Abstract
Background
Individuals who have metastatic cancer experience substantial physical and psychological distress (e.g., pain, depression, anxiety) from their disease and its treatment compared to patients with less advanced disease. As the burden of symptoms varies over time, ecological momentary assessment (EMA) may be used to better understand patients’ symptom trajectories, complimenting traditional longitudinal data collection methods. However, few have used EMA in patients with metastatic disease. The current study adds to the existing literature by exploring interrelated, common cancer-related symptoms of pain, anxiety, and depression and use of cannabis-based products, opioid medications, other (nonopioid) pain medications, and medications for anxiety or depression.
Methods
An eight-day prospective observational feasibility study was conducted among 50 patients with metastatic cancer recruited from seven solid cancer clinics at The Ohio State University Comprehensive Cancer Center. Participants completed a week of interval-contingent mobile EMA, administered daily at 9 a.m., 3 p.m., and 8 p.m., and a comprehensive interviewer-administered questionnaire on Day 8. Participants were queried on their symptom burden and management strategies (i.e., use of medications and cannabis). We considered EMA to be feasible if a priori retention (80%) and adherence goals (75%) were met.
Results
Seventy-nine percent of eligible patients contacted enrolled in the study (n = 50 of 63). Among those enrolled, 92% were retained through Day 8 and 80% completed >90% of EMAs, exceeding a priori objectives. Participants’ average pain, anxiety, and depressive symptoms across the week of EMA ranged from 1.7 to 1.8 (1 to 5 scale). Symptoms varied little by day or time of administration. On Day 8, significant proportions of participants reported past-week use of medications and cannabis for symptom management.
Conclusions
Participants exceeded a priori adherence and retention objectives, indicating that mobile EMA is feasible among metastatic cancer patients, addressing a gap in the existing literature and informing future research. Restricting eligibility to participants with a minimum cutoff of symptom burden may be warranted to increase observations of symptom variability and provide opportunities for future health interventions. Future research is needed to test the acceptability and quality of data over a longer study period in this patient population.
Introduction
Individuals who have metastatic cancer experience substantial physical and psychological distress (e.g., pain, depression, anxiety) from their disease and its treatment compared to patients with less advanced disease.1–8 Approximately 75% of patients with advanced cancer experience pain, and severe pain is one of the most common reasons for emergency department visits among such patients. 9 Up to 65% of patients with metastatic disease also experience anxiety, and 29% experience depression, both of which are significantly associated with adverse patient outcomes including pain, reduced quality of life, and increased mortality.10,11 Patients with advanced disease utilize a variety of strategies to mitigate cancer-related symptoms, including the use of prescription and over-the-counter medication, as well as using cannabis products. Among general oncology populations, prevalence estimates of cannabis product use range from 10% to 25%,12–14 and possibly higher among patients with advanced disease. 12 However, the relationships between symptom burden and symptom management strategies in patients with metastatic cancer are not well understood.
One strategy to measure patients’ symptom trajectory is ecological momentary assessment (EMA), which involves repeatedly sampling participants’ current behaviors and experiences in real-time and in their natural environments. 15 EMA methodology may be particularly useful because dynamic real-time, real-world changes in patients’ symptom burden and its management—each expected to vary substantially—can be readily assessed.16,17 EMA analysis highlights individual differences in patient-reported outcomes and behavior, their distribution over time, factors affecting outcomes and behavior, and mutual associations between these factors. 17 EMA can complement traditional retrospective self-reports by providing real-time context to symptom burden and related management strategies, and can reduce measurement error. 18
Recent data suggests that EMA is a feasible and cost-effective approach in oncology research,16,19 which has been used in >40 studies within cancer populations.20–23 However, four have focused on metastatic patients specifically, 21 and only two focused on patients’ symptoms and their management.24–26 Specifically, previous literature on EMA use among metastatic cancer patients is limited in measuring only one symptom (pain or sleep disturbance) 26 and use of pain medication, 24 thus limiting our understanding in this population. While the reason for this gap is unknown, it may be that patients diagnosed with metastatic disease have been overlooked as candidates for this methodology due to a presumed burden of EMA on this population. 27 Herein, we tested the feasibility—defined here as meeting EMA adherence and retention goals—of conducting mobile EMA in a pilot study of patients with metastatic cancer to inform future research by adopting methodology in this patient population. Secondarily, we aimed to characterize the burden of symptoms and symptom management (i.e., the use of medications and cannabis) in this population. The current study adds to the existing literature by exploring interconnected 28 and common cancer-related symptoms of pain, anxiety, and depression and use of cannabis-based products, opioid medications, other (nonopioid) pain medications, and medications for anxiety or depression.
