Abstract
Background
Children with hypoglycaemia disorders, such as congenital hyperinsulinism (CHI), are at constant risk of hypoglycaemia (low blood sugars) with the attendant risk of brain injury. Current approaches to hypoglycaemia detection and prevention vary from fingerprick glucose testing to the provision of continuous glucose monitoring (CGM) to machine learning (ML) driven glucose forecasting. Recent trends for ML have had limited success in preventing free-living hypoglycaemia, due to a focus on increasingly accurate glucose forecasts and a failure to acknowledge the human in the loop and the essential step of changing behaviour. The wealth of evidence from the fields of behaviour change and persuasive technology (PT) allows for the creation of a theory-informed and technologically considered approach.
Objectives
We aimed to create a PT that would overcome the identified barriers to hypoglycaemia prevention for those with CHI to focus on proactive prevention rather than commonly used reactive approaches.
Methods
We used the behaviour change technique taxonomy and persuasive systems design models to create HYPO-CHEAT (HYpoglycaemia-Prevention-thrOugh-Cgm-HEatmap-Assisted-Technology): a novel approach that presents aggregated CGM data in simple visualisations. The resultant ease of data interpretation is intended to facilitate behaviour change and subsequently reduce hypoglycaemia.
Results
HYPO-CHEAT was piloted in 10 patients with CHI over 12 weeks and successfully identified weekly patterns of hypoglycaemia. These patterns consistently correlated with identifiable behaviours and were translated into both a change in proximal fingerprick behaviour and ultimately, a significant reduction in aggregated hypoglycaemia from 7.1% to 5.4% with four out of five patients showing clinically meaningful reductions in hypoglycaemia.
Conclusions
We have provided pilot data of a new approach to hypoglycaemia prevention that focuses on proactive prevention and behaviour change. This approach is personalised for individual patients with CHI and is a first step in changing our approach to hypoglycaemia prevention in this group.
Keywords
Introduction
Congenital hyperinsulinism (CHI) is a disease of disordered and unregulated insulin secretion which results in severe and unpredictable hypoglycaemia (low blood glucose). The definition of hypoglycaemia for children with CHI has not reached an international consensus but a pragmatic cut-off of <3.5 mmol/L is frequently used in UK centres. 1 Regardless of the cause, recurrent hypoglycaemia episodes (hypos) in early childhood can have a long-term impact on developmental outcomes.2,3 The clinical management of CHI is restricted by therapy choices and by the absence of suitable monitoring solutions that reasonably predict the possibility of hypoglycaemia. CHI is the commonest cause of recurrent and severe hypoglycaemia in early childhood and rates of neurological impairment range from 15% 4 to almost 50%. 5 Non-clinical outcomes from hypoglycaemia are also significant, with effects on quality of life and the healthcare economy.6,7
Given the serious impact of hypoglycaemia on children with CHI and other conditions associated with hypoglycaemia (such as Type 1 diabetes mellitus (T1DM)), prediction and prevention are vital. Standard practice for children with CHI is the use of a limited repertoire of disease-modifying drugs combined with intermittent fingerprick glucose testing to reduce, detect and prevent hypos. 8 However, the low granularity and lack of trend information provided by fingerprick testing result in missed hypoglycaemia between tests and offer little ability to predict future hypos. Consequently, there has been a surge in interest in using continuous glucose monitoring (CGM) and modern algorithmics to accurately predict future glucose levels with an aim to anticipate and prevent hypoglycaemia. The reporting of glucose values every 5 minutes by CGM devices has been shown to improve the amount of time patients spend with glucose in a safe and desirable range (time in range (TIR)) for those with T1DM 9 but not yet in patients with CHI.10,11 Further limitations to the use of CGM for patients with CHI relate to the suboptimal point accuracy and hypoglycaemia sensitivity seen 12 and the burden of alarms and sensor changes reported. 13 The more recent trend to try and prevent hypoglycaemia through the forecasting of future values with modern algorithmics is a somewhat flawed approach that ignores the human in the loop and the importance of considering behaviour change rather than simply providing more information. 14
To this end, we developed
Research motives and objectives
Given the obvious clinical need for a reliable way to reduce hypoglycaemia and the insufficiency of the current approach, there has been great interest in recent years in designing an algorithm to help prevent hypoglycaemia. Most approaches have focused on the generation of increasingly accurate in silico predictions of future glucose values using machine learning (ML) and continuous CGM. We have detailed elsewhere the significant limitations of this approach to predict future glucose values when the aim is to actually prevent hypoglycaemia, 14 and, in the interests of presenting a complete picture, we briefly summarise these below.
