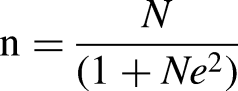Abstract
Background
The use of mobile phone technology for reporting adverse drug reactions (ADRs) in pharmacovigilance is relatively new.
The objective of the study was to explore challenges and facilitators for the use of the Med Safety App for reporting ADRs in Ghana. A comparative evaluation of ADR reports received through the app and the standard paper-based form was also conducted.
Methods
This was a cross-sectional study with a purposive sampling technique. The study population was persons who had downloaded the Med Safety App launched in Ghana 18 months before the study.
Results
Of the 350 participants, 121 provided answers to the questionnaire sent as a Google form, representing a response rate of 34.6%.
Ninety-five (78.5%) of the participants were healthcare professionals, and the remaining were patients. Seventy-five (64.7%) of the participants were using the app after initial installation because they thought it had helpful features. However, only 33 (27.3%) participants used the app to report ADRs, and of these, seven (21.2%) participants indicated that they would continue to use the app because it was easier than the other means of reporting ADRs. Most of the respondents, 109 (94%), indicated that they would recommend the app to someone else. There were some differences between the reports received through the app and between the paper-based Council for International Organizations of Medical Sciences (CIOMS) 1 form and the app, which warrant further exploration.
Conclusion
Most participants indicated that the app is a useful tool and easy to use, and they were satisfied with the features of the app. Given that only just under one-third of participants had used the app to report ADRs, more time and training may be required to fully evaluate the feasibility of the use of the app going forward. The findings will help improve introduction of the app in other countries.
Introduction
Spontaneous reporting is one of the major pharmacovigilance methods for the identification of unknown safety issues with marketed drugs, vaccines, and other health products. However, spontaneous reporting is negatively affected by several drawbacks including underreporting. There have been several interventions to improve underreporting, namely, the introduction of user-friendly reporting systems, 1 availability of the reporting forms, 2 use of web-based or mobile technologies,1,3 and feedback information provided to reporters.4–6
The use of mobile phone technology is a relatively new intervention in pharmacovigilance, with the first app launched in France in January 2015, then in the United Kingdom, Netherlands, and Croatia in July 2015, January 2016, and May 2016, respectively. 7 As of March 2023, mobile app for reporting safety issues has been used in 13 countries in Africa. The World Health Organization (WHO) has supported low- and middle-income countries (LMICs) to roll out the adverse drug reaction (ADR) reporting app, the Med Safety App, in collaboration with technical partners, namely, the United Kingdom Medicines and Healthcare products Regulatory Agency (MHRA) and the Uppsala Monitoring Centre (UMC). The launch of the app follows a successful pilot in Burkina Faso and Zambia to understand the potential benefit and sustainability of the use of this app in low-resource settings. 8 The use of a mobile app is expected to improve the quality and quantity of ADR reporting by patients and healthcare professionals to National Pharmacovigilance Centres and improve safety communication and feedback to reporters. 7
In June 2019, Ghana launched the Med Safety App, which is freely available in the Google Play and App Store for Android and iOS (iPhone Operating System), respectively. 9 As of the end of February 2021, 21 months after the launch, there were 996 downloads with 120 safety reports received through the Med Safety App. Of these 120 reports, 99 (82.5%) were submitted by healthcare professionals, and the remaining 21 (17.5%) were from patients.
The primary function of the Med Safety App is for healthcare professionals and patients to report ADRs to medicines and vaccines. The app also has “News” and “Watch List” features. The “News” and “Watch List” features allow users to access safety-related news articles and alerts from the Food and Drugs Authority and to create a watch list of products of interest, respectively.