Materials and methods
Overview
From October 2022 through February 2023, a convenience sample of adult patients with metastatic cancer was remotely recruited into an eight-day prospective observational study. Eligible patients from seven Ohio State University (OSU) Comprehensive Cancer Center (CCC) medical oncology clinics (breast, cutaneous oncology, gastrointestinal, genitourinary, gynecologic oncology, otolaryngology, and thoracic) were identified through electronic medical records. Trained research staff screened patients who met the study eligibility criteria: a metastatic cancer diagnosis, cancer treatment within the past 12 months, and access to an internet-connected smartphone so that they could complete the EMA surveys sent via a texted web link. Patients were excluded if they were diagnosed with noninvasive or nonmetastatic cancer, could not speak or read English, had a diagnosis of dementia or were otherwise unable to give informed consent, were receiving hospice care, or were unable to provide verbal informed consent to participate in research. The study was approved by OSU's Institutional Review Board. As this study represented observational research with minimal risk, a requirement for consent documentation was waived in favor of verbal consent procedures.
Recruitment
Research personnel called prescreened patients at least 24 h following their in-clinic oncology appointment. Research staff introduced themselves, affirmed eligibility, and further screened patients to ensure they had access to an internet-connected smartphone. If individuals indicated in the affirmative, research staff explained the study via a verbal consent script and verbally consented the participant into the study. Participants were also asked to allow research staff to access their medical records. Upon consent, research staff collected contact information, set up the EMA phone messages to the participant,29,30 and scheduled participants’ Day 8 interviewer-administered questionnaire.
Epidemiologic assessment (Day 8)
An overview of data collection is given in Figure 1. On Day 8, the OSUCCC's Recruitment, Intervention, and Survey Shared Resource conducted a 20–30 min interviewer-administered, phone-based questionnaire to assess participants’ symptom burden, medication use, use of cannabis products, and clinical and demographic information over the previous seven days. This questionnaire was previously validated in cancer patients and is modified with permission. 14 It was previously published as supplementary data. 12 Symptom burden: The Patient Reported Outcomes Measurement Information System® (PROMIS®) Pain Intensity (Version 1.1, short-form 3a), 31 Anxiety (Version 1.0, short-form 8a),31,32 and Depression (Version1.0, short-form 8b) batteries31,33 were used to assess participants’ pain and emotional distress over the past week. The pain scale included three items to assess three pain intensity domains: worst pain in the past week, average pain in the past week, and current pain. 31 Responses to the items used a five-point scale ranging from 1 (no pain) to 5 (very severe pain). The anxiety scale consisted of eight statements on the universal symptoms of anxiety (e.g., nervousness, tension, fear) within the past week and the depression scale included eight statements to assess emotional manifestations of depression within the past week (e.g., feelings of worthlessness, sadness, hopelessness). 33 Responses to the anxiety and depression items use five-point scales ranging from 1 (never) to 5 (always), with higher scores denoting greater anxiety and depressive symptoms.

Overview of the eight-day study's data collection procedures in 50 metastatic patients. Ecological momentary assessments were conducted three times daily for one week, followed by an interviewer-administered survey on Day 8.
Medications and cannabis: A modified version of a cannabis-focused questionnaire designed and validated in cancer patients, and previously used in a similar population was administered.12,14 Patients were asked about their past and current use of cannabis products, including both marijuana and cannabidiol. Patients were also asked to report their use of opioids and nonopioid pain medications, as well as medications used for anxiety, depression, and sleep. Examples of common medications within each category were provided to aid in recall.
Participant characteristics: Lastly, sociodemographic data on patients’ age, sex, race, education, and cancer-specific data (e.g., cancer site, degree of metastasis, and past and ongoing therapeutic and palliative therapies) were collected. Characteristics for participants not retained to the Day 8 assessment were abstracted from their medical records.
