Abstract
Background
Coronary disease is the main cause of death around the world. mHealth technology is considered attractive and promising to promote behavioural changes aimed at healthy lifestyle habits among coronary patients. The purpose of this study is to evaluate the efficacy of an mHealth intervention regarding improved results in secondary prevention in patients after an acute myocardial infarction (AMI) or angina in terms of lifestyle, clinical variables and therapeutic compliance.
Methods
Randomised clinical trial with 300 patients who underwent a percutaneous coronary intervention (PCI) with stent implant. They will be assigned to either the mHealth group, subject to a self-monitored educational intervention involving an internet application installed on their mobile phone or tablet, or to a control group receiving standard healthcare (150 patients in each arm). The primary outcome variables will be adherence to the Mediterranean diet, physical activity, smoking, therapeutic compliance, knowledge acquired, user-friendliness and satisfaction with the application. Measurements of blood pressure, heart rate, body weight, waist circumference (WC) and the 6-min walk test will be taken. Furthermore, the blood lipid profile, glucose and HbA1c will be evaluated. Clinical interview will be conducted, and validated questionnaires completed. The primary quantitative results will be compared using an analysis of covariance adjusted for age and sex. A multivariate analysis will be performed to examine the association of the intervention with lifestyle habits, the control of cardiovascular risk factors (CDRFs) and the results after the hospital discharge (major adverse events, treatment compliance and lifestyle).
Conclusions
The study will provide evidence about the effectiveness of an mHealth intervention at improving the lifestyle of the participants and could be offered to patients with coronary disease to complement existing services.
Trial registration
NCT05247606. [https://ClinicalTrials.gov]. 21/02/2022
Introduction
Cardiovascular diseases (CVDs) are the main cause of death worldwide. 1 Coronary artery disease (CAD) is the most common kind of CVD, and, while significant advances have been achieved in both its diagnosis and treatment, it is the main individual cause of mortality and loss of disability-adjusted life years worldwide. 2 Consequently, strategies focusing on optimising secondary prevention measures are of growing interest since, despite the well-known benefits of adopting healthier habits, a large majority of the patients that overcome an acute myocardial infarction (AMI) do not reach the recommended secondary prevention goals established in international clinical practice guidelines, 3 and many patients are observed to follow an unhealthy diet, be physically inactive and take up smoking again in the year following the AMI. 4 There is robust evidence that cardiac rehabilitation (CR) programmes are one of the cornerstones of managing patients with CAD, these programmes being associated with improvements in both morbimortality and quality of life. 5 Over the years, they have evolved to include several domains ranging from guidance about lifestyle, physical exercise, the optimisation of cardiovascular risk factors (CVRFs) and psychological support to nutrition education and treatment compliance. However, although there is a general agreement regarding the main role of CR programmes in the management of CAD these days, several barriers exist regarding how best to put them into practice.6,7
Outpatient CR often begins weeks or months after the coronary event. 8 This delay is a missed opportunity to promote the importance from the very beginning of making lifestyle and physical activity changes. Moreover, delaying participation in CR programmes has been associated with an increase in sick leave duration. 9 For the patients, the process of adopting a healthier lifestyle by making changes in their behaviour is complicated as it involves making several changes at once, such as wrestling with uncertainty and in some cases being overprotected by relatives. 10 Therefore, homebased CR is a valuable option for managing these patients. A recent systematic review has shown that the risk of adverse events during home-based CR is very low. These results should encourage post-AMI patients to regularly follow the exercise prescribed by their cardiologist regularly and be more active in their environment. 11
