Abstract
Objective
This study aims to assess the accuracy and stability of smartwatches in predicting acute mountain sickness (AMS).
Methods
In locations exceeding an altitude of 2500 m, a cohort of 42 subjects had their Lake Louise AMS self-assessment score, blood oxygen saturation (SpO2), heart rate, and perfusion index measured using smartwatches, with the data seamlessly conveyed to the Huawei Cloud.
Results
A significant decrease in SpO2 was observed in individuals positive for AMS compared to those negative (p < 0.05), with the mild AMS group exhibiting significantly lower SpO2 levels than the non-AMS group (p < 0.05). Furthermore, SpO2 emerged as a significant, independent predictor of AMS [β=−0.086, p < 0.01, OR (95% CI) = 0.92 (0.87–0.97)], indicating that each unit increase in SpO2 decreases the probability of AMS occurrence by 8.6%.
Conclusion
The Huawei smartwatches have demonstrated efficacy in diagnosing and foretelling AMS at elevations exceeding 4000 m, showcasing significant reliability and high precision in SpO2 measurement.
Keywords
Introduction
The growing popularity of high-altitude activities, such as tourism, outdoor pursuits, and scientific endeavors, has exposed more non-acclimatized individuals to the challenges of elevated environments. Concurrently, the maturation of transportation infrastructure has dramatically amplified the ease with which these regions are accessed, culminating in a swift surge of individuals toward lofty locales. However, this augmented accessibility has inadvertently exposed a larger contingent of non-acclimatized individuals to the formidable challenges inherent at elevated altitudes. The unique environmental conditions, typified by attenuated atmospheric pressure and curtailed oxygen levels, mandate a cascade of physiological stress reactions that are imperative for acclimatization to such environments. 1 An abrupt ascent to high altitudes without adequate acclimatization can trigger a medley of symptoms associated with acute mountain sickness (AMS), manifesting within 6 to 12 h post-ascension to altitudes surpassing the 2500 m threshold. 2 Neglecting AMS or persisting in the ascent may exacerbate the condition, risking progression to perilous complications such as high-altitude cerebral edema and high-altitude pulmonary edema.
Previous studies have used physiological and psychological indicators, such as blood oxygen saturation (SpO2), heart rate (HR) variability, and baseline anxiety,3–5 for diagnosing and predicting AMS. SpO2 serves as a direct reflection of blood oxygen concentration, 6 rendering the fingertip pulse oximeter a relevant tool for assessing and anticipating AMS. However, the efficacy of pulse oximeters may be affected by factors such as fingertip temperature, measurement duration, device functionality, and the surrounding environment, 7 potentially diminishing their accuracy and impeding the precise diagnosis and prediction of AMS.8,9 The escalating demand for real-time exercise tracking, health monitoring, and disease prediction has bolstered the popularity of smartwatches equipped with monitoring capabilities. 10 Research has substantiated the accuracy of smartwatches in gauging physiological indicators such as SpO2 and HR.11,12 Smartwatches offer a broader applications scope compared to fingertip pulse oximeters, being more convenient, less prone to interference, and offering an integrated suite of functionalities, thereby enhancing user-friendliness. 13 In scenarios where pulse oximeters are unavailable, smartwatches can be effectively employed to measure SpO2 levels for diagnosing and predicting AMS. 14
While the accuracy of smartwatches in measuring physiological indicators has been extensively documented,11,12,15 studies have predominantly examined devices from well-known brands such as Apple, Samsung, and Garmin,11,12,16 with limited attention given to other smartwatch models. The application of smartwatch-based physiological measurements has been utilized in disease diagnosis and prediction,15,17 including the use of maximum oxygen consumption (VO2 max) and SpO2 for diagnosing and predicting AMS.15,18,19 Smartwatches research has primarily focused on the accuracy of their SpO2 measurement capabilities, with fewer studies exploring their correlation with AMS. While the SpO2 measurement features of Apple and Garmin watches have received independent validation,12,20 there remains a paucity of literature on the Apple Watch's measurement precision in high-altitude environments, and the reliability of Garmin watch measurements in such conditions remains uncertain. A study by Lauterbach et al. 20 utilized the Garmin fēnix® 5X Plus watch to measure SpO2 and HR in normobaric hypoxic conditions simulating a range of altitudes (12,000, 10,000, 8000, 6000, and 900 feet). When compared to a standard medical-grade pulse oximeter, the results indicated that at a simulated altitude of 12,000 feet (3657.6 m), the Garmin watch's SpO2 readings had a mean difference of 3.3%, with a range of −1.9% to 8.6%. For other simulated altitudes, the discrepancies in SpO2 measurements were comparatively minor, ranging from 0.7% to 0.8%. Another study conducted at an altitude of 4559 m reported that the watch's SpO2 measurements were less effective for diagnosing AMS, indicating its limitations in monitoring and preventing the condition. 18 Given the limited research on smartwatches’ ability to measure SpO2 for AMS prediction and the uncertain diagnostic precision and reliability of these devices at elevated altitudes, further in-depth investigation is imperative.
