Abstract
Objective
Fibromyalgia (FM) is a prevalent pain syndrome with significant healthcare and societal costs. The aim of the SMART-FM-SP study is to determine the effectiveness, cost-utility, and physiological effects in patients with FM of a digital intervention (STANZA®) currently marketed in the United States, which delivers smartphone-based, fully self-guided Acceptance and Commitment Therapy (Digital ACT) for treating FM-related symptoms.
Methods
A single-site, parallel-group, superiority, randomized controlled trial (RCT) will be conducted, including a total of 360 adults diagnosed with FM. Individuals will be randomly allocated (1:1:1) to treatment as usual (TAU), to TAU plus 12 weeks of treatment with Digital ACT, or to TAU plus 12 weeks of treatment with digital symptom tracking (i.e. FibroST). Participants will be assessed at baseline, post-treatment, and 6-month follow-up. An intention-to-treat analysis using linear mixed models will be computed to analyze the effects of Digital ACT on functional impairment (primary outcome), as measured by the Fibromyalgia Impact Questionnaire Revised at 6 months from the inception of the treatment. Secondary outcomes include impression of change, symptoms of distress, pain catastrophising, quality of life, cost-utility, and selected biomarkers (cortisol and cortisone, immune-inflammatory markers, and FKBP5 gene polymorphisms). The role of ACT-related processes of change will be tested with path analyses.
Conclusions
This study is the first RCT that tests Digital ACT for Spanish patients with FM. Results will be important not only for patients and clinicians, but also for policy makers by examining the cost-utility of the app in a public healthcare context.
Keywords
Introduction
Fibromyalgia (FM) is a chronic condition characterized by widespread musculoskeletal pain, fatigue, stiffness, cognitive impairments, sleep disturbances, and emotional distress.1,2 This condition exhibits a high prevalence in the general population, affecting around 1.8% of people worldwide. 3 Recent advances in understanding FM have highlighted its association with central sensitization, a concept encompassing an amplification of neural signaling within the central nervous system (CNS) leading to pain hypersensitivity. 4 Central sensitization, documented in various rheumatological pain conditions, including FM as flagship condition, represents a discrepancy between the extent of tissue damage and the magnitude of pain and disability, thereby posing a diagnostic challenge. 4
The role of central sensitization in FM underscores the need for a nuanced understanding of pain processing perturbations. While peripheral factors such as tissue damage and inflammation might not fully account for the pain severity in FM, their interplay with central mechanisms is crucial. The concept of nociplastic pain, introduced to describe pain arising from altered sensory processing in the periphery and CNS, resonates with FM clinical presentation. 5 Nociplastic pain manifests as multifocal pain, more widespread or intense than expected based on identifiable tissue or nerve damage, and is often accompanied by fatigue, sleep disturbances, cognitive issues, and mood problems. 5 Given this complex etiology, it is also pertinent to address the role of immune pathways in FM pathogenesis and maintenance. Evidence suggests an imbalance in pro-inflammatory cytokines in FM, potentially leading to chronic low-intensity inflammation. 6 This aspect aligns with the broader understanding of the roots of FM as multifactorial, where both central and peripheral factors contribute to its manifestation and persistence.
Regarding the pharmacological management of FM-related symptoms, there are three drugs approved by the U.S. Food and Drug Administration (FDA) for the treatment of FM (pregabalin, duloxetine, and milnacipran). However, European regulatory authorities rejected the indication of these three drugs due to small effect sizes in various studies and associated adverse effects.7–9 In contrast, numerous non-pharmacological interventions, such as physical exercise 10 and electrical neuromodulation 11 among others, have shown significant positive effects, including psychological, neuroendocrine, and immunological changes, in pain-related conditions, including FM. 12 Among these treatments, modern forms of cognitive behavioral therapy, including mindfulness- and acceptance-based interventions, have shown evidence of effectiveness in patients with FM. A recent meta-analysis of 21 randomized controlled trials (RCTs) demonstrated that Acceptance and Commitment Therapy (ACT) improved anxiety, depression, and pain in patients with pain sensitization syndromes, such as FM, showing superior outcomes compared to non-intervention conditions (i.e. waiting lists), as well as pharmacological and psychoeducational interventions. 13 In addition, recent systematic and narrative reviews have provided evidence suggesting that ACT holds promise in terms of cost-utility for managing chronic pain in general, and specifically for FM.14,15
ACT aims at reducing the dominance of pain in a person's daily life through increasing psychological flexibility. Psychological flexibility is “the ability to contact the present moment more fully as a conscious human being, and to change or persist in behavior when doing so serves valued ends.” 16 In turn, the aim of increasing psychological flexibility is not symptom reduction but improved daily functioning. 17 Despite the demonstrated benefits of ACT for patients with FM, access to this therapy is often limited due to barriers such as a scarcity of trained providers, limited referral pathways, geographical distance from treatment centers, inadequate insurance reimbursement, and high costs. 18 Offering a self-guided digital ACT therapeutic could increase the accessibility of this non-pharmacological approach for the individuals with FM. A self-guided digital ACT therapeutic could empower patients to take an active role in their healthcare and in managing FM in particular. Recently, a smartphone-based, self-guided, prescription, digital therapeutic application (STANZA®, Swing Therapeutics, San Francisco, CA, USA) has been developed in English language and received market clearance by the United States Food and Drug Administration (US FDA) for the treatment of FM symptoms in adult patients. Its content was adapted from a web-based ACT program for FM validated by researchers affiliated at the University of Manitoba. 19 Catella and collaborators 20 examined the feasibility and preliminary effectiveness of an earlier generation of STANZA® compared to an active symptom monitoring condition, in patients with FM, the between-arm effect size for the change from baseline to 3 months in the Fibromyalgia Impact Questionnaire Revised (FIQR) was 0.44 for the intention-to-treat population and 0.54 for the per-protocol population, corresponding to least-squares mean differences of −5.7 (SE, 3.16; 95% CI, −11.9 to 0.6; p = .074) and −6.8 (SE, 3.30; 95% CI, −13.4 to −0.3; p = .042) for the ITT (intention-to-treat) and PP (per-protocol) populations, respectively. 20 High engagement and low attrition were seen in both arms with no treatment-related adverse events.
