Abstract
Objective
Most smokers who achieve short-term abstinence relapse even when aided by evidence-based cessation treatment. Mobile health presents a promising but largely untested avenue for providing adjunct behavioral support for relapse prevention. This paper presents the rationale and design of a randomized controlled trial aimed at evaluating the effectiveness of personalized mobile chat messaging support for relapse prevention among people who recently quit smoking.
Methods
This is a two-arm, assessor-blinded, randomized controlled trial conducted in two clinic-based smoking cessation services in Hong Kong. An estimated 586 daily tobacco users who have abstained for 3 to 30 days will be randomized (1:1) to intervention group or control group. Both groups receive standard-of-care smoking cessation treatment from the services. The intervention group additionally receives 3-month relapse prevention support via mobile chat messaging, including cessation advice delivered by a live counselor and access to a supportive chatbot via WhatsApp. The control group receives text messaging on generic cessation advice for 3 months as attention control. The primary outcome is tobacco abstinence verified by an exhaled carbon monoxide of <5 parts per million or a negative salivary cotinine test at 6 months after randomization. Secondary outcomes include self-reported 6-month prolonged tobacco abstinence, 7-day point-prevalent abstinence, and relapse rate. The primary analyses will be by intention-to-treat, assuming participants with missing data are non-abstinent. This trial is registered with ClinicalTrials.gov (NCT05370352) and follows CONSORT-EHEALTH.
Conclusion
This trial will provide new evidence on the effectiveness of mobile chat messaging as a scalable and accessible intervention for relapse prevention.
Introduction
Relapse is the most likely outcome of any smoking cessation efforts, even when aided by evidence-based smoking cessation treatment. Most relapses occur in the early phase of abstinence, with up to 70% of abstainers who are using proven pharmacotherapy like nicotine replacement therapy (NRT) resuming smoking within the first month after quitting. 1 Maintaining abstinence during the early phase may have a substantial impact on long-term abstinence outcomes. A study shows that half of those with high abstinence probabilities in the first month (early quitters) could achieve biomedically validated abstinence at 6 months. 2 Interventions to prevent early relapse are crucial to promote abstinence.
Currently, there is no proven intervention for preventing relapse among people who are actively quitting. Mixed evidence suggested providing extended courses of pharmacotherapy, especially varenicline, can promote long-term abstinence.3,4 However, extended pharmacotherapy, which can only be given after the end of standard pharmacotherapy (typically 8–12 weeks), cannot prevent early relapse in the active treatment phase. Adjunct behavioral interventions can be delivered concurrently with other active cessation treatments. However, a Cochrane review did not find traditional approaches, including self-help materials, telephone counseling, and group therapy, effective in promoting long-term abstinence. 3
Mobile health (mHealth) offers a potential platform for providing additional behavioral support to complement conventional cessation treatment. A Cochrane review has shown the effectiveness of text messaging interventions delivered through Short Messaging Service (SMS) in enhancing smoking cessation outcomes compared with minimal cessation support or when added to other cessation treatment. 5 Driven by the widespread ownership of smartphones, mobile instant messaging apps such as WhatsApp, Facebook Messenger, and WeChat have emerged as alternative platforms to SMS for mHealth interventions. These apps utilize media-rich messaging (e.g., voice, image, video) and user-friendly interfaces, offering more interactive and conversational experiences than SMS. Several trials have shown the effectiveness of mobile instant messaging-based interventions for smoking cessation.6–8
Research on mHealth intervention for relapse prevention is still limited. Emerging pilot trials showed the feasibility and acceptability of text messaging 9 and WhatsApp and Facebook online group discussions for relapse prevention. 10 A recent trial did not find WhatsApp online group discussion more effective than text messaging for relapse prevention in increasing abstinence, 11 but the trial did not use a control without mHealth support.
By adapting a previous program for cessation,6,12 we developed a mobile chat messaging intervention specifically for relapse prevention among recent quitters. Our pilot trial among 108 recent abstainers has shown high trial feasibility (recruitment rate = 81%), and 80% of the participants in the intervention group responded to the messages at least once. 13 By intention-to-treat (retention rate = 85%), the 6-month biochemically validated tobacco abstinence was higher in the intervention (32%) than control group (22%). In light of the promising results, this study protocol presents the rationale and design of a fully-powered randomized controlled trial aimed at evaluating the effectiveness of mobile chat messaging for relapse prevention in promoting 6-month biochemically validated tobacco abstinence among people who recently quit smoking in Hong Kong. Additionally, the trial will include a mixed-method process evaluation and an economic evaluation of the intervention.
