Abstract
Objective
The growing attention towards the psychosocial characteristics of mild cognitive impairment (MCI), a possible prodromal stage of dementia, contributed to the spread of patient-centered approaches to the care of age-related cognitive decline. Within these new care models, including Case-Management approaches, technology-based treatments showed to improve quality of life and psychosocial functioning of people with MCI. However, studies testing technologies to treat functional well-being of MCI patients are still few. Moreover, whether Case Manager supervised or unsupervised technological interventions lead to comparable results is so far unexplored. This study protocol aims to test the superiority of a 1-year physical and cognitive training treatment supervised by a Case Manager in improving the quality of life, functional decline, treatment adherence, cognitive impairment of MCI patients and the mental well-being of informal caregivers compared to the same yet unsupervised treatment.
Methods
In a multicentric, randomized, two parallel arms controlled clinical trial MCI patients ≥65 year-old will be randomized to receive a technology-based treatment either supervised or unsupervised by a Case Manager. Regardless of the study group, patients will perform cognitive and motor exercises with the monitoring of a specialist only during the first 6 weeks of intervention. Three and two assessment points will be considered during the intervention and follow-up period, respectively. The quality of life will be measured as primary outcome at 6 months after baseline.
Conclusions
The results of this study will provide an evidence base to use Case Management as an adjuvant approach for technology-based treatments of psychosocial characteristics of MCI.
Introduction
The term mild cognitive impairment (MCI) refers to a clinical syndrome characterized by the presence of a subtle cognitive deficit, not related to physiological age associated cognitive modification or dementia. The deficit can involve one or more cognitive functions and has a minimal impact on autonomy in daily life 1 (see Supplemental Materials).
The prevalence of MCI ranges from 3% to 42% in the general >55-years-old population, reaching 85% in clinical samples. 2 It was estimated that MCI progressively converts to dementia over time with a rate of 10% per year3–8 and with the 80% of MCI patients progressed to dementia after about 6 years. 5 The prevalence of MCI increases with age. 9 The exponential growing rate of older population both in developed and developing countries 10 is leading to an increasing rate of age-related syndromes: the MCI development rate is about 5.3% per year, with a prevalence of 3.5% and of 7.2% in the seventh and eighth decade respectively, and the prevalence of dementia is supposed to increase up to 23.5% by 2050 in the cohort of people >60 years-old.4,9
Although memory and executive functions deficits commonly characterize MCI,11–13 also limitations in complex instrumental activities of daily living (IADL) and depression seem to have a high prevalence in older adults with MCI.14,15 Such kind of impairments, including difficulties with personal hygiene, medication intake, organizing personal belongings, anhedonia, sleep problems, worthlessness feelings, and suicidal ideation, have a strong and long-lasting impact on patients with MCI. Indeed, depression was found to mediate the conversion rates from MCI to dementia. 16 Consequently, clinicians and researchers have recently moved their attention towards psychosocial characteristics of MCI.17,18 This has been also motivated by the increasing evidence of reduced quality of life in patients with MCI. 19 A recent study showed that autonomy, self-confidence, social connection and intimacy were particularly affected in individuals with MCI. 20
This scenario contributed to lead the WHO to lately propose the concept of “intrinsic capacity,” that is the set of individual's physical and mental capacities that, interacting with environment and interpersonal connections, determine the healthy ageing status.21,22 This radical change in the definition of “healthy ageing” has opened the way for the development of new care models that, compared to the classical models, do not aim to treat acute conditions within a disease-oriented approach, but rather focus on the maintenance of intrinsic capacity, functional abilities and well-being within an integrated and patient-centered approach. 21
Technologies, such as Information and Communication Technologies, have been found to be particularly useful in these new care systems. Indeed, data suggested that technologies facilitate integration between different care settings,23–25 ameliorate prevention strategies 26 as well as improve individuals’ independence and quality of life. 27 Moreover, several studies highlighted that computerized cognitive training can be effective on global cognition, selected cognitive domains, and psychosocial functioning of people with MCI. 28
However, a recent review underlined that only few studies, in so far, tested interventions aiming at improving psychosocial aspects of MCI and that, among these, studies using technological solutions are actually underrepresented. 29 Moreover, it is not clear whether supervised and unsupervised administration of technological interventions obtain similar outcomes.28,30
Our multicentric, randomized, two parallel arms controlled clinical trial aims to fill this literature gap. The study will compare the quality of life (primary outcome), functional decline, treatment adherence, cognitive performance/impairment and informal carers’ mental well-being (secondary outcomes) between a group of MCI patients receiving a web-based motor and cognitive exercises program or a group of MCI patients receiving the same web-based treatment program supported by a human coach, called Case Manager, over 1 year. We hypothesize that the group exposed to the integrated intervention, compared to the group exposed only to the physical and cognitive training, will show an improvement in our primary and secondary outcome variables even at the follow-up measures.
