Abstract
Objective
Epileptic seizures are neurological events that pose significant risks of physical injuries characterized by sudden, abnormal bursts of electrical activity in the brain, often leading to loss of consciousness and uncontrolled movements. Early seizure detection is essential for timely treatments and better patient outcomes. To address this critical issue, there is a need for an advanced artificial intelligence approach for the early detection of epileptic seizure disorder.
Methods
This study primarily focuses on designing a novel ensemble approach to perform early detection of epileptic seizure disease with high performance. A novel ensemble approach consisting of a fast, independent component analysis random forest (FIR) and prediction probability is proposed, which uses electroencephalography (EEG) data to investigate the efficacy of the proposed approach for early detection of epileptic seizures. The FIR model extracts independent components and class prediction probability features, creating a new feature set. The proposed model combined integrated component analysis (ICA) with predicting probability to enhance seizure recognition accuracy scores. Extensive experimental evaluations demonstrate that FIR assists machine learning models to obtain superior results compared to original features.
Results
The research gap is addressed using combined features to improve the performance of epileptic seizure detection compared to a single feature set. In particular, the ensemble model FIR with support vector machine (FIR + SVM) outperforms other methods, achieving an accuracy of 98.4% for epileptic seizure detection.
Conclusions
The proposed FIR has the potential for early diagnosis of epileptic seizures and can significantly help the medical industry with enhanced detection and timely interventions.
Introduction
Epileptic seizures are a neurological disorder characterized by abnormal and excessive electrical activity in the brain. 1 Seizures can manifest in various ways, including convulsions, loss of consciousness, and altered sensations, depending on which area of the brain is affected. Epileptic seizures can be a result of genetic factors, brain injuries, infections, and tumors. These seizures can have significant adverse effects on health and may lead to mortality. 2 People with epilepsy face a higher risk of premature death compared to the general population. This risk is partly due to the potential for fatal accidents during seizures, such as drowning, falls, or car accidents. Additionally, epilepsy is associated with an increased risk of developing comorbidities such as depression, anxiety, and cognitive impairment, contributing to reduced quality of life and increased mortality rates. In fact, epileptic seizures are a significant cause of mortality in people with epilepsy, accounting for ∼1 in 1000 deaths each year.3,4 To address this issue, an efficient machine learning model is needed for the early detection of epileptic seizures to improve the management and overall quality of life for individuals with epilepsy.
Advancements in artificial intelligence (AI) have recently transformed the field of medical diagnostics, providing exciting opportunities for improved identification and treatment of neurological diseases. AI techniques, such as deep learning, fuzzy logic systems, and ensemble methods, have demonstrated impressive effectiveness in detecting many illnesses, including schizophrenia, attention deficit hyperactivity disorder (ADHD), autism spectrum disorder (ASD), and epilepsy.
A review is performed using deep learning techniques for neuroimaging diagnoses such as ASD. 5 Schizophrenia disorders are detected using EEG signals by implementing hybrid deep learning techniques such as convolutional neural networks (CNNs) and long short-term memory (LSTM) (CNN–LSTM). This technique, combined with the functional connectivity feature, shows the most promising results. 6 Another advanced method is the adaptive neuro-fuzzy inference system (ANFIS) classifier used for the detection of epileptic seizures using EEG signals. This is tested on different datasets and implemented multiple feature extraction techniques. 7 The computer-aided diagnosis system used to automatically detect epileptic seizures using EEG signals is proposed by Shoeibi et al. 8 The data is preprocessed, and features are extracted after machine learning and deep learning models are applied; the proposed CNN and recurrent neural network (RNN) (CNN–RNN) model shows the most promising results for the diagnosis of epileptic seizures. A review of deep learning techniques is performed for detecting and forecasting COVID-19 tweets. 9 The real-time computer-aided diagnosis system based on advanced deep learning techniques works well for real-time detection. The fusion approach is used for rapidly diagnosing the COVID-19 virus on X-ray images. 10 The CNN, support vector machine (SVM), and Sobel filter are used for detection.
AI involves using machine learning algorithms that enable computers to learn from data and make predictions based on that data. 11 One application of AI in healthcare is the detection of epileptic seizures using electroencephalography (EEG) signals. EEG signals are commonly used to detect epileptic seizures, and machine learning algorithms have shown promising results in identifying seizure onset and classifying seizure types. 12 EEG signals are recordings of the brain's electrical activity, providing valuable information about the occurrence and duration of seizures. AI algorithms can analyze EEG signals in real time to detect the onset of a seizure and promptly alert healthcare providers, thereby improving patient safety and the quality of care by enabling timely intervention during a seizure.
Similar to the prognosis of other diseases like cardiovascular disease, kidney disease, and lung disease, a large approach automatically classifies abnormal and normal EEG signals. The ANN outperformed the other algorithms with 97% accuracy. Similarly, Shen et al. 18 proposed an ensemble method for detecting epilepsy seizures through EEG brain signals. Two different publicly available datasets UB and CHB–MIT were used for training and evaluating the applied models. The study evaluated the results using machine learning and ensemble learning algorithms, demonstrating that a 97% accuracy score was achieved using ensemble learning techniques. However, there is still a need for improvement to ensure accurate diagnosis of epilepsy seizures.
The authors collected a dataset in Varone et al. 13 from different medical centers in Italy during 2016 and 2019 for epileptic seizure classification. The authors applied various machine learning algorithms including SVM, linear discriminant analysis (LDA), and multilayer perceptron (MLP) both as stand-alone models and ensemble models. The proposed optimized MLP achieved a 91% accuracy for seizure detection. Despite the optimization of models, the obtained accuracy is low, and further enhancements are needed.
The study 19 focuses on predicting epileptic seizures using advanced deep learning models. The researchers utilized the American Epilepsy Society Seizure Prediction Challenge dataset to develop and evaluate the models. The authors proposed five deep learning models 1-CNN, 2-CNN, 3-CNN, 4-CNN, and 5-CNN, along with a transfer learning ResNet50 model. Among these models, the 5-CNN model achieved the highest accuracy score of 95%. However, there is still room for further performance improvement, especially from the perspective of epilepsy diagnosis. Similarly, Halawa et al. 20 focuses on epileptic seizure prediction using deep learning models. Results are based on the analysis of the pre-ictal and inter-ictal signal dataset known as CHB–MIT. Important features are extracted from the signals and fed into the proposed 1D CNN model for classification, which demonstrates an accuracy of 97%. Nevertheless, there is a need for further improvement.
The epilepsy classification using parameters from wearable sensors is investigated in Kadu et al. 21 The authors utilized the wearable sensors’ health record dataset to develop machine learning and deep learning techniques. Experimental results show that the proposed EMLR–FLIS model achieved an accuracy of 97% outperforming existing approaches. The study 14 focuses on the prediction of epileptic seizures using deep learning methods. The authors utilized the publicly available CHB–MIT dataset and employed several advanced deep learning techniques for experimentation. The proposed hybrid deep learning model, DenseNet–LSTM, performed well, achieving an accuracy of 93%. However, it is worth noting that the model's performance was lower compared to the baseline accuracy.
The prediction of epileptic seizures can be made using scalp signals, as demonstrated in Dissanayake et al. 15 The benchmark CHB–MIT–EEG dataset is employed to evaluate and predict body of work can be found on epileptic seizures.13-16 For example, the study 17 utilized EEG signals to detect epileptic seizures. Experiments were conducted using publicly available EEG data from Bonn University. Genetic algorithms were combined with four different machine learning algorithms k-nearest neighbor (KNN), SVM, ANN, and naive Bayes (NB). The performance of applied models is evaluated. An advanced deep learning Siamese model is trained to learn strategies for pattern verification from various brain signals. The authors report that the proposed model achieves a notable 91% accuracy score; however, its performance is relatively poor compared to other approaches. Similarly, Dissanayake et al. 22 proposed the geometric deep learning (GDL) technique, which improves the prediction of epileptic seizures to a 95% accuracy.
The UCI Epileptic Seizure dataset is utilized in Rahman et al. 23 for epileptic seizure detection. Various machine learning techniques including RF, SVM, and MLP are employed. Hyperparameter tuning is performed using random search cross-validation. Different performance evaluation metrics are utilized, such as accuracy, precision, recall, F1 score, receiver operating curve (ROC), and specificity. The results indicate that SVM exhibits good performance with a 97% accuracy. Along the same directions, Varnosfaderani et al. 16 focuses on epileptic seizure prediction using the Melbourne dataset and various deep learning models. The authors employed a two-layer LSTM with the swish activation function for classification. The proposed model achieved the highest accuracy score of 86% and a sensitivity score of 85% in this study. However, the performance scores obtained were relatively low in comparison to other methods. The study 24 focuses on epileptic seizure detection using the publicly available CHB–MIT dataset. The researchers employ deep learning techniques, specifically 1D CNN and CNN with LSTM, to conduct experiments. The proposed models, 1D CNN and CNN–LSTM, demonstrate promising results with an accuracy of 94%.
Research gap
The following points comprehensively outline the research gaps identified in the studies reviewed in the literature:
Primarily, binary class classification of epileptic seizure detection was solved. Classical feature engineering techniques were used for EEG signal data feature extraction. The performance scores reported in previous studies were low. Previous studies used classical algorithms; there is a need for ensemble learning models for the classification of epileptic seizures.
Table 1 shows a comparative overview of the discussed research works on epileptic seizure detection. Despite the use of different machine learning and deep learning models,25–28 it can be observed that further improvements should be made to improve seizure detection accuracy. In particular, the feature engineering aspect of machine learning models is not investigated very well. So, this study focuses on designing a novel feature engineering approach to enhance the accuracy of machine learning models.
The summary analysis of analyzed literature studies.