Multiple groups have shown good accuracy in forecasting future glucose values but only when tested in silico and thus provide no indication of their ability to actually reduce hypoglycaemia in vivo.15–18 Decision support systems (DSSs) have demonstrated reductions in hypoglycaemia but also in silico 19 or with vastly complex inputs required by patients. 20 Those DSSs that have been evaluated in vivo mostly fail to demonstrate a reduction in hypos.21–24 Liu et al. 25 did demonstrate a reduction in hypos in patients in free-living conditions from their DSS which recommended insulin doses, but this has no applicability to patients not receiving therapeutic insulin such as those with CHI.
While the use of in silico testing is just a gap in the current evidence which could (and likely will) be filled, there is also a fundamental flaw in the underlying theory of such approaches. They all ignore the essential fact that simple provision of information does not necessarily translate into action and a resultant improvement in outcomes. A vital determinant of health outcomes is health behaviours 26 and, in particular, a capacity to self-manage. The Fogg behaviour model (FBM) outlines three components required to change behaviour: motivation, ability and trigger (prompt); all must converge at the same point for behaviour change to occur, 27 in this case, action to prevent or terminate a hypo. Provision of information such as a predicted future glucose value serves as a simple prompt (if the user is even motivated to look at it) and is unlikely to successfully change behaviour if the approach fails to address motivation and ability. We have demonstrated in previous work 28 that, despite the provision of hypo prompts through real-time CGM (live values displayed constantly and alarms available) to patients with CHI, there were still significant numbers of prolonged hypos. Therefore, future work must focus on all aspects of behaviour change rather than simply provide more complex algorithms for the generation of slightly more accurate prompts.
To this end, we developed HYPO-CHEAT: an algorithm to aggregate CGM data and present this to patients with the express intention of changing behaviour and subsequently reducing hypoglycaemia. We have described the technical development of the algorithmics behind HYPO-CHEAT in a separate publication 14 and discussed the evidence-based methodology of its development as a data aggregation tool. In this paper, we aim to describe the essential behaviour change and persuasive technology (PT) elements of HYPO-CHEAT that were designed to ensure that the information provided to patients translated into a change in behaviour and ultimately improved outcomes. HYPO-CHEAT was designed through the selection of behavioural theory-informed behaviour change techniques (BCTs) 29 and the subsequent use of the O/C (outcome/change) matrix and PSD model 30 to ensure the technical delivery of the system was considered and persuasiveness was maximised.
When evaluating behaviour change interventions, Klasnja et al. 31 highlight the difficulty in demonstrating a definitive change in behaviour without large-scale studies performed over long periods of time. In the absence of such studies, it is vital to measure proximal outcomes to assess if and how the system is working, as well as seeking stakeholder feedback to understand why the system is or isn’t working and how it could be improved. 31 This allows researchers to better understand the relationships between intervention and mediator, and mediator and outcome, so that both failed and successful interventions can provide more useful information. 32 Kwan et al. 33 highlighted this when reporting that nudge interventions improved the distal outcome of HbA1C (biochemical surrogate marker of diabetes complication risk 9 ) but not glucose monitoring behaviours (proximal outcome). Thus, the mechanism for improvement was not clear as the intervention changed the outcome but not via the predicted mediator.
With this in mind, we provide the results of an initial evaluation of HYPO-CHEAT's ability to change behaviour measured through both proximal and distal outcomes, as well as a thorough and independent report of patient feedback on the system via interview and thematic analysis. By reporting multiple measures of our approach's efficacy, we aim to better describe how and why our approach works in a pilot setting and thus contribute useful information to multiple areas of the knowledge base and future large-scale trials, rather than simply reporting a final distal outcome of our system with no understanding of how this was arrived at.
