Abstract
Objective
Establish a relationship between digital health intervention (DHI) and health system challenges (HSCs), as defined by the World Health Organization; within the context of hazard identification (HazID), leading to safety claims. To improve the justification of safety of DHIs and provide a standardised approach to hazard assessment through common terminology, ontology and simplification of safety claims. Articulation of results, to provide guidance for health strategy and regulatory/standards-based compliance.
Methods
We categorise and analyse hazards using a qualitative HazID study. This method utilises a synergy between simplicity of DHI intended use and the interaction from a multidisciplinary team (technologists and health informaticians) in the hazard analysis of the subject under assessment as an influencing factor. Although there are other methodologies available for hazard assessment. We examine the hazards identified and associated failures to articulate the improvements in the quality of safety claims.
Results
Applying the method provides the hazard assessment and helps generate the assurance case. Justification of safety is made and elicits confidence in safety claim. Controls to hazards contribute to meeting the HSC.
Conclusions
This method of hazard assessment, analysis and the use of ontologies (DHI & HSC) improves the justification of safety claim and evidence articulation.
Keywords
Introduction
The World Health Organization (WHO) defines digital health interventions (DHIs) as digital health functionality that enable improvements in patient safety and health strategies and address health system challenges (HSCs). 1 DHIs provide a classification scheme or taxonomy, enabling a digital function to be aligned to HSCs, for example, the transmission of health events to specific population groups, diagnostic results, or screening services. The HSC is a health service problem or need to be addressed (e.g. lack of access to information or data, poor patient experience). The DHI is the class of technology intervention that aims to address the health service challenges. The adoption of digital tools within the health industry is hindered by the disparity between health datasets, interoperability between technology solutions and societal influences of a lack of expertise in the use of these technologies available. 2 The pace of technology versus a perceived inflexible regulatory legislation impacted the modernisation of the health economy. The influence of digital technology users (healthcare professionals and patients) on DHI development and implementation has improved digital transformation efforts post COVID-19 pandemic, particularly in the development and implementation of DHIs.3,4 Post COVID-19 pandemic influences on data sharing, improvements in interoperability and data linkage have accelerated the progress being made. The regulatory challenges presented prior to the pandemic are lessening through the influences of improvements in health data access and legal reform.3,5 As these health industry challenges are addressed it is natural to expect that insight and influence from other industries are key. There is a greater volume of interventions available to be implemented and used within society, to meet these challenges.
Technology advances, health industry demand and post pandemic digitisation has increased the availability of DHIs. The Internet of Medical Things (IoMT) through expended network availabilities across cloud-based infrastructure enhancements and improved networks such as 5G. Big data analytics and blockchain improving health record interoperability, clinical decision-support analysis and operational support to health organisations. Artificial Intelligence, specifically improving symptom checking clinical decision-support, patient health and well-being mobile health apps. The digital health industry is predicted to grow at a compounded annual growth rate of 23.3%, from $452.79 billion in 2023 to $1965.30 billion by 2030.6,7
The problems of understanding the limitations of a DHI in regard to its intended use (associated safety claims) and assessing the quality of these safety claims (high-level assertions or statements of quality and safety) become a much larger scale issue. The fundamental principles of the DHI safety claim and the legislative and regulatory challenges can be addressed partly through the improvement in communication between stakeholders – regulator, health organisation, patient and health-care professionals, throughout the lifecycle of the DHI. The safety (freedom from unacceptable risk) of a DHI can be declared (as safety claim) and is supported by evidence (incorporating risk management activities performed during the lifecycle of the DHI). Risk is a combination of the probability (or likelihood) of occurrence of harm and the severity of that harm.8,9 Probability includes the exposure to a hazardous situation and the potential to avoid or limit the harm. Evidence aligned to safety claims may originate from the product's intended use, performance and the justification of risks presented by the implementation and use of the product through risk management methodologies. The safety claim forms part of standards and regulatory based compliance across many sectors in health software (including medical devices). The relationship between DHI and HSC, when used within the context of hazard identification (HazID), leading to safety claims, can offer insight and affirm or establish confidence that the DHIs are safe and fit for purpose.
Safety, security and useability
The use of standards within the development of DHIs provides a consistent approach to complying with legislation. In many instances, standards are harmonised with legislation; for example, medical device legislation utilises this approach internationally for quality and risk management, design and development lifecycles. 10 The goal of DHI manufacture and placement into the market is one of providing a product that is safe to use. This is underpinned by the security of the product (vulnerabilities in computer security as stand-alone or connected systems), effective and consistent approach to its operation within the context of intended use and the user interface. 11
We define terms associated with safety and risk assessment within this research to provide a common understanding in support of the contributing factors and the methodology promoted (Table 1).
Terms and definitions associated with risk assessment of DHIs.
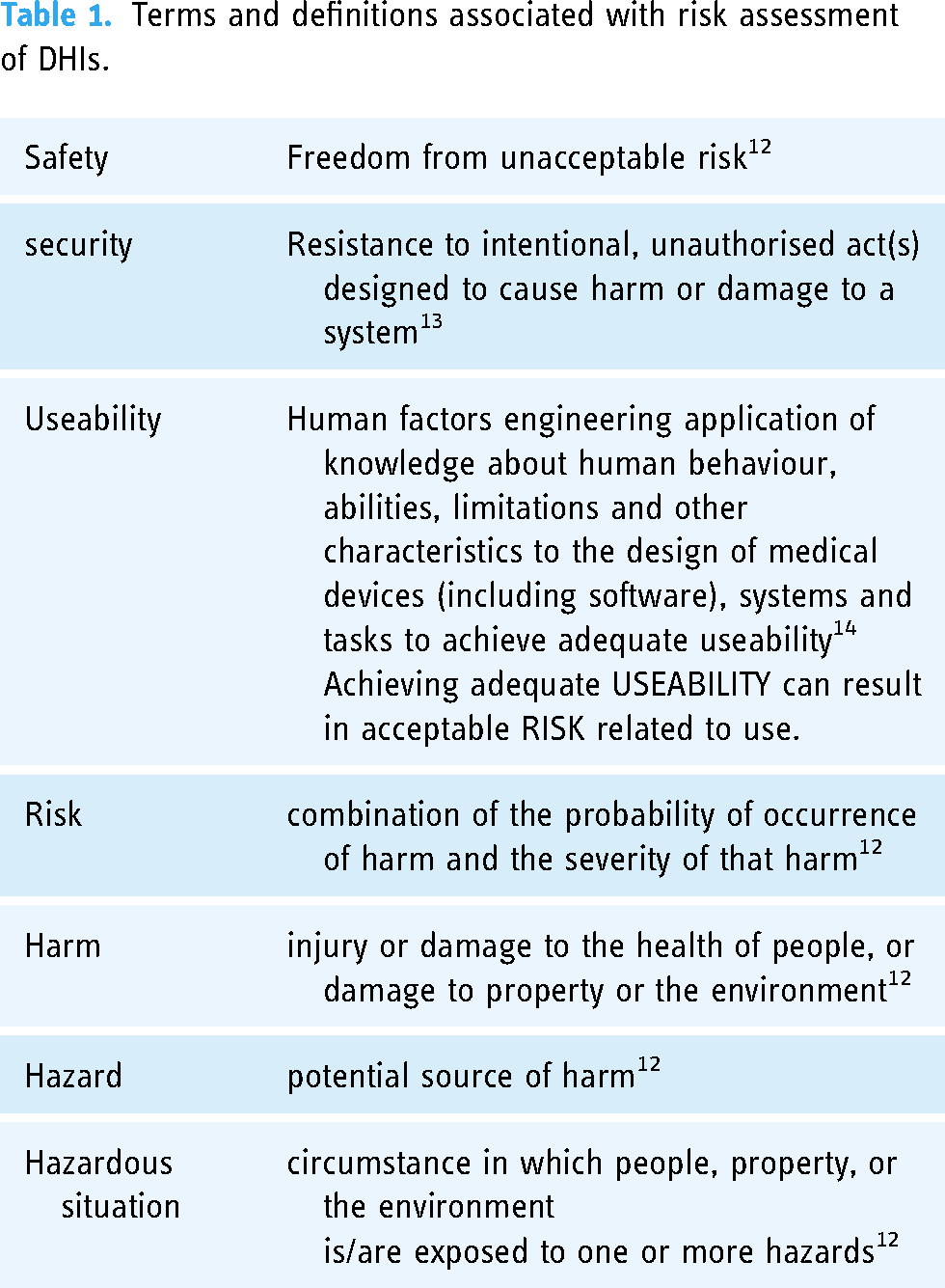
Security is one part of the overall safety claim, as are other domains defined in standards and legislation relating to health software and medical device development, implementation and use. The definition of ‘safety’ in ISO 14971 and underlying ISO and IEC guidance, safety incorporates harm that affects health, property and environment. Safety is not just related to the health of patients, that definition of harm also includes any adverse outcome of malicious attacks we assess when we discuss security. The risk management standard ISO 14971 requires the examination of harm even if the device is not used according to its intended purpose, in case of foreseeable misuse. It is required to consider the environment the device will be used in, in this regard. So, malicious attacks are not excluded. Security vulnerabilities can compromise operations of DHIs, accessibility, integrity and availability. 15
An important subject or domain that influences the effectiveness of DHIs and the associated safety claim is the science of human factors and useability (Figure 1). Human factors refer to the interaction of people with the systems surrounding them, including the technology they use. The development or design of DHIs has an essential part to play with the safe implementation and use of these technologies. As with the placing of a product in the market place or industry (i.e., digital health) by a manufacturer, the intended purpose of the DHI is critical. This is explored in more detail relating to the methodology used in this research. Intended purpose, patient cohort, user competency (including cognitive behaviour) and environment all play a part in this domain. Human factors within the Digital Health industry and healthcare in general has learned from safety critical industries and has become an essential domain within the development of DHIs, underpinning safety claims. As such within the industry we encompass terms such as ergonomics and useability. For the purposes of this research, we use the term useability engineering. We define the term Ergonomics (or human factors) as the scientific discipline concerned with the understanding of interactions among humans and other elements of a system and the profession that applies theory, principles, data and methods to design in order to optimise human well-being and overall system performance. 16