The objectives of the SMART-FM-SP study are (a) to analyze the effectiveness of a digital ACT delivered through the app STANZA® (hereafter Digital ACT) compared to a digital symptom tracker (hereafter FibroST), in each case added to treatment as usual (TAU), and compared with TAU alone, for Spanish patients diagnosed of FM; (b) to examine the cost-utility of Digital ACT from a healthcare and societal perspective in Spain; and (c) to measure a set of immune-inflammatory, neuroendocrine and genetic biomarkers alongside the RCT to investigate physiological markers of the impact of Digital ACT and to identify potential mediators of treatment response.
Methods
Study design
This RCT protocol was registered in ClinicalTrials.gov (NCT05962437) and it has been developed following the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guidelines. 21 SMART-FM-SP is a single-site, parallel-group (1:1:1 allocation ratio), superiority, RCT that includes three study arms: TAU, TAU + Digital ACT, and TAU + FibroST. While all three arms receive usual care, Digital ACT and FibroST serve as co-adjuvant treatments to those provided by the Spanish Health System. Due to the nature of behavioral interventions and psychological therapies, it is not possible to provide a true sham or placebo control for evaluation of new digital psychological tools. As a result, the SMART-FM-SP study will use a “blind to hypothesis” design, in which prospective participants will be informed they would be randomized to one of two potentially effective therapies for FM management. For ethical reasons, all study participants who are randomly assigned to TAU alone will be offered the use of the Digital ACT tool at the end of the study Figure 1.

Flowchart of the SMART-FM-SP study.
Treatment arms
TAU. In Spain, usual care of FM is mainly carried out by general practitioners and rheumatologists. Participants randomized to this arm will not receive additional active treatment over the study period but will continue with their regular pattern of medication (if any). Usual pharmacological management of FM typically includes analgesics, anxiolytics, opioids, antidepressants, and/or anti-inflammatories, recommendations for practicing aerobic exercise regularly plus some minimal explanation of the main characteristics of the syndrome and its potential etiology. Considering the recommendations of the American College of Sports Medicine (ACSM), 22 the following exercise regimen is proposed in our hospital (40 min once or twice a week): Warm-up (5’) - activation and mobility exercises; Therapeutic exercise (25’): moderate aerobic-cardiovascular (marching/walking) and muscle strengthening (upper body and lower body) exercises combined with some balance and coordination exercises performed in a playful manner, where the level of difficulty gradually increases; Cooling-down (10’) - flexibility and relaxation exercises.
TAU + Digital ACT. The participants will receive TAU with the Digital ACT application, which consists of 41 structured ACT lessons, incorporating mindfulness practices and daily skills training exercises to facilitate behavior change and promote gradual improvement in daily activities. The core content was designed to be completed within 8 weeks, with a 4-week maintenance period thereafter to strengthen skills. ACT is delivered in 15- to 20-min daily sessions over the course of a 12-week program without the involvement of healthcare providers. The program consists of interactive training materials that promote development of ACT skills which are reinforced experientially via mindfulness, relaxation exercises, and values exploration and identification. Values-based assignments follow each session to assist patients in incorporating ACT skills into their daily lives. Additionally, Digital ACT offers conventional condition management activities, including self-guided physical exercise and pacing daily activities via a stepwise gradual approach.