Study contributions
It can be observed from the discussed literature that, predominantly, the existing approaches are marked by low performance for epileptic seizure detection. This study aims to develop an accurate and efficient approach that can identify seizure activity with high sensitivity and specificity.
For this purpose, this study designs a novel feature fusion approach to enhance the performance of machine learning models for diagnosing epilepsy. Several advanced machine learning and deep learning approaches are implemented, and performance is optimized using hyperparameter tuning. The primary contributions of the proposed research for detecting epileptic seizures are as follows:
A novel ensemble approach, FIR, is proposed, which extracts independent components and class prediction probability features, creating a new feature set. Advanced machine learning and deep learning approaches are implemented to investigate the performance of the proposed approach. Logistic regression (LR), SVM, Gaussian NB (GNB), random forest (RF), gated recurrent unit (GRU), and LSTM models are utilized for experiments. Performance is validated using k-fold cross-validation. Additionally, we have performed computation complexity analysis to assess the efficiency of the applied models with FIR. Study results show that using the proposed approach, the applied method outperformed state-of-the-art approaches.
The remaining research is categorized into the following sections. The proposed method for the diagnosis of epileptic seizures is presented in
Material and methods
The materials and methods for diagnosing epileptic seizures are analyzed in this section. The dataset used and the applied models are also described.
The stepwise workflow of the adopted methodology is illustrated in Figure 1. A novel ensemble approach is proposed, which extracts independent components and class prediction probability features, creating a new feature set. We utilized the EEG brain activity signals dataset for experiments. Experiments are performed using an 80–20 train-test split. We employed several advanced machine learning and deep learning models. Models are evaluated using several performance evaluation metrics.

