Abstract
Objective
Selected patients with large vessel occlusion (LVO) strokes can benefit from endovascular therapy (EVT). However, the effectiveness of EVT is largely dependent on how quickly the patient receives treatment. Recent technological developments have led to the first neurointerventional treatments using robotic assistance, opening up the possibility of performing remote stroke interventions. Existing telestroke networks provide acute stroke care, including remote administration of intravenous thrombolysis (IVT). Therefore, the introduction of remote EVT in distant stroke centers requires an adaptation of the existing telestroke networks. The aim of this work was to propose a framework for centers that are potential candidates for telerobotics according to the resources currently available in these centers.
Methods
In this paper, we highlight the future challenges for including remote robotics in telestroke networks. A literature review provides potential solutions.
Results
Existing telestroke networks need to determine which centers to prioritize for remote robotic technologies based on objective criteria and cost-effectiveness analysis. Organizational challenges include regional coordination and specific protocols. Technological challenges mainly concern telecommunication networks.
Conclusions
Specific adaptations will be necessary if regional telestroke networks are to include remote robotics. Some of these can already be put in place, which could greatly help the future implementation of the technology.
Introduction
Acute ischemic stroke (AIS) can be defined as an interruption of the blood supply to the brain. This mainly happens when a clot obstructs one of the intracranial arteries, damaging the brain tissues and causing language disorders, paralyses, and even death. AIS is, therefore, a major cause of disability worldwide. 1 Some AIS cases can be treated with an urgent administration of intravenous thrombolysis (IVT) to dissolve the clot. The effectiveness of IVT is largely dependent on how quickly the patient receives treatment: each minute of delay equates to 1.8 days of disability. 2 Because only a few specialists in vascular neurology can provide this treatment, many regions across the world have developed telestroke networks with telemedicine tools to cut inter-hospital transfer times. This method is effective in providing patients with IVT3,4 because the drug can be directly administered by the distant hospital after a remote evaluation by a specialist in vascular neurology. It relies on strong regional organizations that are able to adequately select patients and run patient awareness programs to provide equity of stroke care, even in remote hospitals.
More recently, it has been shown that patients with large vessel occlusion (LVO) strokes can benefit greatly from endovascular therapy (EVT). 5 However, EVT also loses much of its effectiveness with treatment delays: each minute of delay equates to 4.2 days of disability. 6 Currently, LVO patients who are referred to remote hospitals for emergent strokes use the existing telestroke network to be selected for EVT and are then transferred to EVT stroke centers. The delay caused by inter-hospital transfer results in higher morbidity and mortality rates. 7 It is, therefore, necessary to find ways to provide this treatment to all populations with as much equity as possible, considering the limited human and material resources that EVT requires.
Recent technological developments have led to the first neurointerventional treatments using robotic assistance,8–10 which opens up the possibility of performing remote stroke interventions.11,12 Robotics could (1) reduce the time to treatment by removing the patient transfer step and (2) allow provision of EVT in regions so remote that the inter-hospital transfer time necessarily excludes patients from receiving EVT. The current technical barriers to the complete adoption of endovascular telerobotics include not only the technical requirements of neurovascular devices, but also the validation of a regional organization of telestroke networks, including dedicated infrastructures for teleoperations. A previous study has shown the remaining challenges if neurointerventions are to include remote robotics; 13 however, there is still a need to define and address specific challenges for telestroke networks. The aim of our work is to propose a comprehensive framework for telestroke centers, including candidates for telerobotics, according to the resources currently available in these centers. This should help the centers to prepare for the arrival of this technology by anticipating logistical difficulties and beginning to overcome them. Moreover, we aim to highlight the specific organizational and technical constraints for each type of center.
