Abstract
Introduction
Drug prior authorization (PA) imposes a bureaucratic and economic burden on healthcare service providers and payers. A novel automated PA system may improve these drawbacks.
Methods
An historical cohort study from a large health maintenance organization in Israel, comparing manual versus automated PA mechanisms for diabetes mellitus (DM) drugs: sodium-glucose co-transporter-2 inhibitors (SGLT2i) and glucagon-like peptide-1 analogs (GLP1-A). We compared patients with DM, whose first drug applications were approved using the automated system, with similar patients whose first drug applications were approved by manual PA. The primary endpoint was the time elapsed from application approval to prescription filling (accessibility time). Secondary endpoints included the prescription filling rate at 7 and 30 days.
Results
In total, 1371 automated approved prescriptions and 1240 manually approved prescriptions were included in the analysis. Median accessibility time was one day (interquartile range (IQR) 0–5) with automated PA for both GLP1-A and SGLT2i, compared with four days (IQR 1–9) and three days (IQR 1–8), respectively, with the manual PA (
Conclusions
Automated PA system improved access time to SGLT2i/GLP1-A seven days post-approval compared to manual PA.
Keywords
Introduction
The Israeli national health insurance law mandates that all citizens are entitled to public insurance that covers a uniform list of services (the health-basket). Four health maintenance organizations (HMOs) provide these services, and each citizen is insured by one of them. Every year the health-basket is further expanded with new medicines and technologies via a mechanism called the “Basket Committee”—a dedicated committee appointed by the government whose task is to determine which of the suggested technologies will be added to the health-basket under public funding. 1 This committee defines eligibility criteria for the newly added technologies based on the patient's medical condition, considering national economic, equality, and ethical aspects to balance budgetary constraints and the need for high-quality, advanced therapies. Diabetes mellitus (DM) drugs such as Sodium-Glucose co-transporter 2 inhibitors (SGLT2i) and glucagon-like peptide-1 analogs (GLP1-A) that were added to the health-basket in recent years represent technologies with proven clinical benefits, and challenging economic burden due to a large number of eligible patients. Eligibility criteria for SGLT2i and GLP1-A in the Israeli health-basket are detailed in Appendices A and B, respectively.
Like many other healthcare insurers worldwide, Israeli HMOs operate prior authorization (PA) centers in which physicians, pharmacists, and administrative staffs review each application for funding of drugs and ensure it complies with the health-basket's criteria. Healthcare organizations and payers worldwide use this mechanism to control drug expenditure under different reimbursement policies. 2 Meuhedet, the third largest HMO in Israel, which insures 14% of the Israeli population, operated five PA centers (a central headquarter and four regional centers) that examined and provided prior approvals to tens of thousands of applications annually. Roughly 10% of the publicly funded drugs in Israel require prior approval, and the expenditure on these drugs accounts for over 70% of total drug spending in Meuhedet.
Nevertheless, authorization centers have been shown to impose a significant bureaucratic obstacle on both patients and caregivers, often depriving patients of essential treatment.3–6 A recent survey among physicians in the US found that 92% of drug applications are delayed by PA, leading to a 75% treatment abandonment rate among patients, often leading to adverse clinical outcomes. In addition, PA has been related to a significant overload in physicians’ and staff's labor. 7 Also, operating PA centers poses an additional economic burden. A Council for Affordable Quality Healthcare (CAQH) report published in 2019 concluded that the US healthcare industry could save $13.3 billion on administration through automation, with potential annual savings of $454 million by transitioning to electronic PAs. 8 The above has led to the publication of a conjoint Consensus Statement on Improving the PA Process by the American Medical Association and other organizations. 9 Several automated solutions have been previously suggested, focusing mainly on automating the submission phase rather than automation of the entire approval process. 10
Meuhedet had developed and launched an automated, innovative system that responds immediately to the applying physicians. The system includes an algorithm-based SAP software engine, which interfaces with various databases, including electronic medical records, laboratory test results, and pharmacies’ drug acquisition data. These inputs translate into an immediate decision upon applying for a drug: approval (if all the criteria are met), rejection (if not all criteria are met), or request for data completion (in case of missing data). Once approved, a digital prescription is recorded, and the patient can purchase the reimbursed medication at the pharmacy; in case of rejection, the patient is not entitled to public drug funding.