Materials and methods
Trial design
For our protocol we followed the SPIRIT reporting guidelines. 31 The design is a multicentric, blinded for outcome evaluation, randomized, two parallel arms controlled clinical trial, with an allocation ratio 1:1.
Study setting
Participants will be enrolled in centers specialized in assessing, treating and following elderly with cognitive impairment (MCI, Alzheimer's disease, etc.). These centers are part of the Italian Aging Network, a network established by the Italian Health Ministry in 2017.
Randomization
Participants will be randomized with an application available online using permuted blocks of random size, after assessment of eligibility criteria and informed consent signing. Randomization will be stratified by center and MCI subtype (amnestic vs. nonamnestic MCI, 32 independently by etiology). The allocation rate will be 1:1. The allocation between the two study groups will be concealed for the patients, treating specialized neuropsychologist and/or a physiotherapist and Case Manager until the randomization is performed by a password known only by an independent staff member.
Blinding
A single-blind approach will be used so that the assessors (clinicians/statisticians) of outcomes will be blinded to the group allocation. During data analysis, blinding of outcomes assessors will be guaranteed by using numerical codes to indicate the Case Manager supervised or unsupervised Program.
Eligibility criteria
The study will enroll patients with MCI (for a description of MCI and diagnostic criteria, see the Supplemental Materials) meeting the following inclusion and exclusion criteria.
Inclusion criteria:
Age ≥ 65; Diagnosis of MCI to DSM-5 criteria for mild neurocognitive disorder
33
; Clinical dementia rating (CDR),
34
total score = 0.5; Living at home. CDR total score = 0 or >0.5; Previous or actual major psychiatric illness (e.g., schizophrenia or bipolar disorder); Previous or actual major neurological illness other than MCI (e.g., major stroke, multiple sclerosis, brain tumor, traumatic brain injury); Presence of serious comorbidities that affect the functional autonomy (e.g., severe chronic obstructive pulmonary disease, severe heart diseases, severe chronic kidney failure); History of drugs or alcohol abuse; Severe and not corrected by specific rehabilitation training sensory impairments (mainly visual, auditory, etc.) that prevent to perform cognitive and motor exercises; History of intellectual disability and/or other developmental diseases; Life expectancy less than 1 year as judged by the clinician following the Gold Standard Framework prognostic indicator guidance
35
; The absence of an informal caregiver is not an exclusion criterion.
Exclusion criteria:
The study minimum dataset
The minimum dataset (MDS) of the study represents the minimum set of instruments (tests, scales, questionnaires, etc.) needed for describing the target population in our study. The definition of a MDS common to all test sites is needed to standardize the data that each clinical partner will collect and therefore allow for comparisons across the test sites.
The details of how each instrument should be used (e.g., which version, what kind of score, etc.) is also part of the Minimum dataset.
The set of instruments that compose the MDS has been chosen by taking into consideration the objective of our project on the one side, and sustainability issues on the other (to minimize the impact on current organizational processes in the different test sites). The MDS is therefore composed of instruments already used in the organization responsible for the pilot sites, with the exception of the Camberwell Assessment of Need for the Elderly—Short form (CANE-S) instrument that has been considered essential to capture the needs of patients and subsequently match those against the technologies to be provided. The instruments composing the minimum dataset are reported in Table 1.
The minimum data set of the study protocol.

SOB: Sum Of Boxes.
The Montreal Cognitive Assessment (MoCA36,37) is a widely used screening tool for distinguishing individuals with MCI and Alzheimer's disease from those who are cognitively unimpaired, consistently recognized as a sensitive instrument to discriminate the severity and progression of cognitive impairment and to follow the course of cognitive changes in an individual over time. In our study, we will use a telehealth version delivered by video. 38 The score not adjusted for age and education will be used. 36 The reasons for that are twofold: most published research and surveys in literature use raw scores, and adjustment indexes need to be validated in the local population.
