Abstract
Objectives
To synthesise the literature on clinical decision support (CDS) systems for chronic obstructive pulmonary disease (COPD). We aimed to (1) describe existing COPD CDS systems that have been designed, developed or are being used in practice, (2) describe the impact of COPD CDS systems on outcomes and (3) identify barriers and facilitators to implementation of COPD CDS systems.
Methods
Five databases were searched to identify relevant studies. All studies in English that described clinician-facing COPD CDS systems designed for, or implemented in, hospitals and hospital-in-the-home settings were included. A qualitative narrative synthesis was undertaken, guided by the RE-AIM framework (Reach, Effectiveness, Adoption, Implementation and Maintenance).
Results
Twelve studies reporting the use of CDS in hospital (n = 7) and hospital-in-the-home (n = 5) settings were included. Implementation efforts to reach target users were scantly reported, and low-to-medium adoption rates were observed. The reported effectiveness of the CDS systems was mixed. Only one study reported facilitators to the implementation of CDS systems, none reported on barriers to the implementation of CDS systems, and none reported any information on successful strategies to maintain implementation of CDS systems.
Conclusion
The use of CDS systems in the management of patients with COPD in hospital-related settings is an important emerging field of research. Evidence suggests that the field has largely focused on systems targeted at physicians, often with incomplete descriptions and limited evaluations. Many opportunities to optimise and evaluate the implementation and use of COPD CDS systems in hospital settings remain, including robust evaluation of their impact on patient, clinician and health service outcomes.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a complex, multi-component, heterogeneous disease characterised by airflow limitation and a range of symptoms of varying severities.1–3 Globally, there are 300–400 million people living with COPD, and the disease results in approximately 3 million deaths annually.4–6 Acute exacerbations (worsening of respiratory symptoms) in patients with COPD can result in hospitalisation and other healthcare use and, in severe cases, death.7–9 There has been a recent focus on the design, development and implementation of clinical decision support (CDS) systems for the early detection and prevention of COPD exacerbations through routine assessment of disease severity. 10
CDS systems have the potential to enhance health-related decisions and actions by providing relevant, structured clinical knowledge and patient information to improve patient health. 11 Mechanisms for delivering decision support include computerised alerts and reminders; order sets; patient data reports or dashboards; and links to guidance, documentation templates and clinical workflow tools. 12 CDS systems may be standalone or integrated into broader existing clinical information systems. Whilst there is evidence suggesting that CDS systems have the potential to improve safety, quality and efficiency of care in other clinical domains, 13 including asthma, 14 the evidence regarding the use of CDS systems for patients with COPD in hospital or hospital-in-the-home settings has not been systematically reviewed. 14 Previous reviews of COPD CDS have considered CDS systems within primary care settings 14 or have focused on the algorithms and performance of COPD CDS systems in tele-monitoring settings. 10
Previous studies have indicated that the implementation of CDS systems for complex conditions in hospital settings is often hampered by poor user acceptance and uptake and inadequate integration into existing workflows.15,16 However, the extent to which studies have focused on the uptake and successful implementation of CDS systems for COPD has not been systematically examined. The overarching aim of this review was to synthesise research literature examining the use of COPD CDS systems across the design, development and implementation cycle. In particular, we aimed to (1) describe existing clinician-facing COPD CDS systems that have been designed, developed or are being used in practice; (2) determine the impact of COPD CDS systems on patient, clinician and health service outcomes; and (3) identify barriers and facilitators to implementation of COPD CDS systems.
Methods
Overview
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Supplementary Appendix A) were followed for the method and reporting of this review. 17 The protocol for the review was registered with PROSPERO (CRD42022328309) and can be accessed at https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=328309. As this was a systematic review of literature, no ethics committee approval was required.
Study selection
Eligibility criteria
Eligible studies described CDS systems targeting COPD that focused on supporting an aspect of individual patient care in hospital(s), including remote monitoring (i.e. hospital-in-the-home) settings. We included studies that described the design, development, evaluation or implementation of CDS systems provided the CDS system comprised a clinician-facing component such as dashboards, alerts, user interfaces and computerised order sets. Studies describing only the development or evaluation of algorithms, in the absence of a clinician-facing system, were excluded.