The workflow of the adopted methodology.
EEG brain activity signals data
In this study, we utilized a publicly available benchmark EEG brain activity signals dataset for experiments. 29 The original dataset was collected at the University Hospital Bonn, Germany, and reported in Andrzejak et al. 30 The dataset comprises 11,500 observations, with each observation consisting of 178 data features representing 1 second of EEG recording. Data collection was performed in a real-time environment by placing subjects under observation in the hospital and recording their EEG signals. The dataset contains five types of brain activity, including eyes closed, epileptic seizure, eyes open, tumor brain area, and healthy brain area. The dataset is balanced with each class having a sample size of 2300. All target classes have an equal number of data samples, indicating a balanced distribution of records.
Proposed feature fusion approach
This study proposes a novel feature fusion approach called FIR. The working architecture of the proposed approach is shown in Figure 2.

Novel proposed feature engineering approach analysis.
Independent components using FastICA
First, the EEG signal data is input to the FastICA model, 31 which extracts independent component features. FastICA is a computational technique used in signal processing to separate a multivariate signal into additive, independent components. It is commonly used for extracting independent components from mixed signals. The primary goal of FastICA is to find a linear transformation of input data, such that the resulting components are as statistically independent as possible. Then, class prediction probability features are extracted using the RF model. Finally, the features extracted from both approaches are fused together to create a new feature set. Study results show that, by using the proposed approach, the applied method outperformed state-of-the-art approaches. Algorithm 1 shows the step-by-step flow of the proposed feature fusion approach.
Advantage of using ICA features
The inclusion of ICA features in this study confers a critical advantage in the preprocessing and analysis of EEG data. ICA allows for the decomposition of multichannel EEG signals into statistically independent components, each representing a distinct underlying source or process. This separation is crucial in isolating relevant seizure-related activity from noise and artifacts, which are prevalent in EEG recordings. By employing ICA, we are able to extract meaningful features that are directly related to the epileptic seizure phenomenon, thereby enhancing the signal-to-noise ratio and improving the overall performance of our detection algorithm.
Class prediction features
Furthermore, the integration of class prediction features complements the ICA-based preprocessing approach. These features are derived from a comprehensive analysis of both time-domain and frequency-domain characteristics of the EEG signal. They encapsulate essential information regarding the dynamic changes and patterns exhibited by the brain during different stages of an epileptic seizure. By incorporating class prediction features, our algorithm gains the capacity to discern subtle variations in the EEG signal that may be indicative of pre-ictal, ictal, and post-ictal states, thus enabling more accurate and reliable seizure detection.
Algorithm 1 FIR Algorithm