The Clinical dementia rating (CDR) will be used to measure the severity of cognitive decline. The score adjusted according to Morris 34 will be used. It considers the impairment level as the decline from the subject's usual level of functioning: CDR is equal to the memory score unless three or more of the secondary categories scores are above or below the Memory score. In that case the CDR equals the majority of the secondary categories.
The Katz index 39 will be used to assess the basic activities of daily living (BADL).
The Instrumental activities of daily living (IADL) is an interview format scale of eight-items covering instrumental activities, which are required for independent living: ability to use telephone, shopping, food preparation, housekeeping, laundry, mode of transportation, responsibility for own medications and ability to handle finances. The score ranges from 0 (low function, dependent) to 8 (high function, independent) for women and 0 through 5 for men to avoid potential gender bias. 40
The Neuropsychiatric Inventory—Questionnaire (NPI-Q 41 ), a tool for the brief assessment of neuropsychiatric symptomatology in routine clinical practice settings, will be used to evaluate the behavioral and psychological symptoms of dementia (BPSD).
The Clinical Frailty Scale (CFS) is a tool to summarize the overall level of fitness or frailty of older adults. 42 It is a 9-point scale that describes the frailty status of a patient from “very fit” (score = 1) to “terminally ill” (score = 9).
The Assessing Quality of Life in Alzheimer's Disease (QoL-AD43,44) questionnaire evaluates the health-related quality of life. The total score of the QoL-AD questionnaire is a 13-item measure, which has been specifically designed to measure QoL in individuals with dementia from the perspective of both patients and the informal carer. It includes questions related to the interpersonal, environmental, functional, physical, and psychological status, and thus, it is a global measure for QoL. QoL-AD will be assessed via an interview and via self-completion by informal carers. For the purposes of the present study protocol, the QoL-AD total score will be considered as primary outcome in our analysis (see the paragraph below).
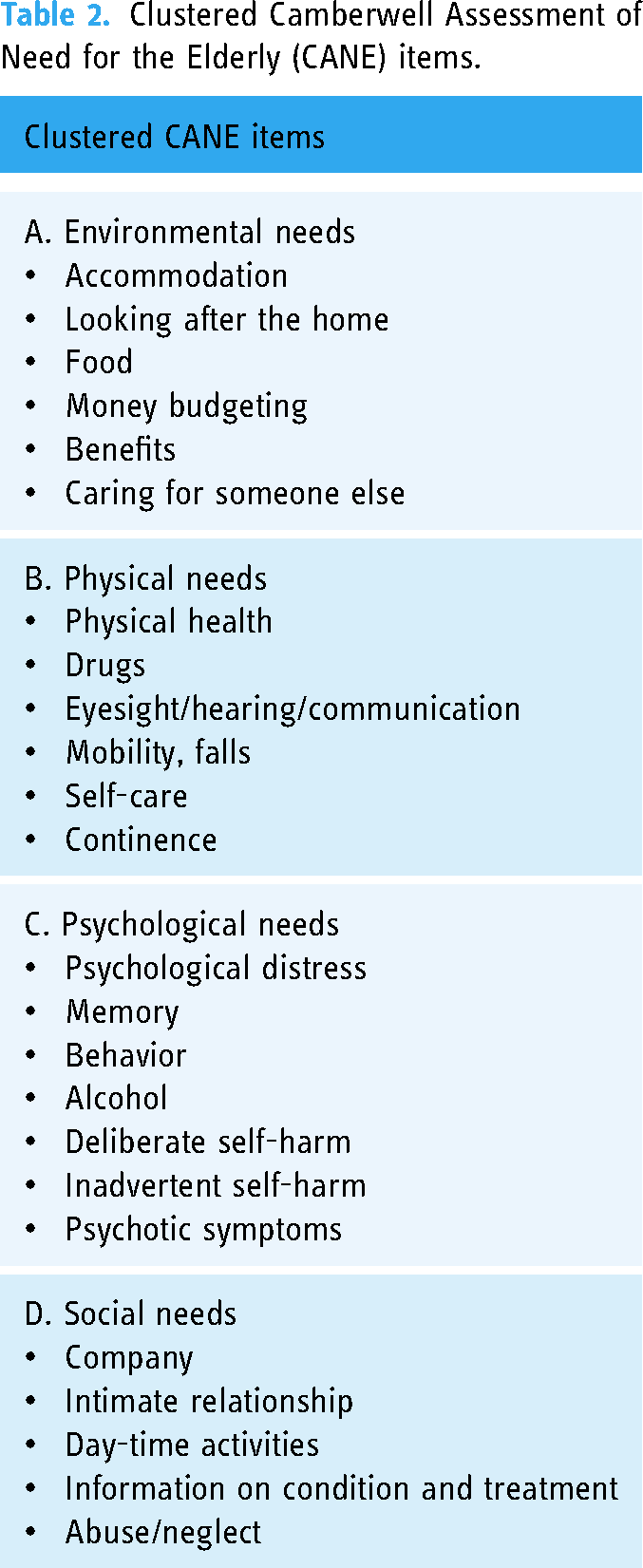
The short form of the Camberwell Assessment of Need for the Elderly (CANE) instrument 45 will be used to assess patients’ needs. For aggregated analysis items will be clustered as described by 46 (see Table 2).