Despite the significant improvement expected from our novel automated process, it remains unclear whether it translates into faster patient access to the requested drug compared to the current manual PA. To the best of our knowledge, this is the first study to examine this issue.
In this study, we aimed to examine whether using an automated real-time authorization system improves the performance of PA in terms of accessibility, adherence to treatment, and staff worktime overload compared to a manual PA, using SGLT2i and GLP1-A drugs as representative cases.
Methods
A historical cohort study based on Meuhedet's database compared two mechanisms for PA of DM drugs: manual versus automated.
Study population and inclusion criteria
We identified data of all patients with DM for whom a first SGLT2i or GLP1-A application was approved. The “automated group” included all patients whose applications were approved using the automated system, whereas the control group consisted of patients whose applications were approved by manual PA. Patients from both groups were aged 18 years and above with DM who had been prescribed SGLT2i or GLP1-A for the first time during the study periods. We included only GLP1-A/SGLT2i naïve patients to discard requests for treatment extensions that patients may fill with a delay since they still have enough doses of the prescribed drugs at the time of prescription renewal.
Meuhedet launched the automated approval for SGLT2i on March 2020; hence the automated group included applications from 15 March 2020 to 31 December 2020 and the control group from 15 March 2019 to 31 December 2019. Meuhedet launched the automated approval for GLP1-A on November 2020; the automated group included applications from 15 November 2020 to 15 July 2021, while the control group included applications from 15 November 2019 to 15 July 2020.
While health-basket criteria for GLP1-A were the same during 2019 and 2020, the 2019 health-basket criteria for SGLT2i were expanded in January 2020 to include DM patients with reduced renal function with albuminuria (Appendix A). The automatic PA system was adjusted accordingly to fit this update.
For each group, we collected demographic and clinical data: age at treatment initiation, gender, geographic residency, socioeconomic status (SES), duration of DM, applying physician's specialty (primary care or diabetes specialist), laboratory tests, weight and body mass index before beginning the treatment with SGLT2i/GLP1-A (HbA1C, eGFR), diabetes medications used before SGLT2i/GLP1-A initiation, and the number of drugs used in combination with SGLT2i/GLP1-A. We also extracted information regarding the patient's co-morbidities: ischemic heart disease, stroke, peripheral vascular disease, heart failure, chronic kidney disease, chronic lung disease, and dementia; last hospital admission date before drug prescription and hospitalization code, number of visits to a primary physician from the moment of receiving the drug prescription until its purchase.
Study endpoints
As our primary endpoint, “accessibility time,” we used the time elapsed from the approval of the physician's application for SGLT2i or GLP1-A to the drug's first dispense to the patient at the pharmacy.
The secondary endpoints included: the percentage of prescription filling at 7 and 30 days, the change in the volume of manual applications after the automated system's introduction, and the evaluation of direct cost savings attributed to operating the automated PA system.
Statistical analysis
We described the categorical variables as frequencies and proportions. Using histograms, we evaluated the continuous variables for normal distribution. We described the normally distributed variable as means and standard deviations, and non-normally distributed as medians and interquartile ranges (IQR).
We analyzed SES as a three levels categorical variable: low (1–3), medium (4–6), and high (7–10).
We used the chi-square test for comparing categorical variables and the Mann–Whitney test for non-normally distributed continuous variables. We used logistic regression to analyze the contributing variables for prescription filling within 7 and 30 days. All the statistical tests were two-sided, with a
We used R version 4.1.0 (R Foundation for Statistical Computing) for all statistical analyses.
This study was exempt from informed consent by Meuhedet's Institutional Review Board, approval number 04-24-08-20.