The limitations of traditional measuring tools underscore the need for efficient, portable, and precise AMS diagnosis to facilitate appropriate adjustments in treatment plans or travel itineraries. Smartwatches provide an innovative avenue for diagnosing and predicting AMS, yet their practical performance and data collection in extreme environments are as of now restricted. Moreover, no studies to date have demonstrated that SpO2 measured by smartwatches can effectively diagnose or predict AMS, mandating further comprehensive research. The aim of this study is to monitor SpO2, HR, and perfusion index (PI) in real high-altitude environments using smartwatches to diagnose and predict AMS, thereby further investigating the accuracy and reliability of smartwatches in such scenarios.
Materials and methods
Participants
This was an observational and non-interventional study conducted in July 2022. The study enrolled a cohort of 42 volunteers, including 36 males and six females. The participants consisted of 32 members of the expedition team, four local residents, five individuals who had recently moved to Yanshiping Town, and a single passerby. The demographic profile of the participants had an average age of 34.1 ± 13.4 years, height of 170.4 ± 7 cm, and weight of 63.9 ± 9.2 kg, with 12 individuals having prior experience at altitudes exceeding 5000 m. Prior to their departure, the team members engaged in a 12-week physical training program on campus, situated at an altitude of 23 m, with each week comprising five sessions of 1.5 to 2 h duration. The exclusion criteria were stringent, ruling out participants with a history of high-altitude exposure at ≥2500 m within the preceding 3 months, those afflicted with acute or chronic physical or mental illness, individuals with SpO2 levels below 95% at sea level, and those with habits of smoking or alcohol abuse, or any physical impediments in their upper extremities that could affect the measurements. Participants were thoroughly informed about the study's objectives, methodology, and potential risks to the research personnel prior to their voluntary consent. They were all required to read and sign an informed consent form.
Research design
This study was conducted on the Tibetan Plateau, involving two groups of participants who stayed at high altitudes for 7 and 8 days, respectively. Other participants were selected through a random process. Given the varied demands of the field tasks, the expeditionary force was divided into two groups. A schematic representation of the daily itineraries and the corresponding altitudes for each group is delineated (Figure 1).

Elevation gain profile.
Data aggregation commenced on the participants’ third day of the plateau sojourn for both groups. The measurements were conducted 30 min post-prandially, with a daily frequency of one to three sessions, in tranquil and serene settings. The absence of mobile network signals in select regions of the Tibetan Plateau precluded the automated transmission of data to the cloud, necessitating manual data recording in the interim. This data was subsequently uploaded to the cloud upon the re-establishment of network connectivity. Regrettably, the second group's foray to more elevated altitudes on fieldwork days 7 to 8, where a pervasive lack of network signal prevailed, precluded the execution of the requisite measurements.