We included studies of CDS systems where findings for COPD patients were reported separately from other respiratory diseases. Studies with broader CDS systems were included if they provided a description of the CDS system component(s) and separate COPD CDS data. For studies with CDS systems that targeted both clinicians and patients, we included those with separate findings focused on clinicians (e.g. uptake).
We included published and peer-reviewed original research studies that described studies of any research design, including case studies and conference proceedings that were available in English language.
Information sources
Online bibliographic databases Medline, Embase, CINAHL, Scopus and Web of Science were searched with the assistance of an academic liaison librarian. Two sets of keywords and subject headings relating to (1) CDS systems and (2) COPD were defined and searched with ‘or’, and the sets were combined with ‘and’. With the help of the librarian, the authors developed search queries consisting of a comprehensive set of keywords and MeSH (Medical Subject Headings) terms relating to the chosen keywords for each database to capture relevant studies (Supplementary Appendix B). We did not restrict the date range of our search. The final search was conducted on 5 April 2022.
Study selection process
The study selection process is shown in Figure 1. The search results were imported into EndNote X9 referencing software, and duplicates were removed. The remaining studies were imported into Covidence 18 where further duplicates were removed. Using Covidence, titles and abstracts were independently screened by two researchers. A total of 82 studies were full text screened, with disagreements discussed until consensus was reached.

Study selection process.
Quality assessment
Two Joanna Briggs Institute (JBI) Critical Appraisal Checklists were used to assess the risk of bias of included studies: the checklist for quasi-experimental studies (non-randomised experimental studies) and the checklist for analytical cross sectional studies. 19 Although the Mixed Methods Appraisal Tool (MMAT) tool was selected a priori (see PROSPERO registration), we deviated from the protocol as we did not identify any qualitative studies that met our inclusion criteria. Two authors (ABA and DB) independently assessed all included studies. Disagreements were resolved by discussion until consensus was reached. Following precedence on cut-offs, 20 studies with a percentage score of >80% were considered to have a low risk of bias, 60–80% a moderate risk of bias, and <60% a high risk of bias. Studies were not excluded based on quality. Quality assessment scores were presented descriptively.
Data extraction and analysis
For each included study, descriptive information about the article, study design and setting, description of the CDS system, user group, CDS system uptake by providers, barriers and facilitators to implementation, effectiveness of the CDS system and strategies for sustaining the use of the CDS system were extracted independently by two researchers. Disagreements discussed until consensus was reached. We considered undertaking a meta-analysis of studies that investigated the effect of order sets on length of stay, as these studies21–24 used similar CDS systems and included the same outcome measure. However, we were unable to obtain all necessary information regarding censoring of participants and the distribution of length of stay data and, therefore, could not be confident in conducting a methodologically sound meta-analysis from the included studies.
Narrative synthesis 17 was guided by the RE-AIM framework, which includes five dimensions: Reach, Effectiveness, Adoption, Implementation and Maintenance. The COPD CDS systems were assessed at both the individual (i.e. end-user) and organisational (i.e. health service) levels. 25 The RE-AIM framework describes various factors in the design, dissemination and implementation process that contribute to achieving intended population-based impact. 26
We conducted a qualitative narrative synthesis using open coding and grouping into RE-AIM framework themes to identify the following: Who the COPD CDS systems had reached or planned to reach (end users) [Aim 1], the effectiveness of the CDS systems or what patient/clinician/health service outcomes were targeted (outcomes) [Aim 2], what settings the CDS systems was adopted or designed for (including roles of targeted clinicians) [Aim 1], what barriers and facilitators impacted implementation [Aim 3] and what factors were recommended or in place to maintain the CDS systems or support sustained use [Aim 3].
Results
Study selection and quality
The systematic search returned 2020 papers. After removing duplicates and screening, 12 studies met the inclusion criteria and were included for data extraction. Eleven of the included studies were assessed using the JBI critical appraisal tools. Five studies22,24,27–29 were considered to have a low risk of bias (scoring 83% or 89%); one study 30 had a low-to-moderate risk of bias (score of 80%); and five studies21,23,31–33 had a moderate risk of bias (scoring 60% or 67%). Item-level quality assessment scores are reported in Supplementary Appendix C. Risk of bias in quasi-experimental studies was largely due to participants in the pre- and post-intervention groups being sociodemographic or clinically different and based on their statistical analysis, which did not account for sociodemographic or clinical differences between the pre- and post-participant groups (see Supplementary Appendix C). Most observational studies did not state a participant or patient inclusion criteria, with four of the five studies assessed scoring poorly on this item (see Supplement 1). One study 34 was not assessed, as all items on the JBI cross-sectional analytical tool were deemed not applicable.
