Abstract
Objective
As highlighted by the COVID-19 pandemic, identifying strategies for home-based patient management is crucial. As pain is highly prevalent and imposes significant burdens, interest in its remote management is steadily increasing. Transcranial Direct Current Stimulation (tDCS) seems promising in this context.
Methods
This systematic review and meta-analysis aimed to determine the effectiveness of home-based tDCS in pain management (PROSPERO, CRD42023452899). The extracted data included clinical conditions, interventions, comparators, outcome measures, adverse effects, and risk of bias; the Grading of Recommendations Assessment, Development and Evaluation (GRADE) assessment was carried out.
Results
12 records (9 randomized controlled trials [RCTs], 446 participants, 266 undergoing tDCS) were included in the systematic review. The meta-analysis showed that home-based tDCS might produce large and clinically relevant improvement in chronic pain intensity at the end of the intervention (standard mean difference [SMD] −0.95, 95% CI −1.34 to −0.56; p < 0.01; 404 participants, low certainty), as well as small clinically unimportant improvement at short-term follow-up (SMD −0.50, 95% CI −0.82 to −0.19; p < 0.01; 160 participants, moderate certainty). A subgroup analysis showed that it might clinically improve the chronic pain related to fibromyalgia and knee osteoarthritis. Moreover, home-based tDCS seems to modulate pressure pain threshold, heat pain threshold, and heat and cold tolerance at the end of the intervention. Notably, tDCS appeared to be generally safe, well-accepted and easily applied at home.
Conclusions
Low to moderate certainty evidence suggests that home-based self-administered tDCS is a safe and effective tool for managing various types of chronic pain. Further well-designed, large-scale RCTs are warranted.
Keywords
Introduction
Pain, described by the International Association for the Study of Pain (IASP) as an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage, is a complex and multifactorial phenomenon involved in several disorders. 1 Considering the high prevalence of pain in a variety of pathological conditions and that conventional treatments are often limited in their effectiveness, pain results in significant health, economic, and psychological costs.2,3 Therefore, a crucial goal of current research is to find cost-effective solutions to improve its management. In this context, noninvasive brain stimulation (NIBS) represents a promising technique for managing the pain of different aetiology.4,5 Indeed, through neuroplastic mechanisms, NIBS allows a transient modulation of the activity of specific brain areas or networks by applying electrical (e.g., transcranial direct current stimulation, tDCS) or magnetic (e.g., transcranial magnetic stimulation) stimuli to the scalp in a safe way. 6 Among NIBS techniques, tDCS involves the application of a direct low-amplitude current over a short period between two electrodes, the positive anode and the negative cathode. It is applied to manage many pathological conditions, particularly in pain treatment.7,8 The tDCS mechanism of action can be explained by the regulation that direct current, even if applied to superficial regions, exerts on specific brain networks involved in pain processing, modulating their activity and promoting downward pain suppression mechanisms.5,9–11 Crucially, this evidence supports the efficacy of tDCS for pain management and opens the door to its use in reducing polypharmacy, which is frequently related to a variety of adverse effects.11,12 The recent COVID-19 pandemic and the restrictive measures aimed at limiting contagions highlighted several limitations characterizing the healthcare systems worldwide. Indeed, most health services require direct access to healthcare facilities that otherwise cannot be offered. 13 Therefore, there is a great interest in treating patients directly at home.14–16 Home-based patient management yields many beneficial effects, reducing direct and indirect medical costs, such as hospital visits, travel, and time expenses, and allowing prioritization for the most critical patients. 17 In light of the ease of tDCS protocols, recent studies have demonstrated the possibility of administering treatments directly at home by the patient and caregiver after specific training or under remote supervision.18–20 Thus, it is reasonable that home-based tDCS might be a viable and cost-effective alternative for the management of painful conditions. However, evidence of its application at home primarily stems from single studies on small and heterogeneous samples with limited generalizability of findings. To the best of our knowledge, no recent systematic review confirmed the effectiveness of home-based tDCS for treating pain. Here, we performed a systematic review and meta-analysis on the effectiveness of home-based tDCS (alone or in combination with or as augmentation for other noninvasive treatments) in managing various types of pain compared to other noninvasive (home-based or not) interventions.
Materials and methods
This systematic review and meta-analysis followed the updated Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 21 The study was registered in the ‘International Prospective Register of Systematic Reviews’ (PROSPERO, CRD42023452899).
Literature search
The following electronic bibliographic databases were searched for eligible articles: MEDLINE (via PubMed), EMBASE, Scopus (via EBSCO), Web of Science, and the Cochrane Library (Cochrane Database of Systematic Reviews, Cochrane Central Register of Controlled Trials (CENTRAL), Cochrane Methodology Register), from inception to 30 September 2023. Grey literature was screened through research from institutional repositories or online platforms. Specific search strings were used for each queried database, and the studies retrieved were listed in Ryyan (//rayyan.org, Rayyan Systems Inc, Qatar). The search strategy included only terms relating to or describing the intervention, combined with Boolean operators (see Supplementary Appendix 1 for the search strategy). In the first step, studies were selected by screening the titles and abstracts by three independent blinded reviewers (A.A., A.B. and G.F.), and duplicate studies were removed. In the second phase, full articles were reviewed by two blinded authors (A.A. and S.S.) for inclusion in the review. Disagreements were resolved by discussion or, when needed, by a third reviewer (A.B.) for final consensus.