Specifications of stroke networks and infrastructures
Territorial need for telestroke networks
AIS treatments have two main constraints: they are highly time-sensitive, and they require specific expertise. When patients call the emergency phone numbers or present to emergency departments, an acute neurological deficit must be recognized by ER teams and characterized as “code stroke.” The experts in stroke neurology and neurointervention are then contacted to discuss stroke management, including reperfusion by IVT and EVT. Expertise in stroke neurology and neurointerventions is relatively rare. For this reason, regional telestroke networks have been organized using telecommunication technologies to provide 24/7 stroke neurology expertise to remote regions, including clinical examination through high-resolution cameras. In Europe, the European Stroke Organisation (ESO) formalized regional organization into stroke units, which can provide in-hospital stroke care, and stroke centers, which integrate a stroke unit into the full clinical pathway of stroke patients, including neurointervention and neurosurgery. 14 The ESO later defined three types of hospital center. A comprehensive stroke center equipped with telemedicine is called a telemedicine stroke center (TSC). A telemedicine-assisted stroke unit (TSU) is a stroke unit without 24/7 on-site stroke expertise. A telemedicine-assisted stroke-ready hospital (TSR) takes in stroke patients transferred after hyperacute management, including IVT. 15 In addition to these existing categories, we would like to introduce the telerobotic stroke unit (TrSU) as a center equipped with a “leader–follower” endovascular robot, and wherein an EVT procedure for an acute stroke could be performed with the aid of a remote neurointerventionist from the TSC. Figure 1 displays a fictional example of such a stroke network, including the new TrSU.

Regional stroke network organization in a fictional area. The aim of a telerobotic stroke center would be to cut out the time needed to transfer to an EVT stroke center (white dotted line). IVT: intravenous thrombolysis; EVT: endovascular treatment (thrombectomy).
Assessment of regional needs
An essential step in the development of a stroke care program is the epidemiological assessment of the needs of the population served by the stroke network. A telerobotic program requires a remarkable amount of human and financial resources, and the first centers selected for its implementation should be chosen based on objective criteria. We suggest that each stroke network should evaluate each of its centers that are candidates for telerobotics. Ideally, a network should be able to provide up-to-date statistics on the number of patients presenting with LVO. Based on this, they should calculate the number of candidates for mechanical thrombectomy per year for each of the centers in the network. In addition, each TrSU candidate should provide the inter-hospital transfer time for its current EVT access. A hierarchy of the centers that would benefit most from telerobotics can then be created, which would help establish recommendations for telerobotics implementation. The most important clinical need will be for those centers with the highest number of EVTs/year and the longest inter-hospital transfer time. A recent study showed that a loss of 10 min for a patient eligible for EVT amounts to a loss of USD 10,593 in the United States. 16 The total time lost per year can, therefore, be presented in parallel with these medico-economic studies to justify the implementation of telerobotics programs to policymakers. These data can help us find out relevant criteria in terms of minimum time saved and minimum case volume for TrSU selection, although these figures could be very different according to each country/region's willingness to pay “quality-adjusted life year” (QALY). A recent retrospective analysis of a large EVT registry showed that longer inter-hospital transfer times result in poorer outcomes: patients with less than 120 mn transfer time had better outcomes compared to those with 120–180 mn, who had better outcomes than patients with >180 mn transfer time. In addition, Sanmartin et al. created a cost-effectiveness model demonstrating that telerobotics would be cost-effective for one patient if it saves a 20 mn delay for an additional cost of 40,000 USD. 12 Minimum volume cases can then be deducted from the robotic cost and should take into account the minimal cases for adequate expertise.
After the implementation of a telerobotic center, a continuous clinical evaluation should be performed preferably to provide high-level evidence of efficacy possibly in a randomized controlled trial. Different workflow strategies to treat stroke have been successfully evaluated in this way.17,18
Alternative care pathways
A specific analysis of regional needs appears to be fundamental to discussions of such huge and onerous changes as robotic EVT. It is likely that some regions would benefit more from the intra-hospital optimization of a workflow than from telerobotics, especially if the inter-hospital distances are not so long. Different workflow methods have also been evaluated to reduce time to treatment in distant hospitals and might be more suitable than robotics in specific regions. The “drip and drive” model 19 and its modified “Rendez-Vous” approach (a cardiologist starts the procedure, while a neurointerventionist travels to the peripheral hospital center), 20 the “flying intervention team,” 21 and the rotation of teams between the remote center and the expert center can provide huge decreases in time to treatment for selected regions. However, they all require a second on-call dedicated team to physically go to the remote center, while the expert center still needs to be staffed. A comprehensive approach to a specific stroke network should discuss every method to provide rapid stroke care to patients.