In summary, the combined utilization of ICA features and class prediction features synergistically enhances the efficacy of our approach for epileptic seizure detection. ICA aids in the robust separation of relevant signal sources, while class prediction features capture critical temporal and spectral information crucial for accurate classification. This integration contributes to a more comprehensive and discriminating analysis of EEG data, ultimately leading to improved diagnostic outcomes for individuals with epilepsy. Despite these advantages, ICA can be a computationally intensive process, especially in the context of EEG signal analysis. However, the primary objective of this study is to explore the feasibility and efficacy of an alternative approach, specifically one that operates without preprocessing of EEG signal data. The proposed methodology leverages a novel framework that bypasses the traditional preprocessing steps typically associated with ICA. By doing so, we aim to streamline the analytical process and reduce the reliance on subjective researcher judgment. This alternative approach has been designed to offer a more direct and efficient means of extracting meaningful components from EEG data.
Applied machine learning and deep learning models
Machine learning and deep learning techniques have shown great promise in the domain of epileptic seizure diagnosis using EEG brain activity signals. These advanced methods32–34 allow for the automated analysis and classification of EEG data, aiding in the early detection and accurate diagnosis of epileptic seizures. By extracting relevant features from EEG signals and employing sophisticated algorithms, these techniques can provide valuable insights to healthcare professionals, leading to improved patient care and better management of epilepsy.
Logistic regression
LR is a widely used and effective method for epileptic seizure diagnosis using EEG brain activity signals. 35 LR uses a sigmoid activation function to calculate the probability of a seizure occurring. Each input characteristic is assigned a weight coefficient that is learned throughout the training process. LR utilizes a threshold to determine binary classification judgments based on the estimated probabilities, allowing for the prompt identification of seizures. It offers a simple yet powerful approach to classify EEG data into seizure and non-seizure states, enabling accurate and timely detection of epileptic episodes. By leveraging the features extracted from EEG signals, LR can provide valuable insights for clinicians in making informed decisions about patient treatment and care. The interpretability, speed, and robustness of LR make it a helpful tool for assisting in the management of epilepsy.
Support vector machine
SVM has emerged as a powerful model for epileptic seizure diagnosis using EEG data. 36 This converts the properties of EEG signals into high-dimensional space. A hyperplane is created to maximize the distance between different classes indicating the difference between seizures and non-seizure classes. SVM determines the best hyperplane during training which are the best points closely located to the decision boundary. This position of the hyperplane is very important for effectively generalizing the new unseen data. SVM distinguishes between seizure and non-seizure EEG patterns successfully by utilizing the acquired hyperplane, enabling precise and early identification of epileptic seizures. Accurate classification results can be achieved by leveraging the SVM's ability to find an optimal hyperplane that separates the classes. Moreover, the SVM model is well-suited for high-dimensional data like EEG signals, making them a promising tool for enhancing epilepsy diagnosis and providing valuable insights for effective medical interventions.
Gaussian Naive Bayes
GNB model is employed to detect epileptic seizures by modeling the probability distribution using EEG brain activity signals for detecting both seizure and non-seizure occurrences. 37 The GNB algorithm effectively follows Gaussian distribution within each class by assuming that each feature is independent. GNB estimated the mean and variance of each feature for both classes during the training. GNB efficiently estimates the probability distribution by assuming feature independence within classes by employing the Bayes theorem and choosing the class with the highest posterior probability to classify new instances. Employing GNB can efficiently handle the high-dimensional data and assume independence across features the model can effectively differentiate between seizure and non-seizure data.
Random Forest