Studies that evaluated the use of mobile apps to report ADRs assessed factors that influenced their use. One such study with healthcare professionals and patients from three countries, namely, Netherlands, Spain, and the United Kingdom as participants, found that the factors that affect the use of a mobile app to report ADRs included the type of feedback given on reported ADRs, how ADR reports are stored, and the type of news provided. Other factors mentioned were related to the functions of the app itself including ease of use, language, source of safety information provided through the app, security of the app, layout, the operating systems on which the app can be used, and the cost. 10
In a second European study, researchers assessed healthcare professionals’ and patients’ interest in an app for two-way risk communication and their preferences and perceptions toward specific app characteristics. 11 The findings from this study suggested that about half of patients and more than 60% of healthcare professionals were interested in using an app to report ADRs. The benefits outlined were a faster means of reporting ADRs and easier access to a reporting form. Additionally, the study indicated that healthcare professionals and patients who already use a health app were particularly interested in the app for reporting ADRs. 11
To the best of our knowledge, there is currently no study that documents challenges and facilitators for the use of a mobile app for reporting ADRs in LMICs. One year after the launch of the Med Safety App in Ghana, we deemed it essential that the use of the Med Safety App is evaluated to obtain user opinion on its functionality, usefulness, and how the usage could be encouraged in Ghana and other LMICs.
The objective of this study was therefore to determine the challenges and facilitators for the use of the Med Safety App in Ghana.
Methodology
A purposive sampling technique with a cross-sectional design from 14 December 2020 to 15 January 2021 was carried out to explore challenges and facilitators for the use of the Med Safety App to report ADRs in Ghana. Purposive sampling technique was used for practical reasons because the focus of the study was on those who had downloaded the Med Safety App. This was necessary to maximize responders who could provide the best information on engagement with the app based on actual use.
Questionnaire design
The semi-structured questionnaire used for the data collection was developed by the study team based on an earlier study. 11 The questionnaire was pre-tested with 10 participants who were representative of the target population but not included in the final analysis. The questionnaire was made up of five sections, namely, Demographics information of participants, Knowledge on the use of the Med Safety App, Usefulness of the app, Features of the app, and Additional information (when available). The questionnaire is included as Appendix I.
Sample size determination
The sample size for the study was determined using Slovin's formula
12
with a study population of 996 persons being those who had downloaded the Med Safety App.
To allow for lost to follow-up and non-response, the questionnaire was sent to 350 persons who downloaded the Med Safety App.
A serious ADR is defined as any event or reaction that results in death, a life-threatening adverse event, inpatient hospitalization or prolongation of existing hospitalization, a persistent or significant incapacity or substantial disruption of the ability to conduct normal life functions, or a congenital anomaly or birth defect. 13
Data collection
The questionnaire was sent by email as a Google form to 350 participants selected by random sampling from among the 996 who had downloaded the app, with follow-ups conducted by phone and/or email. Contact details list were available from those who downloaded the app through their registration.
App user data analyses
Data collected during the study was entered into MS Excel and imported into Stata, version 14.1 by StataCorp, College Station, 77845, Texas, USA, after data cleaning.
Descriptive statistics was used to summarize the demographic characteristics of the participants, knowledge of the Med Safety App, and opinion on the features of the app. Useful features and challenges associated with use of the app were also assessed.
Pearson's chi-square (χ2) and Fisher's exact tests were used to determine associations between variables such as age, gender, level of education, employment status, and responses about knowledge on ADR reporting. Fisher's exact test was used when the number of counts in the contingency table was less than 5. 14 The significance level was set at p ≥ 0.05.
Comparison of ADR reports received through the app and paper-based CIOMS 1 form
ADR reports received through the Med Safety App (n = 122) and the FDA's paper-based CIOMS 1 form (n = 6825) throughout the study period were compared in terms of completeness of the reports, the sex, age, seriousness, and reporter details. This comparison was focused on larger differences (>20%) between reporting methods, rather than a formal statistical analysis, given the limited number of reports received through the app. We further considered the distribution of reports by first level Anatomical Therapeutic Chemical (ATC) classification and System Organ Class (SOC) using the Medical Dictionary for Regulatory Activities (MedDRA).
Ethical consideration
Ethical approval for the study was granted by the Council for Scientific and Industrial Research Institutional Review Board dated 18 August 2020 with approval no. RPN008/CSIR-IRB/2020.
Participants were informed that a response to the email inviting them to participate in the study represents consent.
Results
A total of 121 participants took part in the study out of the 350 randomly selected persons who had ever downloaded the app, representing a 34.6% response rate. A non-response was defined as participants to whom the questionnaire was sent, and who failed to respond after three follow-ups by email and/or telephone calls within intervals of 3 days.