Network infrastructure for telestroke and robotic telestroke
The current infrastructure of a regional stroke network performing telestroke includes connections between hospitals and plays a critical role in the treatment a patient receives, especially IVT reperfusion. This infrastructure should involve the following: voice communication, video transmission of the patient's clinical examination—possibly including remote control of a secure camera—rapid transmission of diagnostic medical imaging, transmission of surgical imaging, and transmission of robotic control. A set of guidelines detailing the recommendations for telecommunication in the context of video teleconsultation for a telestroke network has already been produced. 9 These guidelines recommend the use of a virtual private network (VPN), a connection protected by the Advanced Encryption Standard (AES) up to 256 bits and a broadband speed above 1 Mbps. The technical characteristics of video teleconsultation systems have been described elsewhere. 15
As with any other data transfer, robotic telestroke networks must pay attention to data security and bandwidth. Network reliability is of vital importance because any interference with communications during a telerobotic procedure has a direct impact on patient safety. The bandwidth is also crucial because it is directly linked to the latency between the robotic commands and the live video. Interventional procedures that are guided by imaging, as is the case for endovascular neurological interventions, require a live manipulation of the catheter position based on X-ray images. Latency criteria must, therefore, be considered alongside broadband speed as latency can cause the operator significant problems when manipulating the catheters. These criteria were studied during the first remote robotic surgery by Marescaux et al. in 2001, during which they used the first fiber-optic broadband connections. 22 The maximum latency considered to be safe by the surgeon was 330 ms. Multiple endovascular telerobotics procedures using preclinical cardiology models recently demonstrated the feasibility of telerobotics. These procedures used infrastructures already available in the US, such as hardware firewalls and VPNs. 23 In the procedures that used wired or 5G connections, the latencies were imperceptible to the operator and measured 120 ms for the wired connection and 160 ms for the 5G connection. This preliminary study, therefore, seems encouraging for the pre-existing infrastructure in developed countries. However, specific tests for neurological interventions are still necessary as they often use two flat-panel X-ray detectors, whereas cardiological interventions only use one. It is, therefore, likely that the neurological interventions require a higher broadband speed to transmit two live images simultaneously.
The regional network will also have to prepare for technical failure of the communication network. Although real-life remote surgeries have shown that such procedures are feasible in different regions around the world,13,22,24,25 it is necessary to prepare for rare and potentially dangerous network interruptions. These could be managed by network failover strategies 26 that aim to switch from one impaired connection to another when necessary. In addition, backup generators are crucial for both TrSU angiosuite and CSC cockpit. The network connections between the site of the potential TrSU and the TSC should, therefore, be assessed carefully before setting up the TrSU. It would seem helpful to include tools for the continuous evaluation of the connection quality from the outset of the project in order to rapidly identify issues and provide solutions if necessary, as is already recommended for teleconsultation systems. 15
Digital data management
Robotic endovascular therapy, both local and remote, should require a new approach to the digital data. These data include medical imaging acquired during the procedure from the two X-ray flat panels (digital subtracted angiographies and live fluoroscopies). They also include robotic commands and vocal communications between the distant robotic operator and the bedside team. These data should be acquired during the procedure time and documented as mean 90 mn. 5 Monitoring systems specific for operating rooms have already been developed, as has a “black box” system 27 that can be used for workflow studies and improvement, surgical study assessment, future robotics developments, and automation. Of course, these are considered as clinical and identifying data; hence, it would be necessary to follow and update a specific local legislation for storage and use in addition to specific, secured infrastructures. Patient privacy should be respected, and data registration should be considered in both centers: the TrSU and the TSC.