Selection criteria
We used the PICO model to establish the selection criteria: (i) Population: patients affected by pain (regardless of type and underlying pathology). (ii) Interventions: home-based tDCS (both self-administered or remotely supervised, alone or in combination with or as augmentation for other noninvasive treatments) for the management of various types of pain. The term ‘combination’ refers to tDCS treatment initiated with another type of noninvasive intervention simultaneously, while ‘augmentation’ refers to tDCS treatment add-on to other therapies (including drugs). (iii) Comparator: patients who underwent a (home-based or not) treatment with sham tDCS (i.e., consisting of an active stimulation for a few seconds to mimic the sensations produced by active tDCS to keep participants blind to the intervention). (iv) Outcomes: The primary outcome was the self-reported pain (severity), measured using quantitative responses within a continuous scale (e.g., Visual Analog Scale (VAS) or Numerical Rating Scale (NRS) of pain) and quantitative sensory testing. Moreover, we considered, as evaluated by standardized tools, cognitive status, e.g., as evaluated by the Trail Making Test (TMT), the impact of illness on daily life, e.g., as assessed by the Fibromyalgia Impact Questionnaire (FIQ), the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), and the Headache Impact Test (HIT-6), and mood, e.g., as evaluated through the Beck Depression Inventory-II (BDI-II). Finally, we examined adverse effects, patient-reported symptoms, and adverse effects of tDCS.
The exclusion criteria were: non-randomized trials, people affected by pathological conditions other than pain, an intervention other than home-based tDCS, trials that used some invasive intervention, and wrong publication type (e.g., conference abstract, letter to the editor, pre-print).
Data collection
Three reviewers (A.A., A.B. and G.F.) independently extracted the data from a properly developed electronic spreadsheet. The following data were extracted: identification of the authors; year of publication; study design; characteristics of the participants of both the intervention and the control group (type of pain, pain-related diseases, sample size) details on the home-based tDCS intervention (e.g., stimulation site, time, frequency, duration, and intensity) and control condition (e.g., type, frequency, duration); all measures related to primary (i.e., pain) and secondary outcomes; any reports and characteristics of adverse effects related to home-based tDCS; information for the assessment of the risk of bias.
For the meta-analysis of continuous outcome, mean change from baseline was estimated if not provided in the primary studies. 22 When data were provided as median and range, we converted the median and IQR to mean and standard deviation using appropriate statistical formulas. 23 WebPlotDigitizer (https://apps.automeris.io/wpd/, Automeris, USA) was used to extract numerical data from figures. Missing data were handled according to established methods. 24 If the data could not be extracted from the selected reports, the authors were contacted twice within 2 weeks to request the necessary data.
Data synthesis and analysis
Data from the included articles are reported as numbers and percentages for categorical variables, with mean and standard deviation for continuous variables. A pairwise random-effects meta-analyses were conducted using the Restricted Maximum Likelihood method for primary and secondary outcomes reported in at least two studies. In order to allow comparison between studies, we calculated the mean difference (MD) of mean change from baseline and post-intervention effects between groups for studies that used the same outcome measure, whereas standard mean difference (SMD; Hedges’ g) of mean change from baseline and post-intervention effects between groups for the studies that did not use the same outcome measure to evaluate the same construct. The following thresholds were used: g = 0.2 (small), g = 0.5 (medium), and g = 0.8 (large) to interpret the effect size. The pooled SMD of pain was re-expressed on the visual analogue scale (0–10, low values = less pain) as weighted mean difference (WMD) following the Cochrane guidelines to inform clinical practice and guide future research on the effectiveness of home-based tDCS compared with sham tDCS. 25 The WMD was estimated from the SMDs, utilizing a baseline standard deviation (SD) of 1.81 as the reference from the included study. 26 The estimated WMD was then compared with the minimal clinically important difference (MCID) for change in pain VAS, with a cut-off set at 1.5 points for both the overall effect estimate 27 and for subgroups such as fibromyalgia 27 and knee osteoarthritis. 28 The MCID for the FIQ, WOMAC and BDI-II scores were set as 14, 8.5, and 5, respectively.29,30 Heterogeneity was assessed via the I2 statistic and prediction intervals. The 95% prediction interval was also estimated if there were ≥3 studies in a pairwise analysis to determine a range of potential values which could be assessed in future trial prediction intervals. All statistical analyses have been conducted in R using the meta package (R Foundation for Statistical Computing, Austria, Version 6.5.0). 31 An alpha value of 0.05 was used for all analyses.
Risk of bias and certainty of evidence assessment
The Risk of Bias (RoB) tool was used by two reviewers (G.F. and A.A.) to assess the risk of bias and applicability of the studies. 32 This tool was used to determine the risk of bias in patient selection, blindness, and randomization, and a three-way classification (low, some concerns, high risk for five domains) was provided for each included study. The Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach was used to assess the certainty of evidence. 33 The certainty of the evidence was rated as ‘very low’, ‘low’, ‘moderate’, high’.