Study characteristics
Study characteristics are presented in Table 1. Two studies described the same CDS system resulting in 11 unique CDS systems from 12 studies.28,30 Four of the 12 studies focused on electronic order sets.21,22,24,31 Most CDS systems were delivered as part of a clinical information system, such as an electronic health/medical record (n = 7), and half were in their pre-implementation stage (i.e. not yet used for patient care in real-world settings). Most studies were conducted in Europe (n = 6) or North America (n = 4).
Study characteristics.

Note. CDS = clinical decision support; CIS = clinical information system; CPOE = computerised provider order entry; EMR = electronic medical record.
Aim 1: Details of existing CDS systems (RE-AIM: reach and adoption)
Details of existing CDS systems, specifically who the CDS system had reached or planned to reach (end users) and the settings the CDS system was adopted or designed for, are presented in Table 2.
Reach and adoption of existing clinical decision support systems.
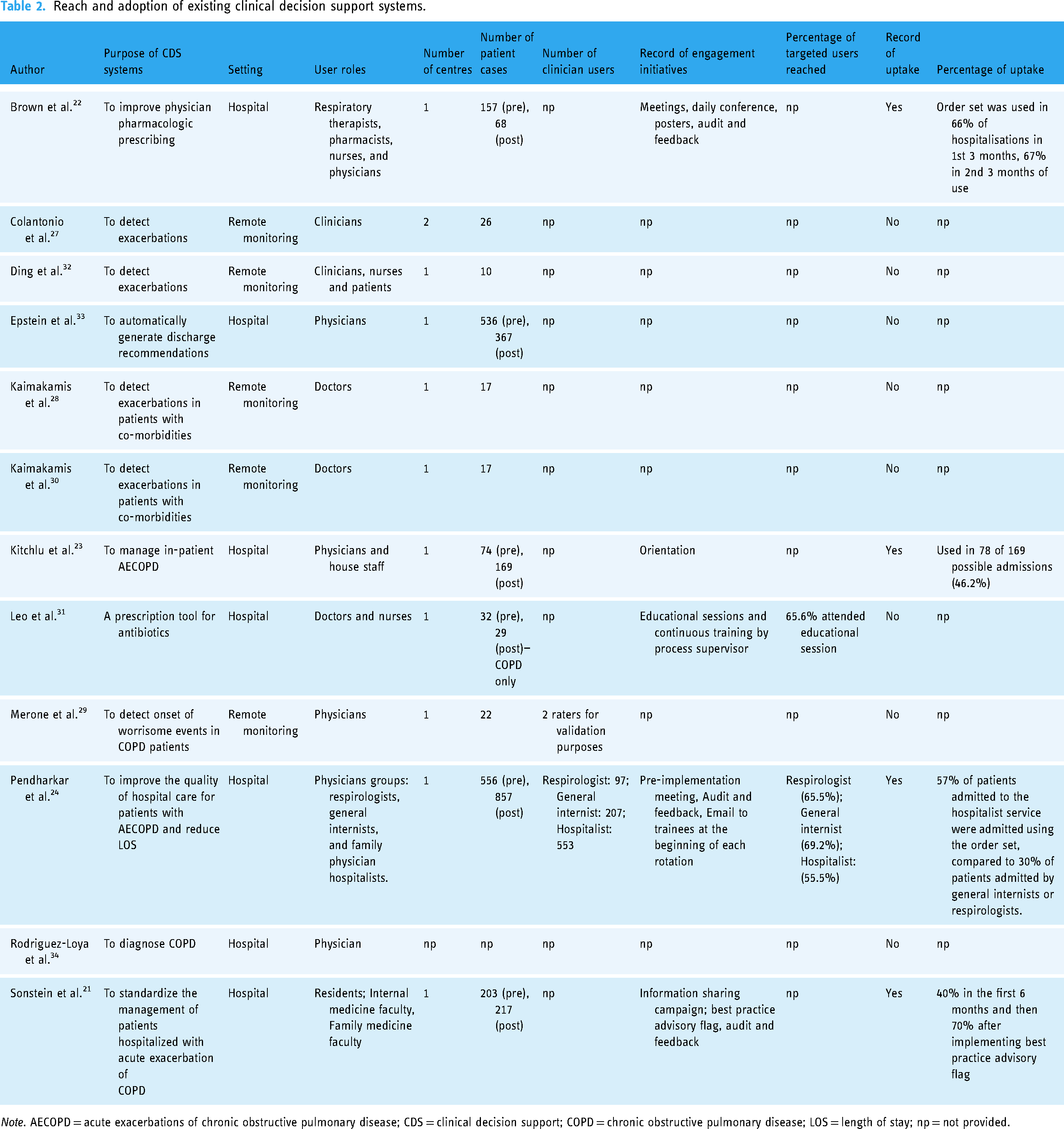
Note. AECOPD = acute exacerbations of chronic obstructive pulmonary disease; CDS = clinical decision support; COPD = chronic obstructive pulmonary disease; LOS = length of stay; np = not provided.
