Abstract
Background
The advance of digital health technologies has created new forms of potential pathology which are not captured in current clinical guidelines. Through simulation-based research, we have identified the challenges to clinical care that emerge when patients suffer from illnesses stemming from failures in digital health technologies.
Methods
Clinical simulation sessions were designed based on patient case reports relating to (a) medical device hardware errors, (b) medical device software errors, (c) complications of consumer technology and (d) technology-facilitated abuse. Clinicians were recruited to participate in simulations at three UK hospitals; audiovisual suites were used to facilitate group observation of simulation experience and focused debrief discussions. Invigilators scored clinicians on performance, clinicians provided individual qualitative and quantitative feedback, and extensive notes were taken throughout.
Findings
Paired t-tests of pre and post-simulation feedback demonstrated significant improvements in clinician's diagnostic awareness, technical knowledge and confidence in clinical management following simulation exposure (p < 0.01). Barriers to care included: (a) low suspicion of digital agents, (b) attribution to psychopathology, (c) lack of education in technical mechanisms and (d) little utility of available tests. Suggested interventions for improving future practice included: (a) education initiatives, (b) technical support platforms, (c) digitally oriented assessments in hospital workflows, (d) cross-disciplinary staff and (e) protocols for digital cases.
Conclusion
We provide an effective framework for simulation training focused on digital health pathologies and uncover barriers that impede effective care for patients dependent on technology. Our recommendations are relevant to educators, practising clinicians and professionals working in regulation, policy and industry.
Introduction
A digital technology that breaks while connected to, or implanted in, a patient's body, may initiate a cascade of physiological changes that induce novel symptoms presenting as previously unseen clinical phenomena.1–3 The increasingly ubiquitous presence of digital systems in patient care means that clinical phenomena related to technological failures are likely to rise; yet, at present, practitioners are not trained to respond to cases emerging from digital causes (e.g. harm from hacked medical devices a.k.a ‘medjacking’).1–7
Technological failures can manifest as adverse clinical events 2 ; researchers have described respiratory compromise resulting from malfunctioning vagal nerve stimulators, 8 cardiac arrests due to faults in ventilators 9 and patient deaths from errors in diabetic pumps. 10 A seminal work from Dameff et al. described high-fidelity clinical simulations based on pathologies secondary to ‘hacked’ medical devices, highlighting the risks of compromised bedside infusion pumps, automated internal cardioverter defibrillators, and insulin delivery devices. 1 Since this research, real-life cases have been reported in the media, including an incident in the UK of a domestic assault enacted via a Bluetooth-connected insulin pump. 11
In our research on digital health pathologies, we go beyond the domain of medical cybersecurity, and expand our scope to encompass clinical presentations stemming from non-malicious digital events (e.g. unintentional faults in medical devices) and wider consumer technologies that can have health effects (e.g. complications of consumer smart implants). 3 In considering clinical cases relating to digital technology, we adopt the term ‘biotechnological syndromes’ from a recent systematic review of 372 cases that identified clinical phenomena emerging at the intersection of human health and digital technology. 3
In the digital fabric of our healthcare institutions, the pathways to digitally mediated harm are diverse and may include devices that directly interface with a patient (e.g. a ventilator) or seemingly distal systems (e.g. cloud-based remote care platforms).1–6 At the individual level, cybersecurity researchers have documented extensive vulnerabilities in implantable medical devices, which if exploited have the potential to incur severe morbidity including blindness, paralysis, pain and cardiac arrhythmia.12–17 Attacks on the wider medical infrastructure may also cause morbidity and mortality, exemplified by the death of a woman requiring transfer in Dusseldorf due to a hospital cyberattack.18–20
Biotechnological syndromes emerge at the intersection of the traditionally siloed domains of clinical medicine, engineering, computer science and cybersecurity. 3 Thus, understanding how to treat the patient experiencing end-point symptoms of these multifaceted pathologies requires a clinical and technical lens, and a skillset that extends beyond traditional medical training.1–3 Currently, a great deal of research into digital health harms focuses on the role of manufacturers and regulatory agencies in improving device oversight, however these interventions can be slow to implement, and need to be paired with changes on the floor of clinical practice.21–28 Given that research has demonstrated that 96.6% of reports on regulatory platforms such as FDA MAUDE database are made by manufacturers (as opposed to treating clinicians), it is perhaps unsurprising that a clinical perspective has been lacking when designing a response to these events. 23 In our research we utilise simulation methods in order to centre the clinician–patient interaction during digital events, and examine the gaps in knowledge, training and resources that impede effective medical care during these scenarios.