With the advancement of mobile phone technology and its widespread use, mHealth and the use of mobile applications are considered appealing and promising tools to encourage behavioural changes among these patients. 4 Several recent studies12,13 have analysed the effectiveness of using mHealth as a tool for improving healthy habits and preventing CVRF in different populations of individuals with CAD. In this sense, a recent meta-analysis conducted by our group involving 4535 patients that had suffered from a coronary event examined the effectiveness of different ways of providing mHealth programmes about changing lifestyle habits, treatment compliance, the control of modifiable CDRFs and psychosocial outcomes. The study concluded that mHealth technology has a positive effect on patients in terms of their exercise capacity, physical activity, adherence to treatment and physical and mental quality of life. It can also reduce the number of readmissions due to all causes or cardiovascular causes. However, no improvements were observed regarding blood pressure, blood lipids, glycosylated haemoglobin and baseline blood sugar in people with diabetes, body composition or anxiety and depression. 12 Another meta-analysis investigated the efficacy of mobile health in decreasing risk factors related to the onset and development of CAD. The results of this study showed that mHealth interventions may result in decreases in body mass index (BMI), waist circumference (WC), total cholesterol, low-density lipoprotein cholesterol (LDL-c), diastolic blood pressure and depression and lead to increases in high-density lipoprotein cholesterol (HDL-c), although no improvements were observed in systolic blood pressure and anxiety levels. 13 However, variations exist in the methodology of published clinical trials analysing the effects of mHealth interventions on coronary disease patients, including the studied variables, duration of follow-up, and intervention components. Therefore, different results have been observed, emphasising the need for engaging mHealth tools that provide the required level of support and knowledge to achieve long-term changes in behaviour. The support received by patients with the digital intervention and the interaction with the app could keep motivation to change their behaviour. Professional assessment and goal setting at entry to the programme are crucial to initiate mHealth use. Enabling self-monitoring and self-assessment is critical in real-world use. 14 Moreover, the use of smartphone technology represents progress compared to previous technology based on text messages (SMS) or telephone calls as a programme based on a smartphone app can begin as soon as the patient is admitted to the hospital and continue for longer. In this sense, in the present project, in addition to including an early start to the invention during the patient's admission, the follow-up will be extended to the last 12 months, with face-to-face monitoring after 3, 6 and 12 months.
Our hypothesis is that the use of the mobile application (eMOTIVA) by patients with coronary heart disease will improve lifestyle, CDRFs, mood, motivation and clinical outcome after discharge from hospital compared to patients who do not use this tool.
The aim of this study is to evaluate the efficacy of an mHealth intervention involving a mobile phone application regarding the improvement in the results of secondary prevention in patients after an AMI or angina compared with standard healthcare in terms of improved lifestyle (adherence to Mediterranean diet, physical activity, smoking cessation and therapeutic compliance), the control of CVRF (BMI, WC, blood pressure, levels of total cholesterol, LDL-c, HDL-c and triglycerides), mood characteristics (anxiety, depression, stress and quality of life), motivation and clinical course after hospital discharge (onset of major adverse cardiac and cerebrovascular events (MACCE), visits to A&E and hospital readmissions during the study period).
Material and methods
Design
A randomised controlled clinical trial (RCT) with a parallel group design will be conducted on patients with CAD submitted to percutaneous coronary intervention (PCI) with stent implantation after myocardial infarction or angina. The study will take place in the cardiology unit of a public speciality hospital in the province of Cádiz, Spain, in which around 1500 coronary interventions are performed every year (Figure 1).

Flow diagram.
Randomisation and blinding
The randomisation and allocation to each group will be 1:1, mHealth or standard care, using computer-generated random numbers. The researchers responsible for the study will not be involved in the allocation of the participants to each group. Due to the study's nature, blinding of the participants is not feasible. To minimise the risk of bias, statistical analyses will be conducted by researchers who were not involved in the intervention.
Participants
Patients will be evaluated according to the eligibility criteria, and they will be offered the chance to participate in the study during admission in the Cardiology Clinical Management Unit. Patients at low, moderate and high risk according to the American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR) criteria will be included in the study. 15
Inclusion criteria
Patients under 75 years of age who have had myocardial infarction or angina pectoris and who have undergone a percutaneous coronary intervention and have a smartphone or tablet with internet connection during the study period and are able to manage the software.
