Abstract
Objectives
To provide a comprehensive review of the use of electronic patient-reported outcomes measures (ePROs) as digital health tools to assess health-related quality of life (HRQoL) in women with breast, ovarian, cervical, and endometrial cancers.
Methods
A systematic review was conducted to identify studies that used ePROs to evaluate HRQoL in women diagnosed with breast and gynecological cancers. The review followed the 2020 update of the PRISMA guidelines and a pre-registered protocol in PROSPERO (CRD42024516737). Inclusion criteria encompassed studies focusing on ePROs for HRQoL assessment in the specified cancers, without language restrictions, and published between January 2000 and December 2023. Studies were retrieved from PubMed, Web of Science, and Scopus. Two reviewers independently screened titles, abstracts, and full texts to identify eligible studies.
Results
The search yielded 4978 articles. After removing duplicates, 900 articles were assessed for eligibility by screening the titles and abstracts. After screening the full text of 168 articles, a total of 16 studies were included in this systematic review. These studies were mainly conducted in Europe and the Americas and included different study designs such as randomized controlled trials (four articles), prospective studies (seven articles), and feasibility and validation studies (five articles). The majority of the studies focused on breast cancer (87.5%), with fewer studies addressing ovarian and cervical cancers. A variety of ePRO tools were used, including the FACT and EORTC QLQ. Findings show that ePROs enhance therapeutic management, treatment adherence, and HRQoL through improved symptom monitoring and communication between patients and providers.
Conclusion
The integration of ePROs in oncology care facilitates a patient-centered approach, enhances communication between patients and healthcare providers, and supports personalized treatment strategies. These findings underscore the importance of incorporating ePROs into routine cancer care to improve overall patient outcomes and HRQoL.
Introduction
Gynecological and breast cancers are a major public health concern worldwide because of their prevalence and their impact on women's reproductive organs and other parts of the body. 1 According to the International Agency for Research on Cancer (IARC), in 2020, more than 2.2 million women were diagnosed with breast cancer, making it the most common cancer among women worldwide. Similarly, almost 662,000 women were diagnosed with endometrial cancer, over 420,000 with cervical cancer, and more than 324,000 with ovarian cancer. The number of deaths recorded in the same year was significant, with more than 666,000 women succumbing to breast cancer, almost 349,000 to endometrial cancer, around 98,000 to cervical cancer, and nearly 207,000 to ovarian cancer. 2
The high incidence and mortality rates associated with these cancers underscore the importance of comprehensive cancer control programs, including vaccination, early detection through screening, access to quality treatment, and ongoing research to advance our understanding and management of these diseases.3,4 In recent years, advances in medicine have significantly improved the therapeutic management of women's cancers. Several treatment modalities are available, depending on factors such as the specific type of cancer, its stage, individual patient characteristics, and medical practices.5,6 This therapeutic progress has significantly contributed to an increase in life expectancy. 7 However, despite the rise in survival rates, women undergoing treatment may experience side effects across physical, somatic, psychological, and social dimensions, raising concerns about their health-related quality of life (HRQoL).8,9
Patient-reported outcome measures (PROMs) serve as a vital tool in evaluating health-related quality of life (HRQoL) by directly capturing patients’ perceptions of their health and well-being. 10 The use of PROMs contributes to a more patient-centered approach to clinical decision-making and an overall improvement in the quality of healthcare. 11 In the era of evolving information and communication technologies in healthcare, PROMs have undergone a major transformation with the advent of digital health devices. Electronic patient-reported outcome (ePRO) tools are designed to collect patient voices via information technology (IT) platforms, such as mobile applications, online portals, or electronic health record management systems. ePROs provide a digital approach to PROMs data collection, enabling more efficient capture, transmission, and analysis of patient-reported information. 12 The integration of ePROs in oncology is emerging as a crucial tool for improving patient care, enhancing communication between patients and healthcare providers, and ensuring continuous monitoring of symptoms and quality of life throughout the treatment pathway.13–16
To date, there has been no comprehensive review detailing the utilization of ePROs in the context of female cancers. This review aims to fill this gap by meticulously reporting how ePROs are used and analyzing their impact on the assessment and management of HRQoL in various types of female cancers. The findings hold significant implications for clinicians, researchers, and decision-makers at various levels of the healthcare system, as they shed light on the implementation and effectiveness of ePROs in female cancer care.