Simulation-based research (SBR) has been described as an effective tool for improving quality and safety in healthcare by replicating real-world situations.29–34 The improved realism of simulation techniques (e.g. through manikins, actors and props) has facilitated the creation of increasingly complex clinical simulation environments, capable of mimicking real-world scenarios and unearthing real-time challenges that occur during difficult clinical scenarios and in complex patient care.29–32 Researchers have detailed the benefit of simulations for observing healthcare teams’ responses to unanticipated crises as they unfold, which is not possible in actual patient care situations and thus offers a powerful tool for improving patient safety and ensuring readiness for rare events (e.g. bioterrorism attacks, natural disasters).31–33 Furthermore, simulation has been demonstrated as an effective way for upskilling practitioners in neglected areas of clinical medicine (e.g. transgender healthcare) and in response to changing population health needs (e.g. palliative care).34,35 SBR can therefore be a means for identifying barriers to patient care and improving clinical practice in a safe setting.29–35 In our research, we explore the value of clinical simulation for improving patient care with regards to adverse digital events.
Our research uses simulation for two purposes:
To uncover the key barriers impeding effective patient care in digital clinical cases. To evaluate the value of simulation training for improving the ability of clinicians to respond to digital clinical cases.
We consider issues with both medical devices (including software and hardware issues) and consumer technology. For consumer technology, we focus on the potential implications of smart implants, and the patient safety implications of tracking devices that have been described in cases of technology-facilitated abuse (TFA).36–39 Across the four clinical simulation scenarios defined below, we explore the barriers to effective patient care, identify the key challenges faced by clinicians, and explore opportunities for improving future clinical care and ensuring the safe implementation of digital health innovations.
Methodology
Between 1 February 2023 and 1 July 2023, a series of four simulation sessions were run at three different hospital sites in the United Kingdom. Healthcare staff (nurses, doctors and clinical medical students) were recruited via National Health Service (NHS) Trust emails and advertisement flyers (n = 14). Ethics approval was obtained from University College London (UCL) Research Ethics Committee and all participants provided written informed consent for their contributions to be used in the research study. Sessions were run using simulation suites fitted with audiovisual (AV) capabilities and prototypes of devices were used throughout sessions (Figure 1). The full materials used throughout the simulation sessions, including the scenario descriptions, actor and doctor briefs, and participant feedback forms are provided in the appendices and Supplemental Material to facilitate the reproduction of our work.

A series of photos demonstrating the set-up of the clinical simulation. The actor and participant are presented in (a), an example of the props used (anatomical hand and RFID chips) in (b), while (c) and (d) demonstrate the use of the simulation suite's AV system. The simulated clinical encounter in (b) was streamed live to the participants in the audience next door (d).
Simulation scenarios
Table 1 provides a summary of the four clinical scenarios that were designed based on existing literature and published case reports.3,40 Detailed instructions for actors and sim facilitators were provided for each scenario to ensure consistency across sites. We focused on four different themes of technological failures (full details of scenarios are published in Appendix 1 for reuse):
Scenario 1: Software failure in a medical device. Scenario 2: Medical presentation of TFA. Scenario 3: Complication of consumer technology. Scenario 4: Hardware failure in a medical device.
Clinical simulation scenarios utilised across the four hospital sites (full details of case scenarios can be found in Appendix 1).

Details of study participants.
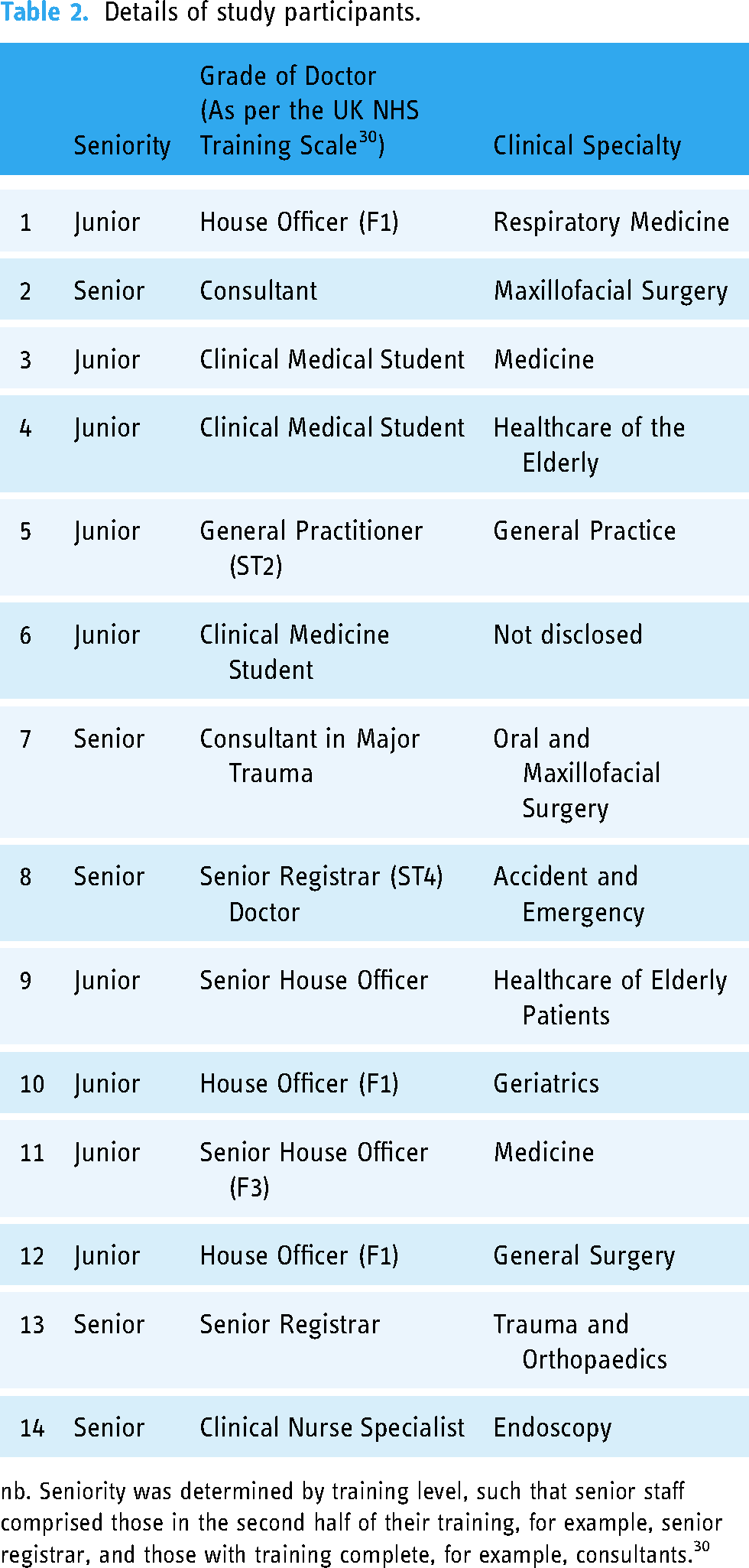