Results
We have extracted 1184 digitally approved and 944 manually approved prescriptions for a GLP-1A medication. We excluded 158 (13%) of the digitally approved prescriptions and 98 (10%) of the manually approved which were not filled within 90 days (
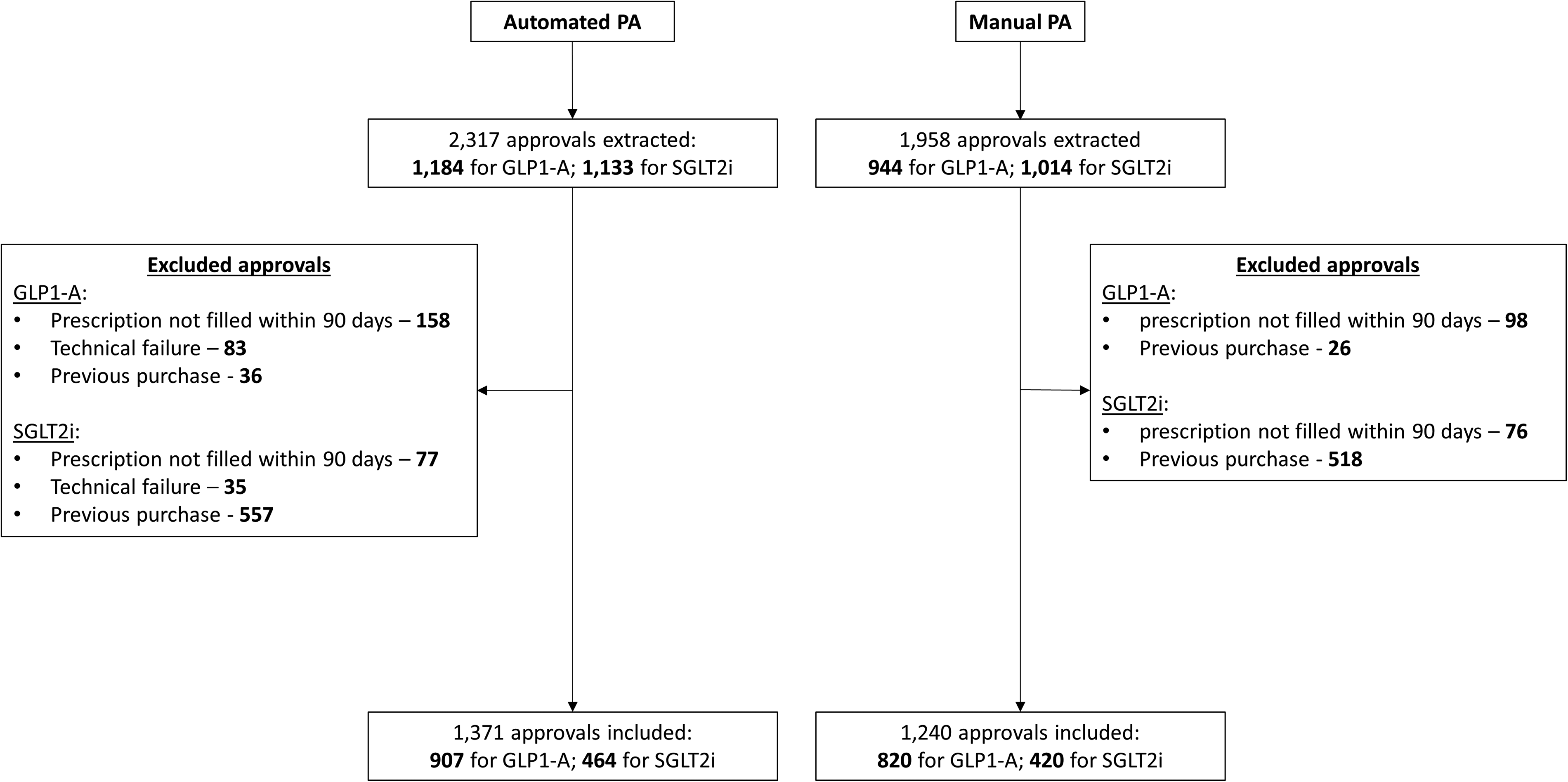
A flow chart of automated and manual applications for GLP1-A and SGLT2i. GLP1-A: glucagon-like peptide-1 analogs; SGLT2i: sodium-glucose co-transporter-2 inhibitors.