Methods
We conducted a systematic review to identify the use of electronic patient-reported outcomes measures (ePROMs) and health-related quality of life (HRQoL) in women with breast, ovarian, endometrial, or cervical cancer. The review was conducted following the 2020 update of Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) (Supplementary file 1) 17 and in accordance with a protocol registered in PROSPERO (CRD42024516737). 18
Eligibility criteria
The predefined inclusion criteria for selection of studies were as follows: (1) cohort, case-control, randomized controlled trials (RTCs), or cross-sectional studies; (2) patients diagnosed with breast cancer, ovarian cancer, cervical cancer, and/or endometrial cancer; (3) electronic patients-reported outcomes (ePROs); (4) health-related quality of life (HRQoL) evaluation; (5) without language restriction; (6) published from January 2000 to December 2023. Following the first search, duplicates were removed with the “revtools” R package. 19 Each reviewer independently confirmed that the electronic search captured all relevant articles, by reviewing the reference lists of key studies and discussing any potential missing studies together, ensuring that no eligible studies were inadvertently excluded.
Studies were excluded if the PROs were not evaluated using an electronic method, included patients with none of the four types of cancers cited above, were qualitative studies, or the outcome assessed was not health-related quality of life.
Information sources and search strategy
Two reviewers systematically searched the following databases in February 2024: PubMed, Web of Science, and Scopus.
The research question was developed according to the PICOs statement (population, intervention/exposition, control, and outcomes). 17 Therefore, corresponding free terms were combined in a research equation as follows: (“electronic Patients-Reported Outcomes” OR “ePROs” AND “health-related quality of life” OR “HRQoL” OR “QOL” AND “gynecological cancer” OR “breast cancer” OR “ovarian cancer” OR “cervical cancer” OR “endometrial cancer”).
Data extraction and quality assessment
After duplicate removal, an initial screening of titles and abstracts was carried out using the Rayyan screening system. 19 All identified studies were independently assessed for eligibility by pairs of reviewers (AB/AA, IYM/MK, AM/MB). Uncertainty or disagreements were resolved by a third reviewer (CN, SB, MT). Then, the full text of potentially eligible studies was evaluated by the same pairs. Any disagreements were resolved through discussions with a third reviewer (CN, SB, MT). The references cited in the included articles were also examined for potentially eligible studies that might have been missed in the search strategy.
Data extraction was performed independently by two reviewers (AB and AA). The following information was extracted from the included studies in a standardized form: first author and the year of publication, country/continent, the aim of the study and the participants, type of cancer, the application name, the instrument used for the evaluation of the QoL, and finally the findings.
Two authors (AB and AA) assessed the methodological quality of the included studies using three tools: the Jadad scale for reporting randomized controlled trials (three items), 20 and The National Heart, Lung, and Blood Institute (NHLBI) Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies (14 items), 21 and the Mixed Methods Appraisal Tool (MMAT) for evaluating feasibility, proof-of-concept, reliability, and validation studies (five criteria). 22
Results
Our search strategy evaluating the use of ePROs as a measure of the health-related quality of life in women with four types of cancer (breast, cervical, endometrial, and ovarian) identified 4978 articles (674 in PubMed, 2197 in Web of Sciences, and 2107 in Scopus). After removing 4078 duplicates, we screened the titles and abstracts of 900 unique studies. After this initial screening, the 168 remaining articles were checked for eligibility based on full-text assessment. Finally, a total of 16 studies were eligible for this systematic review (Figure 1).

Flow chart showing the methodology for selecting articles on the ePROs in gynecological cancer.
Characteristics of the included studies
Table 1 summarizes the characteristics of the 16 studies included in this systematic review. Publication dates ranged from 2017 to 2023. Ten out of 16 (63%) studies were conducted in Europe and 25% (4 out of 16) in America. However, only one study was conducted in Oceania and one in Asia. Of the 16 studies included in the final analysis, five were described as randomized controlled trials, three were prospective, two were pilot studies, two were feasibility studies, two were reliability studies, one was a proof of concept, and one was a cross-sectional study.