RF is a powerful model for detecting epileptic seizures by creating an ensemble of decision trees, where each tree is trained by creating a random subset of features from EEG signals. 38 The bootstrap samples of training data are used to create multiple trees and a subset of features on each node is randomly selected. Each decision tree is independently classified by considering the majority class of instances on each leaf node. To classify new instances, RF works based on the probabilities of all the decision trees. By combining the predictive abilities of multiple decision trees, RF provides robustness and accuracy in classifying seizure patterns. Its ability to handle high-dimensional EEG data and automatically select relevant features makes it well-suited for this application. Moreover, RF's capacity to handle imbalanced datasets enhances its performance in detecting rare seizure events. As a result, the RF model holds great promise as an effective tool for improving the accuracy and efficiency of epilepsy diagnosis, contributing to better patient care and treatment outcomes.
Gated Recurrent Unit
GRU model can provide robust and highly accurate results for epileptic seizure detection. 39 GRU is a type of RNN that can effectively capture temporal dependencies in sequential data like EEG signals. GRU consists of gates that control the flow of information inside a network such as an update gate and reset gate. During the training of the GRU model, it acquires the ability to modify and reset its internal states according to the input sequence. By incorporating gating mechanisms, GRU can selectively retain or forget relevant information, enabling to modeling of long-term dependencies in the EEG signals while avoiding the vanishing gradient problem. This can be learned to reflect the temporal dynamics of seizure patterns by processing sequential data through its recurrent connections and gradient processes. This enables accurate identification of seizures within the EEG signals sequence.
Long short-term memory
The LSTM model has emerged as a promising approach for efficiently diagnosing epileptic seizures. 40 Its ability to capture long-range dependencies and process sequential data makes it well-suited for analyzing EEG signals, which are time series. LSTM uses a gating mechanism the input gate governs the flow of new information into memory cells the forget gate controls which information needs to be discarded from the memory cell and the output gate which information flows outward from the memory cell. By effectively learning temporal patterns and discriminative features from EEG data, the LSTM model shows potential in accurately detecting and classifying epileptic seizures, leading to improved diagnosis and better patient care.
Hyperparameter optimization
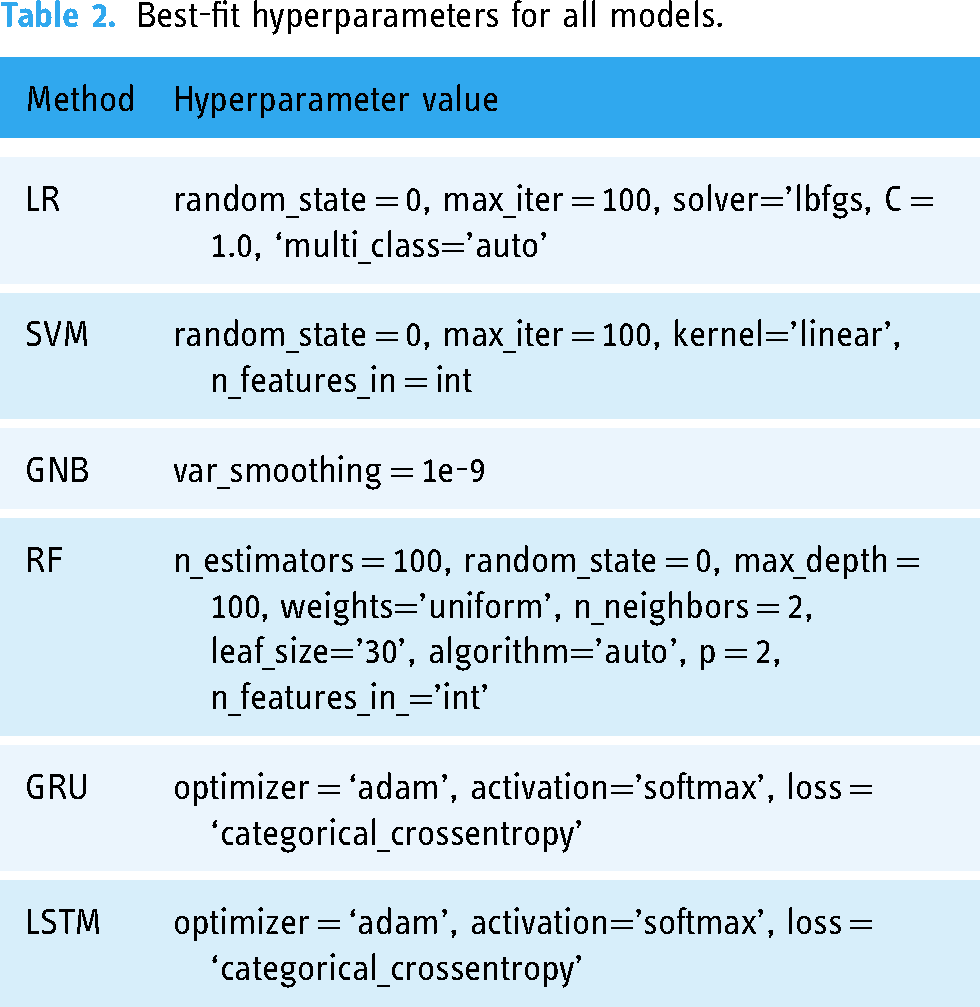
Hyperparameter optimization is an essential component of machine learning and deep learning. 23 The selection of appropriate hyperparameters can be a challenging process that involves a train-and-evaluate approach. A well-tuned set of hyperparameters can significantly impact a model's accuracy, convergence speed, and generalization capabilities. The best-fit hyperparameters are determined for the applied techniques using the GridSearchCV method and are given in Table 2. GridSearchCV methodically examines a predetermined grid of hyperparameters, assessing each combination by cross-validation. This procedure involves splitting the training data into smaller groups and repeatedly training the model in one group while using the remaining data for validation. The performance of the model is evaluated for each combination of hyperparameters using a predefined measure. GridSearchCV systematically explores the whole range of hyperparameters to find the best configuration that maximizes the model's performance on new data. This approach guarantees that our models are effectively tuned to obtain the most beneficial results.
Best-fit hyperparameters for all models.
Results
This section presents experiments involving machine learning and deep learning techniques. The performance of each applied model is evaluated using the epileptic seizure recognition dataset, which consists of EEG brain signals related to epileptic seizures. Experiments are performed using machine learning and deep learning methods with both original features and the proposed feature engineering approach.
Experimental setup
Machine learning and deep learning models are developed using Python 3.0 programming language. All experiments are conducted using an open-source environment, Google Colab, with a GPU backend, 13 GB of RAM, and 90 GB of disk space. The performance metrics accuracy, precision, recall, and F1 score are utilized for evaluating the performance of epileptic seizure recognition from EEG brain signals.
Evaluation metrics
This study employs several well-known performance evaluation metrics to analyze the performance of machine learning and deep learning models. In particular, accuracy, precision, recall, F1 score, and false alarm rate (FAR) are used with the following equations:
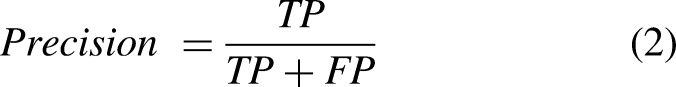