We evaluated HYPO-CHEAT in 10 patients with CHI who were using CGM for 12 weeks in free-living conditions. We discussed the patterns identified by HYPO-CHEAT with patients and their families and explored the behavioural aspects behind these patterns. We monitored changes in fingerprick behaviour as a proximal measure of HYPO-CHEAT's ability to influence health behaviours and patients’ engagement with the system. Finally, reduction in hypoglycaemia (time below range (TBR)) was evaluated as a primary endpoint to assess if HYPO-CHEAT was able to influence more distal outcomes over a short time period and thus potentially change health outcomes in the future. TBR is a practical measure of time spent in hypoglycaemia and, given the UK consensus on hypoglycaemia cut-offs, 8 is a useful outcome marker.
However, defining a minimum clinically important difference (MCID) in TBR has not been described before in CHI as CGM-defined hypoglycaemia has not yet been correlated with neurodevelopmental outcomes and is not immediately translatable to clinical practice but is a retrospective monitoring tool. We, therefore, had to define the MCID for this study. We used the distribution approach 34 based on work by Cohen 35 describing differences based on standard deviation (SD) from the mean as small (0.2SD units), medium (0.5SD) or large (0.8SD). The SD for TBR for patients with CHI was calculated from a large and heterogeneous dataset as 3.05%. 36 A medium difference for TBR, in this patient group, is therefore (0.5 × 3.05%) 1.5%, and this absolute reduction was used as the MCID for this work.
By concentrating on patient behaviour and involving patients from the very start of our approach, we have been able to design a system that aggregates and presents data in a way that is easy to understand. Consequently, it has the potential to change health behaviours and improve health outcomes for patients in free-living conditions in the immediate future.
Our primary objectives were:
Assessment of HYPO-CHEAT's ability to change proximal fingerprick behaviour around times of predicted hypoglycaemia Assessment of the difference in TBR between a period of blinded CGM before using HYPO-CHEAT and blinded CGM after using HYPO-CHEAT (efficacy of HYPO-CHEAT)
Background and related work
As our approach was to use HYPO-CHEAT to change behaviour, here we summarise the background theories and related work from the fields of behaviour change, digital behaviour change interventions (DBCIs) and PT rather than focusing on different approaches to detection and prevention, which are summarised in Section 2 and discussed at length elsewhere. 14
Behaviour change
Vitally and intrinsically linked to health outcomes are health behaviours. 26 Behaviour change is thus a practical target for those wanting to alter health outcomes. Below, we outline the key concepts in understanding the wider context of this work.
Behaviour change theories. The use of an established theory has traditionally been seen as an integral step in the design of an intervention to improve health. 37 This allows the determinants of change to be established, the component BCTs selected29,38 and theoretical mediators of effect to be investigated. 39 However, there are more than 80 behaviour change theories for researchers and developers to choose from, 40 and it can be challenging to choose the most efficacious theory. 41
BCTs. Michie and Abraham developed a taxonomy of BCTs (BCTTv1)42–44 to allow for the provision of a theoretically underpinned approach to intervention design without the need for an in-depth knowledge of all behaviour change theories. The BCTTv1 contains 93 BCTs (in 16 categories) that have the potential to change a behaviour with interchangeable terms to promote accurate replication and faithful implementation of effective interventions. 44
Van Achterberg et al. 45 evaluated 23 systematic reviews and concluded that, while no BCTs demonstrated a consistent effect in all studies, those that were effective most often were self-monitoring of behaviour, risk communication and the use of social support. Closer to our intended patient population are those with diabetes, and within this group, Presseau et al. 46 applied the BCTTv1 to numerous interventions for managing diabetes and found that those utilising BCTs were often effective in changing behaviour and most commonly included: adding objects to the environment, credible source, goal setting and feedback on behaviour.