Demographic characteristics of respondents
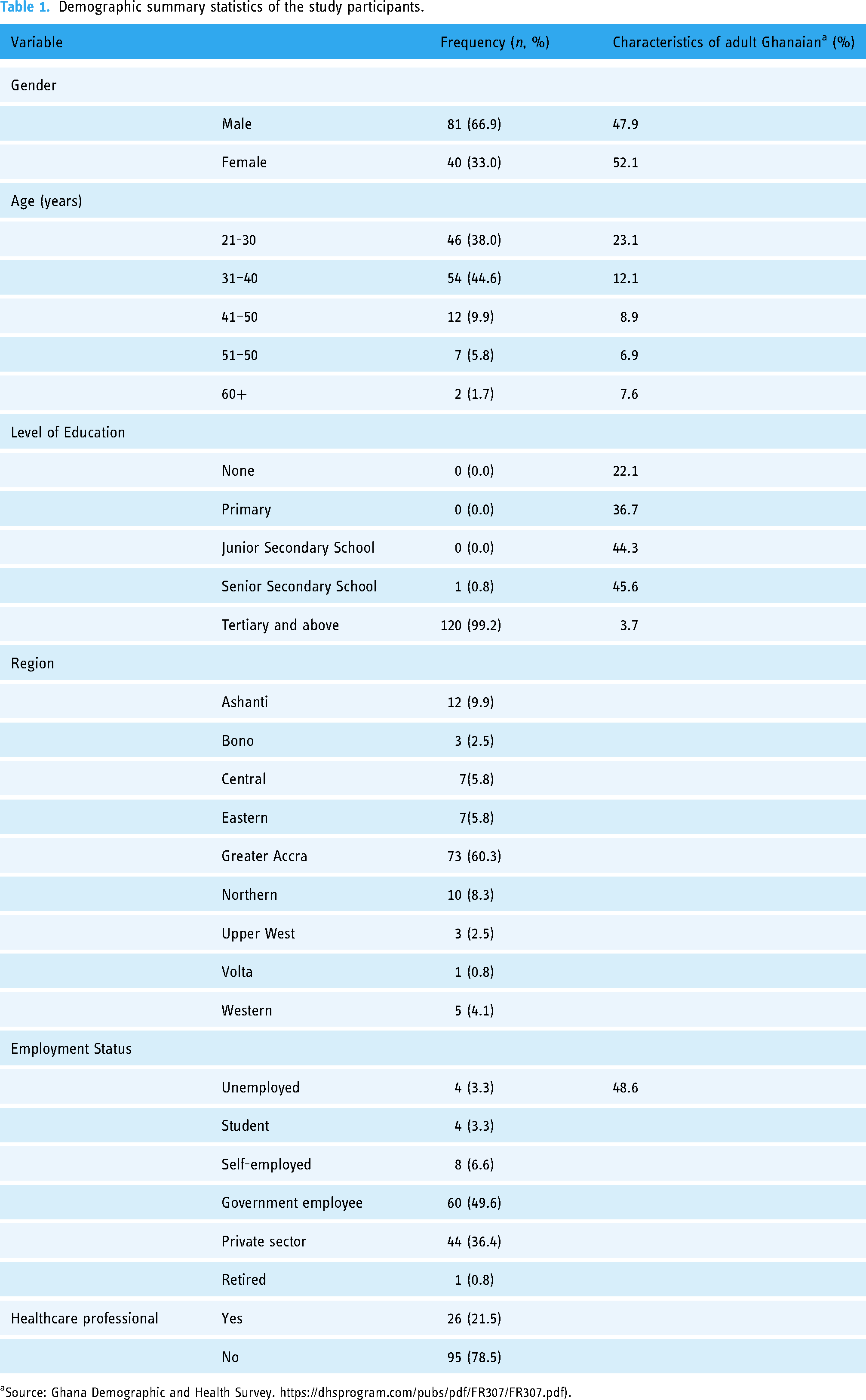
The regions in Ghana with most respondents were Greater Accra and Ashanti regions with 73 (60.3%) and 12 (9.9%) respondents, respectively. There were 81 (66.94%) males with the rest being females. Most respondents were between the age range 31–40 years (54 or 44.6%) and 21–30 years (46 or 38.0%).