Assessment of oxygen saturation, HR, and PI
The study utilized a Huawei WATCH GT3 smartwatch (Shenzhen, China), which employs Huawei's TruSeen™ 5.0+ technology for vital signs monitoring. The smartwatch was securely positioned on the radial aspect of the right wrist, ensuring intimate contact with the skin without interference from movement or attire. Data on oxygen saturation (SpO2), HR, and PI were captured at one-second intervals, stored on the smartwatch, and subsequently transmitted to a mobile device via Bluetooth, culminating in the automatic data upload to Huawei Cloud Space through the mobile network. For the analysis, the mode value was selected as the representative figure for each parameter. The PI is an indicator of peripheral blood perfusion status, denoting the ratio of arterial to venous blood flow. It serves as an important diagnostic tool for assessing pulse oximeter performance, evaluating circulatory system health, and detecting peripheral vascular diseases and other circulatory system disorders, making it significant for clinical monitoring and diagnosis. 21 Smartwatches equipped with photoplethysmography technology can measure the PI by emitting light onto the skin and detecting the changes in the reflected light with a sensor. The PI is calculated from the ratio of the pulse wave's peaks to valleys, providing an assessment of blood perfusion in local tissues. This feature assists in monitoring the wearer's blood circulation and overall health status. 22
Acute mountain sickness (AMS)
The study utilized the revised 2018 Lake Louise AMS Score (LLS) for AMS screening. An AMS diagnosis is confirmed by an LLS score ≥3, including at least one headache score. 2 The LLS encompasses headache, nausea/vomiting, fatigue, and dizziness/light-headedness, each scored from 0 to 3, indicating the absence of symptoms to their severity. The LLS categorizes AMS into mild (3–5 points), moderate (6–9 points), and severe (10–12 points) based on the total score. Integrated within the smartwatch, the LLS facilitates the subsequent measurement of SpO2, HR, and PI. In instances where no communication signal is available during LLS assessment, verbal interviews are employed for data collection. The study focused on high-altitude regions, defined as areas ≥2500 m, thus excluding measurements in lower-altitude zones.
Statistical analysis
The statistical analysis was conducted utilizing G*Power 3.1 software, with a post hoc power analysis applied to the mean for a single independent variable to ascertain the effect size. Given an effect size of f = 0.25, a set significance level of α = 0.05, and 162 data points arrayed across three groups, the resultant statistical power was determined to be 0.81, affirming that the study's sample size was adequate for robust analysis.
Participants were categorized into AMS positive (AMS+) and AMS negative (AMS−) groups based on their LLS scores. Descriptive statistics were used to analyze the demographic information and the varying AMS incidence rates at disparate altitudes within these groups. HR was identified to be normally distributed, while SpO2 and PI, despite not displaying normal distribution, were considered acceptably approximated to normalcy with absolute skewness and kurtosis values falling below the thresholds of 3 and 10, respectively (Table 1). 23 Comparative analyses between AMS+ and AMS− groups for SpO2, HR, and PI were executed using independent samples t-tests, and Welch's analysis of variance (ANOVA) was selected to contrast these variables across varying degrees of AMS severity. Binary logistic regression analysis was further engaged to assess the independent predictive capacity of SpO2, HR, and PI for AMS. The statistical computations and graphical illustrations were accomplished using SPSS 26.0 (IBM, Chicago, IL, USA) and GraphPad Prism 8 (Dotmatics, Boston, MA, USA), with statistical significance being denoted by a p-value <0.05.
Descriptive statistics of K–S test.

K–S: Kolmogorov–Smirnov; SD: standard deviation; SpO2: blood oxygen saturation; HR: heart rate; PI: perfusion index.
*p < 0.05.
Results
The assessment was confined to evaluating the LLS, SpO2, HR, and PI within the elevated regions above 2500 m, as the AMS observations were exclusively made at these altitudes. The average duration for each individual's measurement session was 76.39 ± 12.54 s. Figure 2 depicts the AMS incidence rates among the two groups across various altitudes. The first group recorded a peak AMS occurrence of 38.9% (n = 18) on the fourth day at 4721 m, comprising four participants with mild symptoms and three participants with moderate to severe symptoms. A decline in AMS incidence was then noted on the fifth day, followed by a resurgence to 33.3% (n = 27) on the sixth day, affecting eight individuals with mild symptoms and one individual with moderate to severe symptoms. A progressive decline in AMS incidence was observed as the expedition continued. In contrast, the second group experienced a rapid altitude gain leading to an AMS incidence of 60% (n = 5) on the third day at 5200 m, though no participants were diagnosed with AMS on that particular day. The incidence rates then rose to 20% (n = 25) on the fifth day and 33.3% (n = 21) on the sixth day, with all instances being mild.

Acute mountain sickness (AMS) incidence rates by altitude.