Exclusion criteria
Patients whose survival expectancy is less than 1 year, or who are suffering from severe heart failure, physical incapacity, dementia, severe psychiatric disease, congenital or rheumatic heart disease or triple-artery disease requiring bypass coronary revascularisation surgery or who do not speak the language.
Sample size
The estimated sample size is 128 patients in each arm for a 95% confidence level and a statistical power of 80% to detect a mean effect size, Cohen's d of 0.5 16 regarding adherence to the Mediterranean diet (8.6 ± 2.0 points), 17 physical activity (210.2 METsmin/ d ± 221.8 METs-min/d)18,19 and a 12% decrease in the prevalence of smokers (prevalence of 21% from prior pilot study). A total of 300 participants will be included considering drop-outs with an estimated 15% loss to follow-up (150 in the mHealth group and 150 receiving standard care).
Recruitment
The recruitment and intervention will be conducted by nurses specifically trained for the project. The patients assigned to the intervention group will have access to a mobile app designed to provide support with adherence to healthy lifestyle advice, controlling CVRF and treatment compliance. During hospital admission, a nurse will recruit eligible patients and will arrange programmed face-to-face follow-up visits 3, 6 and 12 months after being discharged. Both groups will be given written recommendations about lifestyle and treatment compliance, 20 and a web page will be available to them containing information about CVRF, in addition to an email address through which they can contact the nurses if necessary.
Mhealth group
Nurses will download the app to the mobile phone of the members of the mHealth group, and the patients will complete a tutorial to learn how to use the app. In this training session, both the patients and their relatives will learn how to use the functions of the application. They will be advised to use the application for at least 15 min per day. Before hospital discharge, all the patients will be encouraged to follow a healthy lifestyle and to comply with their treatment. The stages of change approach will be followed to help patients with behavioural modifications. 21 The participants will be assessed at the beginning, during (3 and 6 months) and at the end of the study (12 months) by means of an interview and their medical records. Prior to the trial, a pilot study was performed with 20 participants to test the application and make any necessary changes and adjustments.
Control group
Patients in the standard healthcare group will receive guidance on medication management and lifestyle modifications, including the Mediterranean diet, physical activity, smoking cessation and therapeutic adherence. Additionally, they will receive written recommendations. Patients in the control group will undergo identical face-to-face follow-up visits.
Technical data of the app and website
The online app acts as an access platform after validating the username and password (Figure 2), and it is managed through the project website. All the information recorded by the patients when they begin a session on the application is transferred to a professional interface accessed by the professionals treating the patient. The application follows the Principles of Universal Design and reference accessibility standards and recommendations.

e-MOTIVA application home page.
Technology development: 1. AJAX web development techniques (asynchronous JavaScript and XML). The app runs on the participant's browser and interacts with the server in the background. 2. Use of sequence commands in PHP (personal home page tools). 3. Bootstrap open-source tools for designing websites and online apps. Operating environment: It is an app with remote access to a MySQL database. For storage, this database is fast enough to deploy web apps. Access privacy: Data is stored on a web server and not on a local computer, with access to them with a username and password. This web server works with anonymous data and is located in Spain to comply with the regulations for the protection of high-level data. The web server performs daily backups of all the files, as well as backups performed by the software on demand. Thus, the data and the programme are doubly protected.
Application contents
The application provides the participants with information and support to: (a) follow a healthy eating pattern based on the Mediterranean diet 20 ; (b) perform physical activity of duration and intensity in line with the recommendations of their cardiologist according to the risk stratification of the AACVPR 15 ; (c) monitor and maintain healthy body weight; (d) maintain suitable treatment compliance; (e) monitor their blood pressure; (f) monitor capillary blood glucose in people with diabetes; and (g) stop tobacco smoking if they smoke.