nb. Seniority was determined by training level, such that senior staff comprised those in the second half of their training, for example, senior registrar, and those with training complete, for example, consultants. 30
Participant feedback: subjective scores
Simulation sessions ran for half a day and began with an introductory lecture on the topic of biotechnological syndromes, with examples that differed from the clinical sims (e.g. seizures in virtual reality, harm from hacked insulin pumps).3,10,11 Participants were asked to reflect on cases in their own practice of patient illnesses stemming from digital technology and engage in group discussion on the topic.
Following this, the simulation sessions began. Participants completed pre-simulation and post-simulation survey questions that evaluated their understanding of syndromes related to technology, focusing on five dimensions of clinical skills: (a) awareness of syndromes, (b) knowledge of syndromes, (c) ability to investigate syndromes, (d) ability to treat syndromes and (e) ability to find appropriate resources. The feedback questionnaires were designed based on existing resources described in the SBR domain, where collecting a combination of qualitative and quantitative data has been demonstrated to facilitate the richest description of the participant's experience.29–33 We collected subjective qualitative scores for the five dimensions of clinical skills described above, which have previously been identified as essential elements of clinical care and provided space for participants to add free text comments (see Supplemental Material). To evaluate the significance of any change between the pre- and post-simulation scores, paired t-tests were carried out across the collected data. We produced Bonferroni corrected p-values to account for the multiple comparisons and evaluate for significance.
Participants were also asked to score how relevant they felt biotechnological syndromes were to their clinical practice. Finally, participants completed scenario-specific feedback, in order to facilitate a comparison of the challenges that differed between the various types of technology and clinical domains.
Examiner marking: objective scores
One participant took part in each clinical scenario, who was scored by one researcher acting as an invigilator. Mark schemes were written to reflect UK medical school ‘Objective structured clinical examinations’ (OSCEs), where each defined point covered an area of clinical competence (e.g. professionalism, clinical examination), for which the participant was awarded a binary score (0/1) depending on their performance and if they achieved the point (mark scheme example and scores provided in Appendix 2).
All participants watched each simulation scenario via AV streaming, which was followed by a 15-minute focused group debrief during which two scribes took extensive notes that were retrospectively analysed through a thematic analysis (Figure 1). The thematic analysis was performed by two members of the research team, who analysed themes that emerged in the qualitative data regarding the key areas of interest set out in the research aims (barriers to effective care and the value of simulation training).
Results
Our results are divided into (a) scenario-specific findings and (b) cross-scenario findings. The research participants were from a diverse range of disciplinary backgrounds (surgical, hospital medicine and community practice), ranging in seniority from clinical students to hospital consultants. The full details of participants are provided in Table 2, which also provides the breakdown by seniority. 30 Participants were categorised as either ‘juniors’ (ranging from clinical medical students to junior trainees) or ‘seniors’ (including speciality registrars, general practitioners [GPs] and consultants). 30
Scenario-specific findings
The scenarios were intentionally broad as we sought to identify challenges across a range of technologies. Scenario 1 and Scenario 4 both focused on medical devices, one with a software failure and one with a hardware fault. Scenario 2 and Scenario 3 focused on technologies outside of the medical device space, and tested the participants’ knowledge of emerging consumer devices and their ability to consider the wider health implications of these tools.