Human factors affect the outcomes of using a medical device. 17
Useability engineering within the design and development of DHI products relate to the safety claims declared. The application of risk management focusses on design flaws, manufacturing failures, electrical issues and the correct or incorrect use of the product (foreseeable misuse). Risk control measures are applied during the design, development and production part of the product lifecycle14,18 The inclusion of useability into the risk assessment of a DHI requires domain specific goals that must align with the safety claim – objectives, roles and target performance indicators. 19
Application of risk management
We postulate the application of risk management methodologies to identify hazards, can be linked to DHI classes and HSC categories thereby improving safety justifications (or claims) made. There are other influencing factors across the domains of Information Governance, Privacy, Information Security, Cyber Security and Useability; our objective is improving quality through the utilisation of these domains during risk assessment, from common terms and structured statements concerning hazard assessment. We acknowledge the influences on DHI safety through the implementation and use, as well as the development lifecycle; the focus of this research is to establish a relationship between DHI and HSCs (HSCs), as defined by the WHO; within the context of HazID, leading to safety claims. To improve the justification of safety of DHIs and provide a standardised approach to hazard assessment through common terminology, ontology and simplification of safety claims. Articulation of results, to provide guidance for health strategy and regulatory/standards-based compliance.
We categorise and analyse hazards using a qualitative HazID study. This method utilises a synergy between simplicity of DHI intended use and the interaction from a multidisciplinary team (technologists and health informaticians) in the hazard analysis of the subject under assessment as an influencing factor. 20 Although there are other methodologies available for hazard assessment, the method chosen enables examination of the research objective rather than the effectiveness of risk assessment methodology. In safety critical industries, multiple methods of risk assessment are utilised to establish both proactive and reactive risk treatment plans. 21 This also includes the utilisation of libraries of methodologies and applicability to system specifications. The steps presented and associated assessments aim to contribute (qualitatively) via the application of a safety analysis and assessment methodology to generate improved safety claims, standardise terms used and correlate towards classifications of DHI and HSCs. We aim to present a direct correlation between HSC, the safety justification and building of structured safety claims using taxonomies through safety engineering methodologies. The benefits of HazID align with a more structured data driven approach where the utilisation or standard terms and categories of risk move towards a more effective assessment.22,23 HazID – hazard identification, is utilised in safety critical industries (including digital health) often requires that all hazards with the potential to cause harm are identified. The methodology provides a structured approach to HazID and the assessment of likelihood and severity. 24
The health industry is influenced by safety critical industry practices where patient safety is paramount and the foundations of safety engineering concepts and methods can be seen to improve quality and safety.25,26 However, safety claims about such interventions vary in quality. Quality improvement concerns the consistency of terminology, repeatability of safety analysis and assessments made and standardisation in the description of safety claims. These quality issues impact the hazard assessments performed and subsequent regulatory evidence supporting compliance.2,27 The communication of the safety claims between technical and clinical experts and other stakeholders, such as users of DHIs is open to confusion, lack of understanding and limited expertise in safety engineering concepts or regulatory requirements.25,28,29 The claims made for DHIs often support regulatory requirements that enable a product to be placed into the market. This aligns with the objectives of good manufacturing practise, health software quality development, medical device manufacturer and the supporting standards and regulations that align to best practise.
The lack of the adoption of the fundamental concepts of clinical risk management (CRM) and safety methods, within health informatics, demonstrates safety's limited influence in the development of digital health technologies.30,31 It has been shown that the foundations of safety engineering concepts and methods can improve quality and safety.
‘Adopting a systematic approach to risk management, building on best practice in system safety, has been beneficial. Much of the benefit has been realised due to the close engagement by the clinicians. However, despite such progress, more work remains in order to mature current safety assurance practices and improve organisational support’. 25
The impact of DHIs on the safety of patients and potential harm exercised by the unsafe actions of clinical users, is not documented openly.31–33 Evidence suggests a lack of rigor within the industry, where strategies for innovation to improve clinical outcomes and advance health using new technologies, overlook the principles of safety.25,28,29 The exponential growth, diversity of DHIs and associated regulatory position are the biggest challenges to the industry. Policy makers, manufacturers, health organisations and digital technology users (healthcare professionals and patients) have different understandings and objectives of DHIs.30,34,35 These strategies often, see rigor as a barrier not an enabler to innovation.29,36,37
In contrast to the digital healthcare industry, traditional safety critical engineering industries have the capability of in-depth analysis and assessment, while they have been established over decades. Traditional safety critical industries integrate quality, benefits and safety objectives in structured and formal methodologies that are more amenable to innovation than the healthcare industry currently exhibits. 38 This work aims to indicate that the domain of safety can be correlated, using a structured methodology to improve the communication of safety claims of DHIs.
Digital health interventions
The benefit and contribution to the communication between stakeholders, for safety claims aligned with the classifications of DHIs is presented in this paper. The safety of a digital health product can be declared (safety claim) and supported by evidence from the intended use of the product, performance and the justification of risks presented by the implementation and use of the product through risk management methodologies (safety case or assurance case).11,39 We introduce the term CRM in a digital context to focus the risk management methodology on digital clinical safety. CRM is the systematic application of management policies, procedures and practices to the tasks of analysing, evaluating and controlling clinical risk. The safety claim forms part of standards and regulatory based compliance 40 in this regard.
The WHO classification of DHIs, 1 and their relationship to HSCs, provides us with an opportunity to establish or affirm safety claims by the application of safety analysis methods. HSC is a health service problem or need to be addressed (e.g. lack of access to information or data, poor patient experience). DHI is the class of technology intervention that aims to address the service problem (HSC). This can offer insight and establish confidence that the DHIs are safe and fit for purpose by applying safety methods. The WHO classification promotes an accessible and bridging language between technical and clinical experts aimed at simplifying dialogue and aiding digital health implementation. The classification represents discrete functionality of DHI, to achieve health sector objectives and meet the HSC. This is aimed at commissioners of digital services. By the application and use of these classifications and HSCs, we are able to identify hazards and construct a safety claim and justification. This, in turn, will bridge the understanding of the health delivery organisation and manufacturer CRM processes by way of guidance for each DHI classification.
The quality of design and development, implementation and the use of DHIs are dependent on the rigour applied from the initial concept or innovative idea to minimum viable product or prototype development and minimum marketable product. The use of relevant standards and industry guidance provides a manufacturer greater opportunity to demonstrate effectiveness and meet health sector challenges and user needs.37,41,42 The use of taxonomies, synonyms and ontologies is an established method of grouping common themes to assist in resolving interpretational issues within the field of health informatics.43,44
The Digital Health Assessment Methodology (DHAM) provides informative guidance and insight to both technical and clinical experts. This will inform the benefits of effective safety claims linked to DHI classifications and HSCs. The framework for synthesis of safety justification, 44 enables the establishment of DHI safety related justifications within the method developed. The safety justifications are created in such a format that we can group the terms used. These taxonomies will allow for the grouping, classifying, grading, ranking, sorting and stratification of the results. The grouping further improves the communication and guidance of safety claims and relate to the HSCs faced.
HSCs provide synergy between intended operational use and the challenge faced in the health system. It is purposeful for examining the relationship between HSC, Hazard, Effect and overall DHI contribution, which is key to establishing safety claims. The HSC also provides a consistent terminology and phrasing that is less open to misinterpretation. This helps to establish consistency and focus to the hazard assessment. 45
Safety engineering and preliminary hazard analysis
DHIs, health software (including medical devices), are safety critical systems presenting immediate patient safety risks during normal and abnormal operating conditions to the user. The foundations of safety engineering concepts and methods are well known in the improvement of quality and safety. 25 The relationship between a defined hazard or hazardous situation and the effect of the hazard will be assessed using this method. We postulate that the use of safety engineering methods, HazID using a defined method, enables a higher quality preliminary hazard analysis to be performed. This preliminary hazard assessment methodology is applicable to CRM requirements within medical device regulatory standards.11,39,46,47 We use a qualitative HazID study due to the cooperation between simplicity of understanding, the use of multidisciplinary teams and the reliance on the experience of the team in the subject under assessment. Although there are many methods available this has been selected due to the preliminary nature of assessment in the overall lifecycle and the objectives of this study (i.e., improvements in the articulation of safety claims).
Digital health interventions and health domains
The classification scheme utilises the term System Category to represent diverse types of Information and Communication Technologies (ICT). The original objective of the WHO classification scheme was to address the need for a shared language for health researchers to enable improvements in the synthesis of evidence for innovation across a complex healthcare landscape.48,49 The DHIs used in the following method cover a common fixed scope of health domains (Table 2), to provide a tenable boundary for analysis and results. Further discussion will acknowledge those DHIs that fall into the category of self-care.
Definitions of included DHIs. 49
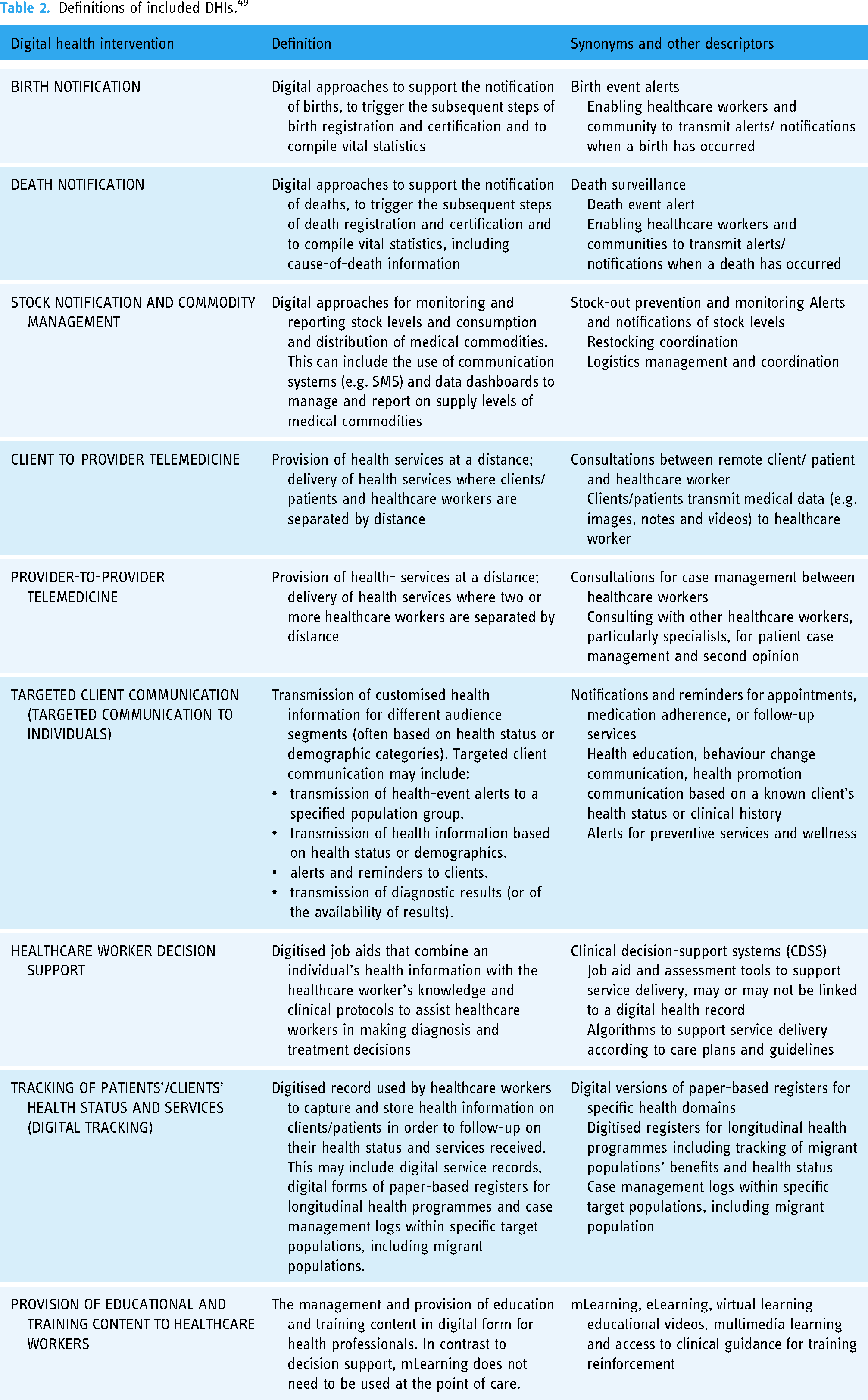
‘The World Health Organization (WHO) defines self-care as the ability of individuals, families and communities to promote health, prevent disease, maintain health and to cope with illness and disability with or without the support of a healthcare worker’. 50
The classification scheme also draws relevance from the international standards community.
‘provides a guide to best practice business requirements and principles for planning the use of information and communications technology (ICT) to support the development, coordination and delivery of healthcare services by countries and subordinate health authorities within a country’. 51
Aligning to the need to support a growing health information system ecosystem and infostructure, these strategies follow the common objectives of providing improvements in communication between stakeholders and removing barriers to standardisation, quality and safe implementation goals. We provide a summary of DHIs to enable further understanding (Table 3).
Included digital health interventions. 48