Machine learning models and original features
Table 3 describes the machine learning algorithm's comparative results using the original features. Results regarding the accuracy, precision, recall, and F1 score are displayed for machine learning models such as LR, SVM, GNB, and RF. The results of five different classes and their average are calculated as a classification report. The analysis demonstrates that SVM achieves poor performance with only a 0.19 accuracy score, followed by LR and GNB. In this analysis, the RF model archives the best performance with a 0.69 accuracy score. Precision, recall, and F1 scores are also the same. It can be inferred from the results that EEG data is more linearly separable, which results in the low performance of linear models.
Performance analysis of machine learning methods with original features.

Deep learning model using original features
Table 4 demonstrates the experimental results of deep learning models GRU and LSTM when used with the original features. The accuracy scores and class-wise performance metrics are also determined. Results show that LSTM shows poor performance with 65% accuracy, compared to GRU. The performance of GRU is slightly better, with an accuracy score of 67%. The results show that the performance of deep learning models LSTM and GRU is inferior to machine learning model RF which shows better performance with a 0.69 accuracy score. There is a need for an advanced feature engineering approach to enhance performance scores for diagnosing epileptic seizures.
Performance analysis of deep learning methods with original features.

Machine learning models with proposed feature engineering approach
To investigate the impact of the newly proposed feature engineering approach, experiments are conducted for both machine learning and deep learning models. The performance of machine learning models with newly generated features is given in Table 5. The results show that the GNB model obtains an accuracy score of 0.97 which is a substantial improvement compared to using the original features. However, LR, SVM, GNB, and RF exhibit better performance using new features compared to the results calculated with the original features. The results demonstrate that LR, SVM, and RF algorithms perform well with a 0.98 accuracy score and exhibit good precision, recall, and F1 scores for each class. It is also observed that the performance of models for target classes 2, 3, and 5 is better than that of classes 1 and 4. The average performance of all classes is also better. We can say that the proposed feature engineering approach significantly improves the performance of all machine learning methods.
Performance analysis of machine learning methods with the proposed feature engineering approach.

Bold values in Table 5 are average values as main results.
The performance of machine learning algorithms using the proposed feature approach concerning correct and wrong predictions is illustrated in Figure 3. The analysis shows that LR, SVM, and RF techniques achieved the minimum error rates. Wrong predictions for LR, SVM, GNB, and RF are 50, 35, 65, and 41, respectively out of a total of 2310 predictions. GNB shows the highest number of wrong predictions and especially shows poor performance for target class 2.

Confusion matrix for machine learning models using the proposed feature engineering approach.
Deep learning models with proposed feature engineering approach
Similar to machine learning models, deep learning models are also trained using the newly generated feature set, and their results are analyzed regarding detection accuracy.
Figure 4(a) and (b) shows the training and validation accuracy and loss for GRU and LSTM, respectively. Performance is shown for 10 epochs of training, evaluating the training loss, validation loss, train accuracy, and validation accuracy parameters. The analysis reveals that both GRU and LSTM achieved low performance scores during the first three epochs. However, through optimization, the models attained enhanced performance. Specifically, the training loss of GRU and LSTM algorithms was high in the first two epochs but gradually decreased over subsequent epochs. Moreover, both training accuracy and validation accuracy showed a positive trend over time.
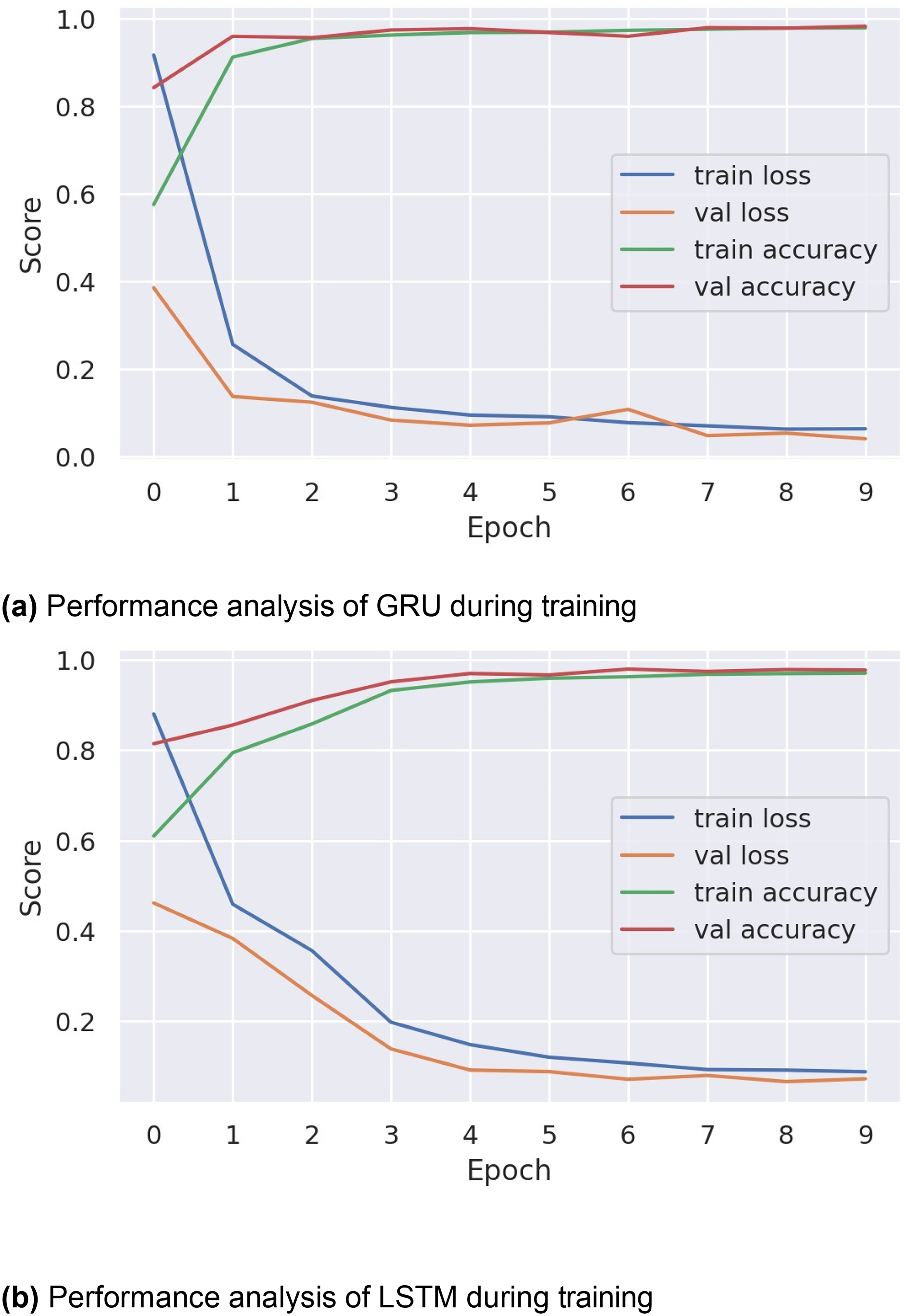
The timer series–based performance analysis of deep learning methods during training.
The results of the deep learning techniques GRU and LSTM with novel features are presented in Table 6. Results demonstrate that the LSTM algorithm yields acceptable results with the proposed features, achieving a 0.97 accuracy score. On the other hand, the GRU performs exceptionally well, reaching a 0.98 accuracy score. Additionally, precision, recall, and F1 scores for all target classes exhibit promising results for both LSTM and GRU. Overall, the application of newly generated features has improved the performance of deep learning models compared to the original features.
Performance analysis of deep learning methods with the proposed approach.