Basic characteristics of the included articles.
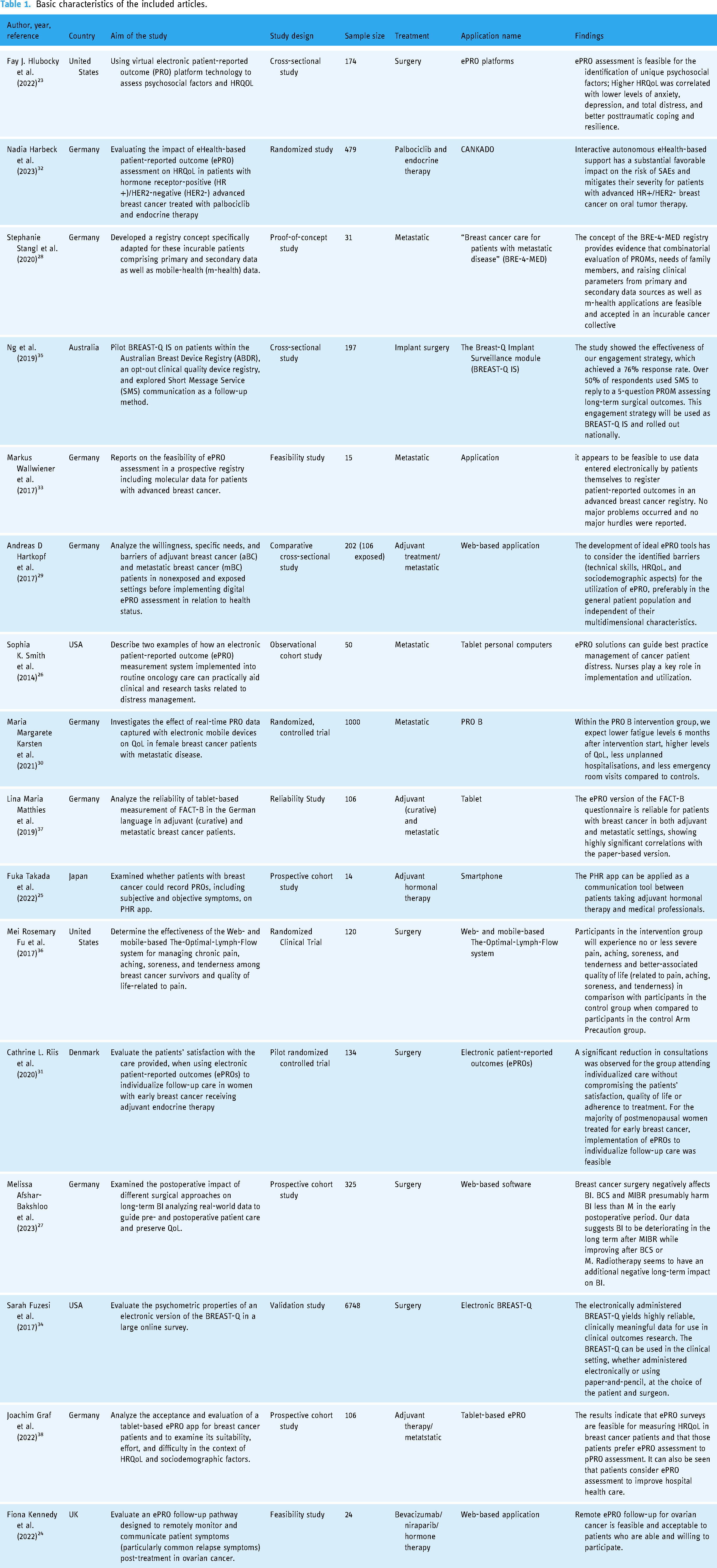
Characteristics of the study population
Fourteen of the included articles used ePROs exclusively for patients with breast cancer. Meanwhile, the remaining two studies used ePROs for ovarian cancer23,24 (Table 2). The number of participants varied significantly, ranging from 14 to 6748 across the different studies. The total number of patients was 9727, of whom 7698 (79.1%) underwent surgery, 517 (5.3%) underwent hormone therapy, 1098 (11.3%) had metastatic cancer and 414 (4.3%) included both patients receiving adjuvant treatment and those with metastatic cancer.