Of the 12 studies, the purpose of five CDS systems was to detect exacerbations in COPD patients,27–30,32 with others focused on patient management and discharge (n = 4),21,23,24,33 physician prescribing (n = 2)22,31 and COPD diagnosis (n = 1). 34 Most CDS systems were designed for use in hospital settings (n = 7) and the remainder (n = 5) for hospital-in-the-home settings.27–30,32 The reported user roles that the CDS system reached or planned to reach included various physician groups23,29,33,34 that were described as respirologists, 24 general internists, 24 family physician hospitalists, 24 residents, 34 house staff, 23 internal medicine faculty, 21 family medicine faculty 23 and doctors and28,30,31 other clinician groups27,32 such as respiratory therapists, 22 pharmacists, 22 and nurses22,31,32 and patients. 32 Most studies (n = 10/12) were conducted at a single site. One study was conducted at two sites, 27 and another did not describe the study site. 34 Most (n = 11/12) studies provided no information on the number of clinicians for whom the system was designed, except for one study 24 that was designed for respirologists (n = 97); general internists (n = 207) and hospitalists (n = 553) and another 29 that was used by two raters.
Many of the studies (n = 7) did not report any engagement initiatives conducted at the study site to reach the planned users of the system. Of those that reported engagement initiatives, most of them described using multiple engagement strategies including meetings,22,24 daily conferences, 22 posters, 28 audit and feedback,21,22,24 educational sessions, 31 continuous training, 29 email to trainees, 24 information sharing campaign 21 and best practice advisory flag. 23 Only two studies provided information on the percentage of targeted end-users reached by the engagement initiatives. One study 31 reported that 65.6% of targeted users attended the educational session, and the other 24 reported that 65.5% of the respirologists, 69.2% of the general internists and 55.5% of the hospitalists were reached by the engagement initiatives (pre-implementation meeting, audit and feedback and email to trainees at the beginning of each rotation).
Of the 12 studies, only the studies that described electronic order sets (n = 4) reported the level of adoption or uptake of the CDS system.21–24 The mean percentage of uptake across the four studies was 54% (range = 30% to 70%) with one 21 reporting an increase from 40% in the first 6 months to 70% after implementing an additional engagement initiative—a best practice advisory flag. This flagged the clinician if the patient presented to the emergency department with a primary symptom that matched predefined criteria, prompting the admitting provider to use the acute exacerbations of COPD order set. 21
Aim 2: Study outcomes (RE-AIM: effectiveness)
Study outcomes are summarised in Table 3. Effectiveness (in a real-world setting) of the CDS system on one or more types of outcomes (i.e. patient, clinician or health service) was reported to be assessed in seven studies. Six of these studies used a quasi-experimental pre–post design, and one 27 was a pilot study. Of those that conducted pre–post studies, the pre-implementation duration ranged from 6 to 25 months, and the post-implementation duration ranged from 3 to 18 months. The patient outcomes assessed were length of hospital stay,21–24 readmission rates,21,23,24 follow-up appointment rates, 21 30-day adverse clinical event, 22 percentage of patients discharged with long-acting medications, 33 duration of antibiotic treatments 31 and detection of worsening symptoms. 27 Three22–24 of the four studies that assessed patients’ length of hospital stay showed an improvement, whilst the fourth study 21 did not show any effect on length of stay. There was no statistical difference in the hospital readmission rates across the three studies that assessed this outcome.21,23,24
Effectiveness of existing chronic obstructive pulmonary disease clinical decision support systems

Note. COPD = chronic obstructive pulmonary disease; CDS = clinical decision support; CPOE = computerised provider order entry; LOS = length of stay; RDD = recommended daily doses; recs = recommendations; PD = patient days; VTE = venous thromboembolism.
