Abstract
Objective
This study aimed to analyze the effect of Smart Cancer Care program on the quality of life and ease of chemotherapy continuation in cancer patients and the effect of additional tele-management on frequency of use and satisfaction with the Smart Cancer Care program.
Methods
‘Smart Cancer Care’ is a mobile program that allows cancer patients undergoing chemotherapy to report symptoms of adverse events and receive remote management. In this study, patients were randomly assigned to three groups: Group A, who received only classical face-to-face management; Group B, who used the Smart Cancer Care program as addition; and Group C, who used the Smart Cancer Care program and received telephone management. After 12 weeks of follow-up, the effectiveness of using the Smart Cancer Care program was analyzed by examining the quality of life, ease of maintaining chemotherapy, and unplanned hospital visits in each group. The frequency of use and satisfaction with the Smart Cancer Care program were also analyzed.
Results
Cancer patients who used the Smart Cancer Care program had 1.93-fold (1.15–3.25) higher overall quality of life than those who did not. This became 2.33-fold (1.34–4.04) higher when phone care was added. Patients with tele-management were significantly more likely to use the Smart Cancer Care program (odds ratio (OR) = 25.80; 95% confidence interval (CI), 11.28–58.97).
Conclusions
A mobile self-reported management program has a positive effect on the quality of life of cancer patients undergoing chemotherapy. Tele-management is conducive to active and effective use of this program.
Keywords
Introduction
Each year, the number of new cancer cases diagnosed worldwide is rapidly increasing from 14.1 million in 2012 to an estimated 21.6 million in 2030. 1 Following the emergence and worldwide spread of COVID-19, there has been a rising popularity of smartphone health apps. The growing interest in utilizing applications for health behavior intervention within the medical field represents an opportunity to improve cancer care and management. 2 These apps can be used to collect objective data about patients’ behavior and behavior monitoring, which could help patients change their behavior, promote self-monitoring of symptoms, and enhance patients’ sense of empowerment and willingness to care for themselves3,4 while allowing them to communicate with their health care team from a distance.5,6 Adverse symptoms are common among patients receiving treatment of advanced cancers and often go undetected. The systematic collection of symptom information using patient-reported outcome (PRO) standardized questionnaires has been suggested as an approach to improve symptom control. 7
This study focused on the usefulness of “Smart Cancer Care (SCC),” a platform established to implement a management program for the side effects of chemotherapy in the Republic of Korea. Previous research on remote monitoring systems in the chemotherapy context highlights their benefits for health-related quality of life, symptom alleviation, avoiding unscheduled hospital admissions, survival, and cost-effectiveness. 8 There is compelling evidence that cancer patients within traditional consultations often do not receive sufficient help with their symptoms and problems, urging them to call for better and more patient-centered models of care delivery. Interactive health communication applications that reduce symptom distress and can provide cancer patients with assistance when and where they need it will make an important contribution to the care of cancer patients. 9
The primary purpose is to help cancer patients reduce their symptom distress, improve quality of life and emotional well-being, and enhance self-efficacy and social support. The e-communication tool allows patients to stay connected with clinicians who can quickly answer questions and concerns. The purpose of this study was to evaluate the effects of Smart Cancer Care on three groups. We hypothesized that there would be a difference between the group using Smart Cancer Care and the group with added telephone management compared to the group using conventional treatment only. Various randomized controlled trials (RCTs) have found that mobile health interventions may be effective for adult cancer survivors. 10 We hypothesized that cancer patients who received web and telephone intervention would have better outcomes compared with a control group that received links to publicly available, cancer-related websites.
Methods
Setting and participants
This study was a feasibility RCT with ethical committee approval from the Institutional Review Board of Ulsan University Hospital (2019-08-034). Assuming that the general treatment completion rate of patients receiving chemotherapy is 70%, we sought to test the hypothesis that the treatment completion rate of patients using ‘Smart Cancer Care’ would improve by 15% to over 85% with a power of 90% and a significance level of 0.05. The required number of participants was 174 in each group, and considering a dropout rate of 10%, a total of 600 participants, with 200 individuals in each group, were recruited.