Results
Database searching identified 712 records. After removing duplicates, 507 were screened for title and abstract, and 462 were excluded. Among the 45 remaining papers, 34 did not meet the inclusion criteria and, thus, were excluded from the collection: specifically, 23 applied a different kind of stimulation protocol, two had a study design other than RCT, and nine had a wrong article type (i.e., congress abstracts). Thus, we included 11 studies according to the inclusion and exclusion criteria. Moreover, following the suggestion of an expert in the field, we added a recent work that the search strategy had not identified, as it was published after its completion. 34 Therefore, we included 12 records (9 trials) in our systematic review (see Figure 1). It is important to note that three records were secondary analysis studies of other works included in this review.35–37 All the studies were published in the last 5 years with very limited geographical distribution: four were conducted in the USA,19,38–40 four in Brazil26,34,41,42 and only one in Europe (UK). 43 All were double-blind RCTs; only two were double-crossover 43 and factorial, 34 respectively.

PRISMA flow diagram outlining the literature review and study selection.
Population
Although we aimed to include studies that assessed the effect of home-based tDCS on both acute and chronic pain, after conducting title/abstract screening and full-text review, the final set of studies focused exclusively on the effectiveness of home-based tDCS for chronic pain conditions. Most studies evaluated the application of tDCS in the treatment of pain related to fibromyalgia,26,34,41,42 i.e., characterized by neuropathic and psychogenic features, and knee osteoarthritis.19,39 In contrast, only single studies have examined its effects on pain in Alzheimer's disease (AD) and related dementia patients (i.e., reasonably due to a combination of osteoarticular, neuropathic, and psychogenic pain), 40 persistent headache attributed to head trauma, 38 and different types of pain due to central or peripheral nervous system injuries (e.g. spinal cord injury, phantom limb pain, central post-stroke pain, neuropathic pain, trigeminal injury) 43 (see Table 1). Table 2 summarizes the RoB evaluation of the included studies.
Summary of studies’ characteristics

Secondary study of Ahn et al., 2019. 19 bSecondary study of Martorella et al., 2022. 39 cSecondary study of Ahn et al., 2019 19 and Martorella et al., 2022. 39
Abbreviations: A: anodal; BAI: Beck's Anxiety Inventory; BDNF: brain-derived neurotrophic factor; BDI–II: Beck Depression Inventory-II; B-PCP:S: Brazilian Portuguese version of the Profile of Chronic Pain: Screen; BP-PCS: Brazilian Portuguese Pain Catastrophizing Scale; C: cathodal; CMAI: Cohen-Mansfield Agitation Inventory; COWAT: Controlled Oral Word Association Test; CPM: conditioned pain modulation; CSI-BP: Central Sensitization Inventory for Brazilian Population; dlPFC: dorsolateral prefrontal cortex; FIQ: Fibromyalgia Impact Questionnaire; fNIRS: functional near-infrared spectroscopy; HADS: Hospital Anxiety and Depression Scale; HbO: oxygenated hemoglobin; HIT-6: Headache Impact Test; HPTh: Heat Pain Threshold; HPTo: Heat Pain Tolerance; ISI: Insomnia Severity Index; M1: primary motor cortex; MMSE: Mini Mental State Examination; MOBID-2: Mobilization-Observation-Behavior-Intensity-Dementia scale; NPI: Neuropsychiatric Inventory; NPS: Numerical Pain Scale; NRS: numerical rating scale; PCL-5: DSM-5 PTSD Checklist; PTSD: post-traumatic stress disorder; PHQ-9: Patient Health Questionnaire; PPT: pressure pain threshold; PSQI: Pittsburgh Sleep Quality Index; QoL: quality of Life; QST: Quantitative sensory testing; RCT: randomized controlled trial; RPQ: Rivermead Post-Concussive Symptoms Questionnaire; SF-36: Short Form-36; TMT: Trail Making Test; VAS: Visual Analogue Scale; WAIS-III: Wechsler Adult Intelligence Scale; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index
Risk of bias (RoB) of the included studies.

1: random sequence generation; 2: allocation concealment; 3: blinding of participants and personnel; 4: blinding of outcome assessment; 5: incomplete outcome data; 6: selective reporting; 7: other bias.
Description: + indicates a low risk of bias; − indicates a high risk of bias;indicates an unclear risk of bias.