TAU + FibroST. The participants will receive TAU with a digital active control application (Daily Symptom Tracker), which is implemented to control for study and treatment engagement, expectations, and healthcare provider interaction biases. Built on the same platform as Digital ACT, FibroST enables patients tracking their symptoms and functioning. Symptom tracking is a commonly used approach in chronic pain management, and several validated symptom tracking apps have been developed. 23 FibroST also provides access to educational materials related to FM but does not include any psychotherapy or involvement of healthcare professionals. By using FibroST as an active control, the study aims to mitigate biases related to expectations and engagement, which are often present in chronic pain studies utilizing passive comparison conditions.
Regarding adherence, both digital applications (Digital and FibroST) are programed to register participants’ use (time connected, number of completed sessions…). Participants in both arms are asked to engage with their assigned treatment 5−7 days per week for 12 weeks, with completion of the core program of 42 sessions considered full compliance Figure 2.
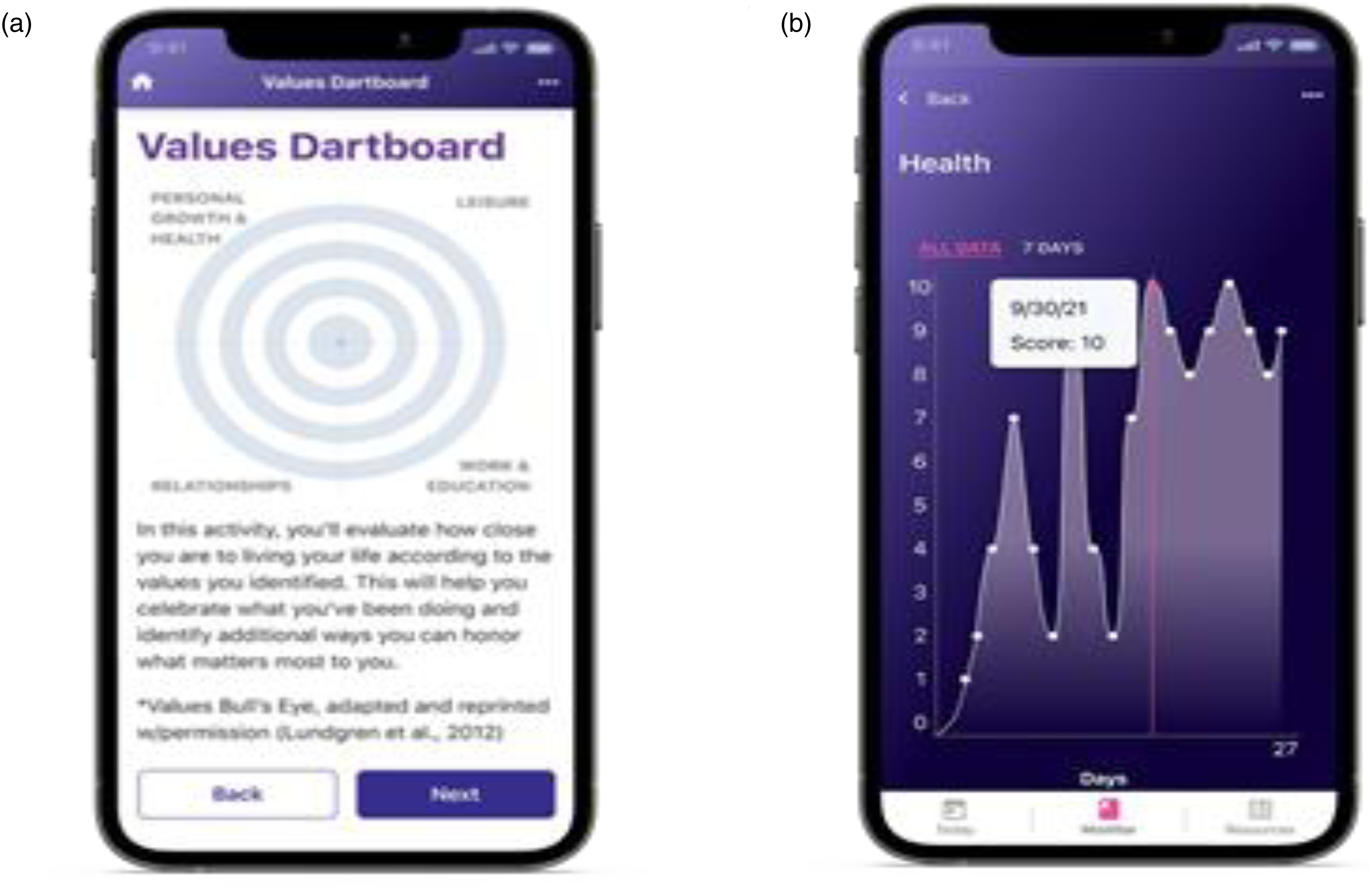
Treatment application design. (a) Screenshot of Digital ACT showing one of the ACT exercises included in the self-guided program (English version). (b) Screenshot of the FibroST active control showing the daily symptom score monitoring display. ACT: Acceptance and Commitment Therapy
Study setting and participants
Patients diagnosed with FM by specialists of the Rheumatology Service at the Central Sensitivity Syndromes Unit (CSSU) of Vall d'Hebron University Hospital are eligible if they meet the full enrollment criteria. Study participants will be allowed to continue ongoing medications for FM and comorbid conditions provided the dose and regimen were stable for 30 days prior to screening and will remain stable throughout participation in the trial.