Materials and methods
Study design
This is a two-arm, assessor-blinded, randomized controlled trial with 6-month follow-ups conducted in Hong Kong, where the daily tobacco use prevalence was 9.4% in 2023. 14 The trial is registered with ClinicalTrials.gov (identifier: NCT05370352) and follows the CONSORT-EHEALTH (Consolidated Standards of Reporting Trials of Electronic and Mobile Health Applications and online TeleHealth). 15 The trial has been approved by the Institutional Review Board of the University of Hong Kong/ Hospital Authority Hong Kong West Cluster (UW 22-026). The trial procedures are largely equivalent to that of the pilot trial. 13
Participants and settings
Participants will be recruited onsite and offsite from smoking cessation clinics under Tung Wah Group of Hospitals Integrated Centre on Smoking Cessation (TWGH) and the United Christian Nethersole Community Health Service (UCN). Both cessation service providers are subvented by the Department of Health, the Hong Kong SAR government, and offer behavioral counseling and pharmacotherapy (typically 8–12 weeks). For onsite recruitment, a trained research assistant will approach smokers in the clinics and screen their eligibility. Flyers with contact information of the research team will be distributed in the clinics to publicize the study. For offsite recruitment, staff at TWGH and UCN will refer potential subjects who joined their “Mail to quit” program to the research team. Users of the “Mail to Quit” received telephone-based behavioral support and mailed NRT without needing to attend the clinics. All participants provided informed consent to participate in the trial before randomization.
Participant inclusion criteria:
Hong Kong residents aged 18 years or above Own a smartphone with WhatsApp installed Enrolled in the smoking cessation program under TWGH or UCN Smoked daily before the present quit attempt Abstained from smoking for 3 to 30 days
Participants will be excluded if they are participating in other ongoing smoking cessation studies, or have been diagnosed with a mental health disorder or are on regular psychotropic drugs. Those who maintain abstinence for less than three days but fulfill other eligibility criteria are encouraged to leave their contact information. A research assistant will re-approach them three days after their expected quit date via phone call to confirm their abstinence duration and enroll eligible subjects in the trial.
The Cochrane review on relapse prevention interventions recommends randomizing participants after they have achieved short-term abstinence to segregate the effect of relapse prevention interventions from the effect of cessation treatment. 3 However, there is no consensus on how to define “short-term abstinence”, and previous trials used varying duration of abstinence (from 24 hours to 16 months) to classify recent abstainers. 3 We will use 3 days to be the lower cut-off for short-term abstinence to be consistent with our pilot trial and other trials of mHealth intervention for relapse prevention to facilitate comparison.11,13 The upper cut-off will be 30 days because the trial aims to address relapse in the early phase of abstinence, given most relapses occur in the first month of abstinence. 1
Allocation and masking
Participants will be individually randomized (1:1) to intervention group or control group, stratified by recruitment method (onsite and offsite) and service providers (TWGH and UCN). The random allocation schemes were generated by an online tool (https://www.sealedenvelope.com/simple-randomiser/v1/lists) using random permuted blocks of 2, 4, and 6. The randomization scheme will be concealed from research personnel involved in participant recruitment to avoid allocation bias, and all baseline procedures are identical between the intervention and control groups. Due to the behavioral nature of the intervention, blinding of the participants and treatment providers will not be possible. However, the participants will not know the intervention contents in the other group. Information about the treatment allocation will be withheld from the outcome assessors and statistical analysts for blinding.
Intervention group
Participants in both groups will continue to receive standard smoking cessation treatment provided by TWGH or UCN. These typically include biweekly behavioral counselling combined with pharmacotherapy, including NRT monotherapy, bupropion and combined NRT, for 8–12 weeks. Varenicline is no longer available since the manufacturer halted production of the drug in 2021 after discovering an impurity in the formulation.
Participants in the intervention group will additionally receive chat messaging support, in which a live counselor (trained in applied psychology and smoking cessation counseling) will interact with a participant in real-time and provide relapse prevention advice via WhatsApp for 3 months after randomization.