The clinician outcomes assessed were as follows: prescriber error rate, 22 educational referral rate, 23 VTE prescription rate, 23 steroid prescription rate, 23 antibiotic prescription rate, 23 guideline adherence, 31 corticosteroid dose administered in the first 48 hours 21 and percentage of recommendations for smoking cessation, vaccination and follow-up visits. 33 There was an improvement in all clinician outcomes with the use of a CDS system. The only health service outcome assessed was rate of antibiotic consumption, which decreased after the intervention (CPOE) was implemented. 31
Aim 3: Barriers and facilitators to CDS system implementation (RE-AIM: implementation and maintenance)
None of the 12 studies reported barriers to the implementation of CDS systems. Only one study stated that using end-user feedback to optimise a best practice advisory flag and order sets facilitated successful implementation of the CDS system. 21 No study reported any strategy used for maintaining the uptake of the CDS system.
Discussion
This study systematically reviewed COPD CDS systems in the published literature, focusing on studies across the system design, development and implementation cycle. Despite the increasing use of CDS systems in clinical domains, we have uncovered key evidence that the use of CDS systems for hospitalised COPD patients, including hospital-in-the-home patients, is still in its early stages with important questions remaining. We identified only 12 studies that met the inclusion criteria, generally with a low-or-moderate risk of bias. Half of the CDS systems described by these 12 studies were not yet used for patient care in real-world settings (pre-implementation stage), indicating a significant gap in the literature and in the implementation of COPD CDS in healthcare.
Most included studies targeted physicians, pharmacists and nurses and were from Europe and North America. Many of the CDS systems were electronic order sets aimed at improving the quality of hospital care for patients with acute exacerbations of COPD. Other CDS systems were primarily designed to detect exacerbations in COPD patients in remote monitoring settings. In a systematic review of factors influencing successful implementation of guideline-based CDS systems, uptake of CDS systems was shown to increase when systems are integrated within clinical information system (CIS) routinely used by clinicians, such as the electronic medical records (eMR) or computerised provider order entry (CPOE).35,36 We found that none of the CDS systems designed for remote monitoring settings were integrated into a CIS. In remote monitoring settings, it has been argued that integrated CDS systems are essential in cases of emergency as a clinician's time to decision can be reduced significantly due to ease of access to patient history. 37 Therefore, as COPD CDS systems evolve within the remote monitoring setting, there is a need for the design and development of more integrated systems to support work in this context.
Consistent with previous studies, 38 there was considerable heterogeneity between the included studies in terms of design, CDS systems and measures used to evaluate the CDS system. For example, some systems focused on physiological monitoring of vital signs to detect COPD exacerbations, whereas others aimed to provide standardised pharmaceutical recommendations (Table 2). These differences along with incomplete reporting precluded the pooled effect of COPD CDS systems on clinician behaviour and patient outcomes from being estimated. Nonetheless, we found that where clinician outcome measures were reported, the COPD CDS system had a positive effect (see Table 3). However, these findings need to be interpreted with caution, as three of the studies21,23,33 that reported positive findings had a moderate risk of bias. Previous work has shown that low use rates of asthma CDS systems contributed to a lack of improvement in outcomes. 39 Given the varying degrees of use reported by studies in the current review, it is somewhat surprising that we found all studies reported positive effects on clinician outcomes with COPD CDS use. Only one study 31 included a health service outcome measure, potentially reflecting the early stage of maturity of CDS systems in this field of research.