Inclusion criteria. Adult patients aged 18–70 years, diagnosed with FM according to the 2016 Revisions to the 2010/2011 FM diagnostic criteria. 24 Additionally, patients must have a FIQR 25 total score within the range of 35–80 at baseline. They should agree to maintain their current pain treatment throughout the study, own and use a smartphone compatible with the study app with an operating system iOS 12 and above (Apple, USA) or Android 7.0 and above (Google, USA), have daily internet access on smartphone, be comfortable with downloading and using smartphone apps, and have a proficient understanding of Spanish.
Exclusion criteria. History of a severe mental disorder (i.e. psychotic disorder, any disorder with suicidal behavior or ideation in the preceding year, history of significant alcohol and/or drug abuse or dependency, etc.), presence of severe medical conditions (i.e. cancer) that could endanger the patient or interfere with the patient's ability to comply with the study protocol. Patients will be also excluded if they are undergoing psychotherapy, currently or in the past 12 months. Exclusion criteria for biomarkers substudy (50% of patients in each study arm): Men, patients with major organ dysfunction or uncontrolled systemic disease, fever (> 38°C) or infection within the last 2 weeks, consumption of more than eight units of caffeine per day, smoking more than five cigarettes per day and having hair length less than 3 cm, recent physical trauma, not being vaccinated during the last week, needle phobia, using oral or local corticosteroids or any anti-cytokine biologic drugs or oral contraceptives.
Sample size
This RCT will compare between TAU plus Digital ACT, TAU plus Fibro-ST, and TAU alone. Taking into account the results from a recent feasibility study carried out by Catella and collaborators, 20 in the present RCT, a sample size of 100 patients/arm and an effect size of 0.50 will provide more than 90% power to detect a difference between active treatments in the FIQR total score analysis (primary outcome) at post-treatment. Considering a possible attrition rate of 20%, we aim to enroll a total of approximately 360 participants in the study to ensure an adequate sample size.
Participants’ recruitment and informed consent
A list of potential participants will be provided to the study team at the Vall d'Hebron University Hospital. Clinicians will conduct screening by telephone with the support of a research assistant. Self-report data will be collected via the Qualtrics software platform (Qualtrics International Incorporated, Seattle, Washington, USA) throughout the 6-month study. Except for the screening, there will not be no contact between the research team and the participants at any assessment time point (baseline, post-treatment, follow-up).
Participants’ identification and randomization
Random assignment of participants to study arms will be executed after baseline assessments as recommended by the CONSORT guidelines. 26 The randomization process will be planned and executed by a statistician with no involvement in screening, enrollment, or treatment processes. After completing the online baseline assessment, each study participant will receive a unique personal code and be allocated into a study arm using a randomization program. To ensure balanced groups while considering the eligibility criteria for the biomarkers substudy, computer-generated randomization will employ a permuted block design. The randomization list will be securely held by the clinical trials committee of Vall d’Hebron University Hospital throughout the entire duration of the RCT. It will be stored in an encrypted file on a password-protected computer in the office of the clinical trials supervisor to ensure allocation concealment. Patients will be informed of their group allocation through telephone calls. Subsequent online assessments will be conducted at the end of the 12-week treatment period (post-treatment) and at the 6-month follow-up. The same procedure as for the baseline assessment will be followed to obtain biological samples for the post-treatment assessment in the biomarker substudy participants.
Study measures
We obtained permission to use all the study measures described below from the respective developers or copyright holders.
Sociodemographic-clinical features and screening
Sociodemographic questionnaire. Data including gender, age, marital status, living arrangements, education, and employment status will be collected with a structured questionnaire.
Clinical data interview. Information regarding the history and duration of FM symptoms, comorbidity with other diagnosed physical conditions or mental disorders will be collected in a brief interview.