To initiate conversation between participants and counselors, 18 regular messages will be sent to the participants over the 3-month intervention period. These messages cover information and advice on managing the five major reasons for relapse based on the US Tobacco Use and Dependence Guideline. 16 These include: (1) lack of support for cessation, (2) negative mood or depression, (3) strong or prolonged withdrawal symptoms, (4) weight gain, and (5) smoking lapses. To enhance participants’ engagement, the messages utilize emojis and infographics and include prompts to promote responses. These messages are scheduled to be delivered at random time between 12:30 pm and 2:30 pm on working days with a tapering schedule frequency (from twice a week in the first month to once a week in the last month) to avoid abrupt withdrawal.
The participants can initiate a chat support session with the counsellor by responding to the message. The chat-based interaction is designed to be dynamic and adaptive, allowing the counselor to have natural conversations tailored to the specific needs and responses of each participant. Based on the needs identified, the counselor will use behavioral change techniques (BCTs) to provide smoking cessation advice and support. 17 These include BCTs that promote motivation (e.g., strengthen ex-smoker identity [BCT code: BM8]), maximize self-regulatory capacity (e.g., advise on avoiding social cues for smoking [BS11]), promote adjuvant activities (e.g., advise on stop-smoking medication [A1]), and other supportive BCTs that facilitate information gathering (e.g., assess withdrawal symptoms [RI4]) and communication (e.g., elicit and answer questions [RC3]). All advice or information will be personalized according to the participants’ sex, age, and abstinent status to be more personally relevant.6,12 For example, female participants may receive additional guidance on managing weight gain that can arise during cessation, while younger participants may receive advice on how to deal with peer influences.
Although participants can send a message anytime, the counselor can only respond during the daytime because providing all-day live support is highly resource intensive. Therefore, the participants can also access a supportive chatbot in WhatsApp, which will provide on-demand smoking relapse prevention support when the counselor is not available (e.g., during nighttime). The information and advice provided by the chatbot will also focus on the five major reasons for relapse in the US Tobacco Use and Dependence Guideline. The chatbot will be introduced to participants via regular messages. Participants can obtain information about smoking relapse prevention in the chatbot by two means. First, the chatbot provides a button-based interface with menu options for participants to navigate the content (Figure 1(a) and 1(b)). The chatbot will guide participants through the conversation according to a pre-defined chatbot flow and provide advice on relapse prevention grounded in BCTs. Second, participants can type their queries directly (e.g., “how to use NRT patch?”), and the chatbot will process the question using pre-set keywords or intents recognized by using natural language processing and answer accordingly (Figure 1(c)).

Interface of the WhatsApp chatbot for relapse prevention with English translation. (a) Greeting and welcoming messages upon entering the chatbot. (b) Main menu of the chatbot. (c) Chatbot navigation by keywords.
Control group
In addition to standard cessation treatment provided by the services, participants in the control group will receive eight text messages via SMS covering generic information about the harms of smoking and the benefits of quitting over 3 months from baseline, with a tapering schedule from once a week to once every fortnight. We have considered and decided against only using a usual care approach because the imbalanced contact time and attention may lead to differential attrition between the two groups, especially when participant blinding is not feasible. In the pilot trial, 13 18 text messages instead of eight messages were used as attention control. However, this messaging frequency was later deemed to be overly intensive, raising the concern that the true effect of the mobile chat messaging intervention would be underestimated.
Data collection and outcomes
Figure 2 shows the schedule of data collection at baseline, 3-month follow-up, and 6-month follow-up. We will collect baseline data via face-to-face questionnaire for participants recruited onsite and telephone survey for participants recruited offsite. Follow-up data will be collected via telephone calls by a research assistant blinded to treatment allocation.