Clustered Camberwell Assessment of Need for the Elderly (CANE) items.
All tests of the MDS will be administered to participants in a baseline assessment session. The following tests will be administered also at different timepoints as primary and secondary outcome variables: QoL-AD, IADL, and MoCA. Regarding the baseline assessment session, we will not include a more extensive cognitive and motor assessment for two reasons: (1) with our study protocol we do not aim to primarily test changes in cognitive or motor performance, but rather changes in functionality, in terms of quality of life and independence living. Therefore, a more extensive cognitive and motor assessment would have been beyond the scope of our study protocol. (2) Applying the same battery of cognitive and motor tests for all study centers would be costly and time consuming. Therefore, we decided to simplify the assessment procedures by including a brief, but in our opinion complete, set of tests that are already in use in all study centers. Informal caregivers’ mental well-being and burden will be also measured at baseline and at follow-up timepoints (see “Timeline” paragraph for details) by administering specific questionnaires (see “Outcomes” paragraph for details).
Recruitment
Recruitment for this study will occur in three ways:
Targeted/clinician-based recruitment:
Each clinical center involved will recruit for the trial through the identification of viable patients in collaboration within their clinical services. Sites will email community and network physicians who refer interested patients to recruitment staff. Broad-based recruitment/public awareness strategies:
Social media and hospital/clinical center advertising. Consortium partnership:
Partners of the Italian Aging Network.
More in detail, viable patients will sign privacy policy documents and a preliminary consent form reporting the description of the study rationale and procedures to be referred (i.e., forwarding of personal details—name/surname—and contacts—phone number/email address) to the recruitment staff by clinical services of the study network physicians or other Partner of the Italian Aging Network.
Interventions
First, at initial recruitment, trained clinicians will check for inclusion and exclusion criteria for all potential study participants. Then, informed consent and privacy policy documents will be obtained from participants who meet the inclusion criteria, and their caregivers. Caregivers will be people (one per participant) who live with participants, or who take care for the most of the day. The trained clinicians who perform the recruitment are also in charge of collecting the informed consent and privacy policy documents. Consenting participants and caregivers will be enrolled in the study and will undergo a baseline assessment in which all tests of the MDS are administered. They will also receive a teaching session, delivered by clinicians involved in the study, to show how the programs will be performed and optimize adherence.
After the teaching session, the study participants will be randomly assigned to the Cognitive and Motor Program or to the Case Manager Program, while any other concomitant interventions the study participants receive will not be prohibited. For both programs, the intervention will be splitted in two phases. During the first phase, cognitive and motor exercises, specifically defined by a specialized neuropsychologist and/or a physiotherapist for each participant according to her/his cognitive and motor needs and functional limitations (e.g., falling risk), are delivered 3 days a week during a period of 6 weeks. In the first phase, the specialized neuropsychologist and/or a physiotherapist will supervise participants once a week. Then, in the second phase, the specialized neuropsychologist and/or a physiotherapist will assign specific tasks that participants will carry out by themselves three times a week until the end of the experimental period (12 months). The defined set of cognitive and motor exercises for each participant will be modifiable on request if participants need change during the experimental period. Depending on the assigned condition, participants will receive or will not receive the support of a Case Manager throughout the intervention (first and second intervention phase). Participants in the nonsupervised group can contact a technician in case of technical problems. The Case Manager is an important figure in the healthcare context to provide population-based care to chronic conditions, guiding and supporting patients in their illness.47,48 The Case Manager can be a trained clinician (e.g., medical doctor, neuropsychologist, physiotherapist, etc.) who evaluates options and services to satisfy individuals’ health requirements through communication and available resources to produce quality cost-effective outcomes. In particular, in our study protocol, the Case Manager will follow participants’ symptoms, providing information and support regarding medical and other treatment options. The Case Manager will also supervise training activities, monitoring the exercises platform to give evaluations about cognitive and motor activities as well as feedback on improper use.