The type of cancers studied in the included articles.

 Type of cancer included in the study;
Type of cancer included in the study;  Type of cancer not included in the study.
Type of cancer not included in the study.
Type of ePRO tool used
The ePRO tools used in the 16 studies are detailed in Figure 2. The most commonly used ePRO questionnaire was the Functional Assessment of Cancer Therapy (FACT), which was used in seven studies (FACT-G, n = 1; FACT-B, n = 4; FACT-O, n = 2).23–26 The second most commonly used ePRO questionnaire was the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ), which was used in six of the included studies (EORTC QLQ-C30, n = 4; EORTC QLQ-BR23, n = 1; EORTC QLQ-2, n = 1).24,27–31 Other commonly used ePRO questionnaires were the EuroQol-5D (EQ-5D, n = 3),24,32,33 Breast Q (n = 2),34,35 and Quality of Life in Adult Cancer Survivors (QLACS) (n = 1). 24 Seven studies used ePRO tools other than specific quality of life measures, including Hospital Anxiety and Depression Scale (HADS), 23 International Physical Activity Questionnaire (IPAQ), 33 Center for Epidemiological Studies of Depression (CES-D), 33 Patient Health Questionnaire (PHQ-9), 26 Pain Impact Questionnaire (PIQ-6), 36 the Patient Experience Questionnaire (PEQ), 31 and Patient Activation Measure (PAM). 24