The application is based on the phases of change theory (attention, retention, memory, action and motivation) 21 and on making the process pleasing.22,23 Furthermore, the application includes the use of effective behavioural strategies to achieve changes in habits, such as keeping daily and weekly records 24 of food eaten, physical exercise performed, weight, treatment compliance, capillary blood glucose and tobacco use, and it establishes goals and provides positive feedback according to the data recorded. 25
The application includes a Classroom section, which is a space for participation that, using information based on scientific evidence, encourages the user to main a healthy lifestyle and to reach the treatment goals recommended in clinical practice guidelines. The app addresses four cornerstones of the secondary prevention of CVDs: (a) healthy lifestyle habits, diet, physical activity and recommendations; (b) risk factors, arterial hypertension, cholesterol, obesity, diabetes, tobacco and stress/anxiety; (c) compliance with treatment; and (d) goals to be reached regarding diet, physical activity, body weight, blood pressure, blood sugar, tobacco and medication. Each section comprises online interactive videos (about physical activity both indoors and outdoors, the correct self-measurement of blood pressure and WC, the treatment of cardiac events and a guided mindfulness relaxation audio). In addition, the classroom provides documents that can be downloaded and printed such as weekly menus and graphics with information (food pyramid, heart health, characteristics and benefits of physical exercise and recommendations about healthy lifestyle, stopping smoking and controlling stress) (Figure 3). Each section includes a questionnaire to complete to obtain feedback about the knowledge acquired in the learning sessions.

Training classroom module.
The application also has a messaging section with which the patient can contact the nurses on the project and resolve any queries (Figure 4).

Weekly traffic light and feedback messages, automatic text message reminders, chat and gamification (from left to right).
For self-monitoring, the app has the following components and interfaces:
Diet: where a record is kept of the food eaten each day classified into food groups (fruit, vegetables, fish, pulses, white meat, red meat, eggs, butter, pastries and cakes, ready meals, nuts and soft drinks) and the number of portions eaten every day. The app shows an image outlining the portions of each food group recording during the day and the cumulative portions in the week in progress (Figure 5). Physical activity: where the patient selects the kind of activity performed each day (walking, walking fast, cycling, swimming, etc.). The application also contains a step counter that patients start when they begin walking; it records the distance covered, the time taken, the speed and the number of steps. Moreover, it also shows a summary of the last records (date, minutes and steps) and a graphic showing the progress made over the last 8 weeks (Figure 5). Treatment: where the patient can see their personalised treatment list as prescribed by their cardiologist, where they must mark and record taking each medication with their meals: breakfast, lunch, afternoon snack and dinner. The patient can also consult the list of their latest treatment records (date and dose taken and completed) (Figure 5). Tobacco: where participants that smoke have this tab enabled to record the number of cigarettes they smoke each day and where they can also see a summary of the latest records (date and the number of cigarettes) and a progress chart of the last 8 weeks (Figure 5). Body weight: where it is possible to record body weight and WC once a week. The patient's height is recorded when they start using the app. This section also shows a summary of the last data recorded (date, weight and WC), an image of progress made over the last 12 weeks, and another with the patient's BMI classified into low weight, normal weight and overweight, and the three classes of obesity according to the WHO classification for adults
21
(Figure 6). Blood pressure: where participants can record their systolic and diastolic blood pressure and heart rate once a week and see recent records, as well as a progress chart of the last 8 weeks (Figure 6). Capillary blood sugar: The patients with diabetes mellitus have this function activated to record their fasting capillary blood sugar 2 h after lunch, twice a week. In addition, the interface has a summary box with their last eight records and a progress chart (Figure 6).

Modules on nutrition, physical activity, treatment and tobacco (from left to right).