The subjects were voluntary participants among adults over 18 years of age who were undergoing chemotherapy at the Department of Hematology and Oncology, Ulsan University Hospital. A 12-week follow-up was carried out by dividing the groups into three: the group using the existing treatment (A); the group using the platform (B); and the group using the platform and telephone management (C). This study evaluated the quality of life,11,12 medical treatment other than chemotherapy, discontinuation of chemotherapy, application usage rate, and application satisfaction (Figure 1).

Study flow.
The following inclusion criteria were applied: cancer patient being treated at Ulsan University Hospital; age > 18 years; a histologically confirmed diagnosis of cancer; Eastern Cooperative Oncology Group (ECOG) performance status 0 or 1; 13 a life expectancy of ≥ 6 months; and the possession of and ability to use a smartphone.
Evaluation of platform development and feasibility
The Smart Cancer Care platform is divided into the following: 1) the application for patients that enables the self-evaluation of symptoms to be managed based on the cancer type and administered anticancer drug, and references management guidelines according to the results,; and 2) the dashboard for medical staff whereby the results entered by the patient can be assessed in real time. 14 In order to evaluate the availability of the developed application for patients and the dashboard for medical staff, Smart Cancer Care was used for 2 to 3 weeks by cancer patients and medical staff involved in cancer treatment. Their experience using the platform was then investigated. A survey on the patients and medical staff was conducted to quantitatively evaluate the usefulness of Smart Cancer Care in the cognitive, psychological, written, and social aspects (a total of 12 items), and suggestions to improve Smart Cancer Care were collected using open-ended questions. The usefulness was evaluated on a five-point scale (0: strongly disagree, 1: disagree, 2: neither agree nor disagree, 3: agree, 4: strongly agree). Higher scores indicate greater usefulness, while negative questions were reverse-scored. 15
Evaluation symptoms and effectiveness
The symptom stages were divided into stages 1 (preventive management), 2 (self-care), and 3 (consultation with medical staff or medical institution visit) after consulting previous studies. The guidelines were developed accordingly.16,17
The pain input portion includes position, intensity, number of breakthrough pain instances, or sleep disturbances caused by pain. The pain intensity was evaluated using a numeric rating scale (NRS) ranging from 0 (no pain) to 10 (worst pain imaginable).
Statistical analysis
Categorical variables are expressed as number and percentage, and continuous variables are expressed as mean and standard deviation (SD). Chi-squared tests were used to compare percentages, and independent sample t-tests were used to compare the mean values. All significance levels refer to two-sided tests. A p value < 0.05 was considered significant. All statistical analyses were performed using SPSS for Windows software, version 18.0 (SPSS Inc., Chicago, IL).
Results
Patient characteristics
A total of 363 patients were screened between February 2015 and December 2016, and 348 were included in the study. Ten patients were excluded due to refusal to participate (n = 8), inability to use a smartphone (n = 1), or cognitive impairment (n = 1). The follow-up duration (mean ± SD) of 348 patients included in the final analysis was 12 weeks. The baseline characteristics of the patients are listed in Table 1. Patients entered their SCC using their smartphones.
Baseline characteristics of the study population.