Specifications for telerobotic thrombectomy
Requirements for a remote EVT center
Once the indication for EVT has been agreed upon, the patient should undergo mechanical recanalization as quickly as possible. In the context of a telerobotics procedure, a significant amount of logistics must be mobilized. The neurointerventionist must go to the cockpit of the “leader–follower” robotic system at the TSC (Figure 2). The TrSU must gather a team of technologists who are experienced in the use of robotic assistance, as well as an on-site support physician. The procedure would, therefore, be performed under the joint responsibility of the remote operator and the on-site support physician. The concept is similar to the current performance of remote IVT reperfusion, for which the medical responsibility is shared between the on-site emergency physician and the remote neurologist. The on-site support physician team should establish clear protocols in accordance with the remote team regarding peri-operative environments, considering the local guidelines and legal environments. These include anesthesia techniques that may require conscious sedation or general anaesthetic. 28 The bedside team should also be prepared to manage all the other non-robotized aspects of the procedure, such as the following: getting the patient into position; preparing the puncture site and performing the arterial puncture (in the future, this step may also be robotized29,30); preparing the robot and inserting the sterile equipment; maneuvering the table and the flat panel detectors; injecting the contrast agent; and acquiring the digital subtraction angiography images. This new setup requires a new role for bedside technologists or nurses, who would also be responsible for running the angiography unit between the technical management of the procedures. Endovascular interventions may require specific equipment. Particular attention should be paid to maintaining the stocks, including those materials that are not often used, but remain essential in certain situations, such as angioplasty balloons, carotid artery stents, and intracranial stents. This stock should be identical to that used in the TSC according to the telerobotics operator protocols. The rapid technological progress made in this area, along with the common use of off-label techniques, 31 makes the close collaboration between the remote operators and the on-site medical device coordinators all the more important. In light of the recent progress concerning non-mechanical EVT, it is also necessary to maintain a stock of emergency antithrombotic agents (e.g. Alteplase, Tirofiban, Cangrelor, etc.), which could be necessary during EVT.32–34

Illustration of a generic leader–follower robotic system for the remote endovascular treatment of acute ischemic stroke.
Constraints for a trSU center
However, before telerobotic thrombectomy can be fully implemented, there are other potential challenges, which we can already begin to tackle. The maintenance of the expertise of the TrSU will depend on the volume of cases. Some centers will be selected because of their remoteness, and they may have a low volume of cases, making it difficult for the local team to maintain their reactivity and competence. It is known that a higher volume of interventions is associated with a higher expertise. 35 Patients transferred to high-volume centers (>132 cases per year) had lower mortality rates in a large retrospective study. 36 Maintenance of expertise concerns every potential TrSU and is the primary barrier to setting up the neurointerventional infrastructures in a center that is not the largest TSU. One solution could be setting up simulation labs to carry out regular mock remote procedures. This would allow the TrSU's bedside team to perform the required number of procedures to ensure high-level EVT of AIS. The remote operators usually deal with a high volume of cases by treating the population of the TSC, but the bedside team at the TrSU also has specific technical skills that must be maintained. As such, we suggest the installation of a simulation lab that allows the bedside robotic technologists to regularly practice their skills. Simulation labs have already been shown to be useful in the adoption of new neurointerventional technology 37 and are likely to also be useful in the long-term maintenance of expertise.
In terms of telecommunication, the literature on telestroke networks suggests that it is important to assess the latency in addition to the connection speed. Existing networks in high-income countries seem able to provide such low-latency live imaging, 23 but latency should be a basic prerequisite for a TrSU program.