Real tDCS treatment group
The sample size ranges from 10 to 60 patients. In most cases, the anode (i.e., the active electrode) was placed on the primary motor area (M1), while the cathode (i.e., the inactive electrode) on the contralateral supraorbital region.19,39,40,43 In other studies, the anode was instead placed on the dorsolateral prefrontal cortex (dlPFC), while the cathode was on the contralateral homologous area26,41,42 or, in only one case, on the occipital pole. 38 Only one study also used a cathodic stimulation in addition to an anodal one, reversing the previously described positioning on M1 and the contralateral supraorbital cortex. 43 Finally, only one study applied active stimulation to both M1 (while the cathode was positioned on the contralateral supraorbital cortex) and, in a second group, on the dlPFC (with the cathode in the contralateral homologous region). 34 All studies applied a current intensity of 2 mA. In comparison, only one used a lower value of 1.4 mA, 43 and all used a 20-min stimulation session except for one study that employed 30-min sessions. 26 While all included studies used stimulation protocols characterized by five sessions/week, the number of weeks varied widely from one 40 to 12. 26 Only two studies combined tDCS with a behavioural treatment, such as mindfulness.19,38 All studies provided remote patient supervision except for two.26,43 However, in one of them, the telephone number of the investigators was available, and in-person visit at half of the stimulation protocol was scheduled. 26
Comparator treatment group
Even in this group, there is considerable variability in the sample size, from 10 to 60 patients. All the studies used a home-based sham-tDCS protocol, a montage similar to the real treatment without the actual application of the continuous current. Moreover, two included the application of mindfulness, one in sham modality (without specific instructions but with the only indication to relax and breathe deeply), 19 and one as applied in the experimental group. 38 In addition, most studies involved the application of a current, usually of 2 mA (only one study applied a current intensity equal to 1.4 mA), 43 for a few seconds at the beginning and end of the protocol. This was done to mimic the sensations produced by the actual treatment and to avoid the knowledge of the subject regarding the allocation group. Similar to the real group, all sessions included 5 days a week of 20 min each; only one study provided 30-min sessions. 26 The number of weeks varied similarly to the real treatment group, from 1 40 to 12. 26
Outcome measures
Pain outcome measures
Pain was evaluated using several methods, ranging from scales and questionnaires to subjective threshold assessments. Notably, some studies assessed pain before and after treatment and during follow-up, usually for several weeks.38–40,43 Specifically, most studies employed an intensity assessment by using a numerical (e.g., Numerical Pain Scale, NPS, and Numerical rating scale, NRS) or visual (Visual Analogue Scale, VAS) measurement scales, which allow the patient to express the subjective perception of the pain experience.19,26,34,39–43 Furthermore, most of the included works used quantitative sensory testing (QST) to objectively assess the patient's threshold to painful stimulation of different modalities. Between them, pressure pain threshold (PPT),19,26,41 thermal pain thresholds (Heat Pain Threshold, HPTh, and Heat Pain Tolerance, HPTo),26,34,42 conditioned pain modulation (CPM),19,34,41 as well as the sensitivity to cold pain were assessed.19,42 Finally, disease-specific tools were used, such as the WOMAC scale for osteoarthritis-related knee pain19,39 or specific headache-related information (number of moderate-to-severe headache days, total number of headache days, headache disability [HIT-6]). 38 Of note, two secondary-analysis studies performed a functional near-infrared spectroscopy (fNIRS) evaluation, a noninvasive technique using scattered light to monitor changes in local oxygenated haemoglobin concentrations (HbO) induced by brain activity. Specifically, they assessed HbO at the level of superior motor and somatosensory cortices co-located with the stimulating tDCS electrode 35 or to evaluate functional connectivity during thermal and punctate stimulation. 36
Other outcome measures
Several studies evaluated secondary outcomes related to pain. Specifically, some works explored depression through questionnaires, such as the BDI-II.26,41,42 Furthermore, cognitive performance was evaluated using the TMT and the Controlled Oral Word Association Test (COWAT). 41 Other works used specific assessment tools to assess the impact of fibromyalgia on daily life, such as the FIQ.34,42 Finally, several works considered patients’ experience related to home-based tDCS treatment (satisfaction, feasibility, device usability, and compliance with the treatment) through the use of specific questionnaires or a number of completed sessions.19,34,38,43
Adverse effects
All studies evaluated the presence of adverse events related to tDCS stimulation. Notably, three studies reported no treatment-related adverse events in all recruited patients.19,39,40 The other studies reported the presence of some treatment-related adverse effects, mainly classified, when information was available, as mild and transient sensations.26,34,38,41–43 Specifically, included studies have generally reported the presence of tingling of the forehead/scalp, redness, tiredness, headache, exacerbation of paresthesia, warm sensation, itching, burning, neck pain, mood swings, and concentration difficulties.26,34,38,41–43 Interestingly, although most of the studies documented comparable frequency and severity in terms of adverse effects between the active and the sham group,38,41,43 burning,34,42 tingling and redness 42 were significantly more frequent in the active one. Of note, only two studies reported patient withdrawals due to a burning sensation, which was classified as severe only for patients in the active group.34,42 One study reported a single withdrawn due to an increase in pain intensity following sham stimulation. 43 Similarly, other works showed a higher frequency of headache, neck pain, mood swings, concentration difficulties, tingling, warm sensations, and itching in the sham group compared to the active one.26,38 No studies reported long-term adverse effects after home-based tDCS, except for an increase in paresthesia already present at the 4-week follow-up. 43 These data are summarized in Table 3.
Summary of adverse events (AEs) and acceptability/feasibility/compliance about home-based tDCS interventions in the included studies, broken down by group (active vs sham).

Abbreviations: A: anode: C: cathode; DLPFC: dorsolateral prefrontal cortex; M1: primary motor area; SD: standard deviation.
Home-based tDCS feasibility and satisfaction
Most of the included studies collected data on the feasibility of home-based tDCS. In particular, they assessed the adherence/retention rate (the number of completed sessions) and/or patient satisfaction concerning the protocol modalities.19,34,38–43 Notably, the adherence rate was very high in all the included studies, ranging from 83% 41 to 100%, 40 with an average between 85% and 95%.34,38,42,43 Moreover, the questionnaires used to evaluate usability and satisfaction with the home-based protocol (i.e., device preparation and easiness of use) showed that most patients considered the intervention simple and easy to apply.19,39,40,43 Furthermore, questionnaires highlighted that patients appreciated the possibility of remote supervision.19,39,40 One study highlighted that all difficulties patients encountered while using the device could be solved by a simple telephone conversation. 43 These results demonstrate that home-based tDCS is a NIBS protocol that patients themselves could easily apply at home with high satisfaction. Table 3 summarizes these data.