In the medical device scenarios, participants gave lower confidence scores for managing the software failure, compared to the hardware fault (3.5 vs. 5.2 juniors; 5.75 vs. 7.0 seniors), also reporting that medical education has prepared them less well for the software scenario (2.7 vs. 5.0 juniors, 2.0 vs. 5.25 seniors; Figure 2). One participant described this stating ‘for some reason, I find hardware issues easier to understand/more covered in our training than software’, another shared ‘This case may be less challenging to clinicians as it is a hardware problem which is easier to spot with imaging’. Hardware faults in technology can be picked up on existing radiological modalities and can be understood through the anatomical teaching that medics receive. However, software faults and changes in device settings are defined by shifts in voltage, electrical stimulation and may be influenced by electromagnetic interference, all of which are mechanisms that do not form part of healthcare training.
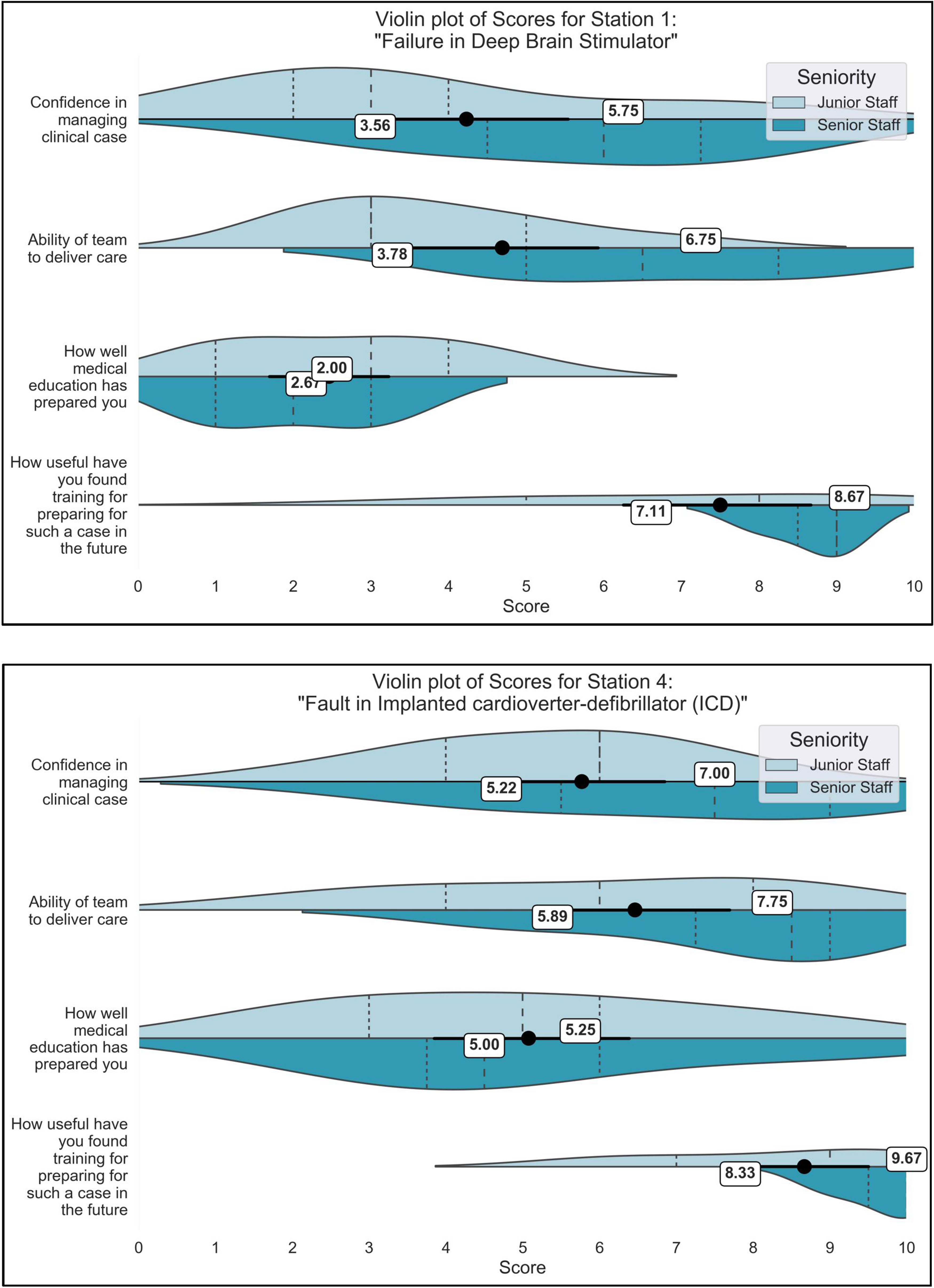
Comparison of Scenario 1 (Software failure in Deep Brain Stimulator) and Scenario 4 (Hardware failure in Implanted cardioverter-defibrillator). Violin plots are split by seniority, such that junior participants (clinical medical students to junior registrars/ST3) and senior participants are visualised side-by-side on plots, with mean scores for each group provided alongside. Significant differences between the junior and senior doctors are marked with asterisks (*).