Participants in the Case Manager Program will perform cognitive and motor exercises but also will receive the support from a figure, the Case Manager, who guides them during the entire process. During the first phase of intervention (first 6 weeks), the Case Manager will supervise participants once a week, while during the second phase (until the end of 12-month program) he/she contacts participants once a month in order to monitor their medical conditions, the maintenance of routine exercises, supporting them regarding medical or other treatment options and helping participants meet their goals. The Case Manager may contact participants if access to the platform is not as planned, in particular he/she will call participants if they do not practice for two consecutive weeks. The Case Manager monitoring aims to guarantee the participants’ adherence to the programs.
Brainer
For cognitive and motor exercises we will use an existing platform: “Brainer, Professional Brain Trainer” (Brainer Professional Brain Trainer. Available online at: http://www.brainer.it/caratteristiche/).
Brainer is a Class I medical device with a relative CE mark clinically tested for the cognitive rehabilitation of people with mild-stage dementia. It consists of a web platform (www.Brainer.it), designed to provide cognitive and motor training. The exercises may be used on any personal computer or tablet with an internet connection.
Brainer offers 77 exercises divided in five cognitive domains: attention, executive function, procedural and semantic memory, language, and visual-spatial skills. Each task offers three levels of difficulty. Moreover, the platform provides a set of ecological tasks played in a house, supermarket or station and a package composed of real-world situations (i.e., the shopping day and the day before the holiday). A total of 138 physiotherapic videos are available to train motor ability, including coordination and balance exercises, postural steps, sequences and exercises on the carpet. Moreover, the platform allows the management of patients’ data concerning for example clinical history, demographic data, pharmacotherapy and result of their tests. Therapists, but not patients, may monitor progress and save the changes in patients’ activities, keeping track of the work carried out.
In particular, in our study, the cognitive activities aim to reinforce multiple cognitive domains by reproducing the basics of neuropsychological tests that are usually used for diagnostic purposes. The motor activities involve aerobic physical exercises consisting of performing simple movements, for example, shoulder rotations, arm rotations, lower limb flexion, and neck, shoulder, or arm stretching. Each physical activity starts with a brief explanatory training conducted by a physiotherapist that also shows the correct execution of the exercises. Then, participants start the movements mirroring the therapist. Both the cognitive tasks and the physical activities are structured into different levels of difficulty that adaptively increase on the basis of the subject's performance.
Based on the evidence that exercise programs with shorter session duration and higher frequency may generate the best results, also in terms of treatment adherence, 49 in both intervention phases of both Cognitive and Motor Program and Case Manager Program, we propose sessions three times a week lasting 45 min for each cognitive and motor session.
Outcomes
Our primary outcome will be quality of life from baseline to sixth month as measured by the total score of the QoL-AD assessed by interviewing participants.
Our secondary outcomes are:
Functional decline as measured by IADL; Proportion of attendances at healthcare appointments and admission to health care institutions; Adherence to the treatment as measured by the proportion of the attended activities with respect to the total intended activities; Progression of cognitive impairment over time as measured by the MoCA;36,37 Change in informal caregivers’ mental well-being and burden. We will measure the caregiver quality of life by the EuroQol-5D-5L (EQ-5D-5L), a self-completion questionnaire consisting of 5 questions plus a scale rating the caregivers’ health state on a scale of 0 to 100. EQ-5D has been shown to correlate well with QoL-AD, indicating that the two measures are compatible and can be used side by side.43,50 We will also measure the caregiver burden by the Zarit Caregiver Burden Interview (ZBI-1251,52), a 12-item scale with each answer chosen from a 5-point Likert scale. It will be administered via interview with the informal carer.