Effectiveness of home-based tDCS
The results from meta-analyses and certainty of assessments are presented in Table 4.
Summary of results from meta-analysis and GRADE evaluation.
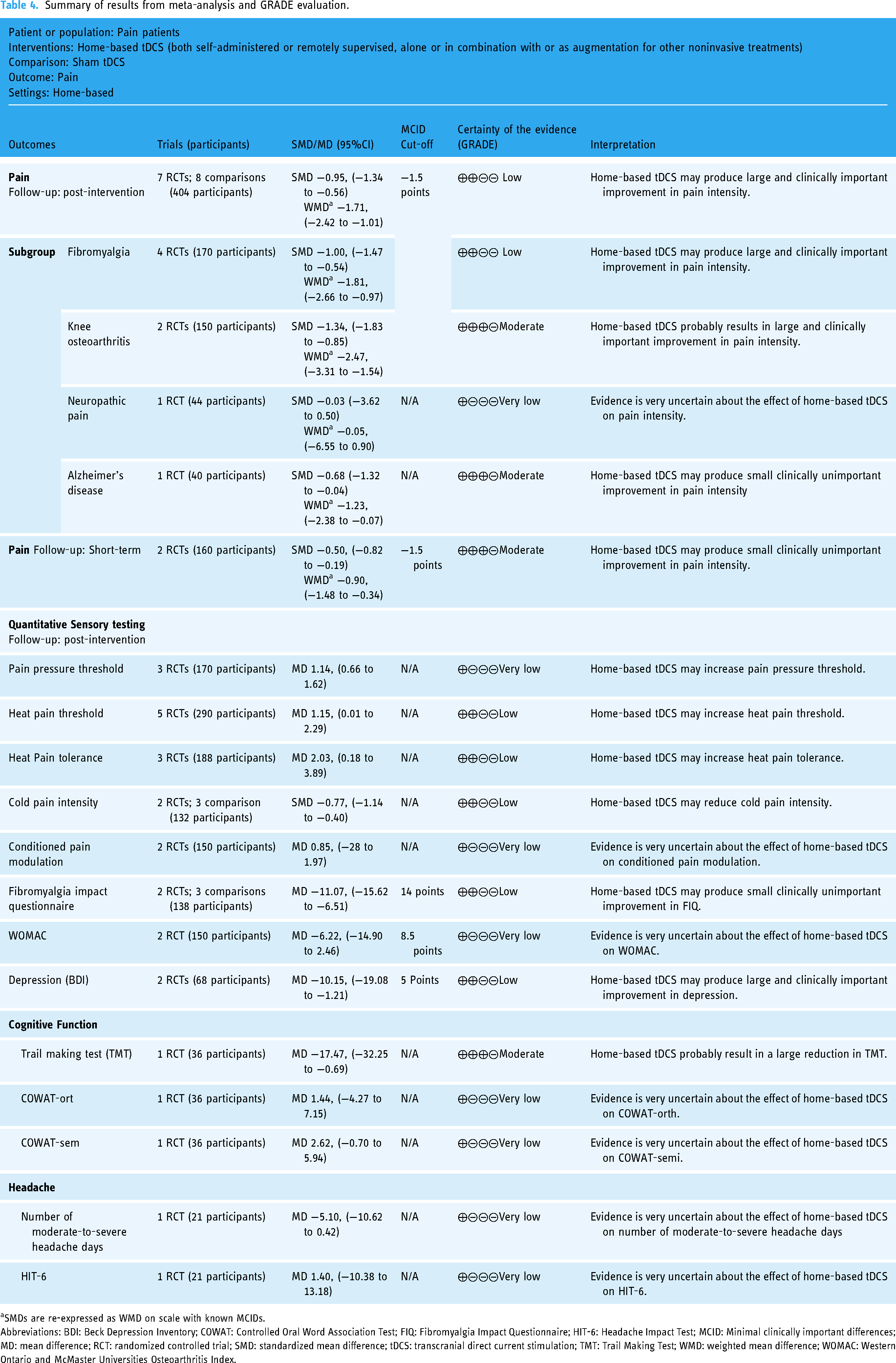
SMDs are re-expressed as WMD on scale with known MCIDs.
Abbreviations: BDI: Beck Depression Inventory; COWAT: Controlled Oral Word Association Test; FIQ: Fibromyalgia Impact Questionnaire; HIT-6: Headache Impact Test; MCID: Minimal clinically important differences; MD: mean difference; RCT: randomized controlled trial; SMD: standardized mean difference; tDCS: transcranial direct current stimulation; TMT: Trail Making Test; WMD: weighted mean difference; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
Pain intensity
Seven studies (8 comparisons) assessed the impact of home-based tDCS on pain. The meta-analysis found that home-based tDCS may produce large and clinically important improvement in pain intensity at the end of intervention (SMD −0.95, 95% CI −1.34 to −0.56; 95% PI: −2.16, 0.26; p < 0.01; I2 = 66%; 404 participants; Low certainty; see Figure 2, first section) and may produce small clinically unimportant improvement in pain intensity at short-term follow-up (SMD −0.50, 95% CI −0.82 to −0.19; p < 0.01; I2 = 0%; 160 participants; moderate certainty; see Figure 2, second section).