Medical process of stroke patients surrounding telerobotic thrombectomy
Pre-hospital patient care
An in-depth discussion of pre-hospital resources or prevention programs is beyond the scope of this work. However, it is important to note that the smooth functioning of the pre-hospital pathway is mandatory for any stroke care program. Before launching a telemedicine or telerobotics project, it is necessary to perform an epidemiological study to evaluate the population and the logistical possibilities open to the pre-hospital teams in order to bring the stroke patients to the correct hospital centers in time. Pre-hospital emergency notification to the receiving hospital is also essential in the reduction of in-hospital treatment time. 28 Ensuring the smooth running of this pathway is even more important for centers that previously only required neurological and diagnostic radiology teams and that would now also need to manage the logistics related to the angiosuite and the pre-operative team.
Clinical examination and initial neurological consultation
The diagnosis of LVO requires, at a minimum, a clinical neurological examination as soon as the patient is admitted to the emergency department and an expert opinion in stroke neurology. This opinion can be provided by an on-site physician team or an on-call specialist. It may also be provided remotely via telemedicine from an expert center. We believe that in the context of telerobotics, it is important that the telemedical consultation be performed by the same partner center that will perform the remote telerobotic act. When the neurological opinion is provided by telemedicine, specific software should be used to allow a more informed decision to be made than in a telephone-only consultation. 38
Diagnostic imaging
The clinical examination should be supplemented by emergency brain imaging, which is available 24/7. This imaging has three aims: first, to confirm the diagnosis of cerebral infarction and exclude brain hemorrhage; second, to detect the presence (or absence) of LVO (these two items are sufficient for the indication for EVT in patients admitted in the 6 hours following the symptom onset); and third, advanced imaging (perfusion computed tomography or magnetic resonance imaging) is used in patients admitted more than 6 hours after the symptom onset to include them in medical or mechanical recanalization procedures, where appropriate.
Different centers may use different brain imaging protocols, and there is debate around which is the most effective.39,40 In the context of telerobotics, in order to optimize the decision-making process and facilitate adhesion to the telerobotic program, we believe it is reasonable for the TrSU to work in partnership with the expert stroke center that provides the teleconsultation and ideally to have access to the same types of imaging with the same availability as at the TSC. This could be supported by the use of AI-powered automated software to automatically detect potential EVT candidates or compute advanced imaging if remote centers do not have local stroke imaging expertise.41,42
Based on the clinical and imaging data, the on-site or remote neurologist should make a decision regarding recanalization treatments. When there is an indication for thrombolysis, this should be performed on site as quickly as possible, even when there is an indication for EVT. 43
Setting up remote thrombectomy networks
Collaboration between the different stakeholders involved in telerobotics is particularly important and may prove challenging. First, some TrSUs may be required to choose between multiple expert centers for the neurological consultation, the neuroradiological diagnosis, and the telerobotics operator. The physical proximity and pre-existing collaboration between the expert neurological team and the neurointerventionists should facilitate decision-making by mimicking the decision-making process used in the pre-existing “drip-and-ship” paradigm.43,44 To support the collaboration, we suggest that, as far as possible, the diagnostic imaging protocols used by the expert center should be adopted by the TrSU. This is particularly important for the small SED-type centers that already make use of remote neurological consultations.
Post-operative treatment
After EVT, patients may require multiple levels of care. First, the criteria necessary for a unit to be suitable for AIS patients and to be registered as an SU have been published previously. 45 Second, patients might need additional care outside the SU. If a patient underwent general anesthesia, continuous post-procedural monitoring is required. This must, therefore, be possible in each TrSU providing it, 46 possibly in intensive care units (ICU). Patients who underwent conscious sedation may be rapidly transferred to a stroke unit. If there is a SU on site, we suggest admitting the patient to the SU and contacting the neurologist who treated the patient via telemedicine if there are any problems outside of working hours. If there is no SU, it seems reasonable to temporarily admit the patient to the local ICU, and then to transfer them to the expert center that gave the indication for EVT and performed the operation as soon as the patient stabilizes. This would allow the patient to benefit from the improved recuperation and reduced mortality rate provided by SUs 47 and would improve the consistency of care.
Specifications of telerobotics centers
Based on the requirements previously identified, we produced a set of specifications for the TrSUs and expert TSCs, which we summarized in Table 1. Based on this set of specifications and our categorization of the centers that admit stroke patients, we identified the gaps that must be addressed for each kind of center if it is to become a TrSU.