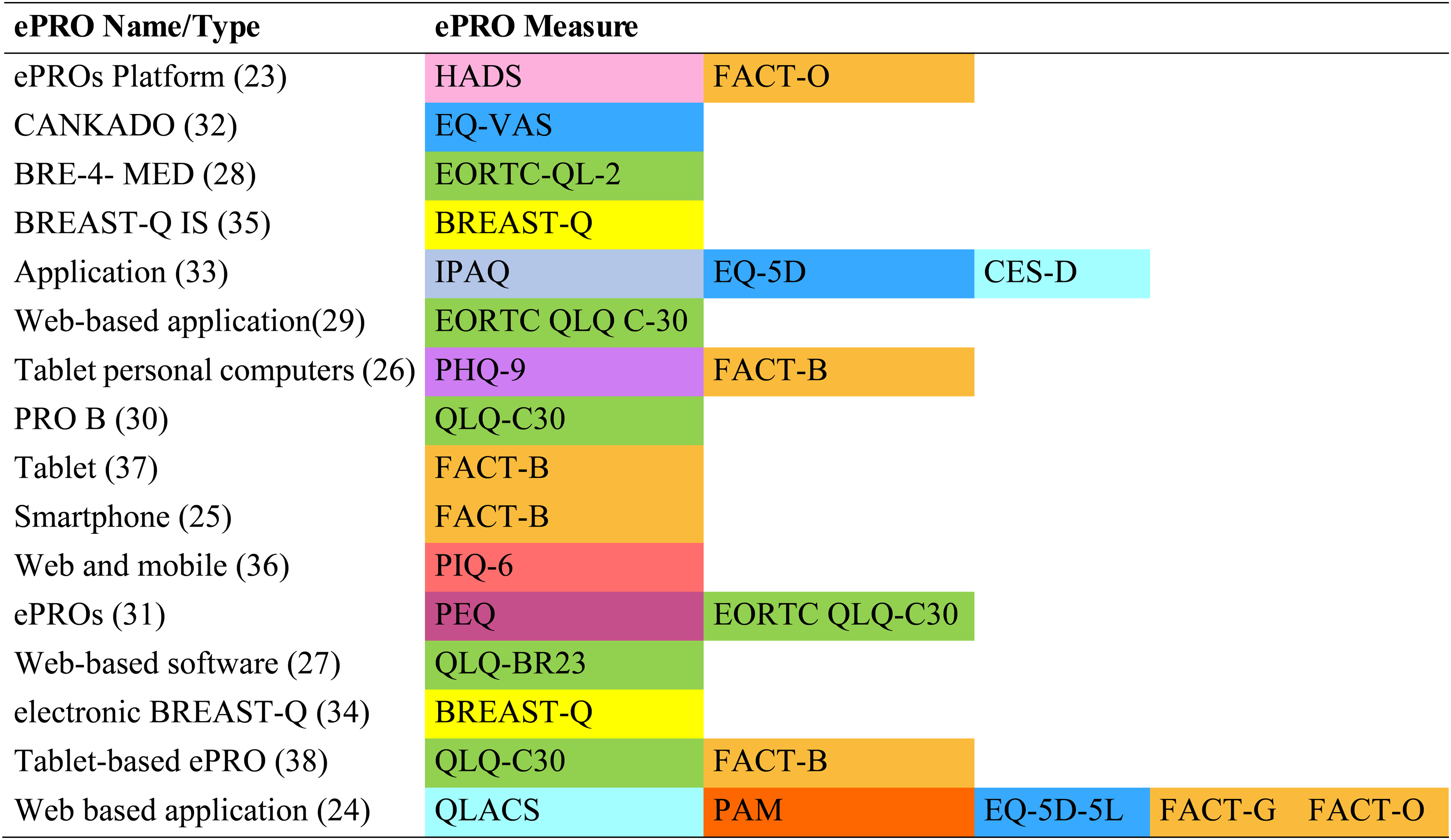
ePRO tool used as a measure of the health-related quality of life in women's cancers.
Data collection method
Regarding the collection method, six studies reported using a web-based assessment method to collect ePROs, the tablet was used in four studies, and e-health applications in two studies. Each of the following has been used in only one study: software system, SMS, Web and Mobile-Based assessment, and platform (Figure 2). Furthermore, among the 16 studies reviewed, 8 utilized remote data collection methods. Three studies collected data in-clinic or at the hospital, while the five other studies employed a mixed approach, combining both in-clinic and remote methods.
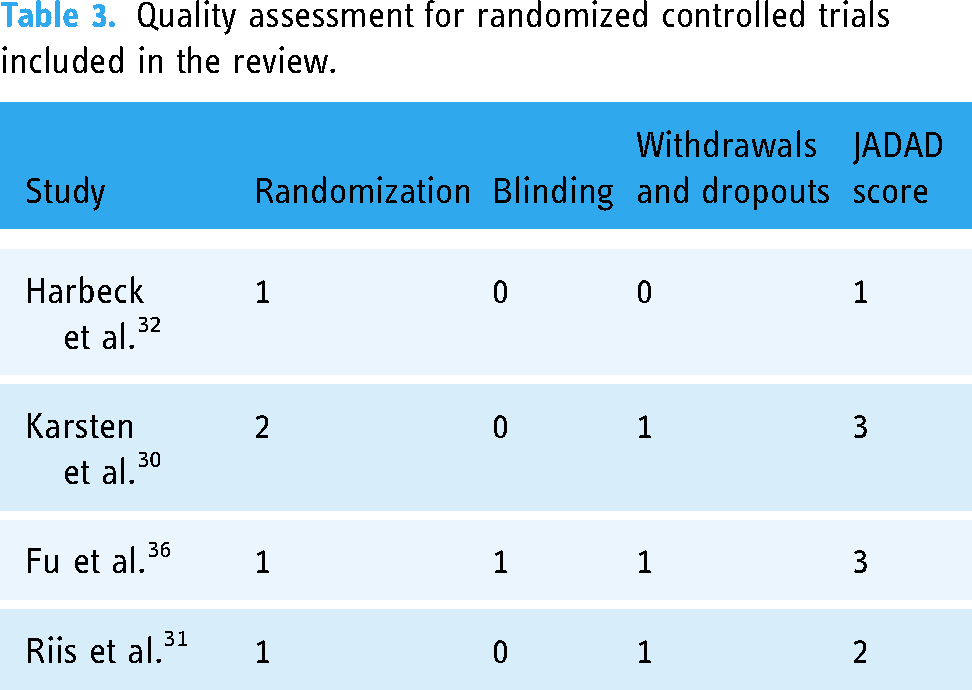
Quality assessment for the included studies
Table 3 presents the quality assessment of the four randomized controlled trials (RCTs) included in this review. Based on the JADAD score, the methodological quality was moderate in two studies30,36 and low in two others.31,32
Quality assessment for randomized controlled trials included in the review.