Method – DHI hazard assessment method (DHAM)
The DHI Hazard Assessment Method (DHAM) has four steps in which the DHI can be classified (Figure 2), analysed and resultant safety measures be established (Figure 3). The DHI classification scheme was used to generate guidance on the effectiveness for DHIs.
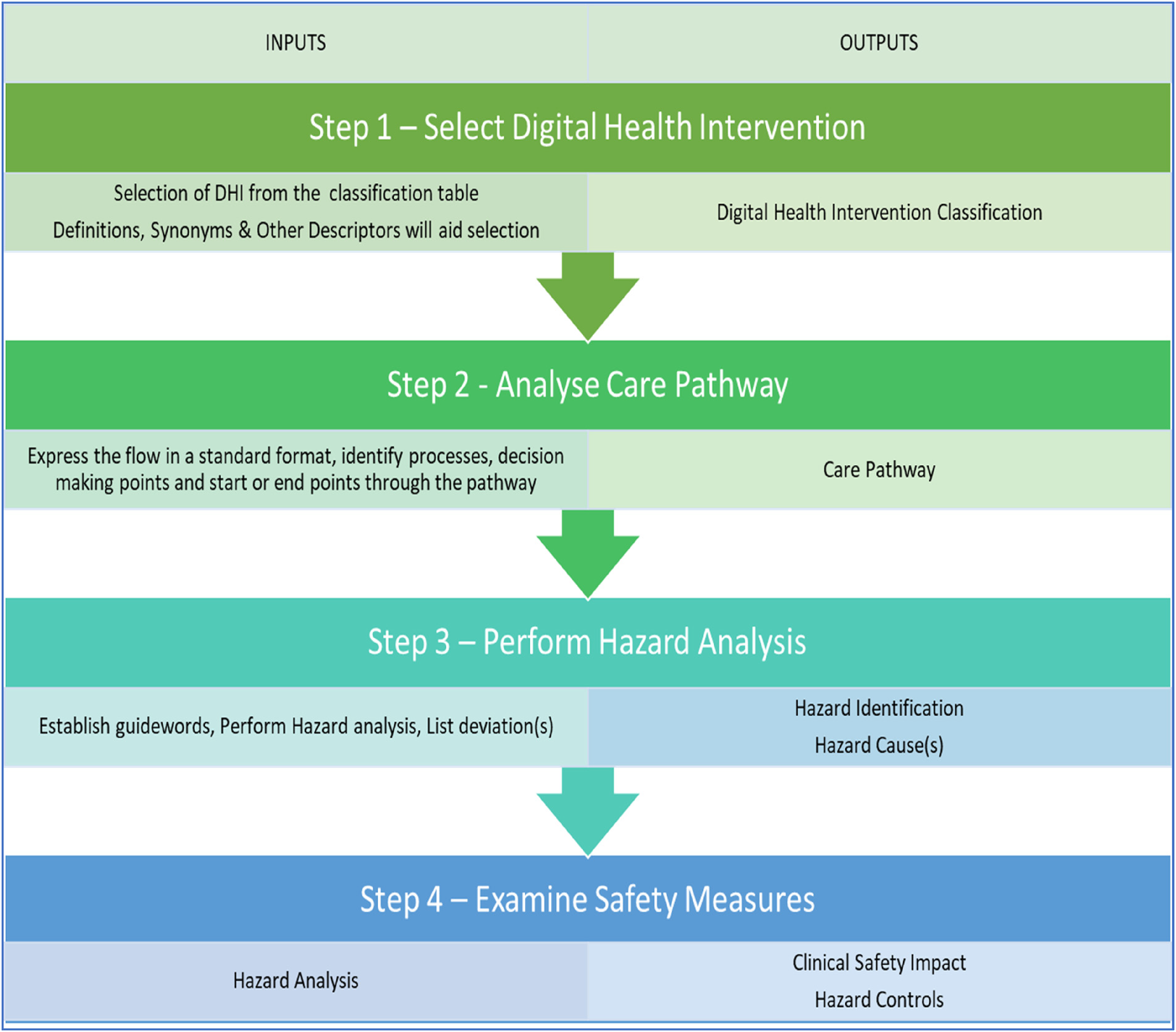
DHI Hazard Assessment Method (DHAM).