Figure 3 provides the participant feedback scores for the scenarios based on technology outside of medical device regulation – Scenario 2 (technology-facilitated abuse) and Scenario 3 (Adolescent with RFID chip). In the tech-abuse scenario, senior participants reported higher confidence in themselves to treat the patient (7.3 seniors vs. 4.0 juniors) and in their teams (7.3 seniors vs. 6.0 juniors) in managing the case. In Scenario 3 with the RFID chip, the gap narrowed for both individual confidence (5.3 juniors vs. 6.8 seniors) and confidence in the team (5.6 juniors, 7.0 seniors). Age and experience may play a role here, such that the more junior participants may be more familiar with the consumer smart implants included in Scenario 3.

Comparison of survey scores for scenarios involving consumer technologies – Scenario 2 (technology-facilitated abuse involving spyware and GPS tracking) and Scenario 3 (RFID chip implants). Mean scores across all participants (n = 14) are provided for junior and senior doctors separately, for each survey question. Significant differences between the junior and senior doctors are marked with asterisks (*).
Examples provided by participants
Participants provided examples of biotechnological syndromes they had encountered within their own practice throughout the group discussions and individual feedback, which have been collated in Table 3.
Examples of biotechnological syndromes provided by participants during group discussions and debriefs.

Cross-scenario findings: barriers to care
Our simulations revealed the challenges that clinicians faced caring for these patients in real-time, aggregated under themes in Table 4. We first consider factors that prevented the identification of the technology as the cause (delays to diagnosis), and then the factors impeding care once the diagnosis was made (delays to clinical management).
Causes for delay in diagnosis.
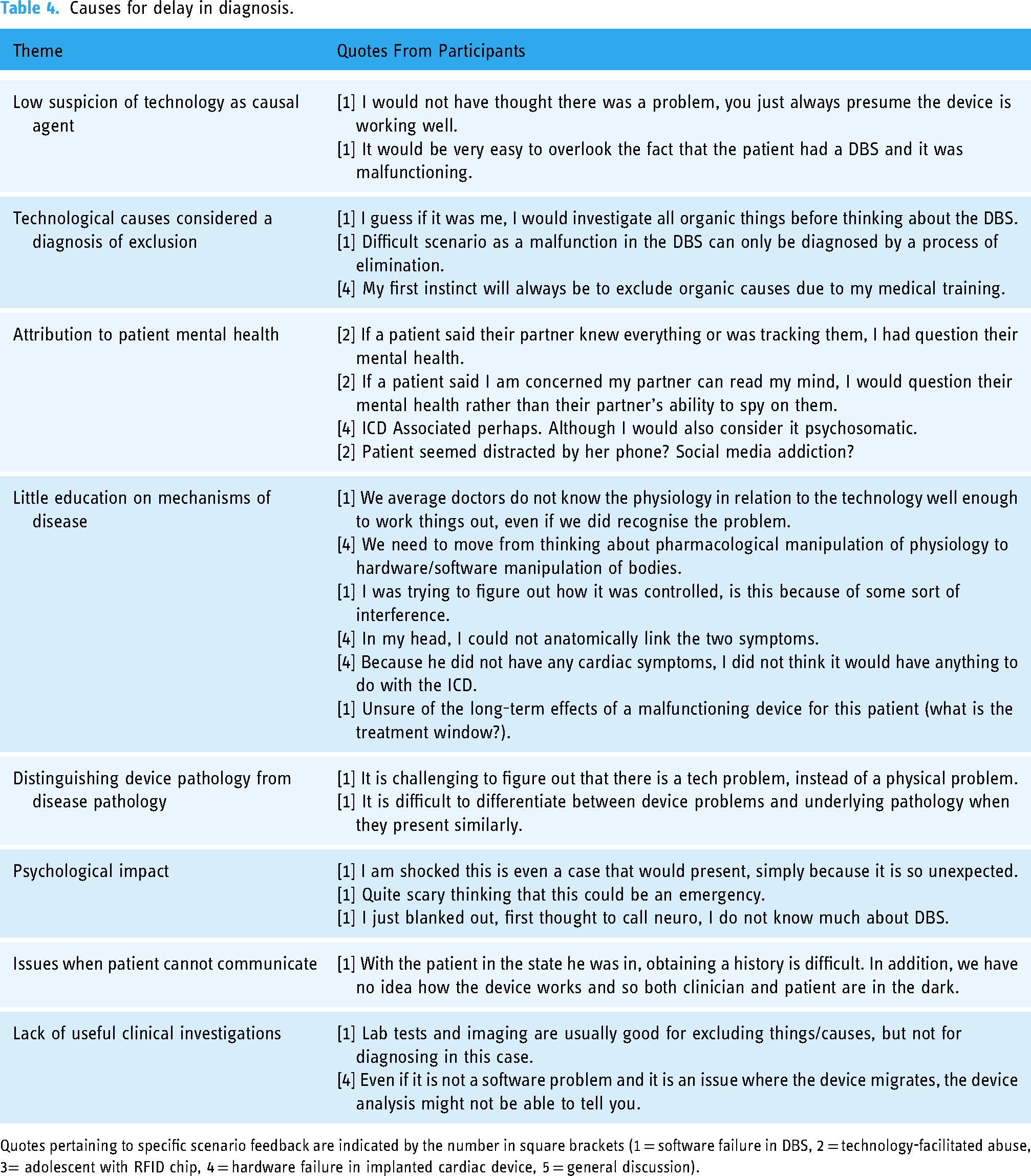
Quotes pertaining to specific scenario feedback are indicated by the number in square brackets (1 = software failure in DBS, 2 = technology-facilitated abuse, 3= adolescent with RFID chip, 4 = hardware failure in implanted cardiac device, 5 = general discussion).