Regarding the cross-sectional and cohort studies, the NHLBI quality assessment tool indicated that two studies (28.6%) exhibited good methodological quality,23,35 whereas five others (71.4%) demonstrated fair methodological quality24–27,29 (Table 4). The primary weaknesses identified across these studies were the lack of sample size justification and power analysis, as well as the absence of long-term follow-up data in cohort studies.
Quality assessment for cross-sectional and cohort studies included in the review.

C/D, cannot determine; N/A, not applicable; N/R, not reported.
Table 5 presents the quality assessment of the five studies included in this review, evaluated using the MMAT. Among the two feasibility studies,24,33 both demonstrated clear objectives and appropriate methods, though limitations in sample representativeness were noted. The proof-of-concept study 28 and the validation study 34 both exhibited strong methodological rigor, with representative samples and well-supported findings. The reliability study 37 was also robust, though its sample size was somewhat limited in representativeness. Overall, these studies provide reliable evidence on the feasibility, reliability, and validation of electronic patient-reported outcome tools in clinical settings.
Quality assessment for feasibility, proof-of-concept, reliability, and validation studies included in the review.

Results of ePROs
Feasibility, reliability, and acceptability of ePROs
Four studies examined the feasibility and reliability of the ePRO tools in the field of women's cancer (Table 1). These studies revealed positive adoption and high acceptability of ePROs. In the PRAEGNANT registry study, which focused on advanced and metastatic breast cancer, 15 out of 17 participants successfully completed the ePRO questionnaires, demonstrating good compliance and ease of use. 33 Another study, the electronic version of the FACT-B questionnaire for breast cancer patients showed high reliability, with strong correlations between the electronic and paper-based formats. 37 The BREAST-Q study, involving over 6748 women, confirmed that the electronic version of the questionnaire produced consistent and reliable data compared to its paper-based counterpart. A prospective two-center trial assessing an ePRO system for monitoring HRQoL in breast cancer patients reported high levels of acceptability among participants. 34 In ovarian cancer patients, ePRO tools used for symptom monitoring achieved compliance rates between 75% and 82% over the study period, indicating their feasibility in a remote follow-up setting. 24
Contribution of ePROs to therapeutic management, treatment adherence, and HRQoL improvement
The studies analyzed in this systematic review highlight the significant impact of ePRO tools on the HRQoL, symptom management, and treatment adherence in women with cancer. The use of ePRO platforms to assess HRQoL and psychosocial experiences reflects a growing trend toward the integration of these technologies for a more holistic assessment of patient well-being. ePROs have the potential to play a key role in improving women's cancer healthcare by providing relevant data on patient voice, improving communication between patients and healthcare professionals, and personalizing care according to individual needs.23,28,29 In addition, interactive e-health systems showed a favorable impact on therapeutic management and the reduction of adverse events, thus contributing to an overall improvement in quality of life.26,30,32 At the same time, the application of e-health technology to improve patient satisfaction and treatment adherence underlines the growing importance of personalized care based on patient-reported outcomes.21,27,32 Overall, these results suggest that the patient voice through ePROs plays a crucial role in improving HRQoL for breast and ovarian cancer patients, by offering a more patient-centered and personalized approach to managing their health.
In some studies, ePROs were also used for continuous symptom monitoring, with some systems triggering real-time alerts when symptoms exceeded set thresholds. These alerts prompted timely clinical actions, such as phone consultations or medication adjustments. 30 In other cases, ePRO data were reviewed during follow-up visits, allowing clinicians to adjust treatments based on evolving symptoms. This real-time feedback improved communication between patients and providers, enhancing symptom management, treatment adherence, and overall HRQoL in women with breast and ovarian cancers.24,25,31
Discussion
The use of ePROs in assessing HRQoL for women with breast or gynecological cancers has shown significant benefits and provided valuable insights into patient experiences and treatment outcomes. This systematic review highlights several key aspects of implementing ePROs in clinical practice, particularly for breast, ovarian, cervical, and endometrial cancers.