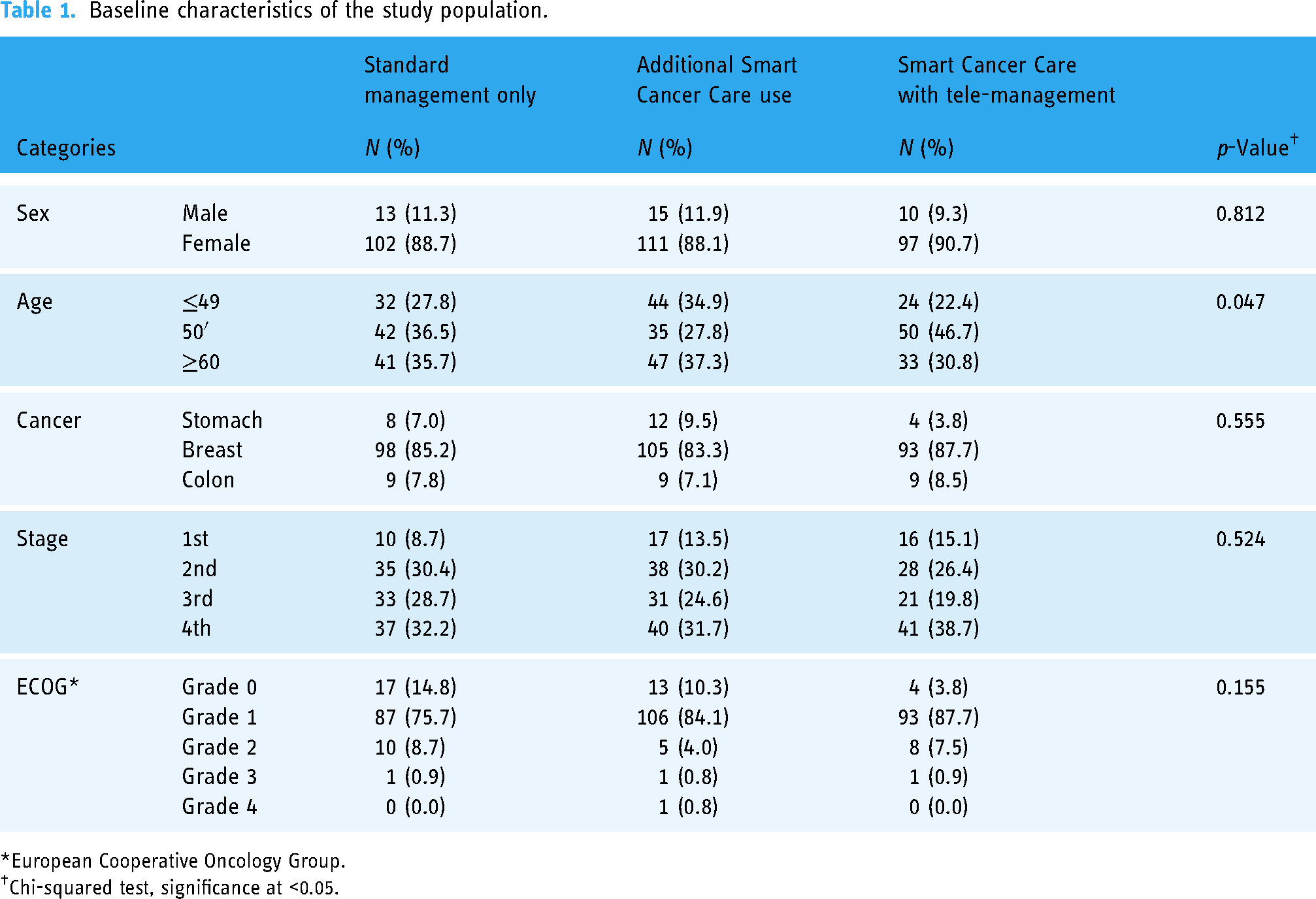
European Cooperative Oncology Group.
Chi-squared test, significance at <0.05.
Quality of life
The degree of pre/post change in the quality of life was observed and categorized as ‘improved: Score increased,’ and ‘unchanged/worsened: Score remained the same/decreased’ was categorized as binary. The odds ratio with Group A as the reference level is presented along with the odds ratio of the quality of life improvement adjusted for gender (male and female), age (under 40, 50, over 60), and stage (stages 1, 3, and 4). However, the quality of life for symptoms is rated as positive as the score decreases (Table 2).
Differences in the quality of life according to patients’ character and management methods.

A: patients with only standard management; B: patients using Smart Cancer Care; and C: patients using Smart Cancer Care with tele-management.
Overall quality of life and function of quality of life are treated as improved when the score increases, and the symptom of quality of life is treated as improved when the score decreases.