Risk matrix.
By utilising the DHI classes we categorise and analyse hazards relating to patient safety in the implementation and use of the DHI function. The safety analysis process incorporates a category of operational and deviation-based risk assessment processes – HazID (Hazard identification), 20 Hazard and Operability Studies (HAZOP), 44 Failure Modes and Effects Analysis (FMEA) and Functional Hazard Analysis. 2 Completed at the early stages of development lifecycles, these methods have the advantage of highlighting risk to multidisciplinary stakeholders with a common level of understanding. A precursor to using this method may be fault tree analysis or code reviews, which are sub-system level and dependant on product experts and technologists. We have also stated that mixed methods of risk assessment performed during the DHI lifecycle are beneficial and this method allows for the constructive interaction of methods utilised.
We perform an initial HazID on a typical DHI and provide a rationale for the justification of DHIs. This four-step method was designed and applied to typical examples of Digital Health services or implementations in healthcare. We apply the method and derive safety justifications to DHIs. Examination of the hazards identified and associated failures are used to articulate potential improvements in the quality of safety claims.
Step one – select the DHI
Select the DHI that best describes the digital health technology from Table 3. This provides an input to the method (Figure 3) and provides the initial DHI classification and ongoing study case for assessment.
Select DHI is completed using the established DHIs (Table 3). These predetermined taxonomies, from mobile applications and more traditional digital health technology solutions, ensure the DHI, definition, synonyms and other descriptors are meaningful and consistent. The objective being to align the digital health technology to the DHI class for further analysis.
Step two – analyse the care pathway
Document the care pathway for the DHI. This pathway should allow for the examination and assessment of potential deviations from the intended or desired process flow. Potential deviations can consider omissions, early, mistake, wrong and other guidewords.44,52 Ensure each DHI example has the required minimum evidence for analysis and assessment – workflow, explanation of intended use, operation and clinical speciality. Noting importantly that we utilise the definition of care pathway published in literature – ‘care pathways as a method for patient care management of a well-defined group of patients during a well-defined period of time’. 51 The care pathway must demonstrate the key components or elements of the patient flow or care journey. Care must be taken to express the flow in a standard format, identify processes, decision-making points and start or end points through the pathway, e.g. a flowchart.
Documenting this way allows us to convey the treatment of the patient in steps, allowing us to analyse care pathway deviations from these steps that may become hazards. Clinical, care, or patient pathways and other terms are used that attempt to define the patient journey (end to end or partial). We use the term care pathway and align to best practise as defined in clinical professions, international standards and health technology guidance.
Step three – perform hazard identification
The formal methodology chosen for hazard analysis must enable an assessment to be performed such that the focus on the care pathway, is independent of technology beyond the classification of DHI. This allows us to perform HazID at a more operational assessment level. The objective of step three will aid in bridging the communication barrier between clinicians and technologists. The HazID methodology 44 enables us to assess credible failures by examination of the use of the DHI and the deviation or divergence from that use.
The inclusion of HSCs provides constructive interaction between intended operational use and the challenge faced in the health system when a DHI does not operate as intended. We use this to highlight the relationship between hazard and defined HSCs.
These can be found summarised in Table 4.
Health system challenges.