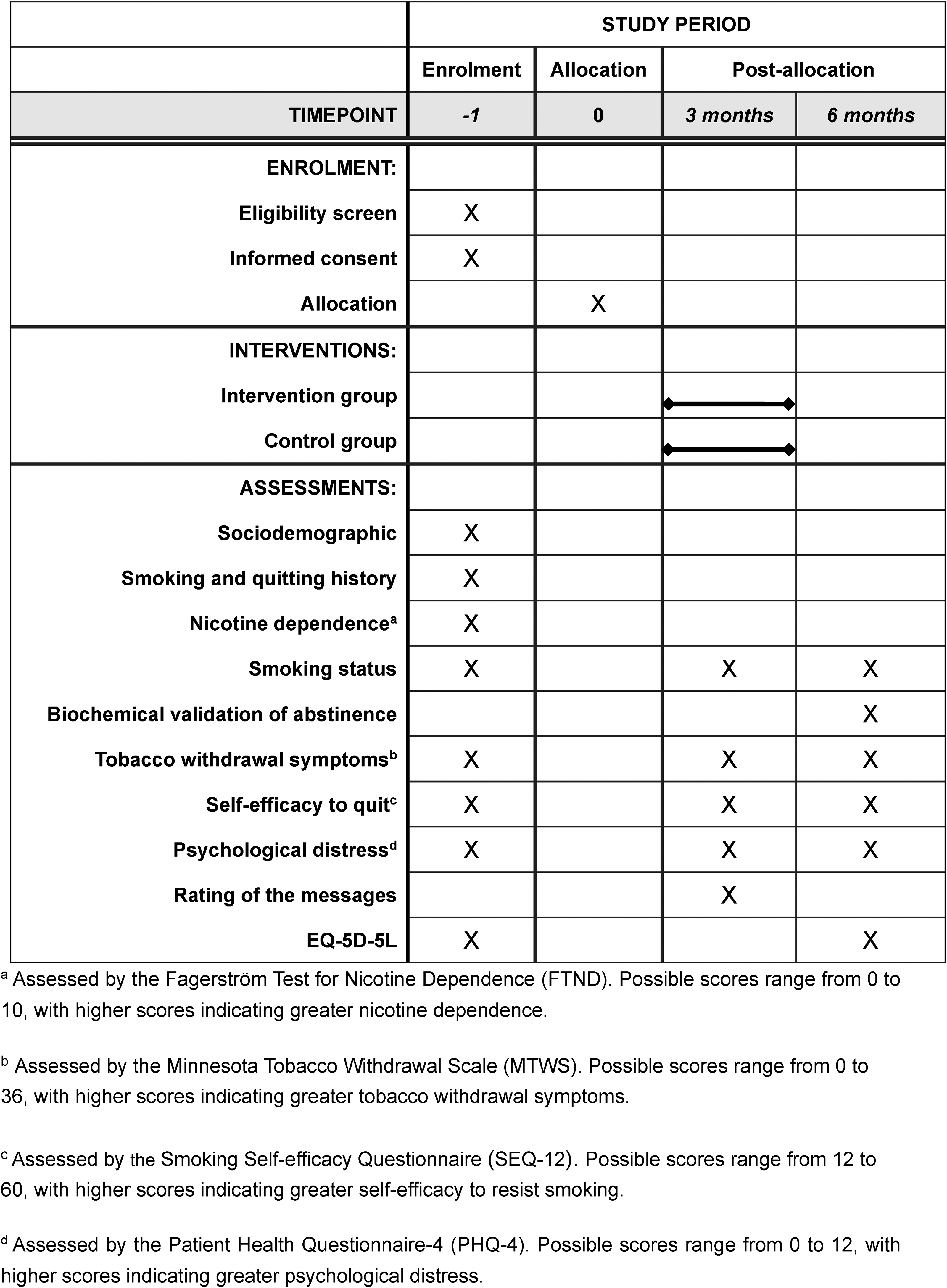
The schedule of enrolment, interventions, and assessments (SPIRIT diagram).
Primary outcome
The primary outcome will be biochemically validated 7-day point prevalent tobacco abstinence at 6 months after randomization, 18 verified via an exhaled carbon monoxide level of lower than 5 parts per million or a negative salivary cotinine test. 19 We will invite participants who self-reported 7-day abstinence at 6-month follow-up to test for biochemically validated tobacco abstinence primarily using a Smokerlyzer (Bedfont Scientific Ltd) for exhaled carbon monoxide testing. iScreen OFD Cotinine device for salivary cotinine test will be offered to those who are unwilling to use the Smokerlyzer due to hygienic concerns. A research assistant, who is blinded to the treatment allocation, will conduct the test at a location suggested by the participant. Those who participated will receive HK$300 (≈US$38) cash as compensation for their time and travel expenses.
Secondary outcome
The secondary outcomes will include the self-reported 6-month prolonged tobacco abstinence (no more than five lapses), self-reported 7-day point prevalent tobacco abstinence at 3 months (end of treatment) and 6 months after randomization, and smoking relapse (tobacco products consumed for 7 consecutive days or longer) at 3 and 6 months. 18 To evaluate the long-term effect, an additional follow-up at 12-month post-randomization will be conducted to assess biochemically validated tobacco abstinence, self-reported abstinence and relapse. We have updated the trial registry record to include the change on April 25, 2024.