We have extracted 1133 digitally approved and 1014 manually approved prescriptions for SGLT2i medications. We excluded 77 (7%) digitally approved and 76 (7%) manually approved prescriptions, which were not filled within 90 days (
The demographic characteristics of all GLP1-A and SGLT2i patients included in the analysis are presented in Table 1.
The characteristics of the patients who received the prescriptions.

DM: diabetes mellitus; GLP1-A: glucagon-like peptide-1 analogs; IQR: interquartile range; SES: socioeconomic status; SGLT2i: sodium-glucose co-transporter 2 inhibitors.
For GLP-1 cohort: 4.6% were missing, for SGLT-2 cohort: 2.4% were missing.
For GLP-1 cohort: 0.1% were missing, for SGL-T cohort: 0.1% were missing.
For GLP-1 cohort: 13.1% were missing, for SGL-T cohort: 26.7% were missing.
For GLP-1 cohort: 1.6% were missing, for SGL-T cohort: 2.5% were missing.
For GLP-1 cohort: 2% were missing, for SGL-T cohort: 2.8% were missing.
GLP1-A accessibility time
The median delay between application and manual PA approval was one day (IQR 0–1), whereas the automated system provided an immediate approval. The median delay between automated approval to prescription filling was one day (IQR 0–5), compared with four days (IQR 1–9) for the manually approved applications (
We fitted logistic regression models for prescription filling within one week and one month, controlling for the patient's gender, age, SES, and diagnosis of COPD. The odds for prescription filling within a week were 2.21 folds higher for patients who received digitally approved prescriptions (
logistic regressions for GLP1-A prescription filling within a week and a month from approval

CI = confidence interval; COPD = chronic obstructive pulmonary disease; GLP1-A = glucagon-like peptide-1 analogs; OR = odds ratio; SES = socioeconomic status
SGLT2i accessibility time
The median delay between application and manual approval was one day (IQR 0–1). The median delay between digital approval to prescription filling was one day (IQR 0–5), compared with three days (IQR 1–8) for the manually approved prescriptions (
We fitted logistic regression models for prescription filling within one week and one month, controlling for the patient's gender, age, SES, having the diagnosis of stroke, number of years since the diagnosis of DM, the specialty of the prescribing physician (diabetes specialist or other), and eGFR < 60 mL/minute/1.73 m2. The odds for prescription filling within a week were 1.48 folds higher for patients who received digitally approved prescriptions (
logistic regressions for SGLT2i prescription filling within a week and a month from approval

CI = confidence interval; DM = diabetes mellitus; eGFR = estimated glomerular filtration rate; OR = odds ratio;
SES = socioeconomic status; SGLT2i = sodium-glucose co-transporter 2 inhibitors
Direct cost savings attributed to operating the automated PA system
Compared to the period before its introduction, the automated PA system reduced the total number of manual PA applications by 40%, which allowed significant streamlining and re-organizations measures, including the closure of four regional manual PA centers, resulting in an annual administrative cost savings of roughly 1.9 million Israeli Shekel (ILS) or US$566,000 (according to 4 May 2022 exchange rates).
Discussion
Although necessary for expenditure control, drugs’ PA centers impose a significant bureaucratic barrier regarding patients’ access to treatment. Our study found that incorporating an innovative, automated drug authorization system improved accessibility time to SGLT2i and GLP1-A drugs, reduced worktime overload of manual PA centers by roughly 40%, and saved as much as US$566,000 annually by efficiency process.
Our study found no differences in overall rates of first prescription filling within 30 days between automated and manual PA approvals. However, automated PA improved the short-term rate of the first prescription filling (within seven days) compared to manual PA for SGLT2i and GLP1-A. A possible explanation for this discrepancy may be that DM is a chronic condition in which most patients comply with the treatment and fill their first prescription. Similar findings in various chronic conditions support these results: Shah et al. demonstrated similar behavior in DM and hypertension patients, where 85% and 83% of first-prescribed antidiabetic and antihypertension drugs were filled within 30 days, respectively.11,12 Somewhat lower rates were found for asthma medication. 13
Short-term purchase of the first prescription might predict the likelihood of long-term persistence with treatment. Jensen and colleagues had developed an algorithm that predicts long-term adherence to medications among patients with different chronic diseases, including metformin for DM, based (among other factors) on the time that elapsed from first prescription writing and its actual filling. 14 Delays in treatment may eventually translate into a substantial risk for clinical sequelae, as previously reported for various conditions.6,15–17
A higher benefit is expected when this system is used for medications for acute conditions, where a short delay in treatment initiation may result in more severe outcomes (e.g. antibiotics, antifungal, and antiviral drugs).