Ecological momentary assessment (Days 1–7)
The EMA portion of the study consisted of one week of mobile EMA surveys, administered three times a day, in the morning (9 a.m.), afternoon (3 p.m.), and evening (8 p.m.). The EMA questionnaire included three questions to measure current symptoms derived from their respective PROMIS pain intensity, anxiety, and depression batteries and was modified (in the case of anxiety and depression) to evaluate current symptoms rather than symptoms in the past week. Patients were asked, “What is your level of pain right now?” (the identical question is within the PROMIS pain intensity short form 3a), and the extent to which they endorsed the statements, “My worries overwhelm me right now,” and “I feel depressed right now.” Responses ranging from 1 to 5 were utilized as above, except for anxiety and depression questions, the response options ranged from “strongly disagree” to “strongly agree” instead of “never” to “always,” to account for items referring to the present moment. A final multipart question asked patients about their past-hour medication and cannabis product use, “Have you used any of the following products in the past hour?” Participants were asked to indicate yes or no to the use of cannabis-based products, opioid medications, other (nonopioid) pain medications, and medications for anxiety or depression. Because anxiolytics can be used to treat depression and antidepressants for anxiety, we did not request that participants specify the type of medication used for these symptoms.
Compensation and incentives
Participants were compensated with a $5 gift card for completing the Day 8 questionnaire, and a $5 or $10 gift card for completing <75% or ≥75% of EMA questionnaires, respectively. Participants who completed ≥75% of EMA questionnaires were additionally entered into a lottery with a chance to win an additional $10 gift card.
Statistical analysis
Rates of accrual, adherence, and retention were estimated. The accrual rate was defined as the proportion of enrolled participants (n = 50) among those contacted who passed screening (n = 63). The adherence rate was defined as the proportion of participants that completed ≥90% of the 21 EMA questions (considered “adherent”). Lastly, the retention rate was the proportion of enrolled participants that answered the Day 8 questionnaire (considered “retained”). The a priori adherence and retention goals for the EMA to be considered feasible were 75% and 80%, respectively.
Participants’ sociodemographic and clinical characteristics were examined by their retention and adherence statuses (yes/no). Symptom burden and symptom management during the week of EMA and on the Day 8 follow-up were summarized using frequencies and proportions for categorical variables; age was summarized using the mean and standard deviation (SD). EMA data on pain and emotional distress derived from PROMIS questions were summarized with means and standard deviations. Symptom data derived from the full PROMIS pain, anxiety, and depression batteries (i.e., rather than a single question used in EMA) were scored using a T-score relative to its reference population with a mean score of 50 (SD = 10). The same individual PROMIS item used for EMA (referring to current symptoms) as used on Day 8 (referring to symptoms over the past week) was examined. We also calculated “minimum symptom” levels, defined here as the proportion of participants who experienced at least one instance of moderate pain (≥3 on the 1–5 scale) or who “agreed”/”strongly agreed” (≥4) with the PROMIS anxiety or depression symptom statements. All analyses were performed using SAS 9.4 (SAS Institute, Cary, NC).
Results
One hundred twenty-two potentially eligible patients were identified in the respective clinics. Among them, 63 were contacted and successfully screened until reaching a total of 50 participants for a response rate of 79% consented to the study. Among 50 participants, 40 (80%) were adherent with EMA, and 46 (92%) were retained through the end of the study
Select sociodemographic and clinical characteristics among 50 metastatic cancer patients enrolled into the pilot study, displayed overall and stratified on retention and adherence status.

Includes medical record data for n = 4 who did not complete Day 8 questionnaire.
Includes n = 1 who completed Day 8 questionnaire but did not complete any EMA.
Education data were missing for n = 3 nonadherent participants.
Overall, the mean age of participants was 65 years, 54% were female, and the majority were white, high school graduates, and self-reported distant metastasis (Table 1). More than half of the participants were currently receiving systemic therapy. Participants’ characteristics did not differ appreciably by their retention or EMA adherence status.