The performance of deep learning models using the proposed features is illustrated in Figure 5. The analysis shows that GRU techniques achieved the minimum error rates with 43 wrong predictions. However, the LSTM approach resulted in a moderate error rate for target class 2, and it has a total of 59 wrong predictions out of 2310 predictions. This analysis validates the effectiveness of applied deep learning for the diagnosis of epileptic seizures.

Confusion matrix for deep learning models with new features.
Results with proposed features after data splitting
This section contains the validation of our proposed feature engineering technique after data splitting in the methodology. First, we split our dataset into training and testing portions. Then, our proposed feature engineering is applied to each portion to extract features. The extracted features serve as input for applied machine learning models, and the results are demonstrated in Table 7. The performance analysis shows that the GNB approach achieved an average accuracy of 0.96. This analysis validates our proposed feature engineering mechanism and demonstrates that our proposed SVM approach achieved high accuracy scores of 0.99.
Performance analysis with proposed features after data splitting.

K-fold cross-validation
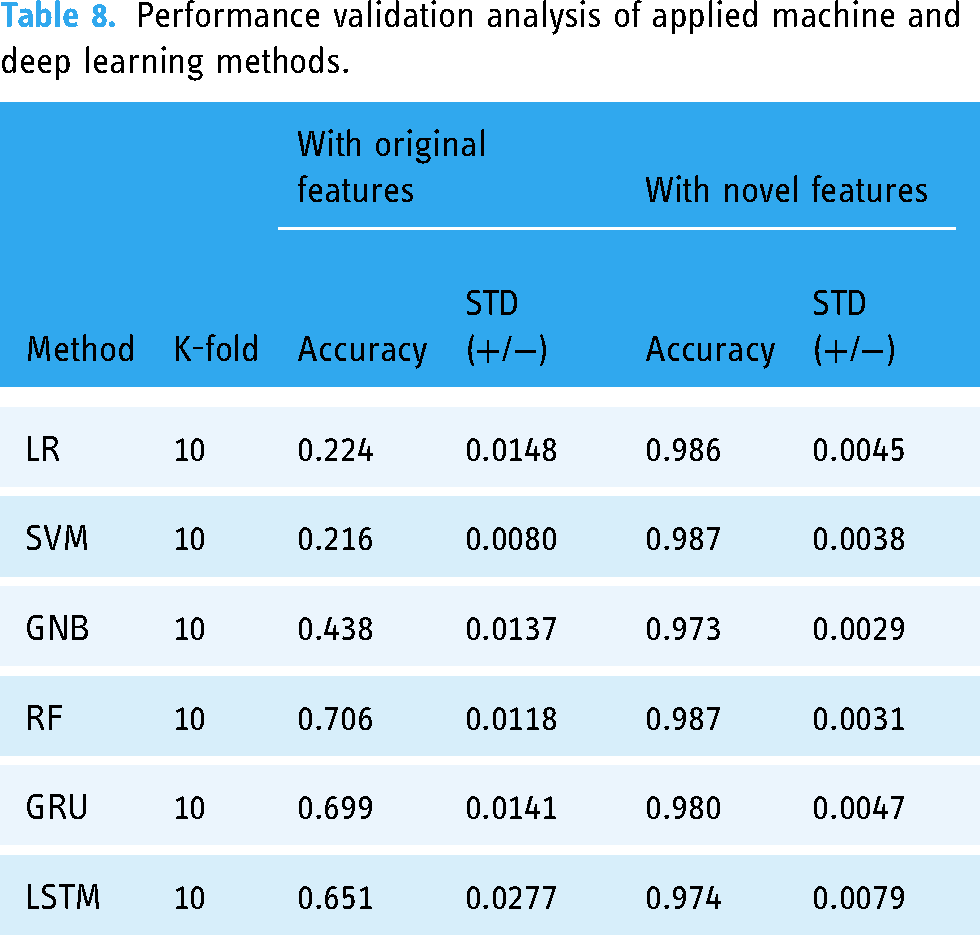
The performance of each applied method is validated with a cross-validation and the results are reported here. The performance validation results, with both original and new features, are analyzed in Table 8. Machine learning techniques and deep learning techniques are evaluated using 10-fold cross-validation. The analysis demonstrates that the accuracy scores of all the machine learning and deep learning techniques with the original features are inferior. However, the highest performance validation accuracy scores are achieved with the newly generated features with minimal standard deviation. The analysis score of k-fold cross-validation confirms the generalization performance of the machine learning and deep learning techniques for the diagnosis of epileptic seizures.
Performance validation analysis of applied machine and deep learning methods.
Computational complexity analysis
The comparative complexity analysis of applied models both with the original feature and the proposed approach is demonstrated in Table 9. The analysis shows that the computation time of all the models is high when using the original features. On the other hand, the computation time of all algorithms with the proposed feature engineering approach is lower. The results show that deep learning models LSTM and GRU take a longer computation time, while the time for machine learning models is shorter. In conclusion, the computation time of all algorithms with the proposed approach is reduced.
Computational complexity analysis of machine and deep learning methods.