Meta-analysis of studies evaluating the effects of home-based tDCS on pain intensity at the end of intervention (first section) and at short-term follow-up (second section).
Sensitivity analysis
Sensitivity analysis after removing the cross-over study 43 does not differ from the primary Analysis. Leave-one-out analysis found no influential study for pain (see Supplementary Appendix 2).
Subgroup analysis
Subgroup analysis according to pain population showed that home-based tDCS may result in large and clinically meaningful improvement in pain intensity in fibromyalgia and knee osteoarthritis. Evidence from a single study indicates that home-based tDCS may produce small, clinically unimportant improvement in pain intensity in AD. In contrast, evidence is very uncertain in neuropathic pain (see Table 4).
QST (central sensitization)
Meta-analyses found that home-based tDCS may increase PPT (MD 1.14, 95% CI 0.66 to 1.62; 95% PI: −3.90, 6.19; p < 0.01; I2 = 52%; 170 participants; see Figure 3), HPTh (MD 1.15, 95% CI 0.01 to 2.29; 95% PI: −2.62, 4.92; p = 0.05; I2 = 67%; 290 participants; see Figure 3), HPTo (MD 2.03, 95% CI 0.18 to 3.89; 95% PI: −18.80, 22.87; p = 0.03; I2 = 66%; 188 participants; see Figure 3) and may reduce cold pain intensity (SMD −0.77, 95% CI −1.14 to −0.40; 95% PI: −3.18, 1.64; p < 0.01; I2 = 0%; 132 participants; see Figure 3) at the end of intervention. The evidence is very uncertain about the effect of home-based tDCS on CPM at the end of intervention (MD 0.85, 95% CI −28 to 1.97; p = 0.14; I2 = 84%; 150 participants; see Figure 3).

Meta-analysis of studies evaluating the effects of home-based tDCS on pain pressure threshold (first section), heat pain threshold (second section), heat pain tolerance (third section), cold pain intensity (fourth section) and conditioned pain modulation (fifth section) at the end of the intervention.
Fibromyalgia impact questionnaire
Three studies assessed the impact of home-based tDCS on the FIQ score. The meta-analysis found that home-based tDCS may produce small clinically unimportant improvement in FIQ at the end of intervention (MD −11.07, 95% CI −15.62 to −6.51; 95% PI: −40.62, 18.49; p < 0.01; I2 = 0%; 138 participants; see Supplementary Appendix 3 and Table 4).
WOMAC
Two studies assessed the impact of home-based tDCS on WOMAC. The evidence is very uncertain about the effect of home-based tDCS on WOMAC at the end of intervention (MD −6.22, 95% CI −14.90 to 2.46; p = 0.16; I2 = 61%; 150 participants; see Supplementary Appendix 4 and Table 4).
Depression
Two studies assessed the impact of home-based tDCS on BDI. The meta-analysis found that home-based tDCS may produce large and clinically important improvement in depression at the end of intervention (MD −10.15, 95% CI −19.08 to −1.21; p = 0.03; I2 = 32%; 68 participants; Supplementary Appendix 5 and Table 4).
Cognitive function
Only one study assessed the impact of home-based tDCS on cognitive performance in fibromyalgia. Analysis indicated that home-based tDCS probably resulted in a large reduction in TMT at the end of intervention (MD −17.47, 95% CI −32.25 to −0.69; p = 0.04; 36 participants; see Supplementary Appendix 6 and Table 4). The evidence is very uncertain about the effect of home-based tDCS on controlled oral word association test orthographic and semantic (see Supplementary Appendix 6 and Table 4).
Headache
Only one study assessed the impact of home-based tDCS on post-traumatic headache. The evidence is very uncertain about the effect of tDCS on the number of moderate-to-severe headache days (MD −5.10, 95% CI −10.62 to 0.42; p = 0.07; 21 participants; see Supplementary Appendix 7 and Table 4) and on HIT-6 (MD 1.40, 95% CI −10.38 to 13.18; p = 0.81; 21 participants; see Supplementary appendix 7 and Table 4).
GRADE
The detailed GRADE assessments for all meta-analyses are in Supplementary (see Supplementary Appendix 8).
Discussion
To the best of our knowledge, this is the first systematic review to evaluate the safety and effectiveness of home-based tDCS for chronic pain management. Low to moderate certainty evidence suggests that home-based tDCS is both safe and effective for managing chronic pain. Low certainty evidence suggests that home-based tDCS may produce large and clinically meaningful improvements in pain intensity at the end of the intervention and small clinically unimportant improvement in pain intensity at short-term follow-up. Home-based tDCS may increase PPT, HPTh, and HPTo, and reduce cold pain intensity at the end of the intervention. It may also positively affect cognitive function, FIQ scores, and reduce depression. However, the evidence is very uncertain regarding the effects of home-based tDCS on CPM, WOMAC, and headache at the end of the intervention. The overall certainty of the evidence is low to moderate, indicating that the true effects could differ significantly from the estimated effect sizes.