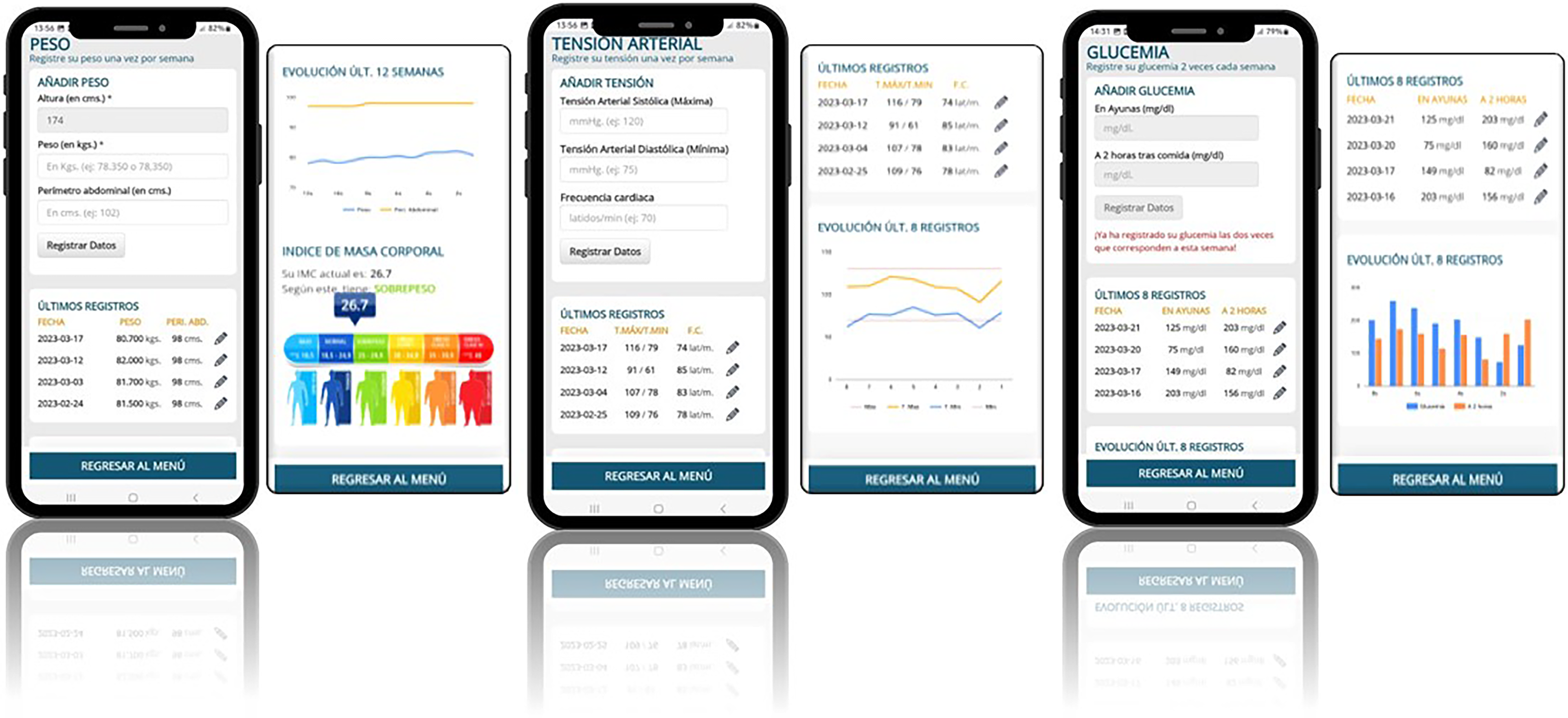
Body weight and BMI, blood pressure and capillary blood glucose modules (from left to right).
All patients will be informed that if they experience changes in their health status or detect abnormal values (indicated by red alarm messages in the app) related to blood pressure, blood glucose, heart rate or weight gain, they should consult their doctor. On the other hand, to motivate the participants to improve and maintain healthy habits, the app includes the following functions: (a) reminders about healthy habits generated at random on a pop-up screen once a week and (b) personalised messages according to the user's achievements and recommendations about aspects to be improved, in accordance with the information recorded during the previous week. These messages appear on Mondays on a pop-up screen when the app is opened and may be green (goal reached), yellow (goal partially reached) or red (goal still to be reached). In this way, the patient receives weekly feedback about their achievements. Furthermore, each icon on the home page of the application appears in the colours mentioned according to the goals reached the previous week as a reminder of their achievements and aspects to improve (Figure 4).