Digital Behaviour Change Interventions (DBCIs)
BCTs are brought together to form DBCIs that ‘use computer technology to promote behaviour change’. 47
Webb et al. 48 analysed 85 DBCI studies including data from 43,000 participants and found those interventions targeting a single behaviour had more effect than those targeting multiple behaviours. The most effective BCTs (not specifically related to abstinence maintenance) were to demonstrate the behaviour, social comparison, goal setting, action planning, feedback and barrier identification/problem-solving. 48 The most effective mode of delivery was automated tailored feedback and access to advisor to request advice. 48 This meta-analysis informed the development of HYPO-CHEAT to select the BCTs and method of delivery with the most evidence for an effect on behaviour change.
Persuasion
As interventions become more interactive and adaptive, current behaviour theories may be inadequate, 49 and the technology itself must be considered. 50 Parallel to behaviour change theory, computer science evolved specific theories such as the technology acceptance model 51 and unified theory of use and acceptance of technology 52 to take account of the computer as an intrinsic part of the system rather than simply a mode of delivery.
In 2003, Fogg coined the term PT 53 with its own background theories upon which interventions can be based. 54 Subsequently, PT systems created by authors such as Fogg and Oinas–Kukkonen allow for the development of evidence-based and replicable PT interventions without a comprehensive study of background theories27,55 while simultaneously considering the computer as a vital component of the system.
Persuasive technology (PT). Human-computer persuasion 56 forms the basis of PT and involves the persuasion of individuals by the computer rather than through the computer (e.g. via email). 57 PT targets automation of behaviour change 27 through influencing the determinants of behaviour, such as attitude, beliefs and social norms, to improve the users’ intentions to perform the target behaviour. 58
PT can operate as a tool to facilitate target behaviour, through the exploration of cause and effect and as a social actor. 53 Each of the three basic components can be further subdivided to provide practical strategies to persuade and bring about behaviour change. Fogg 53 detailed these subcomponents in his original work, and they have subsequently been refined and improved upon by multiple authors as discussed below.
Behaviour change support systems. The implementation of PT is referred to as a persuasive system 57 or a behaviour change support system (BCSS) 55 dependent on the date of publication. The structure for the development of BCSSs has evolved over many years, and here we provide an insight into the major developments and how they relate to our work.
The persuasive systems design (PSD) model describes three stages to designing a persuasive system which is outlined in Figure 1 30 and utilised in Section 4.1.3.

The persuasive systems design (PSD) model. First, one must understand the seven key postulates which exist behind the persuasive system. Second, the persuasion context can be understood as comprising intent, event and strategy. Finally, the persuasive software features highlight the importance of considering the technology as part of the system and provide a structure for the design of a persuasive system. Figure from Oinas-Kukkonen. 55 .