Our findings were inconsistent with those of Lauffenburger et al., who evaluated the impact of implementing an electronic PA system on prescription filling and found that the prescription filling rate at 30 days was significantly lower with the electronic PA system. 10 The different results of the two studies may result from the differences between the two PA systems: our system provided immediate response while Lauffenburger's system facilitated only the first step and informed the physician whether a specific drug required PA, but the approval itself was still done manually.
Our automated PA system decreased manual PA's workload and related economic burden. Numerous studies have evaluated the economic impact of operating manual PAs.18,19 Carroll et al. described the effect on the expenditure for cyclooxygenase-2 (COX-2) inhibitors before and after introducing an automated PA system (SmartPA). 20 This study showed substantial savings on COX-2 inhibitors (mainly through approval of reduced doses) after incorporating SmartPA compared to the previously used manual PA. However, in contrast to our study, they did not examine the system's impact on the administrative costs of PAs.
We could not find studies that examined the effect of an automated system on reducing the workload or economic burden of manual PAs. However, the reduction in the volume of applications for manual PAs and the operational cost savings, shown by our study, were not surprising. The automated system may also achieve various other goals, such as uniformity across organizational units, improving physicians’ and patients’ satisfaction with the PA process, and preventing the growing overload of PA centers, which originates from introducing new medications and technologies. These issues are to be examined in future studies.
Our study had several limitations. First, our automated PA system was launched during the COVID-19 pandemic, a period that was characterized by a general decrease in healthcare services consumption.21,22 We could not adjust for these changes; however, the fact that the accessibility time during this period was lower as compared to the corresponding period before the pandemic, implies that this factor had no significant effect on our results. Another possible limitation relates to the 2020 SGLT2i health-basket expansion for additional renal patients, which might have affected the behavioral profile of these patients who were eligible for these drugs only during the automated PA period. Fifty percent of the patients that received approval for SGLT2i were already treated with a medication of this class; we excluded them from the analysis to avoid bias, as we suspected that they might have some doses of the medication from previous prescriptions. Although our study did not aim to explore this issue, the clinical effect of such intervention remains to be determined. In addition, complementary data on physicians’ and patients’ satisfaction from the automated system remains to be examined.
Conclusions
Our automated PA system reduced delays in prescription filling for SGLT2i and GLP1-A during the seven days post-approval. It also reduced the workload and economic burden of manual PAs.
Contributorship
Shai Moshel contributed to conceptualization, supervision, methodology, investigation, writing—original draft, literature search, figures, study design, data collection, data interpretation, and verification of the underlying data. Shmuel Klang contributed to project administration, conceptualization, and resources. Revital Nikname contributed to data curation, software, supervision, and validation. Katy Bar Shalom contributed to project administration, conceptualizations, and resources. Dov Albokerk contributed to writing—review and editing. Galia Zacay contributed to formal analysis, conceptualization, methodology, investigation, writing-original draft, study design, data collection, data interpretation, and verification of the underlying data.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231203889 - Supplemental material for Automated versus manual prior authorization for diabetes mellitus drugs: A retrospective study from Israel
Supplemental material, sj-docx-1-dhj-10.1177_20552076231203889 for Automated versus manual prior authorization for diabetes mellitus drugs: A retrospective study from Israel by Shai Moshel, Shmeul Klang, Revital Nikname, Katy Bar Shalom, Dov Albukrek and Galia Zacay in DIGITAL HEALTH
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was reviewed and approved by Meuhedet's Institutional Review Board (approval number 04-24-08-20).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Galia Zacay.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