Considering pain, meta-analysis found that home-based tDCS may produce large and clinically important improvement in pain intensity at the end of the intervention. Notably, the positive effect on pain has been shown in different patient populations and chronic pain categories. This evidence suggests that the modulation exerted by this NIBS technique acts independently of the underlying pathophysiological mechanism, reasonably through modulation of cortical areas critically involved in pain processing. 11 This aspect is crucial, as it indicates that tDCS could be a therapeutic aid in the management of chronic pain irrespective of its type since it seems effective on pathologies characterized by different pain physiopathology.44–46 Moreover, tDCS may offer an alternative to the polypharmacy of pain conditions, often ineffective, burdened by numerous side effects and with a potential risk of drug interactions.47,48 Consistently, pain intensity clinically improved in patients with fibromyalgia and knee osteoarthritis, whereas small, not clinically relevant changes were recorded in AD patients. These encouraging results were confirmed even after sensitivity analysis, ruling out a carry-over effect that may partially justify the strong effects of tDCS on pain modulation. Of note, the only study analyzing the effectiveness of tDCS according to pathological conditions (e.g., complex regional pain syndrome, brachial plexus avulsion, radiculopathy) didn’t show any result supporting the intervention. 43 It is reasonable to assume that the presence of mixed, clinically and pathophysiologically heterogeneous conditions may have diminished its overall efficacy.49,50 Furthermore, additional studies with larger samples for each condition are necessary to confirm this assumption. Although only two studies evaluated the effects of tDCS at follow-up,39,40 our results show how it leads to improvements for several days. This finding supports the hypothesis that modulation of cortical areas involved in pain processing is crucial to improve its complex management. 51
Coherently, the most frequent areas of stimulation were M1 and the left dlPFC, two regions commonly employed in NIBS protocols.52,53 Indeed, they both have peculiarities of great relevance in pain perception. On one hand, M1 appears structurally and functionally altered in patients suffering from pain, suggesting its involvement is not limited to motor control.54,55 Consistently, it's established that motor and sensory functions constitute a loop that updates the internal models comparing the expected and the obtained sensory consequences of an action.56,57 Sensory and M1 areas exhibit dense and reciprocal interconnections, facilitating a continuous exchange of information between both sides of the loop.58,59 Therefore it seems reasonable as M1 exerts a top-down control over the processing of afferent information like pain, representing a relevant gateway to endogenous pain modulation pathways. 60 Crucially, NIBS protocols targeting M1 demonstrated efficacy in attenuating pain perception by influencing the descending pathways implicated in pain processing.61–64 However, pain perception also depends on factors other than sensory and motor information alone, such as attention and emotional context.65,66 Notably, the left dlPFC plays a key role in the regulation of both executive functions, in particular, attention and cognitive flexibility, and emotional control.67–70 Consistently, several studies have documented alterations in dlPFC in painful conditions that regress when pain disappears, suggesting its pivotal role in the pain network. 71 Moreover, various research highlighted that its activation correlates with the ability to suppress painful information.72,73 Thus, neuromodulatory interventions aimed at enhancing the activity of the left dlPFC might play a key role in improving the subject's ability to devote the right attentional resources to pain and to influence its emotional state positively.71,74,75 Furthermore, within the previously discussed framework of pain, the dlPFC could be fundamental in integrating incoming painful stimuli with pain expectation, reducing mismatch and, consequently, abnormal perception. 76
In people with fibromyalgia, modulation of the dlPFC might restore the alignment between prediction and outcome, reducing the mismatch that often causes pain and related mood disorders typical of these patients.71,77 Indeed, FIQ changes following tDCS treatment seem to confirm this hypothesis, both when the left dlPFC or M1 were the target.41,42 This evidence highlights that fibromyalgia-related pain depends on not only attentional and emotional impairments but also sensory-motor alterations due to nociplastic changes (i.e., related to altered nociception without evidence of nociceptor activation or lesion of the somatosensory system) frequently observed in these patients.78–80 Importantly, this evidence is consistent with other findings in the literature suggesting that neuromodulation techniques may be pivotal in normalizing specific neurophysiological mechanisms that are characteristically altered in fibromyalgia patients, who experience widespread chronic pain and central sensitization. 81 Consistently, in addition to regulating the excitability of brain areas most involved in processing various aspects of pain, as previously suggested, tDCS may also influence neuroplasticity mechanisms in the CNS to reduce the amplification of pain signals. 82 Furthermore, it might also help to restore the balance between cortical excitation and inhibition, which is crucial for the proper functioning of the body's endogenous pain-inhibiting systems. 83 In addition, although it was not possible to perform a meta-analysis on these data, the tDCS effect was demonstrated through fNIRS investigations, revealing that only participants receiving active tDCS treatment reported a significant increase in HbO activation within the motor and somatosensory cortices, corresponding to the anode site. 35 These findings imply an association between cortical activation and pain perception, suggesting a crucial role of tDCS in pain relief. Montero-Hernandez et al. 36 exploited this technique to characterize functional connectivity in brain areas involved in pain processing. They observed that only subjects who received the active treatment showed a significant reduction in number and strength of connections between the prefrontal cortex, M1 and the primary somatosensory cortex during pain stimulation. This phenomenon underscores the potential of tDCS to induce network-level effects able to mitigate pain perception modulating the complex exchange of information between key areas.84,85
Several studies evaluate the effects of tDCS through QST showing strong effects in people who received the active stimulation. Specifically, they observed an improvement in terms of PPT,19,26 HPTh,26,34,42 HPTol,26,42 and a reduction in cold pain intensity threshold.19,42 The effect of tDCS on CPM was uncertain, probably due to the complexity of this phenomenon and the low number of studies on this topic.19,34,41,86 Taken together, these evidence suggest that M1 and the left dlPFC represent an ideal target for pain-modulating tDCS, enhancing top-down modulation of sensorimotor integration and reducing attentional and emotional demand of pain. In light of this, the potential role of NIBS in restoring properly pain processing takes on significant importance in all chronic conditions where CNS sensitization supports pain maintenance. 87 As highlighted by our results, brain areas involved in pain processing are easily stimulated by simple training or remote guidance, making home-based tDCS protocols particularly suitable for the management of chronic pain conditions. The recent COVID-19 pandemic has critically underscored the importance of implementing protocols that enable patient management within their homes. This approach could not only limit the costs and waiting lists of hospital facilities, but also mitigate patient discomfort and the associated risks of hospital-related complications.15,88,89 Furthermore, several works reported a substantial number of patients affected by COVID-19 developing late-onset pain. Given the magnitude of the just-ended pandemic emergency, it is reasonable to expect a significant increase in pain symptoms in the coming years.90,91 Since most of the included studies documented excellent levels of adherence and patient satisfaction with home-based tDCS protocols, this strategy might play a crucial role in future interventions for home-based chronic pain management.