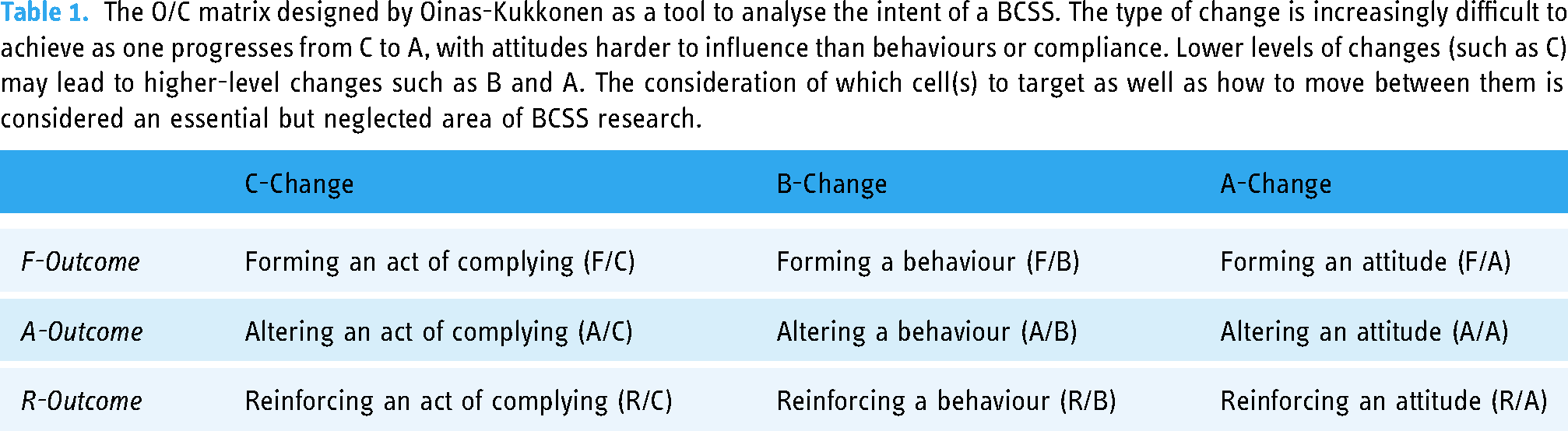
The key component of intent (Figure 1) was further refined by the O/C matrix (Table 1), allowing for an analysis of system persuasive potential. 55 This approach allocates significant importance to the technological platform rather than relying on ‘black-box thinking of software systems’ as is common in the development of DBCIs. 30
The O/C matrix designed by Oinas-Kukkonen as a tool to analyse the intent of a BCSS. The type of change is increasingly difficult to achieve as one progresses from C to A, with attitudes harder to influence than behaviours or compliance. Lower levels of changes (such as C) may lead to higher-level changes such as B and A. The consideration of which cell(s) to target as well as how to move between them is considered an essential but neglected area of BCSS research.
The FBM 27 and behavioural intervention technology (BIT) 59 provide similar structures for the design of a BCSS to that of the PSD model. However, due to the comprehensive nature of the O/C matrix/PSD model, and its explicit consideration for software requirements and implementation, it was felt to offer the most comprehensive model. As such, HYPO-CHEAT was designed using the O/C matrix/PSD model, and the FBM and BIT were not used. Thus, further details regarding FBM and BIT are not provided here.
BCSSs in the healthcare setting
This section discusses examples of the use of PT in the healthcare setting, within which HYPO-CHEAT will be operating. A systematic review of PT in health (mostly physical activity, dental health and disease management) found that there was a fully positive outcome in 64 of 85 (75%) studies and that targets for future work should be participatory design (involving users) and studies targeting children. 60
Interventions targeting glucose control. As HYPO-CHEAT is designed to reduce hypos, here we provide a review of the small number of BCSSs or DBCIs that have targeted a change in behaviour relating to blood glucose control. Jalil and Orji 61 investigated patient perceptions of PT and found that a majority of patients reported a preference for a PT over a human when it comes to advice about food relating to their diabetes.
Knowing that tailored feedback results in more desirable health behaviours, 62 Whelan et al. 63 used functional magnetic resonance imaging (MRI) scanning to investigate the types of feedback that would likely evoke the strongest responses. They discovered that personalised feedback regarding interstitial glucose levels (as obtained via CGM) resulted in greater responses than when feedback related to behaviour and as such recommended that behavioural feedback should be combined with physiological feedback to encourage behaviour change. HYPO-CHEAT aims to do exactly this.
Finally, Kwan et al. 33 undertook a systematic review of 38 studies to investigate the impact of ‘nudge’ interventions on glucose monitoring behaviour. Their results demonstrated that nudges did not affect glucose monitoring behaviours (proximal outcome) but did impact positively on HbA1C (distal outcome). Further analysis revealed that those interventions that included gamification and reminders were the most likely to result in positive outcomes 33 ; this was taken into account when designing HYPO-CHEAT.