Almost all respondents, 120 (99.2%), had tertiary education or above, with 60 (49.6%) of these being government workers.
Demographic characteristics of participants are provided in Table 1.
Demographic summary statistics of the study participants.
Source: Ghana Demographic and Health Survey. https://dhsprogram.com/pubs/pdf/FR307/FR307.pdf).
There was no relationship between the use of the app and the age of the participants (X2 = 2.4, p < 0.118) or gender (X2 = 1.1, p < 0.301).
Ninety-five participants (78.5%) were healthcare professionals, and of these 39 (41.1%) had worked for between 1 and 5 years with 23 (24.23%) having worked for 6–10 years.
Fifty-three (55.8%) of the healthcare professionals were pharmacists, 11 (11.6%) were pharmacy technicians, and nine (9.5%) were nurses. The rest were public health/disease control officers (nine or 9.5%), regulatory officers (three or 3.2%), physician assistants (three or 3.2%), medical doctors (two or 2.1%), and laboratory scientists (two or 2.1%). There were also one (1.0%) each of ayurvedic doctor, medicine counter assistant, laboratory scientist, and a pharma industry representative.
Knowledge on the Med Safety App and its use to reporting ADRs
A total of 116 participants responded to the question on knowledge of the use of the Med Safety App to report ADRs. Of this number, 111 (95.7%) responded that they were aware that the app can be used to report ADRs;
113 (97.4%) respondents downloaded the app themselves with 74 (65.5%) of these indicating that the app was very easy to download. Figure 1 shows the number of responses to the ease of download.

Frequency distribution of ease of download of the Med Safety App (n = 113/121 who downloaded the app themselves).
The reasons given by those who stated that it was not easy to download the app were that they needed help from someone else to download the app, they did not have all the pieces of information needed to fully download, or the app logo did not depict anything concerning drug safety or the National Pharmacovigilance Centre in Ghana or the Food and Drugs Authority and as such the downloaded program was not recognized as the correct app.
Thirty-three of the 121 (27.3%) respondents indicated that they had used the app to report ADRs. Of these, 12 (36.4%) indicated that it was easy to use the app to report ADRs, and three (9.1%) indicated that it was difficult.
With regard to the experience of using the app to report ADRs, nine out of the 33 (27.3%) reported that they easily understood the items required to submit a report with two (6.1%) indicating it was difficult. Seven (21.2%) respondents indicated that they would continue to use the app because it was easier compared to other means of reporting including paper-based reporting (Table 2).
Experiences with the Med Safety App among those who used the app for reporting ADR (n = 33).

Usefulness of the Med Safety App (opinions on the use of the app)
Generally, a large proportion of the respondents agreed that the Med Safety App was a useful tool, and they would recommend it to someone else. At the time of the survey, a total of 75 (62.0%) of the 121 respondents indicated that they were using the app after initial download; however, only 33 of these employed the app to report ADRs. Table 3 provides the responses on the usefulness of the Med Safety App.
Usefulness of the Med Safety App (opinions on the use of the app).

Opinion about the most useful features of the app
There was little difference in perception of the useful features among “news,” “watch list,” and “report side effects.” More importantly, 183 out of 197 found a feature useful, and only eight out of 197 did not found any feature useful. Figure 2 provides the opinions of respondents on the most useful feature of the Med Safety App.

Frequency distribution of responses regarding useful features of the Med Safety App.
The survey also assessed factors limiting the use of the app. Respondents were asked which of the features of the app most concerned them. Options included how visually appealing the app was, how easy it was to understand the information provided in the app, and the font size of the text used in the app. Most respondents making up 87 (75.7%) did not have any concerns with the current design of the app. Thirteen (11.3%), nine (7.8%), and six (5.2%) indicated, respectively, that they had concerns with how visually appealing the app was, how easy it was to understand the information provided in the app, or font size of text used in the app.
Opinion about the use of healthcare mobile technology
Most respondents, 93 (76.9%), indicated that, in general, they were concerned about the privacy and security of their personal information in everyday life when using a mobile health technology.