Other measures
To evaluate the cost-effectiveness of the intervention, the Chinese version of European Quality of Life 5 Dimensions 5 Level Version (EQ-5D-5L) will be assessed at baseline and 6-month follow-up. 20 This scale consists of two parts: the descriptive system and the visual analogue scale. The descriptive system asks respondents to rate their health status on five dimensions and combines their answers on each dimension into a five-digit number that represents their health status. The visual analogue scale asks respondents to rate their overall health on a scale from 0 to 100, with higher scores indicating better health status. Change in self-efficacy to quit smoking will be assessed at baseline, 3 months and 6 months by the Chinese version of the Smoking Self-Efficacy Questionnaire 12. 21 Smoking urges and withdrawal symptoms will be assessed at baseline, 3 months and 6 months by the Chinese version of the Minnesota Tobacco Withdrawal Scale. 22
For process evaluation of the intervention, participants in both groups will be asked to what extent they read the regular messages and their perceived helpfulness of the messages at 3 months (end of treatment). The questions are designed to be non-specific to the mHealth platforms by which the messages were delivered (i.e., WhatsApp or SMS) to blind the assessors and minimize potential biases. To evaluate the blinding mechanism, the outcome assessors will be asked if they can determine the allocation of the participants at the end of the follow-up survey.
Qualitative interviews
Individual qualitative interview will be conducted at 6 months to examine participants’ perception and experiences with the chat messaging intervention. We will purposively sample intervention participants of different sex, age and quitting status to increase the richness of the data. The study endpoint will be based on data saturation. The interview will be guided by an interview guide and tape recorded.
Statistical analysis
Sample size
PS: Power and Sample size version 3.1.6 was used to calculate the sample size for the primary outcome of biochemically validated quit rate at 6 months after randomization. Based on the results from our pilot trial and previous trials of mHealth interventions for smoking cessation,5,13 we assume the intervention effect (RR) to be 1.5 and the validated abstinence rate in the control group to be 20% by intention-to-treat. To achieve 80% power with a significance level of 5% and an allocation ratio of 1:1, a sample size of 586 (293 per group) will be needed.
Primary analyses
All analyses will be performed in Stata/MP Version 15.1 using the intention-to-treat method, in which all randomized participants will be included. Those with missing data at follow-up will be assumed to be non-abstinent. Descriptive statistics will be used to profile the main demographic characteristics of participants. Poisson regression with robust variance estimators will be used to calculate the RR of the intervention effect on the primary and secondary outcomes between the two groups. Number needed to treat will be calculated as the reciprocal of the difference in cessation outcome between the two groups. Four sensitivity analyses will be used to evaluate the robustness of the findings. First, multivariable regression will be used to adjust for baseline factors that are prognostic of cessation outcomes, such as nicotine dependence, duration of abstinence, cessation treatment received. 23 Second, we will use multiple imputation by chained equations to impute missing data. At least 50 imputed datasets will be inferred. Third, complete case analysis to exclude participants with missing outcomes will be conducted. Finally, we will combine the data from the pilot trial 13 to obtain a pooled RR for biochemically validated tobacco abstinence at 6 months.
Subgroup analyses
To examine potential effect modifications, we will examine the primary outcome in subgroups of sex, age, nicotine dependence level before quitting, duration of abstinence pre-randomization, cessation service used (TWGH vs UCN), recruitment method (onsite vs offsite), and if the participants are reapproached. We will also include the multiplicative interaction terms to evaluate the significance of the interaction, adjusting for multiplicity. The analyses will be exploratory since the trial is not powered to examine interaction.
Process evaluation
We will follow the UK Medical Research Councils’ framework on process evaluation of complex interventions, which provides a robust, comprehensive, and universally applicable approach to examine the implementation and mechanisms of the chat messaging intervention using mixed-method approaches. 24 First, we will conduct causal mediation analyses to explore the mediating effect of several potential mediators, including smoking urges, self-efficacy to quit, perceived psychosocial support, and psychological distress. Prior studies showed that these factors mediated the effect of mobile text messaging on abstinence 25 or were predictive of abstinence or relapse and potential targets of the intervention.26,27 Compared with traditional approaches to mediation (e.g., the Baron and Kenny's method), causal mediation analyses are more rigorous because they can decompose the total effect into direct and indirect (mediating) effects in the presence of interactions and non-linearities and allow causal inference of the mediating effects. 28 A bias-corrected 95% confidence interval of the direct and indirect effect will be estimated by using 1000 bootstrap replications. We shall follow the 9-item short form of AGReMA (A Guideline for Reporting Analyses of Randomized Trials and Observational Studies). 29
Second, we will examine how intervention engagement may impact treatment outcomes. Similar to our previous study, 30 we will apply group-based trajectory modeling to identify the engagement trajectories in the intervention group based on their responses to the regular messages. Multivariable regressions will be used to examine the associations between intervention engagement and abstinence outcomes, adjusting for sociodemographic and baseline factors that are predictive of smoking cessation. The trial does not aim to examine the individual effect of live chat and chatbot, but the abstinence outcomes in participants who used either or both intervention components will be compared.