Figure 3 illustrates the variations in physiological parameters across the two groups at different altitudes. Within the range of 2670 and 4721 m, the first group demonstrated a significant divergence in SpO2 levels [84.9 (8.5) vs. 77.6 (7.6), p < 0.05]. Similarly, the second group also presented a marked variation in SpO2 from 4721 to 5300 m [85.4 (3.5) vs. 74.5 (9.7), p < 0.05]. For the first group, HR at 2670 m was significantly distinct from those recorded at the higher altitudes on the fourth and fifth days, peaking at 4721 m [3 vs. 4: 83.2 (13.8) vs. 97.2 (14.7), 3 vs. 5: 83.2 (13.8) vs. 95.8 (14.6), all p < 0.05]. Overall, an increase in altitude was accompanied by a significant decline in SpO2 levels and a concurrent, significant upsurge in HR.
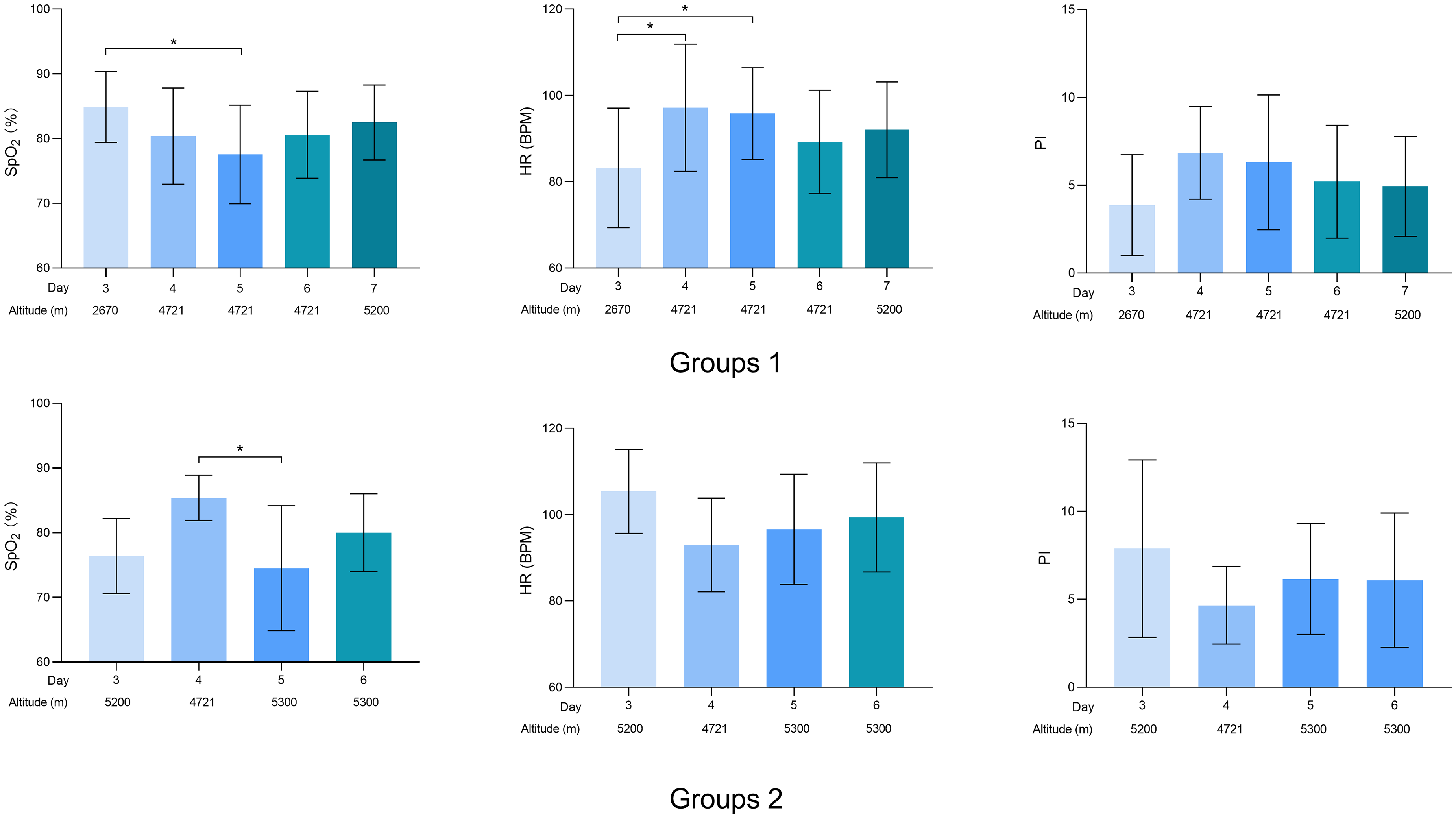
Changes in physiological indices at different altitudes (mean ± SD).