Timeline
The schedule and procedures of the study are presented in Figure 1. After the recruitment, the baseline assessment and the randomization, the participation to the study is designed to cover 1 year of intervention. The first intervention phase involves participants in one of the two Programs for 6 consecutive weeks, while the second intervention phase starts right after the first 6 weeks and lasts till 12 months. The primary outcome, that is the quality of life, will be obtained at the baseline before the start of the two Programs (T_0) and at 6 (T_2) months after baseline. It will be also measured at the end of the first intervention phase (6 weeks; T_1), and at 12 (end of the second intervention phase; T_3), 18 (T_4), and 24 (T_5) months after baseline to monitor the trend of the reported quality of life over the intervention period and follow-up points. Adherence will be measured from T_1 to T_3, whereas the other secondary outcomes will be measured at baseline and at T_3, T_4, and T_5 assessment points.

Protocol timeline.
Sample size calculation
Since we did not find a precise estimate for the standard deviation of a 6 months change in QoL-AD in MCI patients equivalent to our population, we resorted to the definition of “small effect size,” 53 since we expect a rather low change in quality of life (QoL) in MCI patients in 6 weeks and do not expect the new treatment to increase QoL in an important way. In order to take into account the specificity of a pilot trial we did not use a calculation for a standard efficacy trial and followed the recommendation of calculating an upper limit of a one-sided 80% CI which excludes 0.2. 54 This results in an estimate of a sample size of 72 participants (36 per group). Since a maximum of a 10% drop-out rate in 6 months is expected, we will recruit a total of 80 participants.
Statistical analysis for primary and secondary outcomes
Baseline description of the groups
Descriptive analyses will be carried out using classification (number and percentages) in categorical variables (e.g., biological sex) and using graphical methods (histograms) and means, SD, medians and quartiles in numerical variables (e.g., MoCA and IADL).
Primary analysis
Primary outcome (value of QoL-AD at 6 months) will be compared between the two intervention groups using analysis of variance (ANOVA). In case of violation of requirements for ANOVA (and in any case as a secondary analysis) a non-parametric Friedman test will be carried out, comparing the change in QoL-AD between the two groups.
Secondary analyses
QoL-AD will also be analyzed inserting into the ANCOVA analysis any variable of interest (e.g., age, education, frailty status, number of comorbidities) that at baseline will be found statistically different between the two intervention groups.
Change at 12, 18, 24 months after baseline in IADL, MoCA, EQ-5D-5L and ZBI-12 will be analyzed by means of ANCOVA, with the use of Friedman test in case of violation of assumptions for ANCOVA. Covariates included in ANCOVA will be tested at baseline. QoL-AD, IADL, MoCA, EQ-5D-5L and ZBI-12 changes will also be analyzed separately at 12, 18 and 24 months after baseline. Differences in the rates of progression of the disease (IADL and MoCA) between the two groups will also be analyzed using a mixed linear regression. Adherence to the treatment will be compared using logistic regression, while difference in the rate of admission to health care institution will be tested using a Cox proportional hazard model. Due to the anticipated very small change in BADL, this last measure will be only described without a formal test in the two groups.
All participants will be analyzed according to how they were originally randomized using intention to treat analysis. Cutoff for significance is set at 0.05. Data will be analyzed using Stata v. 15 or following version.
No interim analyses are planned to avoid the risk of Type I error and unbalances between statistical and clinical significance, especially for secondary outcomes, due to the small sample size that will be enrolled.
Missing data
Proportion of missing data on principal outcome will be minimized by a predefined possibility to contact the patient directly at 6 months to elicit an answer, even in case the patient/caregiver decides to stop the treatment, unless the patient withdraws his/her informed consent. Missing data eventually present will be treated according to the approach proposed by Jakobsen and colleagues. 55
Data management
An independent Data Monitoring Committee (DMC) of experts external to this study, without conflict of interest, will be established. The DMC will be in charge of keeping all participants’ consent documentation and data in structured electronic files. Moreover, it will assess the progress, safety data and, if needed, critical efficacy endpoints. Confidentiality will be maintained during data monitoring, review and deliberations. Agendas of meetings will be developed by Project Officer (PO) and the DMC. Voting and minutes will be kept confidentially. Open Session and Close Session reports will be prepared, presented and appropriately distributed. Recommendations on continuity, pause or termination of the trial and necessary adjustments will be tracked to ensure prompt and adequate response.