Third, content analyses of the chat conversations will be conducted, using the taxonomy of BCTs for smoking cessation for coding. 17 We shall record the frequency and type of BCTs used in each participant and examine their associations with abstinence outcomes. This will help identify BCTs that are most used and useful in helping the participants, which will inform the optimization of the chat messaging intervention.
Fourth, tape records from the qualitative interview will be transcribed verbatim and analyzed by two independent researchers. Thematic analyses will be used to analyze the qualitative data, 31 which will be used to explain the quantitative findings from the trial. 24
Cost-effectiveness analyses
Within trial incremental cost-effectiveness ratio (ICER) of the chat messaging interventions (vs. control) will be estimated in terms of cost per successful abstainer and cost per quality adjusted life year (QALY) gained. The total cost of implementing the interventions, including the equipment and manpower needed, will be estimated. EQ-5D-5L utility scores at baseline and follow-up will be used to estimate QALYs in both groups using the area under the receiver operating characteristic curve technique. The intervention will be considered cost-effective if the ICER is less than 3 times of the gross domestic product per capita in Hong Kong, as recommended by the WHO.
Discussion
Our personalized chat messaging support is expected to prevent smoking relapse and promote successful smoking cessation attempts among people who recently quit smoking. If found effective, the findings will provide evidence of an effective intervention that can be readily implemented in existing cessation treatment to promote abstinence outcomes. The prespecified mixed method evaluation and cost-effectiveness analyses will provide valuable insights on the underlying mechanisms and value of the intervention.
Although the chat messaging intervention requires a live counselor to deliver, it is much less burdensome than traditional multi-session, in-person counseling. The personalized, chat-based support delivered by live counselor can maintain human touch and foster therapeutic alliance, while mobile messaging enhances treatment reach and scalability. The hybrid model combining live counseling support and mobile messaging optimize access and effectiveness of interventions for smoking relapse prevention.
This trial has several strengths by design. First, we randomize participants after they have achieved short-term abstinence as recommended by a Cochrane review, allowing the separation of relapse prevention intervention effects from cessation treatment effects. 3 Second, the control group will receive regular text messaging with the same duration but less frequency than the intervention group to achieve attention control. This design mitigates potential differential attrition caused by imbalanced contact time and attention received across groups. Third, assessor blinding is implemented by using follow-up questionnaires with equivalent items, even for questions related to participants’ ratings of the mobile messaging. We will also evaluate the utility of this approach in preventing the assessor's awareness of the group assignment. Finally, we will use biochemical validation of abstinence as the primary outcome to address potential misreporting of abstinence status (e.g., due to social desirability bias), enhancing the robustness of the trial's results.
Conclusion
Mobile chat messaging presents a potential intervention to address smoking relapse, the most likely outcome of any smoking cessation attempts. This fully-powered RCT will provide new evidence on the effectiveness of mobile chat messaging in preventing smoking relapse among people who recently quit smoking.
Footnotes
Acknowledgements
We appreciate the support from the Smoking Cessation Research Team, School of Nursing, the University of Hong Kong, and the personnel from Tung Wah Group of Hospitals Integrated Centre on Smoking Cessation and United Christian Nethersole Community Health Service.
Contributorship
TTL, MPW and HCC conceived the study. TTL suprvised the study and obtained ethical approval. TTL, MPW, YTDC, HCC and SYH obtained funding. VW provided administrative and technical support. XS and TTL wrote the first draft. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was granted by the institutional review board of the University of Hong Kong/Hospital Authority West Cluster (Reference No.: UW 22-026).
Funding
This study was supported by the Health and Medical Research Fund, Food and Health Bureau, Government of Hong Kong SAR (grant number 19201341).
Guarantor
TTL