The Fibromyalgia Survey Diagnostic Criteria (FSDC).27,28 The FSDC is a 6-item measure designed to assess FM symptoms based on the ACR 2016 criteria. It consists of 2 subscales: the Widespread Pain Index, which is used to identify the presence of pain in 19 body areas in the last week, and the Symptom Severity Scale, which assesses the severity of the three main FM symptoms (fatigue, “fibrofog” and waking up tired) as well as three additional symptoms (pain in the lower stomach, depression and headache). The total FSDC score ranges from 0 to 31, where higher values indicate greater FM severity. The Spanish version of the FSDC had a Cronbach's α ranging from 0.68 to 0.85, whereas the Kappa coefficient computed to assess inter-criteria agreement for FM diagnosis was 0.73.28
The Columbia-Suicide Severity Rating Scale (C-SSRS). 29 It is a 20-item clinician-administered scale that measures suicidal thoughts and behaviors which are rated for the past 30 days, for the past three months and lifetime. Suicidal ideation severity is evaluated by means of 5 yes/no items summarized in the single item reflecting the most severe level of ideation, scored 0–5 where 0 corresponds to no suicidal ideation, 1 to a wish to die and 5 to active suicidal ideation with a specific plan and intent to act. The intensity of suicidal ideation is assessed only if some degree of severity is reported. The Spanish version is reliable and discriminates between patients with and without suicide attempts. 30
Primary outcome
The FIQR. 25 The FIQR includes 21 items that are answered on a 0–10 numerical scale in which higher scores indicate greater impact or severity of FM symptoms. It is focused over the past 7 days and covers three domains: physical impairment, overall impact, and symptom severity. The item content includes various aspects such as pain, energy levels, stiffness, sleep quality, depression, memory problems, anxiety, sensitivity to touch, balance issues, and heightened sensitivity to noises, lights, smells, or temperatures. The total FIQR score is calculated by summing the scores of the three subscales, resulting in a range of 0 to 100. Higher scores indicate a greater level of impairment. The Spanish version of the FIQR had an excellent reliability (α = 0.94; coefficient H = 0.93) and showed good discriminant validity. 31
Secondary outcomes
The Patient Global Impression of Change (PGIC) and the Pain Specific Impression of Change (PSIC) are commonly utilized as measures to assess significant clinical improvements in chronic pain treatments. The PGIC is one item (on a 7-point numerical scale, from 1 = “Much better” to 7 = “Much worse”) referred to the participants’ perception of global improvement due to treatment. This measure has been validated for use in psychological treatments targeting chronic pain.32,33 Additionally, the PSIC evaluates (also on a 7-point Likert scale) changes in specific domains, including physical and social functioning, work-related activities, mood, and pain. Both the PGIC and PSIC scales will be completed by participants assigned to STANZA® and FibroST treatments.
The Depression Anxiety Stress Scales-21 (DASS-21) 34 measures depression, anxiety and stress in clinical and non-clinical samples. Participants are asked to report their experience of various symptoms during the past week. Each item is evaluated on a scale ranging from 0 (“did not apply to me at all over the last week”) to 3 (“applied to me very much or most of the time over the past week”). The questionnaire comprises three subscales, each with seven items, focusing on: depression, anxiety and stress. Consequently, the total possible scores for each subscale can range from 0 to 21, with higher scores indicating more severe levels of depression, anxiety, and stress. The internal consistency of the Spanish DASS-21 subscales is adequate: 35 α_depression = 0.84, α_anxiety = 0.70, and α_stress = 0.82. In addition, the Spanish DASS-21 showed adequate convergent validity since correlations with similar constructs were high and significant. 35
The Multidimensional Inventory of Subjective Cognitive Impairment (MISCI). 36 The MISCI is a self-reported questionnaire comprising 10 items designed to assess subjective cognitive dysfunction in individuals with FM over the past week. Each item is rated from 1 (“not at all/never”) to 5 (“very much/always”), resulting in a total score ranging from 10 to 50. Lower scores on the MISCI indicate a higher level of cognitive dysfunction. For the Spanish version of the MISCI, reliability analyses yielded a coefficient α of 0.91, and an intraclass correlation coefficient of 0.88, suggesting very good internal consistency and test–retest reliability for the MISCI. 37
The Pain Catastrophising Scale (PCS) 38 is a 13-item instrument that evaluates the frequency of thoughts about perceived catastrophic consequences of pain. It encompasses three facets: Rumination, which is the persistent focus on the sensation of pain; Magnification, which involves overstating the danger associated with pain sensations; and Helplessness, reflecting a sense of inability to manage pain intensity. The total and subscale scores of the PCS are derived by adding the rating of each item. For the purposes of this study, the total PCS score, ranging from 0 to 52, will be used, where higher scores indicate a greater tendency towards pain catastrophizing. The Spanish adaptation of the PCS is recognized for its excellent psychometric attributes, with a Cronbach's α of 0.79 and an intraclass correlation coefficient of 0.84. 39