The app uses gamification, whereby through symbols of achievements, users can get different medals depending on whether they comply with the recommendations set for food and physical activity after a month, 2, 3, 6 or 12 months using the application (Figure 4). These systems with fun rewards, such as awarding digital badges obtained for specific objectives, are related with the participation and motivation of the users in mHealth interventions, and they encourage an initial and sustained commitment among the users to modify CVRF.26,27 What is more, gamification can make the interventions more enjoyable, and this is in line with the self-determination theory, which states that a key part of intrinsic motivation is enjoyment. 28 The application also has fun, colourful warnings and messages, advice, feedback and self-comparisons by means of graphics detailing weekly progress.
Strategies to ensure adherence to the app will encompass goal setting, self-monitoring of lifestyle and CDRFs through measurements and records.
Additionally, users will receive educational content displayed on their screens regarding healthy lifestyles and treatment adherence. Motivation will be fostered through feedback on accomplishments and areas for enhancement, complemented by reminders from healthcare professionals to engage with the app.
Ethical considerations
The project has been approved by the Research Ethics Committee (Reference 002_jun20_PI-RECAMAR-19) and authorised by the hospital. The study will be conducted in agreement with the guidelines and protocols established in the Helsinki Declaration as revised in Fortaleza (Brazil) in October 2013 and complies with Law 14/2007 on Biomedical Research and with European Data Protection Regulations. The written informed consent of all the patients will be requested in agreement with the standard model for this kind of studies used in the hospital.
Results of the study
After the initial assessment during their hospital admission, the participants will be reassessed after 3, 6 and 12 months (final visit) in face-to-face follow-up visits in which they will undergo different medical tests (measurement of blood pressure, heart rate, weight, WC and walk test), and an evaluation will be made of the last blood test (total cholesterol, LDL-c, HDL-c, triglycerides, glucose and HbA1c). A clinical interview will also take place and validated questionnaires will be completed.
Variables collected in the initial, 3-, 6- and 12-month assessments
The data collected in the initial assessment will include the patient's sociodemographic characteristics, educational level, personal history (smoker, tobacco consumption, diabetes mellitus, arterial hypertension, prior cardiovascular events and associated morbidities), the reason for the PCI (stable or unstable angina, ST-segment elevation or non-ST-segment elevation AMI), number of diseased blood vessels, stent implants, Euroscore II classification of the left ventricle ejection fraction (LVEF), 29 days of hospitalisation, major adverse events during hospitalisation and drug treatment on discharge. During all the assessments, measurements will be taken of body weight and height to calculate BMI, WC, systolic and diastolic blood pressure, heart rate and oxygen saturation; also, the plasma level of total cholesterol, HDL and LDL cholesterol, triglycerides, blood glucose and HbA1c will be examined in patients with diabetes. During the follow-up period, records will be kept of MACCE, visits to A&E and hospital readmissions due to both cardiovascular causes and other causes. In addition, adverse effects and harms related to the intervention will be reported. Tobacco consumption will be assessed by the self-reported number of cigarettes per day and the Fagerström nicotine dependence test. 30 Physical activity will be assessed using the International Physical Activity Questionnaire (IPAQ), 31 (METS-min/week) and the hours of sedentary behaviour will be recorded. An objective assessment will also be performed of functional exercise capacity through the 6-min walk test. 32 To assess the foods eaten and dietary intake, validated questionnaires of food consumption frequency and adherence to the Mediterranean diet will be used.17,33 Treatment compliance will be assessed using the Morisky–Green 8-item test.34,35 Levels of anxiety, depression and stress will be measured by means of the validated Depression, Anxiety and Stress Scale (DASS-21), 36 while the quality of life will be evaluated with the validated questionnaires, Short Form-12 Health Survey (SF-12) and European Quality of Life-5 dimensions-5 levels (EQ-5D-5L).37,38 The level of knowledge about CAD, healthy lifestyle and CVRF will be examined using a questionnaire that has been validated for this population. 39 The usability of the app will be assessed in the mHealth group using a System Usability Scale (SUS) consisting of 10-Likert-type questions scored from 1 to 5. Motivation to stop smoking will be measured using Richmond's test. 40 Satisfaction with the app and with the care received, and the motivation to follow a healthier diet and perform physical exercise, will be assessed with specific questionnaires developed by the research team.