Step four – examination of safety significance & control – hazard analysis
Examine safety significance & control of each hazard and

COVID-19 care pathway (flowchart) including hazard identification.
Identify safety controls or safeguards that enable mitigation of hazards identified. The identification of controls is performed by suitably experienced and competent personnel. The expected outcome of this stage will be identification of hazards that correlate to HSCs that form the important connection between DHI implementation and satisfying an outcome based on the HSC. It is expected that this will enable clarification of safety features for the DHI to be effective in this regard and achieve its intended purpose.
Examination of the hazard analysis performed must ensure that terminology used is unambiguous and describes the defined term without risk of miscommunicating statements. Careful reflection on the definitions of Hazard, Hazardous situation; should ensure the context is maintained (consistency and repeatable). Familiarity with the DHI is key in this regard so that explanations of events can be further analysed for grouping or consolidating causes. Finally, HSCs used together with the patient safety impact ensures the terminology is standardised.
Results – DHI implementation using the DHAM method
Example 1 – UK NHS COVID-19 App
The UK NHS COVID-19 app 53 provides contact tracing functionality, a high-level symptom checking and alerting functionality. This example has been simplified from publicly available information, with the test and trace system performed by a separate government health organisation not considered within scope of this example (due to complexity).
Step one – establish the digital health intervention functional description (DHI selection)
Select the functional description from Table 2 that represents the DHI, including the definition, synonyms and other descriptors. Table 5 summarises the functional description for this example DHI.
DHI functional description.

Step two – analyse the DHI-enabled care pathway
Document the care pathway that the DHI is used (Figure 5). Ensure each DHI example has the required minimum evidence for analysis and assessment – product name, workflow (flowchart), explanation of intended use, technical operation and features or functions (clinical speciality). Table 6 provides the summary for this example DHI.