Despite concerns about privacy and security on the use of mobile health technology, 89 (73.6%) (strongly agree and agree) that they would like information provided during the use of the app to be available to others because this would help in the identification of safety issues of medicines. Table 4 provides responses from participants regarding their opinions on the use of mobile health technology.
Opinion about the use of mobile health technology.

Comparison of ADR reports by submission route
A total of 6947 ADR reports were received within the study period. Of these 6825 (98.2%) were received through the FDA's paper-based CIOMS 1 form, and the remaining 122 (1.8%) through the Med Safety App, with no duplicate reports detected.
We compared the completeness and characteristics of the reports received through the Med Safety App with the paper-based CIOMS 1, although not applying formal statistical tests. We found anecdotal differences of >20% in the completeness in age group, indication of the suspected drug/vaccine, and profession of reporters. Comparing the reports by other attributes, we observed that there was a higher percentage of missing information for the age group in reports originating from the app compared to the paper-based CIOMS 1 form. This is in contrast to missing information for the profession of reporters for which there was a greater percentage of missing information for paper-based CIOMS 1 form (29.6%) compared to none from the app. Table 5 shows the differences between the two submission routes.
Completeness and characteristics of reports received through the Med Safety App compared with paper-based CIOMS 1 reporting form.

There were no differences between the SOC and the first level ATC codes for the ADR reports received through the Med Safety App and paper CIOMS 1 form.
Discussion
This study sought to explore challenges and facilitators for the use of the Med Safety App for reporting ADRs in Ghana and to compare the features of ADR reports received through the app and paper-based CIOMS 1 form. We found that majority of participants indicated that the app is a useful tool and were satisfied with several of the features. There were some differences between reports received through the app and paper-based CIOMS 1 form, which warrant further exploration. Challenges with the app were the ease of downloading the app, understanding the information provided within the app, and the font size of text used in the app.
The sex distribution of our study population was similar to the general Ghanaian population; however, the study population age distribution was lower, and the level of education attained was higher than the average in Ghana. These findings were not surprising because both age and education have been found in other studies to be variables associated with use of mobile technology.15,16
Our study also showed that more males (66.9%) participated in the study; however, there was no significant difference between males and females regarding the use of the app (Table 1). This finding is not consistent with others which reported female gender as a predictor for the use of e-health technologies.15,17 The limitation in our sample size does not allow full exploration of gender differences.
The findings from the study suggest that the app is perceived useful, with the majority of the respondents satisfied with its design and appeal to use, which is reassuring regarding the likelihood of uptake going forward.
Findings from the study suggest that ease of using the app was a facilitator with majority of those who had knowledge of the app using it to report a safety concern. This is similar to the findings from other studies where respondents suggested that apps simplified ADR reporting, making it quick and easy, and reducing delays in reporting. 18 Additionally, digital features such as a defined drug list and data file attachment capability made reporting even easier. 8
The news and the watch list features were perceived by participants as useful in the app, suggesting that they like using a tool that allows two-way communication. The news feature provides information on urgent safety issues from the FDA including warnings, alerts, and direct healthcare professional communication to the users of the app. These are urgently needed by both healthcare professionals and patients for the safe use of new medicines and other health products. Indeed, the objective for the Med Safety App is to be a two-way communication tool between users and the National Pharmacovigilance Centre, allowing communication of drug safety information and other related news in a timely manner and strengthening the engagement of reporters’ interest in drug safety. Additionally, immediate acknowledgment of a report submitted will make reporters more committed to reporting as they believed their contribution was important. 8 Further, use of the news and watch list features may have longer-term additional benefits to health care workers or patient users of the app, essentially as educational tool and/or reduction in ADRs through earlier detection.
Participants in our study expressed concern about the privacy and security of personal information and sharing of this information on online, yet they were all willing to share information via the app with others, in order to help in the identification of safety issues. Our finding is the same as what was obtained from earlier studies which found altruism as a motive for reporting ADRs by patients.19,20
The findings in our study are consistent with those from an exploratory qualitative study by Kiguba R et al. 18 among health workers in 12 HIV clinics in Uganda, which also found willingness among the health workers to adopt the Med Safety App for ADR reporting with the majority admitting that they will recommend the app to other health workers. Potential barriers to the uptake of the Med Safety App in this study were perceived lengthy processes of initial app registration and completion of multiple screens during ADR reporting.