We applied an implementation science framework (the RE-AIM framework) to examine COPD CDS systems that have been designed, developed and are being used in practice. With respect to implementation, only one study 24 reported the number of planned users of the system. Of the included studies, most did not report the use of any engagement strategy to ensure planned users of the system were reached. When multiple engagement strategies were reported, the use of meetings and audit and feedback mechanisms were common. Given the importance of an engagement strategy for CDS system uptake and subsequently patient benefit,40,41 it was surprising that when uptake was reported, almost half of the planned users did not adopt the CDS system despite the use of single 23 or multiple 24 engagement strategies in those studies, likely due to high staff turnover and lack of usability evaluation. 23 This suggests that despite the importance of meaningful engagement strategies, there are other factors associated with the successful implementation and uptake of COPD CDS systems, and this warrants further investigation. Our findings of inadequate uptake are supported by previous reviews of CDS systems for similar pathologies of breathlessness 42 and asthma, 39 which identified poor clinician uptake as the main barrier to CDS system patient effectiveness. 42 Future studies need to consider the factors associated with successful implementation to ensure that CDS systems are used by clinicians, so the full benefit to patients and clinicians can be realised.40,41
No studies included in our review identified or reported strategies for maintaining the sustained use of COPD CDS systems, suggesting that most clinician engagement efforts were solely focused on the pre-implementation phase. Whilst engagement of target users in early stages of implementation has been touted as key to successful CDS system implementation, 43 sustained user engagement requires continuous promotion of the CDS system beyond the initial implementation phase. 44 Furthermore, our research found that evidence of factors impeding or facilitating implementation of CDS systems used by COPD clinicians is absent from the scientific literature. Within the COPD domain, limited CDS systems appear to have been implemented and are being used in practice. Therefore, it is unsurprising that there is scarce research into the barriers and facilitators for successful implementation.
Limitations
We summarized published studies in English language only. The findings of this review could be impacted by publication bias and do not consider COPD CDS systems that may be used in practice but not reported in the scientific literature. Consequently, the CDS systems identified in this review may not represent CDS systems adopted in all hospitals. Important differences in study designs, interventions and outcome measures along with incomplete reporting and moderate risk of bias in approximately half the studies assessed prevented the undertaking of a meta-analysis of patient, clinician or health service outcomes. Therefore, we were unable to provide any pooled estimate for the effectiveness of COPD CDS systems. We recommend improvements to the design, evaluation and reporting of the effectiveness of CDS for COPD so that this gap can be addressed in future studies.
Conclusion
This review has uncovered gaps in the use of COPD CDS systems in hospital settings. Our findings highlighted that limited research, generally with a low-or-moderate risk of bias, has focused on the use of COPD CDS systems in hospitals and hospital-in-the-home settings. The use of the RE-AIM framework revealed significant implementation gaps, including poorly documented efforts to reach planned/target users, contrasting results on the effectiveness of the CDS systems, low-to-medium adoption rates, little to no reporting of barriers and facilitators to the implementation of CDS systems and no information on successful strategies to maintain implementation of the CDS systems. CDS systems offer enormous potential to streamline work and facilitate clinical decision-making, but this is yet to be capitalised with respect to the management of COPD. There remain many opportunities to optimise and evaluate the implementation and use of COPD CDS systems in hospital and hospital-in-the-home settings.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231219107 - Supplemental material for Clinical decision support systems for chronic obstructive pulmonary disease (COPD) in hospitals: A systematic review
Supplemental material, sj-docx-1-dhj-10.1177_20552076231219107 for Clinical decision support systems for chronic obstructive pulmonary disease (COPD) in hospitals: A systematic review by Adeola Bamgboje-Ayodele, David N Borg, Steven M McPhail and Melissa T Baysari in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors wish to thank Isabelle Raisin, academic liaison librarian, at the University of Sydney, for her assistance in developing the search strategy.
Contributorship
ABA, MB and SM contributed to the conception and design. ABA and MB with assistance from an academic liaison librarian developed the search strategy. ABA conducted the database searches. ABA and DB conducted the title/abstract and full-text screening. In case of disagreement with respect to inclusion criteria, MB was consulted as a third independent reviewer. ABA and DB conducted the data extraction and assessed the study quality. ABA wrote the initial draft, and DB, SM and MB provided the critical review. All authors approved the final version for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by Digital Health CRC Limited (DHCRC). DHCRC is funded under the Commonwealth's Cooperative Research Centres (CRC) Program. SMM is supported by an NHMRC administered fellowship (#1161138). These funders played no role in data collection, analysis, data interpretation or preparation of the manuscript.
Guarantor
ABA.
Patient consent statement
The authors confirm that the patient consent statement is not required as the study was a systematic review of the literature, i.e. no patients were involved.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