The studies reviewed span various countries and continents, showing a broad interest in ePRO implementation across different healthcare settings. For instance, research in the United States explores using ePROs to evaluate psychosocial and HRQoL outcomes among ovarian cancer survivors. 38 In Europe, studies such as Harbeck et al. (2023) 32 from Germany demonstrate the use of interactive eHealth systems to manage adverse events in breast cancer treatment, illustrating regional differences in technology adoption and healthcare practices. 39
A variety of ePRO questionnaires were used across these studies, each tailored to capture specific aspects of HRQoL. The Functional Assessment of Cancer Therapy-Breast (FACT-B) questionnaire, used in studies like Matthies et al. (2019), 37 is an example of a tool designed to measure HRQoL in breast cancer patients. 37 Other instruments, such as the BREAST-Q used by Fuzesi et al. 34 were validated electronically to ensure reliability and effectiveness in capturing the patient's voice. The context of ePRO implementation varied, providing insights into their versatility and applicability in different clinical settings. For example, Smith et al. 4 showed how an electronic system improved distress management in oncology, highlighting the potential for ePROs to facilitate timely clinical interventions. Meanwhile, Kennedy et al. focused on the feasibility of electronic symptom monitoring during ovarian cancer follow-up, emphasizing patient acceptability and the practical challenges of integrating ePROs into routine care. 24
The reliability and acceptability of ePROs in routine clinical practice are well documented. Matthies et al. validated an ePRO tool for the FACT-B questionnaire, confirming its reliability in assessing HRQoL among breast cancer patients. 37 Wallwiener et al. 33 highlighted the feasibility of implementing ePRO data entry within the PRAEGNANT registry, underscoring the practicality of integrating these tools into clinical workflows. Studies also show that ePROs can enhance patient engagement and satisfaction with healthcare. For example, Riis et al. 31 found that individualized follow-up care using ePROs improved patient satisfaction and adherence to treatment protocols.
One primary benefit of ePROs is their ability to enhance HRQoL and provide psychosocial support for cancer patients. Hlubocky et al. 23 found that an ePRO platform significantly improved the evaluation of psychosocial and HRQoL experiences among ovarian cancer survivors, helping healthcare providers address patients’ unique emotional and psychological needs more effectively. 38 Similarly, Harbeck et al. 32 demonstrated that an interactive eHealth system incorporating ePROs positively impacted managing severe adverse events in breast cancer patients undergoing treatment. This suggests that ePROs can play a critical role in monitoring and mitigating treatment-related side effects, enhancing patient well-being. 39
Despite the benefits, several challenges impede the widespread adoption of ePROs. Hartkopf et al. identified significant barriers, including technological literacy, privacy concerns, and the need for user-friendly interfaces. These barriers highlight the necessity of the need to design ePRO systems that are accessible to all patients, regardless of their technological proficiency. 29 Kennedy et al. emphasized the importance of robust support systems to effectively manage and interpret ePRO data, including training of healthcare providers and ensuring that patients have the necessary technological infrastructure, such as reliable internet access and compatible devices. 24
Logistical challenges also arise when integrating ePROs into routine clinical practice. Reviewing and responding to ePRO data can be time-consuming for healthcare providers. Additionally, ensuring that ePROs seamlessly integrate into electronic health records (EHRs) remains a significant challenge. Studies of Graf et al. and Karsten et al. have proposed solutions such as alarm-based monitoring and mobile app interventions to streamline this process, but further research and development are needed to refine these approaches.30,38
Future research should focus on making electronic patient-reported outcome (ePRO) systems more user-friendly and accessible. This means creating easy-to-use interfaces and providing thorough training for users. Hartkopf et al. highlighted the need for ongoing education for both patients and healthcare providers to help them adopt ePROs effectively. 29 We also need more prospective cohort studies and randomized controlled trials (RCTs) to prove how effective and cost-efficient ePROs are in various cancer care settings. Building on Riis et al., 31 additional RCTs could provide solid evidence to support the widespread use of ePROs in clinical practice. These studies should also look at how ePROs affect long-term patient outcomes and healthcare use.45
Advanced analytics and artificial intelligence (AI) can greatly improve ePROs by providing predictive insights and personalized care recommendations. For example, AI can analyze ePRO data to predict adverse events and suggest timely interventions, helping to improve patient outcomes and make clinical workflows more efficient. 39 It's also important to ensure everyone has fair access to ePRO systems by addressing technology access and literacy gaps among different patient groups. We should develop tailored interventions and support to ensure all patients can benefit from ePROs, no matter their socioeconomic status or tech skills.