For the overall quality of life, the odds (95% CI) was 1.93 (1.15–3.25) for Group B and 2.33 (1.34–4.04) for Group C, showing a statistically significant increase in the odds compared to Group A (p < 0.05). Among the functions in the quality of life, emotional functioning was found to have statistically significant higher odds of 2.43 (1.39–4.24) in favor of Group C compared to Group A (p < 0.05) (Table 2).
Ease of continuing chemotherapy
We estimated the odds of one or more chemotherapy delays and interruptions during the 12-week follow-up. The odds (95% CI) for Group B was 0.65 (0.20–2.15), which decreased compared to the odds for Group A, but was not statistically significant. Group C was found to have a slight increase of 1.31 (0.43–4.00), but there was not enough evidence to be statistically significant. In other words, the odds of delayed and occurring chemotherapy did not differ between groups. The odds ratio for SCC use (groups B and C) was estimated to be 0.92 (0.35–2.44) with no statistical significance (Table 3).
Differences in effectiveness according to patients’ character and management methods.
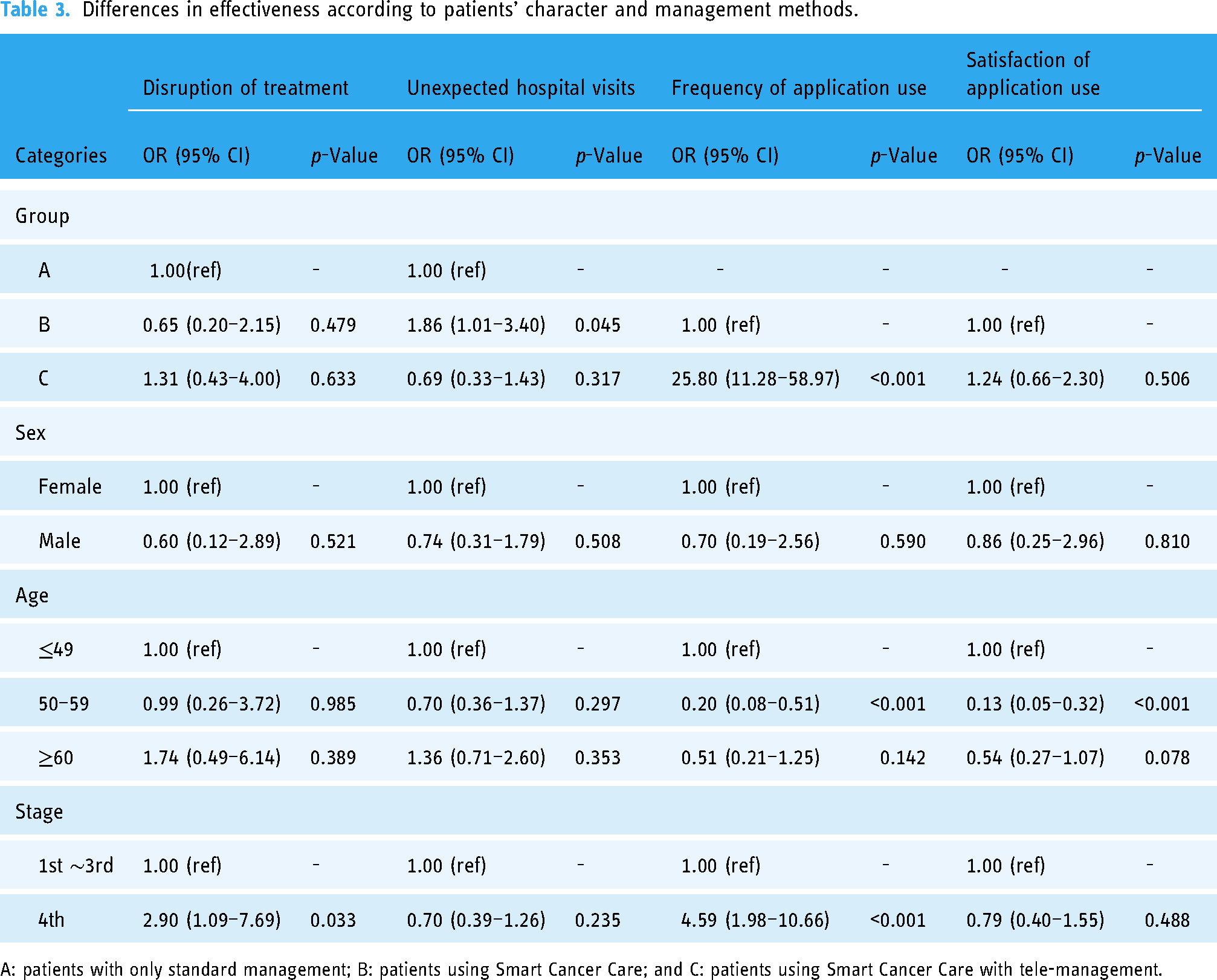
A: patients with only standard management; B: patients using Smart Cancer Care; and C: patients using Smart Cancer Care with tele-management.