Remote patient monitoring flowchart.
DHI product name and technical operation summary evidence.

Step three – perform hazard analysis
Assess credible failures by examining the use of the DHI (process workflow) and the deviation from that use (within the process workflow). Document the findings in the form of a hazard table. Causes, deviations, or failures leading to hazards and hazardous situations are summarised as the following: Loss of function, Partial Loss of Function, Function provided when not required and Incorrect Function. The Hazard Effect must also include HSCs 1 that highlight the problem or challenge should the hazard occur. The objective being to provide establish a connection between hazard and HSC for any given DHI class. See Tables 7–9 for the Hazard Analysis summary. Each of the hazards are also identified numerically in the care pathway (Figure 5).
Hazard analysis summary.

Hazard analysis summary (continued).
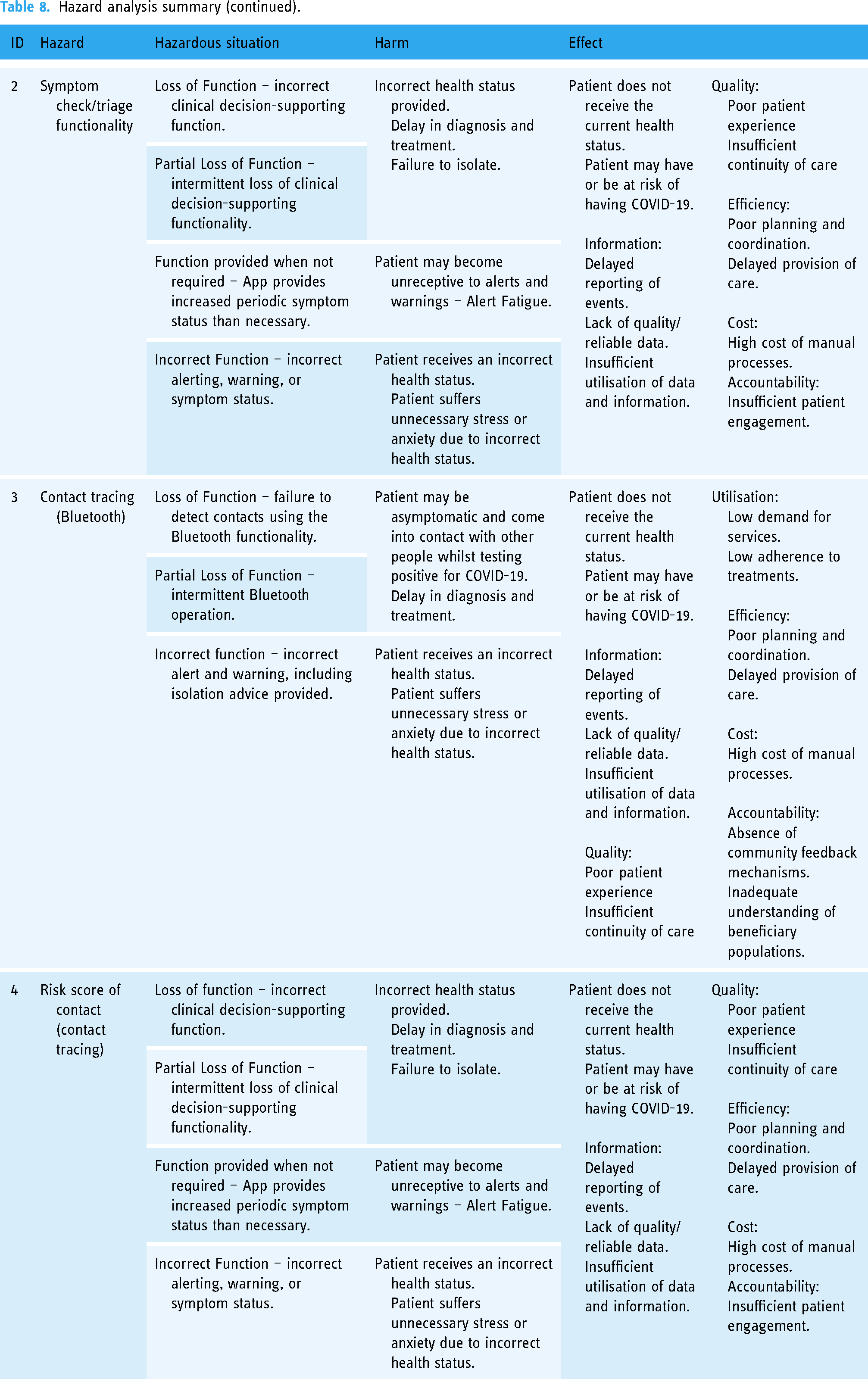
Hazard analysis summary (continued).

Step four – examination of safety significance & control
Examine safety significance of each hazard and identify safety controls. This application of CRM methodology informs the hazard analysis. Examine DHI hazard(s) using likelihood (L) and severity (S) to derive a severity rating (R – safety significance). Identify safety controls that enable mitigation of hazards identified, Tables 10–12 provides the summary results of the examination of causes, controls, initial and residual hazard assessment.
Safety controls.

Safety controls (continued).

Safety controls (continued).

Example 2 – remote patient monitoring
Delivering care to patients within the comfort and safety of their own homes (including care homes) is being enabled by the UK NHS through the launch of the remote monitoring programme. 54 This example considers a remote patient monitoring solution implemented across a region in the UK. The system enables the regular remote capture of patient-reported outcome measures (PROMs) between appointments to support shared decision-making about when patients needed care the most. The solution aligns to a UK government policy and strategy for increasing the scale and capability of digitally enabled care in the home.
Step one – establish the digital health intervention functional description (DHI selection)
Select the functional description from Table 3 that represents the DHI, including the definition, synonyms and other descriptors. Table 13 summarises the functional description for this example DHI.
DHI functional description.

Step two – analyse the DHI-enabled care pathway
Document the care pathway that the DHI is used (Figure 6). Ensure each DHI example has the required minimum evidence for analysis and assessment – product name, workflow (flowchart), explanation of intended use, technical operation and features or functions (clinical speciality). Table 14 provides the summary for this example DHI.

Contributing risk factors conceptual model.
DHI product name and technical operation summary evidence.

Step three – perform hazard identification
Assess credible failures by examining the use of the DHI (process workflow) and the deviation from that use (within the process workflow). Document the findings in the form of a hazard table. See Table 15 for the Hazard Analysis. Each of the hazards are also identified numerically in the care pathway (Figure 6).
Hazard analysis summary.