Participants’ mean pain, anxiety, and depressive symptoms during the week of EMA and on Day 8 are summarized in Figure 2 and Table 2. Participants reported low symptom burden across the week with little variation by day or time of administration (Figure 2). Across the week, the mean (SD) EMA pain score was 1.7 (0.8), and 1.8 (0.9) for anxiety and 1.8 (0.8) for depressive symptom on scales of 1 to 5. On the Day 8 assessment, mean symptoms in the past week ranged from 1.4 to 1.7 (Table 2). Approximately half (47%) of participants reported a minimum pain score ≥3 at least once during the week of EMA. When reporting symptoms more broadly on Day 8, 13% reported at least moderate pain (score ≥3). Over 22% of participants indicated agreement or strong agreement with PROMIS statements of anxiety or depressive symptoms during the week of EMA. Overall, Day 8 PROMIS T-scores for pain, anxiety, and depression were approximately 50, indicating no difference from the reference population.
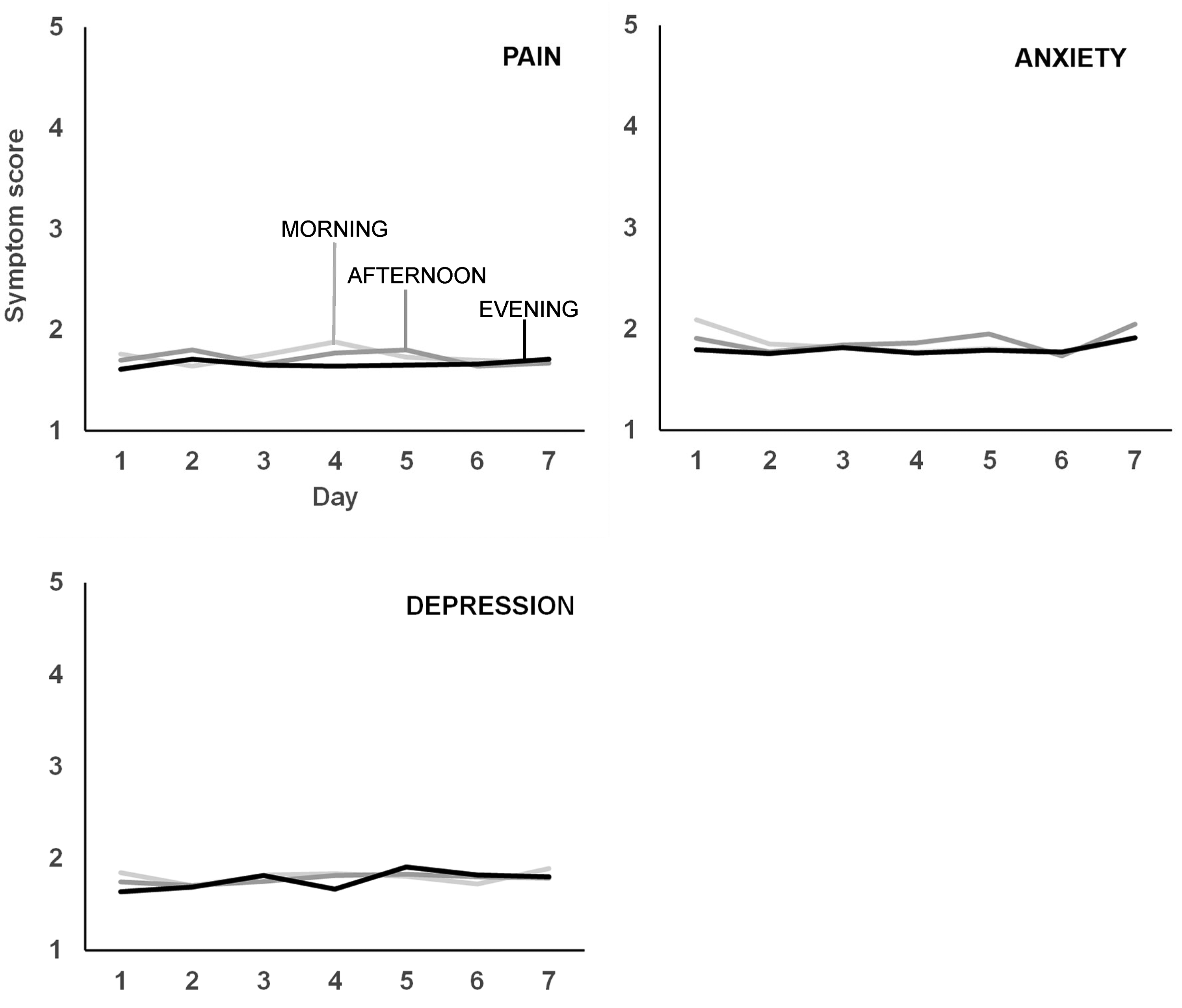
Mean daily pain, anxiety, and depression symptom scores across seven days of mobile, investigator-initiated EMA. Light, medium, and dark gray line shading correspond to morning (9 a.m.), afternoon (3 p.m.), and evening (8 p.m.) measurements. Ordinate scores reflect rankings of 1 (no) to 5 (very severe) pain, and 1 (strongly disagree) to 5 (strongly agree) for statements indicating current anxiety and depressive symptoms. Symptom scores were low and did not vary by time of day or across the week of EMA.