In the statistical tests, the number of cases of chemotherapy delays and interruptions was too small to have sufficient power (Group A: 7 (6.1%), Group B: 5 (4.9%), Group C: 7 (7.4%)). We estimated the odds for outpatient/emergency room visits or hospitalizations for reasons of side effects outside of routine chemotherapy, etc. The odds (95% CI) for Group B was 1.86 (1.01–3.40), which was a statistically significant increase compared to the odds for Group A. By contrast, a decrease in odds to 0.69 (0.33–1.43) was found for Group C, but there was not enough evidence to be statistically significant (Table 3).
Frequency of use and satisfaction with the SCC program
Utilization was calculated according to the number of symptom reports using the SCC per week over a 12-week period, and the odds was estimated for ≥6 (50%). The odds (95% CI) for Group C was 25.80 (11.28–58.97), which was significantly higher than that for Group B and statistically significant. While Group B had 23.2% (29 out of 125) of its users exceeding 50% usage, Group C had 85.2% (75 out of 88) of its users showing high fidelity to the app (Table 3).
Satisfaction was evaluated according to the app system satisfaction tool developed for this study, and the satisfaction level was divided into five grades of A, B, C, D, and F. The odds was estimated by organizing the five grades into two categories (good: A, B; poor: C, D, F). The odds (95% CI) for Group C was 1.24 (0.66–2.30), which showed an increased odds compared to Group B, but was not statistically significant (Table 3).
Discussion
Cancer patients undergoing chemotherapy must manage an overwhelming amount of information to coordinate their treatment and related activities. Comprehensive coordinating efforts are needed to manage complex schedules and adverse effects of chemotherapy treatments. 18 This suggests that the increasing availability of smartphones across socioeconomic groups and regions provides an opportunity to develop digital solutions to reduce health disparities through increasing access to educational resources and symptom monitoring and management. 19
In summary, the odds for the overall quality of life were 2.33 times higher for Group C and 1.93 times higher for Group B compared to Group A, which was statistically significant (p < 0.05). The odds of delayed chemotherapy between groups were not statistically significant (p > 0.05), and the number of events was too small to achieve sufficient power. To answer the question of whether or not there is a difference in the incidence of medical care other than chemotherapy, we compared the outpatient, emergency department, office visit, and hospitalization experiences. For the outpatient/emergency room/hospitalization experience, a significant 1.86-fold increase in odds was found for Group B compared to Group A (p = 0.045). By contrast, a 0.69-fold decrease was identified for Group C, albeit the difference being not statistically significant (p > 0.05).