Step four – examination of safety significance & control
Examine safety significance of each hazard and identify safety controls, Tables 16 and 17 provides the summary results of the examination of causes, controls, initial and residual hazard assessment.
Safety controls.
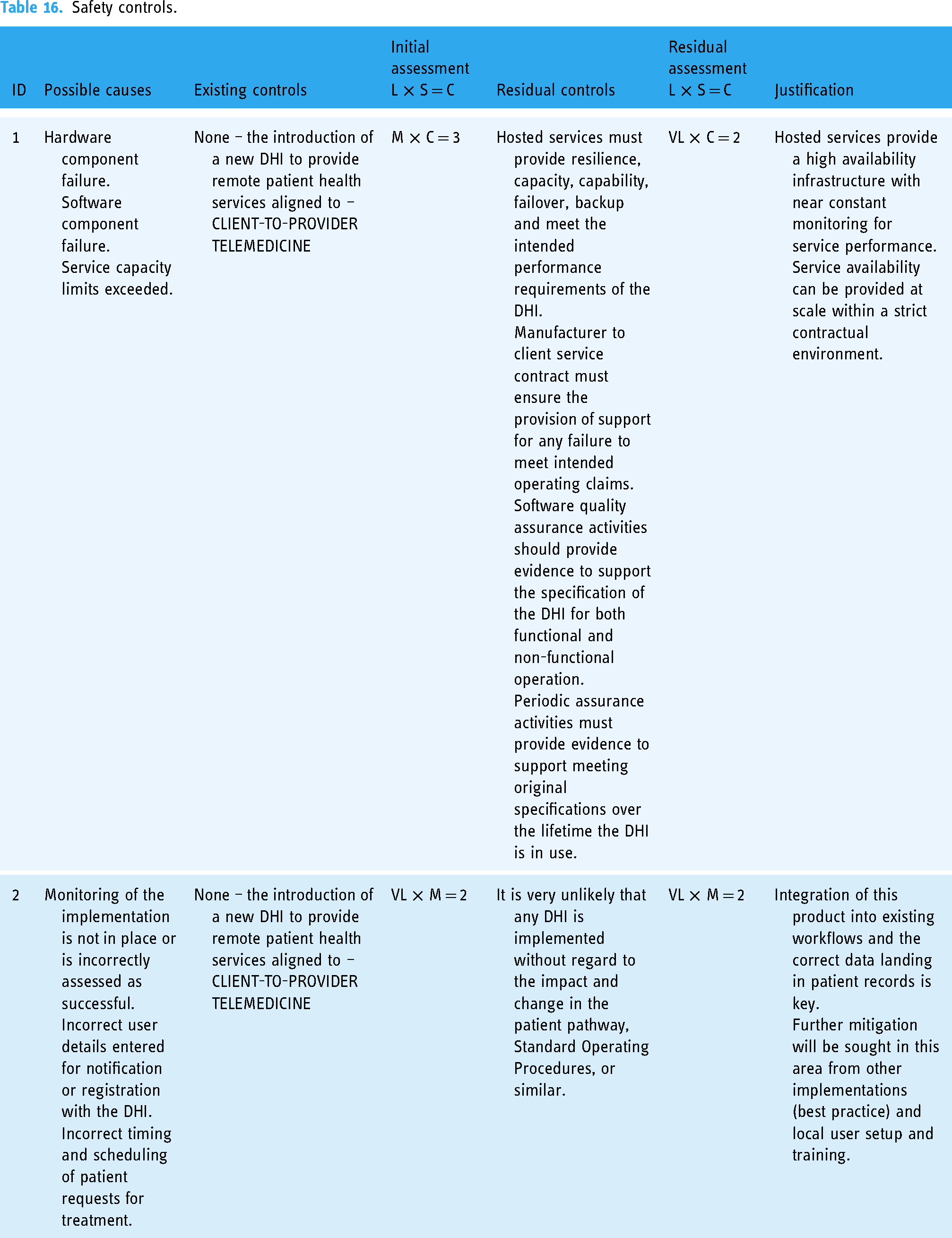
Safety Controls (continued).

Results summary
A qualitative analysis of results was completed based on the authors’ experience. Application of the method used, provided the hazard assessment and the results used to generate an assurance case, thereby providing a justification of safety and eliciting confidence that the DHI is fit for purpose. Avoidance of hazards through mitigating controls contributes to meeting the HSC. From the subject matter experience and suitably qualified personnel performing the HazID and assessment, we can see the impact on patient safety can be defined by either a delay in care or diagnosis or inappropriate care or diagnosis provided. These provide two useful terms that can be used in a more structured context for harm events. The method provides indication that by structuring the hazard assessment this way, including the DHI classes, together with HSCs; ontologies improve the quality of assessment. This is achieved through the use of consistent terminology and enables repeatable assessments to be completed with similar DHIs. Considering the impact on patient safety for a DHI could be misdiagnosis or delay in diagnosis; the impact would naturally be a delay or inappropriate treatment provided. Whilst we acknowledge the individual domains that contribute to the safety of a DHI, the focus of this research is the overall quality of safety claim within the regulatory challenges of compliance or certification and not any single domain such as human factors, useability, security, or other contributory risk factors. The combined contribution is key and underpinned by the flexibility of risk methodology created within this research. We postulate through our method, that improvements in the categorisation, use of consistent language and terminology allows for a broader inclusion of contributory factors and mixed method risk assessments due to the efficiency in risk analysis undertaken.
Discussion
Contributory risk factors
The regulations and standards, including country specific legislation for DHI product safety includes the contributing risk factors of safety, security and useability. Security encompasses cyber security and privacy. Risk analysis methodologies applied within this methodology must incorporate all risk factors that impact the safety of the DHI. Disparate risk assessments have the disadvantage of lower quality safety analysis through a lack of consideration to all contributing risk factors (unstructured safety claims). We acknowledge the contribution that more specialised risk analysis methods bring, specifically to IoMT DHIs, useability engineering, cyber security threat analysis, for example.17,21 With the addition of the classification scheme and HSCs, we provide a hazard assessment that incorporates sociotechnical challenges in a structured and simplified format. When this information is incorporated into the hazard analysis from a conceptual modelling perspective, it is clear to see how each risk factor may contribute to the overall safety claim being made (structured approach). We can see from the conceptual models (Figure 6), that safety claims are dependent on many risk factors and if structured correctly, can provide the essential evidence to support DHI compliance to regulatory requirements.
From a hazard and risk analysis perspective, the methodology allows for the categorisation of risk to contributory risk factors (Table 18) and a direct relationship to DHI classification and HSC.
Examples of categories of DHI hazards.