Symptom burden among 50 metastatic cancer patients.
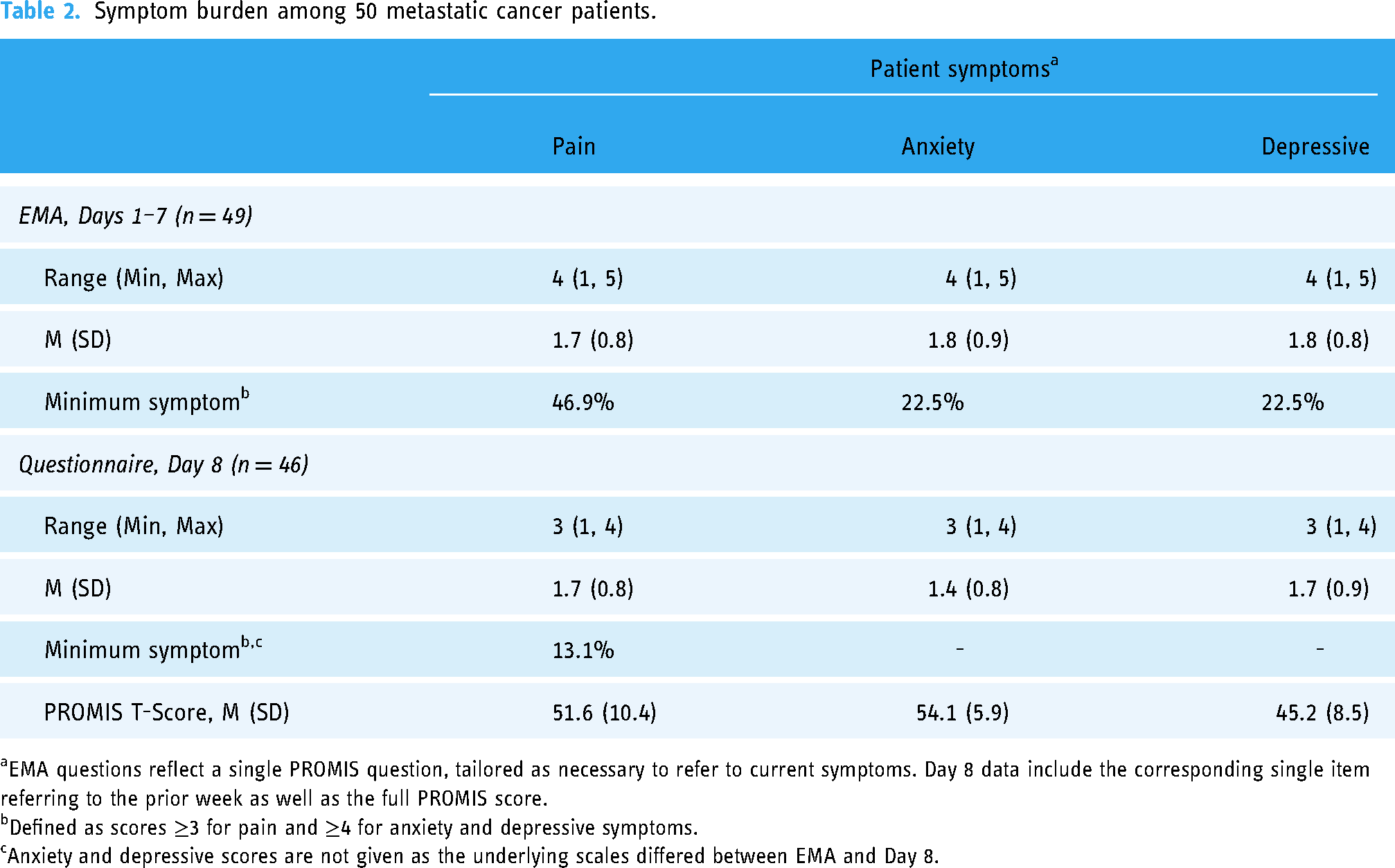
EMA questions reflect a single PROMIS question, tailored as necessary to refer to current symptoms. Day 8 data include the corresponding single item referring to the prior week as well as the full PROMIS score.