Comparison with state-of-the-art studies
Here, we compare the performance of the proposed approach with existing state-of-the-art studies that utilize the EEG signals dataset. The comparative analysis is presented in Table 10. The highest accuracy of 97% is reported in existing state-of-the-art approaches. However, this study shows improvement in the detection accuracy of epileptic seizures with the proposed approach. Performance comparison results show that the proposed approach outperforms the state-of-the-art method, achieving a high accuracy of 98.4%.
Comparisons analysis with state-of-the-art studies.

Bold values in Table 10 are average values as main results.
Discussion
Performance generalization analysis with external dataset
To validate the performance generalization of our proposed research approach, we have taken another EEG brain signals dataset 43 to evaluate the results. The performance results reported in Table 11 show that the proposed approach achieved a high accuracy of 0.97 and is generalized.
Performance analysis of proposed approach for external EEG signal dataset.

Bold values in Table 11 are average values as main results.
Clinical significance of current research
This research has important clinical implications in the field of epilepsy management. The study highlights the potential to transform epileptic seizure detection by introducing a unique feature fusion approach and implementing advanced machine learning and deep learning models. The use of nonlinear features such as the fractal dimension can also be important during features engineering. 44 The proposed method offers improved accuracy and efficiency, which has the potential to enable doctors to diagnose epilepsy promptly and accurately. In conclusion, this research makes a valuable contribution toward enhancing patient outcomes and improving the quality of life for individuals affected by epileptic seizures.
Research limitations
This research has the following limitations:
Machine learning models are trained using old datasets. There is a need for new dataset collection. Classical textual feature representations were used for feature engineering. An advanced transfer feature approach can be applied. Classical machine learning methods were applied, which achieved moderate performance scores. Advanced deep learning models can be applied to achieve high epileptic seizure detection performance.
Conclusions
This study proposed a reliable technique for detecting epileptic seizures using EEG signals. The primary goal is to propose a novel approach for the early warning precautions for individual risks and detection of epileptic seizure disease with high-performance capabilities. The scenario is incorporating our system into the wearable device which is capable of monitoring EEG signals in real time. When these devices identify patterns that suggest a seizure is about to happen, they can send alerts to the person experiencing the seizure, their caretakers, or healthcare experts. This allows for prompt treatments to reduce the effects of the seizure. Instead of using the original features, the proposed approach utilizes RF and independent component analysis to obtain class probabilities from these features. The output from RF and ICA is used to make a new feature set that serves models’ training. Experiments are performed using EEG brain activity data comprising five classes. For performance comparison, experiments are performed using both the original and newly generated feature sets with machine learning and deep learning models. Overall, machine learning models show better performance than deep learning models. Experimental results indicate a substantial improvement in the detection accuracy of epileptic seizures when models are used with the proposed feature engineering approach. Compared to a 69.1% accuracy of RF with the original features, the SVM model obtains the highest accuracy of 98.4% using the proposed approach, besides a low computational complexity of the proposed feature engineering. K-fold cross-validation further corroborates this performance. Additionally, performance comparison with existing studies shows superior performance of the proposed approach. The approach is to improve the quality of life for those with epilepsy by giving timely warnings and enabling preventive measures. Further research collaboration with medical professionals is required to verify and improve our method for practical implementation in clinical environments.
In the future, we intend to expand the feature space by collecting more data samples from hospitals. Furthermore, we will apply transfer learning-based models to enhance the performance of epileptic seizure detection.
Footnotes
Author's note
Adnan Akhtar is also affiliated with the Regional Center, Allama Iqbal Open University, Rahim Yar Khan, 9239166, Punjab, Pakistan.
Contributorship
MK conceived the idea, performed formal analysis, and wrote the original manuscript. AR conceived the idea, performed data curation, and wrote the original manuscript. AA designed the methodology, and performed data curation and formal analysis. FR dealt with software, performed visualization, and designed the methodology. JBB acquired funding and performed visualization and project administration. CLR provided resources, performed investigation, and dealt with software. IdlTD performed the investigation, project administration, and validation. IA supervised the work and performed validation and writing (review and editing) of the manuscript. All authors reviewed and approved the work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study uses a publicly available dataset provided by Rochester Institute of Technology.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European University of the Atlantic (grant number N/A).
Guarantor
Not applicable.
Informed consent
Ethical consent from the patient was not needed.