HYPO-CHEAT approach
When designing HYPO-CHEAT, we followed a structure based on PT and BCSSs rather than behaviour change and DBCIs. This was largely due to the significant focus on the technology as an intrinsic part of the intervention that PT allows but is somewhat ignored by DBCI structures. However, because PT structures are less intrinsically theory-based, we used the BCTTv1 to select BCTs to ensure that our initial idea was evidence and theory-based. Thus, we used a hybrid approach incorporating BCTs from the BCTTv1 into the BCSS structure outlined by Oinas-Kukkonnen in his O/C matrix/PSD approach. 55
The ultimate intention of HYPO-CHEAT was to reduce hypoglycaemia (as measured by TBR) without the requirement for real-time CGM (access to live data). This was so that we could better understand how HYPO-CHEAT changed behaviours based on feedback (rather than live CGM changing behaviours) and also because if we demonstrated efficacy in this approach, it would reduce the need for ongoing CGM in the patient population and thus reduce cost and patient burden. CGM is not funded for patients with CHI in any country and families report significant burden from ongoing wear in relation to sensor alarms and painful sensor changes. 13
The design of HYPO-CHEAT underwent three distinct phases (analysis of intent, selection of BCTs, PSD structure) in order to maximise the likelihood of efficacy based on theory, develop learning points and provide contributions to the field. The usual approach in the field of behaviour change would be to identify a behavioural theory on which to base the intervention. We have opted not to do this, given Riley et al.'s 49 finding that current behaviour theories are not fit for modern adaptive interventions and the intrinsic use of theory within the BCTTv1. 44 Our three phases, therefore, began by analysing our intent using the O/C matrix (4.1.1). In the second phase, we chose BCTs from the BCTTv1 based on available evidence of those with the greatest efficacy (4.1.2). In the third and final stages, we applied these BCTs to our intervention using the PSD structure (4.1.3).
Analysis of intent (O/C matrix)
As suggested by Oinas-Kukkonen, we began our approach by evaluating our intent using the O/C matrix described above. We identified three elements of the O/C matrix that were intended targets for our intervention. These are detailed in Table 2.
The intended outcomes for HYPO-CHEAT are as per the O/C matrix described by Oinas-Kukkonen.

In addition to patients and families, members of the clinical team (doctors, nurses, clinical psychologists and dieticians) were identified as key stakeholders in the development of HYPO-CHEAT. Discussions with these members of the clinical team in a multidisciplinary team (MDT) setting concluded that compliance with HYPO-CHEAT was a very low burden and reinforcing the risks of hypoglycaemia would not be difficult. Further discussions identified that, due to routines patients have built up, often over many years, the two A/B changes were likely to be the hardest to change. As such, much of HYPO-CHEAT was designed to focus on persuasion in this area.
Selection of BCTs from the BCTTv1
Rather than select a specific behaviour change theory which may not have been developed with rapidly adapting, digitally delivered interventions in mind we instead opted to design our intervention using BCTs. This approach has multiple advantages: it is rooted in, and based on, behaviour change theory. It is a modern approach that has been designed with digital delivery in mind, so the established structure allows for easier interpretation of the end result. This offers more information regarding which techniques affect behaviour change in this field and thus contributes more generally to the literature.
Having established which BCTs in the BCTTv1 were most likely to have an effect on our patient population (see Sections 3.1.2 and 3.1.3), we selected 17 BCTs from seven different categories that would contribute towards our aims identified in the O/C matrix. All 93 BCTS from all 16 groups were considered for inclusion and are examined further in Table 3.
BCTs from the BCTTv1 are included in HYPO-CHEAT and the intended target from our O/C matrix. Those categories of the BCTTv1 from which no BCTs were selected are also briefly discussed.

PSD model and practical design of HYPO-CHEAT
Once we had selected the appropriate BCTs for our system, we utilised the PSD approach designed by Oinas-Kukkonen 30 to ensure the persuasive potential of our system was maximised and to allow for a practical design approach that took into account the importance of the mode and method of delivery.
Key postulates behind persuasive systems. There are seven key postulates behind persuasive systems that must be addressed when designing a BCSS. These are discussed in turn along with the subsequent impact upon the design of HYPO-CHEAT.