Barriers to effective treatment.

Quotes pertaining to specific scenario feedback are indicated by the number in square brackets (1 = software failure in DBS, 2 = technology-facilitated abuse, 3 = adolescent with RFID chip, 4 = hardware failure in implanted cardiac device, 5 = general discussion).
As demonstrated by Table 4, the low suspicion for digital failures led to delay in clinical diagnosis and meant that biotechnological syndromes were approached as a diagnosis of exclusion. In the case of medical devices, participants shared that ‘you just always presume the device is working well’, prohibiting early identification of device issues. In addition, the low suspicion regarding malicious consumer technologies meant that participants did not suspect issues of GPS tracking or eavesdropping in the tech-abuse case. Participants shared that ‘tracking/monitoring devices are very subtle, so easy to miss’ and that the main challenges in the case were ensuring patient safety in the context of malicious surveillance and logistical issues of separating the victim from their devices. Across the four sessions, only one participant removed the devices from the consultation to account for potential listening technologies, an intervention that many felt to be useful but that would be challenging in clinical environments – ‘What's the safest thing to do with a patient's belongings/devices if you think they have eavesdropping technology? Should you move them? Or use white noise/opera music?’
Participants shared difficulties in understanding the mechanisms of disease that underpinned these syndromes, such as in the case of the cardiac device hardware fault manifesting in limb signs from a brachial plexus injury (Scenario 4). The difficulty in connecting seemingly unrelated physiological processes that were mediated by technology, challenged participants in the scenarios of software and hardware failures. Lack of pathophysiological understanding may also have influenced the participant's tendency to attribute symptoms to mental health, such as attributing the device complication to psychosomatic features in Scenario 4. In the tech-abuse scenario, participants reported that they had been more likely to raise concerns regarding a patient's mental health than suspect signs of abuse, with participants linking the increased phone activity to social media addiction, and participants concerns about tracking to their mental health.
The participants described their initial shock when encountering these patients, and a sense of bewilderment at experiencing a clinical presentation that fell outside of their training. Lastly, once participants sought to investigate issues related to the device, they highlighted that their usual repertoire of clinical investigations (e.g. laboratory testing) may be redundant in these cases where syndromes act along different pathophysiological pathways.
Cross-scenario findings: barriers to effective clinical management
Once participants identified the technology as the source of the problem, further challenges emerged when forming a management plan (Table 5). In particular, both junior and senior staff felt that their team would lack their domain knowledge to treat these cases (Table 5). The gap in skills amongst healthcare staff was felt to be reinforced by a lack of required technical expertise in clinical settings, such as the need for an on-call programmer equipped to reconfigure devices.
When it came to offering treatment in the clinical scenarios, participants were often hesitant due to their uncertainty of how traditional medical treatments would interact with technological processes (e.g. will pharmacological treatments work for pain stemming from a misfiring implanted electrode?). In addition, participants did not want to offer advice on the RFID chip in Scenario 3, due to uncertainty around the technology. Lastly, clinicians were unsure on the pathways for reporting adverse digital events, stating ‘Whose responsibility is it to report these things? The clinician?’
Suggested interventions for improving patient care
Participants suggested interventions that would have eased the clinical process in these cases, including medical education initiatives (e.g. integrating digital simulations into medical student exams) and the development of clinical support platforms (e.g. a helpline for emergency device failures; see Table 6). The participants also suggested tailored assessments for digital technologies in the clinical workflow, stating that ‘It would be useful to be able to check the integrity of such devices’.
Suggested interventions for improving future practice provided by participants throughout simulations.

Examiner marks and value of clinical simulation training
Simulation training was found to be an effective technique for improving participants’ perceived clinical confidence and knowledge of biotechnological syndromes (Figure 4). Paired t-tests (one-tailed) were performed for all survey scores presented in Figure 4, and across all five questions, there were statistically significant improvements (p < 0.01). These findings are supported by the scores of ‘usefulness’ presented in Figure 2, in which participants consistently rated the usefulness of the session over 8–9/10. Further details on participant mark schemes are available in Appendix 2, which demonstrate participants scored lowest on aspects related to technology in each scenario including (a) managing the digital problem, (b) addresses technical questions and (c) discussion of technology with the patient.