We looked at the weekly app usage to see if there was a difference in the level of adverse event reporting between weeks. When the app usage rate for symptom reporting was greater than 50%, the odds for Group C were found to be 25.80 times statistically significantly higher than that for Group B. Finally, the corrected mean (95% confidence interval) of the app system satisfaction was 62.57 (59.41–65.74), with lower satisfaction among older age groups. Apps can be leveraged by patients to support their efforts to manage their health, coordinate appointments, and keep track of treatment notes and questions. 20 Emerging evidence suggests that health apps can be used by various population groups and may support improvements in pain and fatigue. 21
Currently, smartphone users are increasing worldwide and are expected to reach 6.8 billion by 2023, with a smartphone penetration rate of 53.8%. Furthermore, smartphone apps have natural advantages over websites, SMS text messages, and other similar communication methods owing to their personalized design, rich mobile device features (e.g. cameras, phones, GPS, and contact lists), and timely push features. Therefore, the use of smartphone health apps could be a potentially effective way to improve mental health and social functioning among cancer patients. 22
Based on the experience of patients using Smart Cancer Care, it is believed that patients’ expectations for Smart Cancer Care were high, and they considered the application convenient. Specifically, cancer patients generally experience anxiety about the treatment itself,23,24 but anticipate that Smart Cancer Care can be useful because being able to communicate with medical staff and report side effects could help reduce anxiety about chemotherapy. 8
Remote monitoring and intensified care of symptoms at home extend cancer care to patients where they live and pair care with real-time symptom needs. Automated systems provide efficiencies and judicious use of provider time in follow-up. They also provide an extension of care availability for patients living at a distance from treatment centers and are particularly important for those living in rural communities, where supportive care services are geographically limited. 25
In our study, the two study groups did not report differences in the quality of life, physical health, or mental health. This may be due to the relatively short follow-up period that was done while the patients are still in the midst of completing chemotherapy, which can impair daily function and worsen the quality of life. Future studies should examine the impact of the intervention on patients in different phases of their treatment starting with the initial diagnosis and on longer-term quality of life after they complete their active cancer treatments.
When dividing the comparison group, there is a limitation in that it is difficult to clearly compare what kind of tool makes a difference and in that there is no tele-management alone without the SCC program. Many recent studies have been conducted mainly using websites, PC software, smartphone, and tablet applications rather than using a phone. In 2014, Zapata et al. reviewed empirical usability methods for mobile applications in health, analyzing 22 studies. 26 At the time of that review, the number of medical applications in app stores was estimated at 28,000 (20,000 iOS and 8000 Android). Thus, it is time to re-investigate how usability testing methods for eHealth applications are described in the literature published. 27
Although the goal was to recruit cancer patients in a balanced manner, an overwhelming number of breast cancer patients participated during the study period, which limited the ability to analyze differences by cancer type. In particular, the reason why breast cancer patients had high participation appears to be because many patients showed relatively good results, and the barriers to using smartphone applications were low due to the patient's low age group.
There has been a burgeoning interest in using health-related quality of life (HRQL) assessments in daily clinical practice, with HRQL emerging as a pivotal outcome measure in the continuum of cancer survivorship care. 28 We think we need to discuss which tools are usability testing methods that are being used in the development of eHealth applications. Above all, it should take precedence over the method that various approaches and many studies on methods that are accessible and helpful to patients should be made.
Conclusion
A mobile cancer care program has a positive effect on the quality of life of cancer patients undergoing chemotherapy. Tele-management is conducive to the active and effective use of this program.
Footnotes
Acknowledgments
We acknowledge the insightful guidance and mentorship provided by our colleagues and research advisors, whose expertise and encouragement greatly contributed to the accomplishment of this study. This study would not have been possible without the collective efforts and contributions of all involved parties. Thank you for your invaluable contributions to advancing cancer care and enhancing the lives of patients through this research endeavor.
Contributorship
SJK and DYK conceived and designed the analysis. CS, HK, HSI, and SJK collected the data. HK, HSI, SJK, and DYK contributed data or analysis tools. DYL and DYK performed the analysis. CS, DYL, SJK, and DYK wrote the paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted with exemption approval from the Ulsan National University Hospital Institutional Review Board (IRB number: 2021–05-042-025).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korean Cancer Survivors Healthcare R&D Project through the National Cancer Center, funded by the Ministry of Health & Welfare, Republic of Korea (HA23C0478).
Guarantor
S.-J.K.
Informed consent
All study participants provided informed consent, and the study design was approved by the appropriate ethics review board.