Information technology is never neutral: a BCSS is always on and always offering some persuasion. Thus, HYPO-CHEAT is designed to adapt to changes in behaviour and levels of interaction by feeding back on those to users and encouraging ongoing participation. People like their world views to be organised and consistent: to ensure consistency and commitment, we asked HYPO-CHEAT users to commit to reducing hypos and performing targeted fingerprick checks. Cognitive consistency
54
was used in the following manner: most parents of children with CHI will believe they are doing everything they can to keep their children safe. If they are then presented with evidence that their children are having frequent and predictable hypos (at the same time each week), then this belief must be challenged, and behaviour change will result in a restoration of cognitive consistency. Direct and indirect routes are key persuasion strategies: HYPO-CHEAT employs both: it offers a detailed analysis of CGM data for users’ consideration and reflection (direct route), and it provides a simple ‘heatmap’ with areas of high risk presented in dark red (Figure 3) to convey severity and prompt an immediate emotional reaction of concern worthy of intervention (indirect). Persuasion is often incremental: HYPO-CHEAT only suggests a maximum of three targets for improvement at any one time. This allows users to slowly and incrementally reduce their hypoglycaemia hotspots rather than trying to eliminate them all immediately and reduces the chance of overwhelming the user. Persuasion through persuasive systems should always be open: all users of HYPO-CHEAT were expressly aware of the designers’ desire to change behaviour to reduce hypos. Persuasive systems should aim at unobtrusiveness: since the current version of HYPO-CHEAT does not nudge or prompt users, it is very unobtrusive and is simply available when required by users. Persuasive systems should aim at being both useful and easy to use: HYPO-CHEAT was designed, as far as possible, to be very easy to use (simply read an A4 PDF) and useful in its design to reduce hypos.

Outline of the study structure. Patients were separated into two groups dependent upon the presence (left) or absence (right) of initial hypoglycaemia (TBR > 1%) in the initial blinded period. Subsequent changes in blinding (blue) and unblinding (green) of devices are highlighted in the figure.
Persuasion context. The context of the persuasion is vital and encompasses three key elements: the information is presented to the user; the user must pay attention and comprehend this information; and finally, the user may yield to the position presented, retain this information and, if persuasion is successful, take action to comply with the new position. 65
The intent
The purpose of this is to analyse who the persuader is and thus the type of persuasion that is ongoing. As HYPO-CHEAT was designed and used by the clinical team, its persuasion is both endogenous and exogenous. 66 Users will also likely be keen to reduce their hypos, and so the system is also somewhat autogenous. 66 The intent largely reproduces the O/C matrix described above and is thus not repeated here.
The event
It is assumed in most health-related persuasive systems that users will have the necessary information to act and simply have formed bad habits which prevent them from improving their health. 30 This is an unlikely use context for our users. It is highly likely that users do not have a complete understanding of their patterns of hypoglycaemia, and thus it is the provision of new information that will act to persuade a change in behaviour rather than the usual attitude reinforcement.
The strategy
The strategy of persuasion of a system is built on the concept that there is a central message and the persuader is trying to convince the persuadee of something. 65 This message can act by triggering emotions or appeals to reason and intelligence. 67 The central message of HYPO-CHEAT is to demonstrate that there is a pattern to hypos that users may not have previously noticed and that there is therefore an opportunity to reduce hypos which is currently not being taken. This is both relatively easy to change and vitally important for the long-term health of the patient. The route by which this message is delivered can be via the direct and indirect routes which have both been discussed above.
Design of system features. In the original paper on PSD, Oinas-Kukkonen and Harjumaa acknowledge that much of the design of system features has been adopted and modified from Fogg's original work on PT. 30 However, the benefit of PSD over FBM is the explicit consideration of how the design principles are translated into software features and implementation as actual features in a system. Oinas-Kukkonen and Harjumaa argue that system qualities are not something that should be thought about afterwards but are intrinsic to the persuasiveness of the system as a whole. 30
Once the persuasion context has been evaluated (Section 4.1.3.2), one can select persuasive design principles from four categories: primary task support, dialogue, system credibility, and social support. Because of the significant overlap between these principles and the BCTs provided by BCTTv1, we used the list of persuasive design principles to look for gaps in our approach rather than duplicate components of our system. We assessed our list of BCTs against each category of persuasive design principles in order.
Primary task support
This relates to the methods by which the system will support the user in achieving their primary goal: simplifying behaviour change into simple tasks, guiding this process, tailoring it to the user and providing monitoring and feedback. All of these principles (reduction, tunnelling, tailoring, personalisation, self-monitoring) are covered in detail by a large number of our BCTs.