Comparison of participant survey scores before and after clinical simulation, across factors of clinical awareness, knowledge, ability to investigate/treat/find resources and perceived relevance of content (paired t-tests of pre–post scores gives p < 0.01 for all categories, indicated in the Figure with asterisks [*] to illustrate significance).
Discussion
The advance of digital health technologies has created new forms of pathology which are not captured in current medical curricula or clinical guidelines. The growing gap between potential patient pathology and clinical competency is an urgent challenge that must be addressed to ensure patients receive the care they need when they suffer from digital health harms. Our research provides insights into the gaps in current medical domain knowledge, clinical practice and hospital guidelines, and we offer recommendations for practitioners on the frontline and those working in regulation, policy and enterprise.
Recommendation1: development of resources
Participants described the need for national helplines and a database of biotechnological pathologies, in particular, an app that lists the potential side-effects of all medical devices would be useful. Such a helpline has been proposed before, with the database requiring an interface that is easily accessible and extracts only the most clinically relevant information regarding device failures, so as to avoid clinicians needing to search through individual medical device manuals which are often verbose and dense. 41 The participants also suggested integrating an evaluation of digital devices into the clinical workflow, to ensure digital errors were picked up at the point of patient clerking. These suggestions speak to the wider research on the effective integration of digital technologies within the NHS, from emergency medicine to digital pathology.21,42 In addition, clinicians requested safeguarding guidelines specifically tailored to TFA, that could provide standardised measures for protecting patients in suspected cases of harm.43,44 These guidelines may introduce generic measures such as removing potentially ‘listening’ devices from a consultation room and the use of Faraday bags if there are concerns of geolocation tracking.
Recommendation 2: future research into biotechnological syndromes
When faced with a digital pathology, participants consistently reported ruling out ‘organic’ causes first and raised concerns about not having the tests to identify biotechnological issues. If participants were trained in the halo signs of biotechnological issues, such as respiratory symptoms being indicative of device malfunction in VNS patients, these biotechnological diagnoses would not need to wait for exclusion. To improve the diagnostic capability of clinicians, further research is needed into the onset symptoms and signs of these syndromes. In addition, there is a lack of research on the effective symptomatic management of physiological issues associated with technological complaints, for example, are pharmacological interventions as effective for pain stemming from faults in implantables (e.g. a malfunctioning deep brain stimulator [DBS] 40 ). The growing research that explores the benefits of new medical devices needs to be paired with an equally deep investigation into their potential long-term healthcare implications, the novel pathological pathways that emerge with new implants and the treatments that are effective when they go wrong.
Recommendation 3: education, clinical training and simulation
The scientific language of the life sciences and the physical sciences has historically developed along separate trajectories. However now that human patients depend on physical-digital devices, understanding their health needs requires an interdisciplinary knowledge that intersects these domains. Similarly to the means by which pharmacology became embedded in medical school curricula, clinical students would benefit from basic training in medical technology, covering the foundational components of these tools and the forces by which they function and may fail. AI researchers have advocated for the integration of AI competency training for clinicians in medical schools, we propose that this should be expanded to encompass the wider digital health issues we have discussed including topics such as hardware and software failures, and cybersecurity exploits. 43 Further, we have demonstrated the value of utilising simulation-based methods for this purpose. Our results demonstrate significant improvements in the awareness and confidence of clinicians pertaining to these cases following a half-day exposure to digital health simulations.
Health equity
The burden of digital health complaints may differ between demographic groups. In discussions of TFA, participants raised concerns that some patients may be more at risk including patients with dementia who may be subject to elder abuse and young people at risk of knife crime and gang violence.36–39 Participants also shared concerns about the RFID chip technology that could be imposed on groups without consent, and given the increased investment in smart implant technologies with growing functional capabilities, these may hold significant threats to privacy and bodily autonomy in the future.
Urban–rural health disparities came up as an issue, as participants identified that non-specialist centres were less likely to have required expertise, for example, an out-of-hours implantable cardioverter defibrillator (ICD) expert. Our increasing reliance on digital infrastructure may exacerbate urban–rural health disparities. Furthermore, we noted that clinicians had a tendency to ascribe confusing presentations to psychosomatic issues and mental health complaints. Previous research has demonstrated that these biases often fall along racial and gender lines, such that there is often a delay in investigating and diagnosing illnesses affecting women, and racial and ethnic minorities.44–50 The announcement from the UK Government to deliver an independent review into the equity of medical devices is a welcome initiative for exploring these issues, and further research into these issues specifically within biotechnological syndromes would be welcome. 50
Limitations
Our study is subject to selection bias given that the participants volunteered for the session based on our recruitment email, and we are limited by a low sample size (n = 14). Furthermore, all sites were based in metropolitan areas and therefore we cannot account for differences in urban to rural hospital settings. Lastly, our small sample size (n = 14) restricted our statistical analysis. Identifying significant differences between the junior and senior participants was particularly challenging, as this required stratification into even small subgroups. Further research that replicates our work with larger sample sizes would be beneficial for fully evaluating these effects.