End-points
The primary end-points after 12 months in both groups will be changes in behaviour regarding: (a) healthy diet, adherence to the Mediterranean diet and frequency of consumption of each food group;(b) level of physical activity (MET), number of steps per day, a decrease in sedentary behaviour and exercise capacity (6-MWT); (c) smokers stopping smoking; (d) treatment compliance; (e) knowledge acquired; and (e) usability of and satisfaction with the application.
The secondary end-points will be the different between the two groups after 12 months: (a) BMI and WC; (b) blood pressure; (c) total and LDL cholesterol; (d) HbA1c in patients with diabetes; (e) MACCE, hospital readmissions and mortality due to cardiovascular causes and all causes; (f) quality of life; (g) anxiety, depression and stress; and (h) motivation to maintain a heart-healthy diet, a suitable level of physical activity and to stop smoking.
Statistical analysis
The SPSS v.24.0 software will be used. A descriptive statistical analysis will be performed (mean, standard deviation, 95% confidence interval, frequencies and percentages). For the comparison of means, the Student's t-test will be used (normal distribution variables) and the Mann–Whitney U test (if the variables are non-normally distributed). The chi-squared/Fisher test will be used for the comparison of proportions.
The quantitative primary outcomes from the two groups will be compared using an analysis of covariance adjusted for age and gender. A multivariate analysis will be performed to examine the association of the intervention with lifestyle habits, the control of CVRFs and progress after discharge in terms of cardiovascular events, visits to A&E and hospital readmissions. Intention-to-treat analysis will be used. Data from all patients in the control group and mHealth will be analysed independently of the use of the app. Two separate analyses will be performed for chronic coronary syndrome and acute coronary syndrome. A two-tailed p value < 0.05 will be considered statistically significant. The researchers analysing the results will be blinded to the allocation of the participants.
To ensure the fidelity of the intervention, professionals participating in the study have undergone specific training to implement the intervention. Study variables will be assessed using validated questionnaires, and the application has undergone testing in a pilot study involving 20 patients to address potential errors.
Discussion
This study will evaluate the efficacy of an mHealth intervention in terms of improving the outcomes of secondary prevention in patients after AMI or angina pectoris in comparison with standard healthcare with the aim of promoting adherence to healthy lifestyle guidelines.
This mHealth intervention is designed to be integrated into health services for the care of patients with CAD when they are in hospital and the PCI is carried out. In this way, it will be possible to emphasise and promote a healthy lifestyle from the beginning, when patients are more motivated and hence more likely to change their behaviour. 41
The European Society of Cardiology puts treatment compliance and healthy lifestyle habits at the centre of secondary prevention strategies for CAD.3,42 The ever-increasing burden on the healthcare system likely exceeds the available resources assigned to treating this kind of patients such as prescribing and participating in CR programmes. Therefore, new strategies are required to tackle the demand. Over 6.5 billion people worldwide are estimated to own a smartphone, this being one of the reasons for the growing interest in interventions using mHealth technology to provide guidance and healthcare. 43 There have been many interventions involving mobile tools, but most of them are simple and lack customised content with real-time feedback.44–46 A limited number of trials have addressed many risk factors related with CAD, and most have only focused on one or two health behaviours, such as diet, physical exercise or smoking. 47 However, there is a lack of evidence supporting a decrease in MACCEs with the use of these interventions. 43 Test messages are the most widely used method in mHealth interventions even though it has been proven that applications with visual content such as videos, images, feedback and gamification are more suited to patients’ preferences and may make these mobile health interventions more user-friendly.48,49 This application was developed as a complement to traditional CR. The Covid-19 pandemic resulted in a large waiting list of patients with coronary heart disease who needed CR, but hospital visits were limited due to the risk of infection. In these conditions, our mHealth tool could make it easier to perform CR from home, albeit guided by hospital healthcare professionals.