Defined as scores ≥3 for pain and ≥4 for anxiety and depressive symptoms.
Anxiety and depressive scores are not given as the underlying scales differed between EMA and Day 8.
The use of cannabis and medications during the study period is described in Figure 3. Among 46 participants who were retained to Day 8, eight (17%) reported using cannabis products in the past week. Twelve participants (26%) reported using opioid medications in the past week and 23 (50%) reported using nonopioid pain medications. An additional 12 (26%) participants reported use of medications for anxiety and eight (17%) reported use of medications for depressive symptoms. Participants’ indication of past-hour use of cannabis and medications during the week of EMA was similar to the Day 8 past-week assessment.

Past hour (Days 1–7; n = 49) and past week (Day 8; n = 46) medication and cannabis prevalence among 50 metastatic patients.
Discussion
This study is among the first to report on the feasibility of EMAs in people with metastatic cancer and is the first to measure a variety of symptoms and methods for managing symptoms in this population, including the use of opioids and cannabis. The results of this pilot study of EMA indicate that a week of interval-contingent mobile EMA administered three times daily is feasible in this population. EMAs of cannabis-related behaviors are well-documented in the addiction, psychology, and public health literature;34–37 yet the literature on the use of EMAs to measure opioid and cannabis use among cancer patients is in its infancy. 21
We found high adherence rates among our 50 participants: 40 (80%) were adherent with EMA, and 46 (92%) were retained through the end of the study. Notably, however, we focused on individuals with metastatic disease, and cancer progression (and increased symptom burden) may influence participation in EMA, thus it is important to consider other investigators’ results among similar populations. Five papers from four underlying studies have implemented EMA in patients with metastatic cancer.24–26,38,39 Among them, two used electronic diaries to assess pain and its management, with 68–70% adherence.24,25 Another reported an 88% completion rate for four months of once-daily electronic quality of life reports. 39 Only two studies reported on both compliance to EMA and retention.26,38 Schuler et al. 26 reported 44% completion of a daily sleep EMA among patients with advanced cancer, and near complete retention over five weeks. Vehling et al., 38 assessed intraindividual changes in life engagement and loss orientation in a seven-day study among 17 metastatic cancer patients. Similar to our study, the authors reported an 88% retention rate and high completion (97%) of daily diaries. Taken together, there is growing evidence that EMA is feasible in patients with metastatic disease.
We observed relatively low symptom burden and minimal variability of symptoms over the course of the study, potentially explained in part by successful management strategies as evidenced by a 17–50% prevalence of analgesics, anxiolytics, antidepressants, and cannabis. However, a notable fraction of participants experienced transient, heightened symptoms of pain and emotional distress that might not be identified using traditional epidemiologic measurement strategies, which tend to focus on average, least, or greatest symptom intensities. In contrast, pain varies over time, thus reflecting the value of EMA. Only papers by Badr et al. 25 and Stephenson et al. 24 focused on patients’ pain and its management in a study of metastatic breast cancer patients.24,25 Like our findings, the authors reported low levels of pain, with average daily pain scores of 1.7 to 2.5 on a 0–10 scale.24,25 In contrast, the pervasive use of analgesics in our study contrasted heavily with these prior reports, which found 15–18% analgesic use.24,25
A potential reason for the low symptom burden over the course of the seven-day study period may be a result of our eligibility criteria, which did not require a minimum symptom burden level or adequate medical control of symptoms. Reasons for self-selection in this study could include participants’ current health status possibly resulting in greater participation among healthier individuals. It is also possible that patients who were not adherent to the study may have experienced higher symptom burden. Future research would benefit from use of methods to attain reasons for participation or refusal, perceptions of EMA to measure symptoms, and the participants’ symptom management over the course of their treatment for advanced cancer.