Conclusion
The gap between forms of patient pathology, and clinical understanding, is growing. The new digital technologies woven into both our society and our bodies are changing the way in which we experience health and disease, creating new clinical pictures that require tailored clinical support built from a foundation of digital understanding. In our research we have demonstrated the value of SBR for uncovering barriers to care and latent threats to patient safety that exist in digital clinical scenarios. In addition to demonstrating the value of clinical simulation training, we provide a series of recommendations for clinical practice for improving patient care and ensuring institutional readiness for the rising tide of digital health pathologies.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076241247939 - Supplemental material for Simulation-based research for digital health pathologies: A multi-site mixed-methods study
Supplemental material, sj-docx-1-dhj-10.1177_20552076241247939 for Simulation-based research for digital health pathologies: A multi-site mixed-methods study by Isabel Straw, Joanna Dobbin, Demelza Luna-Reaver and Leonie Tanczer in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076241247939 - Supplemental material for Simulation-based research for digital health pathologies: A multi-site mixed-methods study
Supplemental material, sj-docx-2-dhj-10.1177_20552076241247939 for Simulation-based research for digital health pathologies: A multi-site mixed-methods study by Isabel Straw, Joanna Dobbin, Demelza Luna-Reaver and Leonie Tanczer in DIGITAL HEALTH
Footnotes
Acknowledgements
N/A
Contributorship
IS researched the literature and conceived the study. All authors were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. IS wrote the first draft of the manuscript, all authors reviewed and edited the manuscript and approved the final version.
Declaration of Conflicting Interests
IS is the director of bleepDigital, a non-profit dedicated to technology and medical education. JD, LT and DLR declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was obtained from University College London and all participants provided written informed consent for their contributions to be used in the research study (UCL/CSREC/R/18).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by UK Research and Innovation (UKRI Grant Reference Number EP/S021612/1) who funded the doctoral research of the corresponding author.
Guarantor
IS.
Supplemental material
Supplemental material for this article is available online.
Appendix 1. Clinical scenarios delivered at each site,with references to case reports from which medical information was derived.
Appendix 2. Mark scheme used by examiners during each clinical scenario. Mark schemes were completed at each NHS hospital site,and averages were taken across the four sites to give the average scores for each mark scheme component that is presented in the research paper.

| Binary Score of 0/1 (1 Indicates Participant Achieved Point) | |||||
|---|---|---|---|---|---|
| Mark Scheme Component | Marking Detail | Scenario 1: Software Malfunction in DBS | Scenario 2: Technology-Facilitated Abuse (TFA) | Scenario 3: Adolescent with RFID Chip | Scenario 4: Hardware Fault in ICD |
| Average score across four sites (standard deviation) | Average score across four sites (standard deviation) | Average score across four sites (standard deviation) | Average score across four sites (standard deviation) | ||
| Professionalism | Participant treats the patient professionally and with respect | 1.0 (0.0) | 1.0 (0.0) | 1.0 (0.0) | 0.6 (0.0) |
| History taking | Participant asks appropriate questions and elicits history from the patient | 1.0 (0.0) | 0.5 (0.5) | 1.0 (0.0) | 0.6 (0.0) |
| Clinical examination | Participant undertakes relevant examination of the patient | 0.3 (0.4) | 0.8 (0.4) | 0.5 (0.5) | 0.5 (0.4) |
| Identification of technology | Participant identifies the presence of the digital technology | 1.0 (0.0) | 0.5 (0.5) | 1.0 (0.0) | 0.6 (0.0) |
| Clinical diagnosis | Participant identifies the digital technology as the cause of the patient illness | 0.5 (0.5) | 0.0 (0.0) | 1.0 (0.0) | 0.4 (0.4) |
| Discussion of technology | Participant discusses the underlying issues with the digital technology (i.e. EM interference) | 0.3 (0.4) | 0.0 (0.0) | 1.0 (0.0) | 0.3 (0.0) |
| Treats medical symptoms | Participant provides effective medical care (e.g. to treat symptoms) | 0.0 (0.0) | 1.0 (0.0) | 1.0 (0.0) | 0.4 (0.0) |
| Manages digital problem | Participant provides care to resolve the issue with the digital technology (i.e. contacting the manufacturer/programmer) | 0.5 (0.5) | 0.0 (0.0) | 0.3 (0.4) | 0.3 (0.0) |
| Addresses medical questions | Participant is able to address the patients’ medical questions | 0.5 (0.5) | 0.8 (0.4) | 0.0 (0.0) | 0.4 (0.4) |
| Addresses technical questions | Participant is able to address patient's questions about the digital technology | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) |
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