Table 2 delineates the physiological variable differences between AMS+ and AMS− cohorts. T-tests conducted to evaluate SpO2, HR, and PI among these groups revealed a pronounced difference in SpO2 levels, with the AMS− group registering significantly higher levels than the AMS+ group (p < 0.01). While HR and PI levels were relatively elevated in the AMS+ group, the difference between the two groups was not statistically significant.
Physiological differences between AMS+ and AMS− participants.

AMS+: acute mountain sickness positive; AMS−: acute mountain sickness negative; SD: standard deviation; SpO2: blood oxygen saturation; HR: heart rate; PI: perfusion index.
**p < 0.01.
Table 3 illustrates the physiological variable differences among different severity levels of AMS. Given the heterogeneity of variance for SpO2, Welch's ANOVA was employed, followed by post hoc pairwise comparisons using the Games–Howell test to address heteroscedasticity. 24 This analysis indicates a notable variation in SpO2 across the different severity strata of AMS (p < 0.05), in contrast to HR and PI, which showed no significant differences. Specifically, significant differences in SpO2 were observed between individuals exhibiting mild AMS symptoms and those who were asymptomatic.
Physiological differences across AMS severity levels.
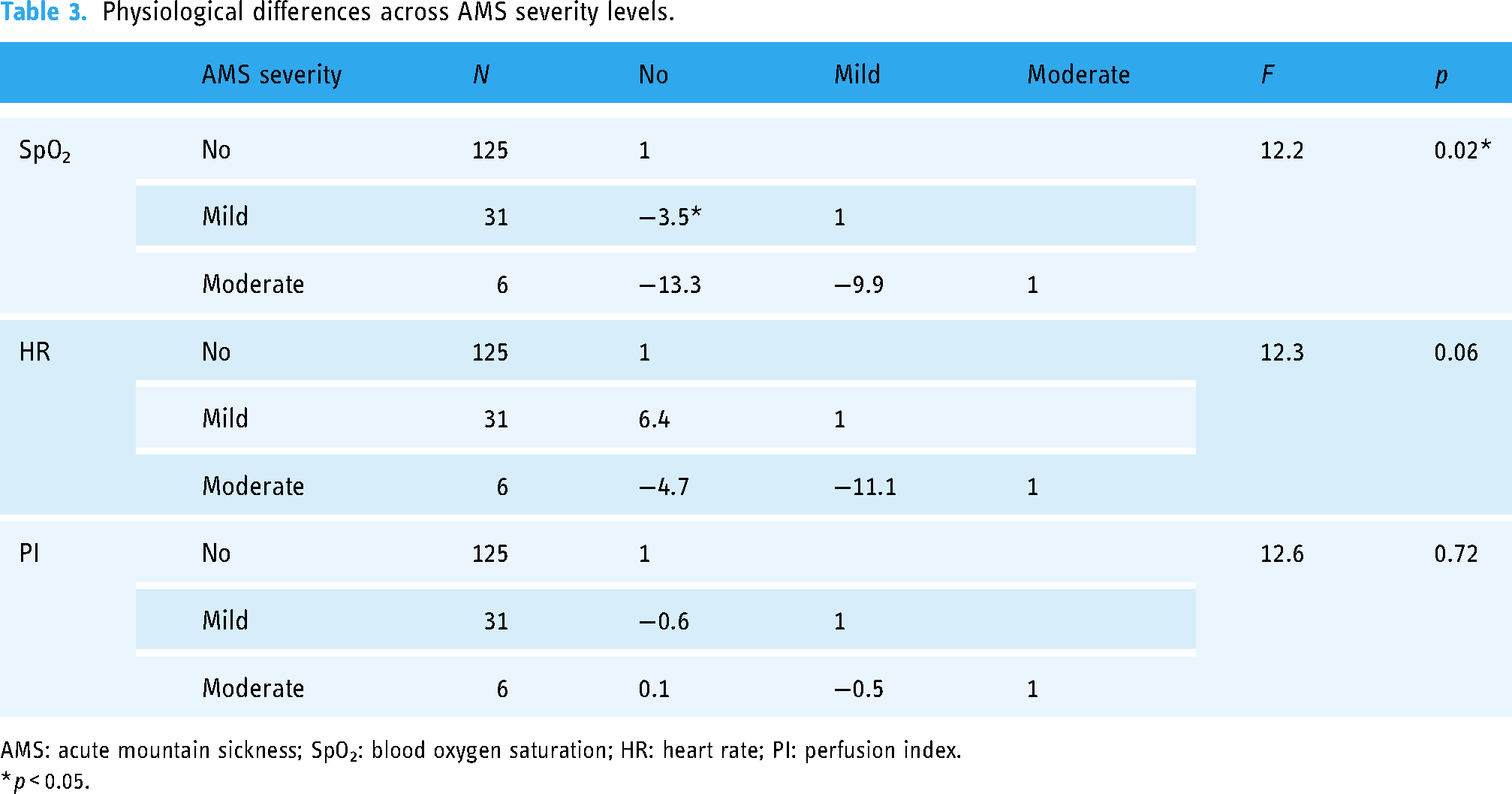
AMS: acute mountain sickness; SpO2: blood oxygen saturation; HR: heart rate; PI: perfusion index.
*p < 0.05.
After conducting a binary logistic regression analysis with SpO2, HR, and PI as independent variables, and the AMS status (positive or negative) as the dependent variables, the model proved to be robust (χ² = 16.05, p = 0.001 < 0.01), with an acceptable fit (χ² = 13.73, p = 0.09 > 0.05). SpO2 was identified as a significant predictor of AMS (β = −0.086, OR = 0.92, 95% CI: 0.87–0.97, p = 0.001 < 0.01). This correlation suggests that an increase in SpO2 by one unit reduces the risk of AMS by 9%, underscoring its role as a protective factor (Figure 4).