Process measures
The Psychological Inflexibility in Pain Scale (PIPS). 40 The PIPS, consisting of 12-item, 34 is a scale developed to measure psychological inflexibility among those individuals who experience chronic pain. The scale is divided into two subscales: one focusing on avoidance and the other on cognitive fusion in relation to pain. Each item on the scale is evaluated using a 7-point Likert-type scale, where 1 signifies “never true” and 7 represents “always true”. Higher scores on this scale are indicative of greater psychological inflexibility concerning pain. In this study, the aggregate score, which can range from 12 to 84, will be utilized, with higher scores reflecting a greater degree of psychological inflexibility. The Spanish adaptation of the PIPS has demonstrated good psychometric properties with a Cronbach α of 0.90 and an intraclass correlation coefficient of 0.97. 41
The Chronic Pain Acceptance Questionnaire (CPAQ-8). 42 The CPAQ-8 is composed of 8 items, categorized into two dimensions: activity engagement (AE: 4 items; e.g. “Although things have changed, I am living a normal life despite my chronic pain “) and pain willingness (PW: 4 items; e.g. “Before I can make any serious plans, I have to get some control over my pain”). Participants rated all items from 0 (never true) to 6 (always true), but the four PW items are reversed for obtaining a total score that can range from 0 to 48. Higher scores indicate greater pain acceptance. The reliability and validity of the Spanish version of the CPAQ-8 is adequate; the Cronbach α ranges from 0.75 to 0.88, and it obtained moderate correlations with committed action scores, supporting convergent validity. 43
Cost-utility measures
The EuroQoL-5D-5L (EQ-5D-5L). 44 The EQ-5D-5L is an instrument for evaluating health-related quality of life. It is composed of two parts: (a) mobility, self-care problems, pain/discomfort, and anxiety/depression; and (b) current perceived health status (from 0 “The worst health you can imagine” to 100 “The best health you can imagine”). Quality-adjusted life years (QALYs) using the utilities obtained with the EQ-5D-5L instrument will be used for cost-utility analyses. The overall Cronbach's α of the Spanish version of the EQ-5D-5L was found to be 0.75, indicating a good internal consistency of the questionnaire. The intraclass correlation coefficient was also excellent (0.86). 45
The Client Service Receipt Inventory (CSRI). 46 The CSRI is utilized for gathering retrospective information on medication usage and service utilization. Data will be gathered on the cumulative days of prescriptions and the daily amount of various FM-related drugs, including analgesics, anti-inflammatories, opioids, antiepileptics, muscle relaxants, antidepressants, and anxiolytics. Regarding service receipt: total visits to emergency services, total days of general inpatient hospital admissions, number of diagnostic tests administered, and the total visits to general practitioner, nurse, social worker, psychologist, psychiatrist, and other community health care professionals. The CSRI will be measured at two different points: at baseline and at 6-month follow-up, both referring to the previous 6 months. The CSRI validated for Spanish populations was used in this study. 47
Other self-reported measures
Adverse events linked to STANZA® or FibroST. Adverse events (i.e. sleep-related changes, headaches, nausea, nightmares, insomnia, anxiety, agitation, and any other type of adverse event) will be recorded using an ad hoc adverse events questionnaire that asks: (a) if the participant has experienced any adverse events as a result of any of the treatments received (yes/no); If any adverse events are reported, participants are asked the following: (b) what adverse events have been experienced; (c) how long the event lasted (hours or days); and (d) how severe the adverse event was (from 0 to 10).
The mHealth app Usability Questionnaire (MAUQ). 48 The MAUQ consists of 21 items categorized into three dimensions: usability and satisfaction (8 items), system information arrangement (6 items), and efficiency (7 items). Responses to all items are given using a 7-point Likert scale, ranging from 1 (extremely strongly agree), to 7 (extremely strongly disagree). A mean value closer to 1 indicates a higher level of the app´s usability. We will only use the usability and satisfaction items for the present work. According to a recent study the MAUQ is reliable (Cronbach α = 0.89), valid, relevant, clear, and comprehensible for Spanish people. 49
Biomarkers
Upon completion of baseline and post-treatment assessments, patients will be contacted 3–5 days later to obtain peripheral blood samples. All subjects will be examined between 8.00 and 9.00am after a 12-h fasting period. Blood and serum will be stored at −80°C in a biobank until the analyses are conducted. To limit the effects of medication on the study variables, patients will be asked to refrain from taking analgesic or anti-inflammatory drugs within 72 h prior to obtaining the biological samples. All the samples will be analyzed in a single analytical batch to reduce inter-trial variability. Serum levels of IL-4, IL-6, CXCL8, IL-10, IL-17A, BDNF, and high-sensitivity C-reactive protein (hs-CRP) will be evaluated. For the quantification of the immune-inflammatory markers, a Simple Plex platform will be used. Hs-CRP levels will be quantified using turbidimetry. The processing, storage and analysis of the samples will be carried out by Laboratorios Echevarne, S.A, Barcelona, Spain.