First, the conversion of a TSU (which by definition includes an SU, but no neurointervention) into a TrSU requires a robot, a team of robotic technologists, and a support physician team. We suggest including a simulation laboratory or a contract with an academic center depending on the expected volume of procedures. It is known that stroke volume per site is associated with the efficacy of the technique,48,49 which could lead to future guidelines for setting a minimum threshold of procedures. This number is thought to be the minimum required in order to maintain a high level of competence, particularly in terms of the procedure time. In addition to the requirements listed above, work on the pre-hospital pathway would be required, as well as identification of a local hospital that can perform the post-operative care. Ideally, consideration should be given to how to provide the same level of care in remote centers as in an SU, and the legislation should be adapted accordingly. This would likely include patient monitoring in an on-site polyvalent ICU coupled with 24/7 availability of a stroke neurology expertise (possibly remotely). Future challenges for TrSU specifications will include a specific certification for telerobotic operators and bedside teams with specific training and a logbook. 13
List of resources necessary in each center for the implementation of a telerobotic stroke network.
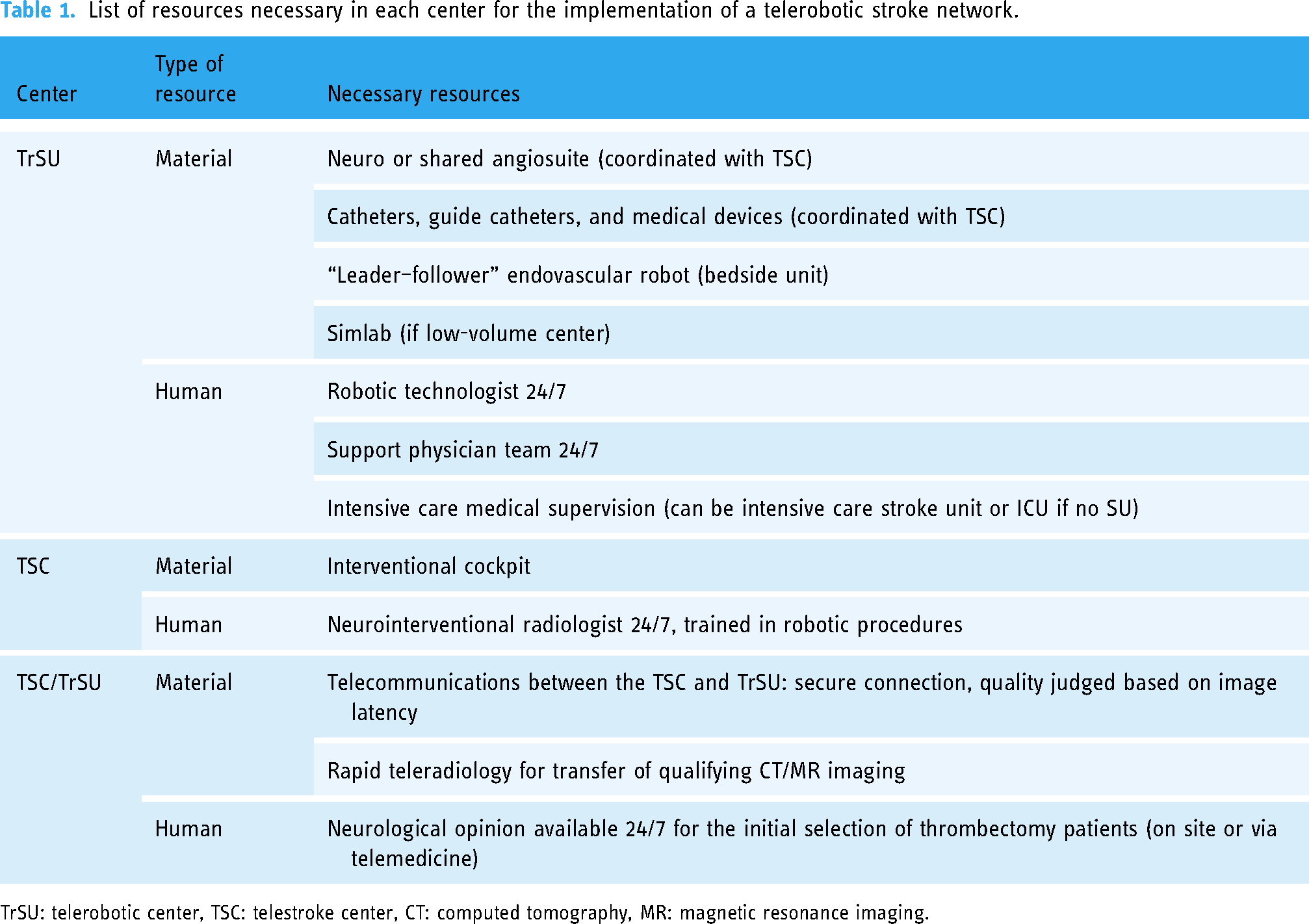
TrSU: telerobotic center, TSC: telestroke center, CT: computed tomography, MR: magnetic resonance imaging.
Overview of the robotic system aiming to achieve remote neurointerventions
The first commercialized robotic platform for endovascular neurointerventions was the Corpath GRX (Siemens, Erlangen, Germany), which was an evolution of a coronary platform. 8 This system protects the operator from ionizing radiation and provides comfortable working conditions in which the operator is not required to wear lead protective clothing. 8 This first generation was able to manage a microcatheter and a microwire and, therefore, the intracranial part of neurointerventions (although not the extracranial). It was limited by the catheter and wires’ working length and the limited types of compatible devices. A recent decision was made by Siemens to close this robotic program to focus on the neuro-field and their next-generation remote neurointerventional robot. Other competitors have announced prototypes targeting remote neurointerventions. Sentante (Novatyvi Medicina, Vilnius, Lithuania) should be able to use regular catheters and wires and be operated