Predictors of acute mountain sickness (AMS) from binary logistic regression.
Discussion
A key finding of our study is the capability of HUAWEI smartwatch-derived SpO2 measurements to precisely diagnose and predict the onset of AMS. Notably, a stark contrast in SpO2 readings was observed between individuals in the AMS+ and AMS− cohorts, along with disparities across varying degrees of AMS severity. Moreover, SpO2 emerged as a reliable prognostic indicator for AMS, with a significant predictive correlation.
The onset and severity of AMS are subject to a multitude of determinants, such as the rate of ascent, the peak altitude attained, the duration of residency at high altitude, the magnitude of physical exertion, and the inherent physiological predisposition of individuals. 25 The abrupt transition from lower to higher terrains subjects the body to acute hypoxia, precipitating a reduction in plasma volume, a surge in hematocrit, and an increase in blood viscosity, all of which culminate in an attenuated oxygen transport capacity of the blood. 26 Consequently, the surveillance of SpO2 provides critical insights into the hemoglobin's oxygen-transporting efficacy within the body, enabling the diagnosis of AMS and the assessment of its severity. 27 Additionally, exposure to high altitude triggers alterations in cardiovascular dynamics, characterized by an initial increase in cardiac output and the onset of physiological symptoms such as tachycardia. Despite cardiac output normalizing following a brief acclimatization period, the HR tends to persist in its elevated state. 26 The advancement in wearable technology has led to the proliferation of smartwatches that offer an array of monitoring capabilities. These devices, by capturing data on altitude, HR, SpO2, and physical activity, offer substantial insights for diagnosing and predicting AMS.