Hair cortisol-cortisone and serum cortisol. Hair samples will be collected from the middle-lower region of the head, ensuring proximity to the scalp. For three days before sample collection, no hair products like gels, lacquers or conditioners will be used. A hair strand approximately10 mm in thickness will be cut from the posterior vertex, wrapped in foil to protect it from light, and stored at ambient temperature. For the hormone determination only the 1 cm of hair closest to the scalp will be analyzed. The extraction of hormones will be performed in the Nutrition and Obesity Laboratory of the Biochemistry and Molecular Biomedicine Department (University of Barcelona, UB). The procedure begins with weighing the hair samples, followed by an initial wash using a 5 mL of isopropanol. After drying the samples at 37°C and minced them, cortisol and cortisone are extracted in 2 mL of HPLC grade methanol. This is then stirred overnight at room temperature, with the addition of deuterated Cortisol-d4 and Cortisone-d8 (Sigma Aldrich solution C-113 and 900170). The methanol is then moved to test tubes and evaporated under a stream of N2 at 50°C in a dry bath. The dried extracts are preserved at −20°C until cortisol and cortisone are quantified using liquid chromatography with tandem mass spectrometry (LC-MS/MS) at the Scientific and Technological Centres of the UB (CCiTUB). Levels of cortisol, ACTH and CBG will be determined using ELISA (DRG EIA-5396, EIA-1887R, EIA-3647, and MyBiosouce MBS2533311).
Polymorphisms in the FKBP5 gene. A 4 mL blood sample will be collected in a vial with EDTA anticoagulant. The analysis of the genetic variants of the FKBP5 gene will be carried out in the Molecular Genetics Laboratory of the Anthropology Unit (UB). The DNA extraction will be performed using the Real Extraction DNA kit (Durviz S.L.U., Valencia, Spain), and its quality will be assessed using the Nanodrop D1000. The genotyping of five specific SNP polymorphisms in the FKBP5 gene [rs3800373 (SNP1), rs9296158 (SNP2), rs1360780 (SNP3), rs9470080 (SNP4) and rs4713916 (SNP5)] will be conducted using TaqMan 5′ exonuclease assay technology (Applied Biosystems) at the Scientific and Technological Centres of the UB (CCiTUB). The PCR reaction will take place in an ABI PRISM 7900HT instrument thermal cycler and genotyping analysis will be carried out using SDS V.2.1 software. To ensure genotyping accuracy, 15% of the samples, chosen at random, will be genotyped twice. Finally, haplotype estimation will be conducted to increase the power of detection of genetic associations. Linkage disequilibrium among the five polymorphisms will be examined by pairwise comparisons of r2 and D’ using Haploview V.4.2. Estimation of the combination of FKBP5 haplotypes per subject will be conducted using a Bayesian approach implemented with PHASE software Table 1.
Time points at which measures and data are collected.

Note: AET: Adverse Effects of Treatments checklist; CPAQ-8= Chronic Pain Acceptance Questionnaire (8-item version); CSRI: Client Service Receipt Inventory; C-SSRS = Columbia-Suicide Severity Rating Scale; DASS-21: Depression Anxiety Stress Scales-21; EQ-5D-5L: EuroQoL; FIQR = Fibromyalgia Impact Questionnaire Revised; FSDC = Fibromyalgia Survey Diagnostic Criteria; MAUQ = mHealth App Usability Questionnaire; MISCI = Multidimensional Inventory of Subjective Cognitive Impairment; PGIC & PSIC = Patient Global Impression of Change and Pain Specific Impression of Change; PCS: Pain Catastrophizing Scale; PIPS = Psychological Inflexibility in Pain Scale
Data analyses
Sociodemographic and clinical characteristics will be summarized by study arm as mean and standard deviation for age and frequency and percentage for other characteristics. Effectiveness and biomarker analyses. ITT analyses will be conducted with multiple imputation for missing values in all variables. Mixed, multi-level, linear models will be created using the restricted maximum likelihood method for the estimation of parameters. The mixed models will include the fixed categorical effects of treatment arm, study time point, as well as the continuous covariate of baseline score. Significance tests will be based on a two-sided alpha of 0.05 and two-sided 95% confidence intervals. These analyses will be conducted for both the primary and secondary outcomes. As sensitivity analysis, we will compute a modified ITT, including all randomized participants that complete a minimum of 42 sessions of their assigned digital treatment. The IBM SPSS version 29 (IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 29.0. Armonk, NY: IBM Corp) will be used for this analysis. Effect sizes will be calculated according to Cohen's d and path analyses will be computed using Mplus 7.4 (Muthén & Muthén, Los Angeles, CA, USA) to create mediation models. Baseline comparisons with t-tests will also be performed to determine differences between treatment responders and non-responders.
Economic evaluation. The incremental cost-utility ratio will be calculated. The costs for 6 months will be modeled and the QALYs obtained in the 6 months following the start of the treatments will be measured using the area under the curve. The effect of the treatment will be estimated using ordinary least-squares multivariate regression, adjusting for the baseline differences between groups. The direct costs will be calculated by adding the costs of FM-related medications (analgesics, anti-inflammatories, antiepileptics, antidepressants, anxiolytics, hypnotics, and muscle relaxants), use of health services (primary care, specialized, emergencies, hospitalizations), and medical tests. The cost of medication will be calculated by determining the price per mg during the study period according to the Vademecum, including VAT. The cost healthcare services and medical tests will be obtained from the eHealth database. Indirect costs will be calculated based on the days off and multiplied by the minimum interprofessional wage in the study period. We will follow the Consolidated Health Economic Evaluation Reporting Standards statement (CHEERS) 50 for reporting the economic evaluation.
