAG facilities and health implications
Our various screening methods showed a little above 10% of the medicines were substandard. Although this agrees with the current reports that one out of ten medicines in LMICs is substandard or falsified,24,25 screening methods have challenges with accuracy and specificity.13,26 A total of 183/199 drug samples were analysed using TruScan™ Raman (RM) analyzer, while 16/199 were not since reference samples were not obtained from the manufacturers or suppliers. Ideally, all failed samples, and ∼10% of passed samples after screening should be subjected to pharmacopoeia testing. 23 However, due to budget constraints, HPLC could only be performed on 21/199 samples (∼11%). Notwithstanding, this is sufficient to draw meaningful conclusions since other studies performed confirmatory testing using 3% 27 and 40%, 28 respectively.
The failure rate of ∼24% using the pharmacopoeia method (gold standard) are lower than the WHO-reported ∼30% and ∼40% substandard drugs in six countries of sub-Saharan Africa and Ghana, respectively. 23 Some or all diclofenac, azithromycin and glibenclamide drug products failed pharmacopoeia analysis, which agrees with previous studies.27,29–31 Substandard antibiotics such as azithromycin may lead to the emergence of resistance, transmission of resistant microbes, bystander selection and negative on combination therapy.32–34 Likewise, substandard glibenclamide will adversely affect the management of diabetes, an NCD. 31
Effect of supply chain platform on the quality of medicines
Inadequate supply chain systems are key contributors to the increased prevalence of substandard medicines.5,25 Since the medicines were sampled from CHAG facilities, either using the Med4All logistic platform from PharmAccess or not, the TruScan™ Raman (RM) analyzer results were also considered based on the logistic platform. This study shows that participation in Med4All did not differ significantly in drug quality using the screening or pharmacopoeia methods relative to non-Med4All facilities. Therefore, involvement in Med4All does not correlate with better drug quality.
This is not surprising since the health facilities on the Med4All platform could still procure medicines outside the Med4All platform, which can compromise the quality of medicines in such facilities. The actual effect of Med4All intervention can only be measured in a follow-up study after a substantial implementation time, in which facilities exclusively procure medicines using the platform with a substantial market share. A future study should use health facilities outside CHAG as its control.
Study limitations
The study acknowledges some limitations. First, the non-homogenous nature of selected health facilities introduced variability in drug product samples obtained from them. For example, since health facilities in the northern sector were mainly health centres/clinics, low numbers of drug products were obtained. Focusing on only CHAG healthcare facilities in Ghana may have introduced some investigator bias. The findings are, therefore, reflective of primary healthcare levels of care.
Second, the number of tracer medicines analysed was not exhaustive and therefore the study results may not adequately represent the wide range of medicines used in the various facilities. However, based on the frequently managed conditions at primary healthcare facilities, the list included commonly prescribed NCDs medicines, antimalarials, antibiotics, NSAIDs and hematinics.
In addition, due to financial limitations, not all failed medicines were tested using pharmacopoeia methods after the screening processes. Therefore, the prevalence of substandard medicines reported for this study cannot be generalized.
Conclusions
Digital screening methods could be relevant for the quality assessment of medicines in resource-limited jurisdictions. Considering the complementary nature of the TruScan™ Raman (RM) analyzer and GPHF-minilab, similar future studies can use both screening methods.
Supplemental Material
sj-doc-1-dhj-10.1177_20552076241299064 - Supplemental material for Quality assessment of medicines in selected resource-limited primary healthcare facilities using low- to medium-cost field testing digital technologies
Supplemental material, sj-doc-1-dhj-10.1177_20552076241299064 for Quality assessment of medicines in selected resource-limited primary healthcare facilities using low- to medium-cost field testing digital technologies by Kwabena FM Opuni, Gifty Sunkwa-Mills, Maxwell A Antwi, Antoinette Squire, George Y Afful, Tobias F Rinke de Wit and Irene A Kretchy in DIGITAL HEALTH
Supplemental Material
sj-xlsx-2-dhj-10.1177_20552076241299064 - Supplemental material for Quality assessment of medicines in selected resource-limited primary healthcare facilities using low- to medium-cost field testing digital technologies
Supplemental material, sj-xlsx-2-dhj-10.1177_20552076241299064 for Quality assessment of medicines in selected resource-limited primary healthcare facilities using low- to medium-cost field testing digital technologies by Kwabena FM Opuni, Gifty Sunkwa-Mills, Maxwell A Antwi, Antoinette Squire, George Y Afful, Tobias F Rinke de Wit and Irene A Kretchy in DIGITAL HEALTH
Footnotes
Acknowledgements
The research acknowledges CHAG, Ghana FDA, field workers, study participants and heads of various institutions for their support in this project.
Contributorship
KFMO, GSM, MAA, AS, GYA, TFRW and IAK conceived and designed the study. KFMO and IAK organized and analyzed the data. KFMO and IAK wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
Data availability
The data collected is either part of the manuscript or the supplemental information.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PharmAccess Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