Structured safety claims
The safety claim provides an argument that a system is acceptably safe for the intended operation and in a digital health context, intended use. These arguments are commonly represented visually in medical devices industries using Goal Structuring Notation (GSN).59–61 The example DHIs assessed using the method, are developed into structured safety claims (Figure 7), that represent the initial HazID and assessment completed.

GSN safety argument for DHI.
The hazard summary tables provide detailed uniquely identified hazards, hazardous situations, harm, effect and further risk control parameters. The GSN arguments highlight the claims made that illicit confidence in the DHI safety through the method applied. Each step in the method and safety claim is supported by evidence in the context of the argument made. The objective of the safety claim and supporting evidence is to provide a compelling and comprehensive case for the safety of a DHI. Inadequately defined evidence will lead to false conclusions. Knowledge gaps has the potential for assurance deficits and directly impact the confidence in claim being made. The quality improvements enabled by the DHAM method are as a result of structure, terminology and clarity of content provided (i.e., ontologies) of the hazard assessment made.
Health system challenges
HSCs provide a terminology that describes hazardous situations derived from the hazards identified. This allows for ontologies to be used for the structuring of hazard assessments. From a CRM perspective, we often see variations in quality of the safety claim with limited context on patient safety harms and effects. 10 Hazard controls have been documented in detail to summarise and to aid the CRM and risk acceptability process. However, we can categorise these controls or group them into areas such as design, process, technical assurance, training, external design and external process. These groupings allow for improvements in risk acceptability, assessing and examining areas where standard termed controls are applied and the completeness of those controls documented. Safety critical industries often utilise database structures 61 to further improve the recording of hazards and provision of a library of terms to prevent duplication of effort during HazID on subsequent systems (new or enhanced) with similar contexts, intended uses or use cases. We postulate that DHI hazard assessment and analysis can be completed more efficiently and to increased quality using structured HazID methods, thus removing ambiguity or subjectivity of claims made.
Conclusion
The method provides indication that by structuring the hazard assessment this way, including the DHI classes, together with HSCs; ontologies improve the quality of assessment. This is achieved through the use of consistent terminology and enables repeatable assessments to be completed with similar DHIs. Considering the impact on patient safety for a DHI could be misdiagnosis or delay in diagnosis; the impact would naturally be a delay or inappropriate treatment provided. This suggests the impact on patient safety can be defined in a structured way, for example:
patient receives incorrect diagnosis delay in the diagnosis and ongoing care of the patient patient deterioration patient receives an incorrect health status patient suffers unnecessary stress or anxiety due to incorrect health status.
Hazard tables or hazard logs, present clarity and concise summaries that support CRM activities already discussed during this method. This method can be applied to many more DHIs leading to further work to assess the contribution of improvements in the quality of assessment of DHIs as a case study, beyond the two examples used in this research. Digital Health regulatory and standards compliance require risk or hazard assessments to be made available for inspection or assessment by those third parties providing certification. This allows us to identify hazards using a more defined terminology and ease regulatory burdens. Although we utilise a HazID methodology, other methods are available and all have their advantages and disadvantages when applied during the various stages of a development lifecycle. The choice of methodology is not under analysis, more the articulation of the assessment results to support any claims made.
There is a dependency on this information to inform the reader to perform various roles – regulatory certification, risk acceptance, implementation and the use of DHI. The use of ontologies aids initial hazard assessments by improving the dissemination of information from different subject areas or contexts and removing communication barriers between individual domain experts (e.g. clinical, technical). This exhibits the similar objectives of communication improvement within the original WHO DHI classification guidance. The quality of initial hazard assessment increases and provides greater insight into the hazardous situations and overall hazard effect. For a DHI to be implemented and used within the health industry, applicable regulatory and standards compliance activities are required. The effort to comply with these regulations and standards is great and often seen as a barrier to digital health innovations. Therefore, standardising the approach of HazID, assessment and analysis in this method and documenting the hazard summary using ontologies such as HSC and categories of controls offers a solution to this problem.
Further work
Further work will examine the qualitative aspects of this method for other DHIs. Additional DHIs assessed will provide the quantitative view. This is intended to assist with creating the ontology and terminology required and the structured language in which to improve the communication of hazards to stakeholders. We intend to apply Model Driven Engineering & object-oriented methodology, to derive these ontologies. Using the conceptual model as a representation of the problem domain. Using concepts, associations and attributes to form part of an overall model, this can be used to interpret the problem. Concepts have categories – physical, specifications, places etc. And an association is a relationship between concepts and indicates a connection between these concepts.
The objective by using model driven engineering and object-oriented methodologies is to investigate and examine the terminologies and concepts used to explore their relationship for further analysis. UML notation format provides a static structural diagram that represents the problem. By following the steps below, we aim to validate the method, the model, metamodel and instances developed.
Analyse and identify the key elements of the problem (system), the conceptual classes within the problem Identify the properties and relationships between these elements Identify any constraints (limitations) and conditions of the model The resultant conceptual model is represented for peer review as a class diagram; and finally Examples (instances) are developed from the conceptual model to validate the metamodel created – object diagram.
Further work is needed, to demonstrate this method across the full DHI classification scheme, providing quantitative analysis and results. The implementation and verification of DHIs, justified this way, provide a direct correlation to the HSC. In turn, through the development of ontologies we can further validate the methodology utilising other risk methodologies including mixed method approaches. The objective being to strengthen safety claims and provide direct correlation to HSCs.
Footnotes
Acknowledgements
This research has been completed with guidance of Prof George Despotou, a former PhD Supervisor.
Author contributions
The method and results of this research were assessed by all authors listed, leading to contribution to the final version of the manuscript. All authors contributed to the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval and consent
Not required. The research did not involve human subjects and/or animals, or case reports/case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was co-funded by the HORIZON.2.1 – Health Programme of the European Commission, Grant Agreement number: 101095448 – Advanced Security-for-safety Assurance for Medical Device IoT (MEDSECURANCE). This work was also supported in part by Engineering and Physical Sciences Research Council (EPSRC) through the grants EP/R007195/1 (Academic Centre of Excellence in Cyber Security Research – University of Warwick); EP/N510129/1 (The Alan Turing Institute); and EP/S035362/1 (PETRAS National Centre of Excellence for IoT Systems Cybersecurity)
Engineering and Physical Sciences Research Council, HORIZON EUROPE Health, (grant number EP/N510129/1, EP/R007195/1, EP/S035362/1, 101095448).
Guarantor
SH.