Pulse oximeters, as ubiquitous noninvasive tools, provide real-time measurements of a patient's SpO2. Nevertheless, their accuracy can be compromised by several factors. Cold temperatures, for instance, induce vasoconstriction—a natural response to minimize heat loss—that reduces blood flow to the extremities, thus impacting the precision of pulse oximeter measurements. Furthermore, reduced perfusion at the measurement site not only skews the oximeter's output but may also slow the device's response, leading to lagging results. 28 Such effects are exacerbated at high altitudes, where the already hypoxic conditions are compounded by the cold-induced reduction in peripheral blood flow. Despite studies reporting the use of smartwatches for SpO2 measurement, the focus has predominantly been on their accuracy,12,29 with limited research exploring their correlation with AMS. The scope of current smartwatch studies is somewhat narrow, with a dearth of investigation into a broader range of devices. To date, only Apple and Garmin smartwatches have received independent validation for SpO2 measurement. 30 However, the Apple Watch's performance at elevated altitudes remains undocumented. Garmin watches, while effective in most settings, 20 have sparked some debate. In a rapid ascent scenario from 1130 to 4559 m, they overestimated SpO2 by 7% relative to arterial blood gas analysis. 18 In a simulated high-altitude study ranging from 3000 to 5000 m, significant discrepancies were noted, with error rates surpassing 50% between altitudes of 3000 to 4800 m and exceeding 80% at higher altitudes. 19 Such overestimation of SpO2 could potentially mislead individuals venturing into high-altitude areas. In research employing maximum oxygen consumption as an indicator for predicting AMS, smartwatch-based measurements have typically deviated from those of cardiopulmonary exercise tests by an average of 6%. 15 These findings suggest that, despite the promising capability of smartwatches to measure SpO2, further confirmation and refinement of their accuracy and reliability at high-altitude environments are essential.
The frigid climate and fickle weather of high-altitude regions may markedly affect data acquisition and integrity. 31 Smartwatches necessitate optimal temperature and humidity conditions for accurate functioning, and extreme environments might result in unreliable readings and fluctuating precision. 32 Consequently, our study mandated that all measurements be taken in indoor settings, such as hotels or tents. Although the Huawei Watch is theoretically designed for SpO2 monitoring between 2500 and 4500 m, our findings indicate its efficacy surpassing this range. Our results are consistent with pulse oximeter data, showing clear distinctions in SpO2 levels between AMS+ and AMS− subjects. 27 It should be highlighted that this study set specific criteria for device fit, positioning, and timing of measurements. While smartwatches offer valuable insights into personal physiological metrics, they are not a substitute for medical diagnosis and should be supplemented with professional AMS diagnostic instruments for precise assessment.
Limitations
The study, undertaken at elevated altitudes, encountered participant recruitment challenges due to the distinctive environmental context, leading to a modest sample size, with a notable underrepresentation of female participants. Moreover, the reliance on optical sensor technology in wearable devices to ascertain SpO2 readings could encounter interference from ambient light and participant movements, risking potential bias in data collection. Considering the physiological variance among individuals, it is advisable to integrate SpO2 measurements with LLS, taking into account individual physiological differences for AMS diagnosis. Furthermore, this research utilized a single smartwatch model for data gathering without cross-referencing its measurements against medical-grade pulse oximeters. Comparisons between smartwatches and medical-grade level portable oximeters should be made at different altitudes in the future. The infrequency of mild and moderate AMS cases might restrict the definitiveness of our findings, impacting a thorough evaluation of the smartwatch measurement precision. Consequently, future research should expand the participant pool and incorporate various smartwatch models for validation to bolster the study's reliability and applicability.
Conclusion
The conclusions drawn from our study suggest that employing smartwatches for SpO2 measurement in altitudes exceeding 4000 m demonstrates commendable reliability and notable accuracy in the diagnosis and anticipation of AMS. However, further research is needed to validate the performance of smartwatches across a broader range of high-altitude environments and in comparison, with medical-grade pulse oximeters.
Footnotes
Acknowledgments
The authors would like to express their sincere gratitude to all the participants involved in this study.
Contributorship
ZZ conceptualized the study, collected the original data, and drafted the initial manuscript. LL, LH, and KW organized and analyzed the data. LL secured funding, reviewed, and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the Academic Integrity and Research Ethics Committee of China University of Geosciences (Wuhan, China) and registered with the Chinese Clinical Trial Registry (registration number: ChiCTR2400088099).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Open Fund of Hubei Research Center for Leisure and Sports Development (grant number 2023B010), the China University of Geosciences (Wuhan) (grant numbers 2023016 and 2023FZB05), and the Talent Development Professional Committee of the China Society for Educational Development Strategies (grant number RCZWH20230028).
Guarantor
LL