To our knowledge, this will be the first study in our country to analyse the effects of an mHealth intervention after an individualised year-long follow-up after a PCI. If the intervention described proves to be effective, it has great potential to become an integral part of the existing healthcare system, and for its use to be extended to other parts of the country.
Limitations
The patients will participate voluntarily in the study and will sign written informed consent, which could imply that these patients are more motivated to adhere to changes regarding a healthy lifestyle compared with those who choose not to participate. This could be a limitation for generalising to less motivated people rather than a selection bias since the study design is a randomised clinical trial. Another limitation could be that to take part in this mHealth intervention, participants must have a smartphone or tablet with internet connection. However, this limitation would seem insignificant since, as mentioned above, over 75% of the world's population has a mobile phone with internet access, while in Europe 99% of people have a mobile phone with internet connection and 86% of homes have an internet connection. 50 Another limitation could be that blinding of participants will not be possible due to the nature of the intervention. However, the coordinators and the researchers who will analyse the results will not know the allocation of the participants. Finally, the trial will take place in an urban hospital and certain differences may exist compared with rural areas. However, it is a reference hospital for the whole province where 1500 intervention procedures are performed every year. Furthermore, in our study, we will not conduct an economic evaluation that could provide useful information to clarify whether the implementation of this mHealth intervention in the healthcare of these patients is cost-effective.
Conclusions
In conclusion, this study will provide evidence about the effectiveness of an mHealth intervention at improving the lifestyle of the participants and could be offered to patients with CAD to complement the existing services. This intervention could also be used with other chronic diseases where adherence to a healthy lifestyle is essential and often poses a challenge.
Footnotes
Contributorship
Conceptualisation: MJSC. Data curation: MJSC, CCC and MABJ. Formal analysis: MJSC, CCC and MABJ. Funding acquisition: MJSC. Investigation: CCC, MABJ, GCP, LG, AGB, DCP, RVG and MJSC. Methodology: MJSC and ARM. Project administration: MJSC, RVG and JAT. Resources: CCC, MABJ, GCP, LG, AGB, GCP and MJSC. Supervision: MJSC, ARM, RVG and JAT. Validation: MJSC and ARM. Visualisation: MJSC, CCC and MABJ. Writing – original draft: CCC, MJSC and MABJ. Writing – review and editing: MJSC. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The project has been approved by the Research Ethics Committee (Reference 002_jun20_PIRECAMAR-19) and authorised by the hospital. The study will be conducted in agreement with the guidelines and protocols established in the Helsinki Declaration as revised in Fortaleza (Brazil) in October 2013 and complies with Law 14/2007 on Biomedical Research and with European Data Protection Regulations. The written informed consent of all the patients will be requested in agreement with the standard model for this kind of studies used in the hospital.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study has a subsidy for the financing of Research and Biomedical Innovation and in Health Sciences, within the framework of the Integrated Territorial Initiative 2014-2020, for the province of Cádiz. Project 80% co-financed by the European Union, within the framework of the FEDER Andalusia Operational Program 2014-2020. Ministry of Health and Consumption, Junta de Andalucía. Reference-Code: PI-0014-2019. JA Tur is funded by CIBEROBN (CB12/03/30038)-Instituto de Salud Carlos III and cofunded by the European Regional Development Fund.
Guarantor
MABJ