The purpose of this pilot study was to examine the feasibility and acceptability of using EMA in an understudied yet vulnerable group of metastatic cancer patients. Results suggest the potential research opportunities using EMA to measure patients’ symptom experience and management. For example, the difference between EMA-measured pain scores and the pain scores reported in the Day 8 survey demonstrate the potential utility of frequent real-time, tech-based symptom assessments within this population. However, the use of EMA as a tool to inform clinical practice is plausible, yet forthcoming. Considering EMA use in a clinical space generates a number of future research questions, including whether this technology is clinically warranted and viable, and the type of technology for symptom monitoring and management that is optimal for both patients and providers.
Strengths and limitations
Our study is among the first to utilize EMA in metastatic cancer populations specifically. Additionally, it is among the first to collect symptom burden of pain, anxiety, and depression and the management of those symptoms including the use of cannabis products. Nevertheless, this pilot study is limited by a relatively small sample size and subsequently limited external validity, as expected from a study of predominantly white, educated individuals receiving treatment at an academic cancer center. There is a potential subset of patients who were not approached for this study including individuals without smartphones and those who are traditionally understudied (racial or ethnic minority groups, individuals of low socioeconomic status, nonmetropolitan/rural populations, sexual and gender minorities, etc.). Further, convenience sampling may not reflect all patients with metastatic disease. This limited generalizability of results warrants the expansion of this study through purposeful and inclusive recruitment methods and partnerships with community and institutional organizations focused on health equity. Lastly, replication of this study within larger, more diverse populations will provide clinicians and researchers with a more comprehensive understanding of symptom burden and management among metastatic cancer populations.
Conclusion
Participants exceeded the adherence and retention goals of this weeklong pilot study, indicating that mobile EMA is feasible among metastatic cancer patients. As we observed relatively little variation in symptom burden in this population over this period, a reduced schedule of EMA intervals may be reasonable. Further, the selection of patients who indicate at least a minimum level of symptom burden may increase symptom variability. Future research is needed to further test the feasibility of EMA methodology in a larger, more diverse cohort of patients with metastatic disease over longer time periods.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241261843 - Supplemental material for Testing the feasibility of mobile ecological momentary assessment for symptom burden and management among metastatic cancer patients
Supplemental material, sj-docx-1-dhj-10.1177_20552076241261843 for Testing the feasibility of mobile ecological momentary assessment for symptom burden and management among metastatic cancer patients by Theodore M. Brasky, Alison M. Newton, Julie A. Stephens, Scott A. Strassels, Roberto M. Benzo, John L. Hays, Erin Stevens, Theodore L. Wagener, Donald Hedeker and Jessica L. Krok-Schoen in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors gratefully acknowledge clinical and research leaders in the participating Ohio State University Comprehensive Cancer Center-Arthur G. James Cancer Hospital clinics. This work would not be possible without their partnership. Stephanie Spielman Comprehensive Breast Center: Dr Bhuvaneswari Ramaswamy, Dr Sagar Sardesai, Ms Jennie Gerardi, and Ms Lindsey Radcliff; Cutaneous oncology: Dr Kari Kendra, Dr David Carr, and Mr Scott Roberts; Gastrointestinal cancer clinics: Dr Anne Noonan, Ms Jackie Lawless, and Ms Krista Maxey; Genitourinary cancer clinics: Dr Steven Clinton and Ms Shireen Desai; Gynecologic oncology clinics: Dr David O’Malley, Dr Floortje Backes, and Ms Molly Meyers; Otolaryngology cancer clinics: Dr Matthew Old and Ms Tina Sowers; Thoracic Oncology Center: Dr David Carbone and Ms Christine Eastep; and the faculty of The Ohio State University College of Medicine, Division of Medical Oncology. We thank the OSUCCC's Recruitment, Intervention and Survey Shared Resource for their interview and REDCap services. Special thanks are extended to Ms. Caroline Gault, Ms. Elizabeth Begle, and Mr Zack Dohar.
Author contributions
TMB, AMN, and JLKS conceived of the study; AMN recruited participants; JAS and TMB analyzed study data; TMB, JLKS, and RMB interpreted findings; TMB, JLKS, JAS, SAS, and RMB wrote the manuscript. All authors reviewed, revised, and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported, in part, by the National Center for Advancing Translational Sciences of the National Institutes of Health under Grant No: UL1TR002733 and by the National Cancer Institute's Cancer Center Support grant P30CA016058.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