Advances in technology and data analytics are expected to significantly enhance the utility of ePROs in gynecological oncology. 40 Graf et al. 38 suggest that future research should focus on using AI and machine learning to predict patient outcomes and personalize treatment plans. 38 Additionally, expanding ePROs to cover a wider range of symptoms and side effects specific to gynecological cancers can make them even more useful in clinical practice. Overall, the potential for ePROs in cancer care is exciting, particularly with new technology and data analytics. Future research should keep exploring how AI can improve predictions and personalize treatments, while also expanding ePROs to include a broad range of cancer-related symptoms and side effects.
Despite the promising findings, several limitations should be noted in this review. First, the geographical distribution of the included studies is predominantly from Europe and the Americas, with limited representation from other regions like Oceania and Asia. This imbalance may introduce publication bias and affect the generalizability of the results. Additionally, the variability in study designs, ranging from randomized controlled trials to cross-sectional studies, also affects the consistency and quality of evidence. The lack of long-term follow-up data in many studies restricts the ability to assess the sustained benefits of ePROs on the HRQoL and patient outcomes over time. Moreover, the predominance of studies focusing on breast cancer, with fewer addressing other gynecological cancers, limits insights into the performance of ePROs across various cancer types and demographics. These limitations underscore the need for further research to address these gaps and enhance the utility of ePROs in cancer care.
Conclusion
The ePROs are a powerful tool significantly contributing to the assessment and improvement of HRQo in women with cancers. Ongoing research and technological advancements are expected to further refine these systems, making them even more effective in addressing the unique needs of women with cancers.
The use of ePROs in managing women's cancers represents a significant patient voice and patient-centered care. While the benefits are evident, continued efforts are needed to overcome implementation challenges and optimize these systems for broader use. As digital health technologies evolve, ePROs will likely play an increasingly vital role in improving the outcomes and experiences of women with cancer. Incorporating patient feedback through ePROs improves the quality of care by empowering patients and supporting a more comprehensive approach to cancer treatment and survivorship. Future research should aim to enhance the accessibility, reliability, and integration of these systems within the healthcare framework.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241297041 - Supplemental material for Electronic patient-reported outcome measures (ePROs) as tools for assessing health-related quality of life (HRQoL) in women with gynecologic and breast cancers: a systematic review
Supplemental material, sj-docx-1-dhj-10.1177_20552076241297041 for Electronic patient-reported outcome measures (ePROs) as tools for assessing health-related quality of life (HRQoL) in women with gynecologic and breast cancers: a systematic review by Amal Boutib, Asmaa Azizi, Ibtissam Youlyouz-Marfak, Malak Kouiti, Mohamed Taiebine, Mohamed Benfatah, Chakib Nejjari, Salim Bounou and Abdelghafour Marfak in DIGITAL HEALTH
Footnotes
Acknowledgments
We thank the Euro-Mediterranean University of Fez and the Higher Institute of Health Sciences, Hassan First University of Settat for supporting this study.
Contributorship
AB, AA, MK, and AM served as a lead for conceptualization, methodology, and resources. AB, AA, IYM, MK, MT, MB, CN, SB, and AM served as a lead for formal analysis and data curation. AB, AA, MK, IYM, and AM served as a lead for writing the initial draft and visualization. AM, IYM, and SB served as a lead for writing–review, editing, and supervision. IYM, CN, SB, and AM served as a lead for project administration. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The data presented in this study are provided at the request of the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Ethical approval
This study involved only a literature review of previously published studies. It involved no primary research on human or animal subjects, or medical records. As such, this work was considered exempt from ethical review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