Ethics approval
The study approval will be required to the ethics committee of each center involved in the patients’ recruitment and data collection. Relevant protocol modifications will be communicated to each ethics committee by amendments of the original protocol.
Discussion
This is a multicentric, randomized, two parallel arms controlled clinical trial, testing the efficacy of combining a web-based motor and cognitive exercises program for patients with MCI with the supervision of a human coach, called Case Manager. Our protocol aims to demonstrate the superiority of the supervised, compared to unsupervised, administration of technological interventions in improving patients’ quality of life, functional decline, treatment adherence, cognitive performance, and informal carers’ mental well-being, as measured at post-treatment and at the follow-ups.
There is a lack of evidence in the literature on MCI for the use of technology-based treatment solutions to ameliorate patients’ psychosocial aspects, that are still not easily targeted by studies in this field. 29 Moreover, the few data collected so far prevent definitive conclusions about the efficacy of supervised administration of technological interventions in MCI.28,30
Although most literature focused on functional variables that characterize MCI,11–13 there is evidence that psychosocial limitations strongly impact on patients with MCI14–20 and that technology-based interventions are effective on such limitations.23–28 Therefore, there is a pressing need to test the efficacy of these interventions and whether a combination treatment supervised by a Case Manager shows greater benefit to MCI patients in a sound randomized clinical trial.
This study will measure the defined primary and secondary outcomes right after the end of the first and second treatment phase and at two follow-up timepoints to compare the short-term effects of the supervised and the unsupervised intervention and test the effects’ maintenance in the long term. Coupled with multicentered recruitment, we believe this study timeline will provide generalizable results.
Limitations are posed by blinding, indeed this study will use a single-blind approach in which only the assessors of outcomes will be blinded to the group allocation. This prevents to control for performance bias that in turn could reduce the study internal validity. Moreover, although multicenter enrollment will increase sample heterogeneity contributing to results’ generalizability, it could determine the possibility that the treatments’ administration modalities vary according to the center. This could impact the results’ interpretation. Finally, uncontrolled sample baseline characteristics could confound the study results, preventing us from reaching definitive conclusions about the treatment efficacy.
This study aims to demonstrate the efficacy of a combination of technology-based treatment with Case Management for MCI patients to improve psychosocial factors that affect this condition and patients’ carers. Ultimately, the evidence generated by this study will influence clinical practice, answering whether Case Management positively impact on remotely delivered treatment protocols.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241258768 - Supplemental material for Case management in a web-based cognitive and motor treatment for patients with mild cognitive impairment: A study protocol of a multicentric, randomized, two parallel arms controlled clinical trial
Supplemental material, sj-docx-1-dhj-10.1177_20552076241258768 for Case management in a web-based cognitive and motor treatment for patients with mild cognitive impairment: A study protocol of a multicentric, randomized, two parallel arms controlled clinical trial by Alessia Gallucci, Cosimo Tuena, Francesca Bruni, Massimiliano Fedecostante, Lorena Rossi, Antonio Greco, Mauro Tettamanti, Fabrizia Lattanzio, Marco Stramba-Badiale, Fabrizio Giunco and Pietro Davide Trimarchi in DIGITAL HEALTH
Footnotes
Acknowledgements
Alessia Gallucci wishes to thank all the authors for providing useful ideas and supervision during the conceptualization, writing, and revision of the manuscript.
Contributorship
All authors contributed significantly to the study conception and design. The first draft of the manuscript was written by Alessia Gallucci and Pietro Davide Trimarchi. All authors commented on previous versions of the manuscript, revised them critically for important intellectual content. All authors read and approved the final manuscript. No other contributors assisted the authors in this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial supports for the research, authorship, and/or publication of this article: This work was supported by the Italian Ministry of Health, under the Aging Network of Italian Research Hospitals (IRCCS) (Project: “Next Generation Promising (NGP): a new network approach to frailty, multimorbidity and age-related disease in the Era of Precision Medicine” RCR-202-223682286), and under Ricerca Corrente.
Guarantor
Alessia Gallucci has the full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Intended registry
The intended registry of the protocol isOSF Registries—Registered Report Protocol Preregistrations; the intended registry of the protocol dataset is the World Health Organization Trial Registration Dataset.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