by simulated catheters and wires from a distant control room. 50 Remedy robotics (San Francisco, USA) and Robocath (Rouen, France) also announced a remote prototype. Additionally, academic engineering breakthroughs could help the adoption of the technology. Recently, Kim et al. described how a magnetically controlled catheter could help the operator achieve remote robotic neurointerventions. 51 Use of a dexterous magnetic robot has also recently been published. 52 Despite the apparent variability of these platforms, they are currently all based on the “leader–follower” design. A bedside unit must be managed by a bedside team, mostly for loading catheters, wires, and devices into the robot. A remote operator uses a controller. Regardless of the next robotic platforms to enter the market, the same questions will need to be considered to prepare existing telestroke networks for robotics. We anticipate that decision makers and clinicians will soon have to face choices regarding which center to target for TrSU, how many of those will be needed and/or cost-effective, and how will they interact with the existing telestroke networks. Our framework could be the basis for making these decisions and could eventually help the adoption of a needed technology.
Conclusion
This study demonstrates the need to address organizational and technical problems that may arise with the adoption of remote robotics. First, site selection requires objective criteria to prioritize the largest population and the most remote sites. Second, some technical challenges can be anticipated, especially regarding telecommunication networks. Finally, new human resources will be needed, and these staff will need to be trained and continuously certified. It is important to lay the groundwork before opening a TrSU in order to anticipate any logistical gaps or difficulties. Advances in knowledge, particularly in the various treatments for stroke, mean that organizations need to adapt as closely as possible to where the patients live and to the largest possible number of patients, whatever the technical complexities that may arise with the implantation of digital health.
Footnotes
Acknowledgements
The authors would like to thank Jennifer Dobson for translating and proofreading this manuscript and Karim Chakroun for the design of Figures 1 and ![]() .
.
Contributorship
TM and GC conceived the study. All other authors contributed equally to the writing process.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No ethical approval was necessary for this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
GC.