This study compared the description of the characteristics and quality of reports received through the Med Safety App and paper-based CIOMS 1 form. There were some observed differences in the completeness of the ADR reports received through the Med Safety App and the paper-based CIOMS 1 form for age group, which had better completeness percentage with the Med Safety App compared with paper-based CIOMS 1 reporting form. A greater number of pharmacists used the Med Safety App to report compared to CIOMS 1 paper form. It is noteworthy to be upfront that these observations should be interpreted with caution, given the limited number of reports received through the app. The reason for the use of the app by more pharmacists could be due to the fact that the app was discussed with pharmacists during the annual general meeting in August 2019, 2 months after the launch of the app, which may explain why pharmacists downloaded the app and using it to report ADRs compared to other healthcare professionals. The reason for receiving a greater percentage of serious reports through the paper-based CIOMS 1 form compared to the Med Safety App is unknown.
Strength and limitations of study
To the best of our knowledge, this is the first study to explore challenges and facilitators for the use of the Med Safety mobile app in Ghana.
The first limitation is the smaller than expected sample size given the response rate of 34.6%. However, while underpowered, we believe that this is the single largest convergent mixed-method study to date exploring the feasibility and acceptability of the use of the Med Safety App in a low-resource setting in Africa. As such, the data collected can help inform especially future implementation research studies and/or approaches to the roll out of the use of the app.
Further, there may have been selection bias with regard to the respondents, since the pool of participants was limited to those who had downloaded the app and importantly, among those, potentially a select group of individuals took part in the study (i.e. among users, respondents may be different from those that did not take part in the study). Additionally, more males took part in the study, which is inconsistent with the male:female ratio in Ghana. However, since this was a first implementation approach and the primary personnel who report ADRs tend to be health care professionals (e.g. pharmacists), as was indeed the case in our study, and that the proportion of males increases with higher education status, these data are not surprising.
Conclusion
The modest number of participants in our study indicated that the Med Safety App is a useful tool, easy to use, and that they were satisfied with the features of the app. The positive findings from this study should be used to broaden the roll out of the app in Ghana and, if successful, to other countries.
To disseminate the Med Safety App and make it attractive for potential users in Ghana, the following is being recommended: First, resources should be invested into advocacy and awareness creation on the benefits of the app to improve uptake and use since majority of participants in our study were using the app at the time of the study. Second, the app should be promoted to patients and healthcare professional groups at their meetings and professional social media platforms to improve its use. Lastly, implementation research focusing on the app should be conducted to scientifically identify interventions which need to be implemented to improve the use of the app by patients and healthcare professionals.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231211276 - Supplemental material for First results from the lessons learnt from the deployment of the Med Safety App for reporting adverse drug reactions in Ghana
Supplemental material, sj-docx-1-dhj-10.1177_20552076231211276 for First results from the lessons learnt from the deployment of the Med Safety App for reporting adverse drug reactions in Ghana by Seth Kwaku Seaneke, Delese Mimi Darko, Edwin Nkansah, Abena Asamoa-Amoakohene, Adela Ashie, Jeremiah Sampson Ewudzie, Phil Tregunno, Marie-Eve Raguenaud, Corinne S. Merle, Branwen J Hennig and George Tsey Sabblah in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to greatly thank all the survey participants who took their time to complete the questionnaire.
Contributorship
GTS contributed to the study design, developing data collection tool, data analysis, writing original draft, and writing (review and editing). DMD, SKS, EN, PT, MR, CM, and BJH contributed to the review and editing of the manuscript, technical support, and supervising. AA, JES, and AAA contributed to the study design, participants’ recruitment, data analysis, and writing the original draft of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The Council for Scientific and Industrial Research Institutional Review Board approved this study (No. RPN008/CSIR-IRB/2020).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Special Program for Research and Training in Tropical Diseases (TDR) through the Access and Delivery Partnership (ADP) program grant. ADP is supported by the Government of Japan and led by the United Nations Development Program, in collaboration with the World Health Organization, TDR, and PATH.
Guarantor
GTS
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.