Finally, we showed that adverse effects were generally infrequent and, when present, they were typically mild and transient. Although several works described a severe burning sensation in some patients who received the active tDCS, causing withdrawal from the study,34,42 other studies showed more frequent adverse effects in patients who received the sham stimulation,26,38 confirming the well-known limitations of patient self-reported measures.92,93 Importantly, no studies documented significant long-term treatment-related adverse effects, except for a modest increase in paresthesia in patients who already complained of this symptom. 43 This finding confirms that tDCS is a safe and easy technique to apply in research and clinical settings. 94 Therefore, home-based tDCS appears safe and reliable also when used by the patient or his caregiver after a simple initial training or under remote supervision, raising interest in its applications in home rehabilitation, as observed in other pathological conditions.18,95,96 In addition, patients expressed considerable appreciation towards remote supervision or the possibility of telephone contact to solve any problems, an aspect that future studies should address.19,39,40,43 Even though further studies with larger sample sizes are needed to confirm these findings, our results encourage using home-based tDCS to improve chronic pain management in heterogeneous patient populations.
Although we performed a thorough systematic review of the literature and a robust meta-analysis of the evidence, this work has several limitations: first, the number of studies is relatively low and implies caution when interpreting the results. Furthermore, the included studies are very heterogeneous regarding pathological conditions and outcome measures (e.g., patient-reported measures, QST). Additionally, the methodological quality of the studies included in the Analysis varied, sometimes lacking rigour. Consequently, the strength of evidence upon which conclusions were drawn ranged from low to moderate, highlighting the need for a cautious interpretation of the results. It is also important to note that, although we did not apply any restrictions on pain duration, our search yielded only conditions associated with chronic pain. This may be because acute pain, by nature, typically resolves with readily available treatments, often without the need for medical intervention, and within a short time frame.97,98 These factors make it potentially challenging to evaluate the application of home-based tDCS for acute pain conditions, at least at present. Once tDCS devices become more widely accessible with standardized protocols, it may be possible that home-based tDCS could become a valuable adjunct to currently used pharmacological therapies. Thus, current findings pertain only to chronic pain conditions and should be generalized to other types of pain with caution. Finally, due to its complex and elusive nature, pain is extremely difficult to evaluate, and it is well known that the experience of the pain symptom is incredibly variable inter-subjectively.99–101 Therefore, further studies with larger samples, better characterized pathological conditions, standardized stimulation protocols, and outcome measures that are as objective as possible (e.g., fNIRS, functional neuroimaging) are needed.
Conclusion
Low to moderate certainty evidence suggests that home-based tDCS may be an effective treatment for reducing pain intensity across various chronic pain populations. It may also increase PPT, HPTh and HPTo, and reduce cold pain intensity. Additionally, home-based tDCS could positively affect the cognitive function, improve FIQ scores, and reduce depression in these populations. Furthermore, home-based tDCS appears to be generally safe, well-tolerated, and feasible for use by patients themselves and/or their caregivers following simple training or remote supervision. However, the overall certainty of the evidence is low to moderate, indicating that further studies with higher methodological quality and larger sample sizes are needed to confirm these findings.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241292677 - Supplemental material for The effectiveness of home-based transcranial direct current stimulation on chronic pain: A systematic review and meta-analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076241292677 for The effectiveness of home-based transcranial direct current stimulation on chronic pain: A systematic review and meta-analysis by Annibale Antonioni, Andrea Baroni, Giulia Fregna, Ishtiaq Ahmed and Sofia Straudi in DIGITAL HEALTH
Footnotes
Acknowledgments
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Contributorship
S.S. had the idea for the work; A.A., A.B., G.F. and I.A. performed the literature search and data analysis; A.A. drafted the first version of the manuscript; all the authors critically revised the work; I.A. and S.S. supervised each step. All authors approved the final version.
Data availability
Datasets used for Analysis are available through the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
S.S. had full access to all of the data and took responsibility for the integrity of the data and the accuracy of the data analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