Discussion
The empirical evidence supporting the effectiveness of ACT for individuals with chronic pain is well-established. 51 However, the universal access to these approaches in a face-to-face manner remains limited, highlighting the need for the development of digital tools. Moreover, as a consequence of the COVID-19 pandemic, digital communication has become a widely popular solution in case of mobility restrictions. 52 Notably, internet-based ACT has consistently demonstrated effectiveness in reducing anxiety and depression, which are commonly comorbid conditions with chronic pain.53,54 In this regard, Rickardsson and colleagues55–57 have set a precedent, showing the efficacy of digital ACT in managing chronic pain through innovative microlearning formats. More recently, Catella and colleagues 20 conducted a pilot study comparing a digital self-guided ACT program for FM, referred to as FM-ACT (an early generation of STANZA®), with an active control group undergoing symptom tracking for FM (FM-ST). The results of this study demonstrated clinically meaningful reductions in the overall impact of FM mainly in those individuals assigned to FM-ACT. 20 Notwithstanding, the current evidence of this digital tool is limited as no studies have been conducted out of the USA yet.
Building upon the existing evidence, this three-arm RCT aims to investigate the effectiveness, cost-utility, and physiological effects of an investigational Spanish version of STANZA®. The SMART-FM-SP study may contribute a significant advancement of knowledge for the development of innovative FM treatment options by evaluating the effectiveness, cost-utility, and physiological effects of a smartphone-based digital therapeutic. The development and comprehensive evaluation of a self-guided digital ACT therapeutic for FM attempts to bypass the barriers that limit access to traditional face-to-face therapy. However, this study design has some limitations that should be acknowledged. First, patients without a smartphone or comfort with the use of technology will be excluded in the selection process. Participants will be recruited at a single hospital, which may not represent the general population of Spanish-speaking patients with FM. By harnessing the advantages of mobile technology-based interventions, this approach has the potential to enhance accessibility, improve treatment outcomes, and broaden the reach of effective psychological interventions for FM in public healthcare context. Another potential limitation concerns the participant burden associated with completing online a long battery of measures. It is estimated that the total time required for survey completion will range from 40 to 60 min. For transparency purposes, participants will be provided with clear instructions regarding the expected duration for completing the Qualtrics (online) survey.
Conclusions
If Digital ACT proves effective, it could offer a scalable, accessible, and cost-effective treatment option for FM, addressing the current limitations in the availability of effective non-pharmacological interventions. The findings may also provide valuable insights into the biological mechanisms underlying FM and the role of psychological interventions in modulating these pathways. This could pave the way for more personalized treatment approaches, integrating psychological, neuroendocrine, and immunological perspectives. The SMART-FM-SP study is poised to make a substantial contribution to the field of FM treatment and research. Its findings could have far-reaching implications for the management of FM, offering new avenues for treatment and a deeper understanding of this complex condition.
Footnotes
Conflicting interests
M.J.R. is an employee of and shareholder in Swing Therapeutics, Inc. J.V.L. and L.M.M. are paid advisors of Swing Therapeutics, Inc.
Contributorship
M.J.R and J.V.L. conceptualized the study. M.J.R., L.M.M., X.B., A.F.-S., M.S. and J.V.L. guided study design. A.G., E.R.C., J.N., and J.P.S.M. will serve as study investigators. F.D. and J.V.L. performed the data analyses plan. All authors contributed substantially to the drafting and revision of the paper. All authors have approved the submitted version and have agreed to be personally accountable for their contributions.
Data availability
Deidentified individual participant study data will be made available to qualified academic investigators for non-commercial research upon reasonable request. Data requests should be directed to the clinical research team at juanvicente.luciano@uab.cat. Code availability of STANZA® and FibroST application code will not be made available.
Ethical approval
This research will be carried out in accordance with the 1964 Declaration of Helsinki and subsequent revisions. The study was approved by the Ethics Committee of the Fundació Sant Joan de Déu (PIC-71-23) on 17 July 2023. None of the patients will receive any financial incentive for participating in this study.
Funding
This study has been funded by the Institute of Health Carlos III (ISCIII; PI22/00829) and has been co-financed with European Union ERDF funds. The funder had no role in the design of the study protocol; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The project also counts on with the support from Swing Therapeutics, Inc., San Francisco, CA. Juan P. Sanabria-Mazo has a PFIS predoctoral contract from the ISCIII (FI20/00034). Jaime Navarrete has a postdoctoral contract awarded by CIBERESP (CB22/02/00052). Estíbaliz Royuela has a research contract linked to a project awarded by the Ministry of Education and Science (PID2020-117667RA-I00).
Guarantor
Dr. Juan V. Luciano