Dialogue support
This focuses on the way the system will interact with users including praise and rewards, which are well covered by BCTs such as 2.2 feedback on behaviour and 10.4 social reward. It also covers the provision of suggestions, which HYPO-CHEAT provides as part of its functionality and is partially captured by BCT 8.6 generalisation of target behaviour (see Table 3). Dialogue support mimics BCTTv1 category 7 (associations) by suggesting the provision of reminders or prompts. Unfortunately, as previously discussed, we did not have the capacity to develop this for the pilot of HYPO-CHEAT, but this will certainly be incorporated into future versions.
System credibility support
System credibility support recognises the importance of the mode of delivery of the intervention and how this impacts the persuasiveness of the system as a whole.
Given that HYPO-CHEAT is to be used by healthcare professionals as part of patient care, the information provided will be truthful and fair, and thus trustworthiness is implicit. System credibility support includes expertise, authority, ‘real-world feel’ and third-party endorsements, which have all been included in HYPO-CHEAT. As users are all patients within a quaternary specialist service (Northern Congenital Hyperinsulinism Service (NORCHI)), they understand the expertise and authority within this system. We ensured that this trust was extended to HYPO-CHEAT through the provision of NORCHI logos within the output as well as multiple references to well-known and highly trusted members of staff within the service. Finally, users were all referred to use HYPO-CHEAT by their consultant, and so third-party endorsement was achieved. Further expertise and authority feel will be achieved in future versions by the provision of references for statements such as ‘We know that hypos can have long-term effects for patients with CHI’.
Surface credibility was considered when designing the system, and much work was undertaken to ensure that the product provided to users was professional in appearance, attractive, easy to look at and can be easily understood and interpreted by the user. If further resources had been available, this could have been improved further using a professional design company.
Social support
Given the clinical nature of the system, it was not appropriate to offer any kind of social support such as cooperation, competition or recognition as part of HYPO-CHEAT. Even if user details had been kept anonymous within a social space, the rare nature of CHI and the existence of widely used family support groups could have easily resulted in the accidental de-anonymisation of certain users by others.
Final design of HYPO-CHEAT
Through the use of all of the above steps, the first version of HYPO-CHEAT was developed. The technical specifics of HYPO-CHEAT are discussed in detail in a separate paper, 14 but a brief summary of HYPO-CHEAT's approach is discussed here. An example output is provided in full in Appendix A.
HYPO-CHEAT aggregates CGM data from users and generates a hypoglycaemia heatmap (Figure 2) based on the frequency, recurrence and severity of hypos throughout the week. This is presented to patients and accompanied by text-based simple interpretations of the heatmap to improve ease of use for those who do not want or are not able to interpret the graph (Appendix A). Further analysis is performed to ascertain the times of week which pose the greatest risk to users and up to three targets are generated. These are presented to users and they are asked to:
Target fingerprick tests to these periods. This provides the dual purpose of both forcing the user to concentrate on the times of most risk as well as generating a second check on the CGM data with a more accurate device. If the user finds that fingerprick tests done at this time are indeed reflective of hypoglycaemia, then they will be prompted to act upon this. If the fingerprick tests refute the CGM-informed hotspots, then the user is praised, and the hotspot is removed at the following HYPO-CHEAT interaction. Reflect on what is different about this specific time within the week. HYPO-CHEAT will only identify areas of outstanding high or low risk, and thus users are provided with a difference on which to reflect.

Example of a hypoglycaemia heatmap from HYPO-CHEAT. This heatmap, generated from 4 weeks (4w) of data, shows areas of high hypoglycaemia risk between 7 a.m. and 9 a.m. and increased further on Friday and Saturday. In HYPO-CHEAT's output, this heatmap would be accompanied by interpretation, and the user would be asked to reflect on the difference between Friday and Saturday morning vs Sunday morning (Appendix A).
Further functionality is provided by assessing if previous suggestions have been followed to (a) perform fingerpricks in certain areas and (b) try to reduce hypos in these same areas. If suggestions have been followed and/or hypos have been reduced, the user is congratulated. If attempts have been made, then this is encouraged. If no attempts have been made, then the user is reminded of the clinical importance of reducing hypos. Further iterative feedback is provided on one-off hypos and their evolution to repeat hypos as well as previous hotspots that have successfully been removed.
As key stakeholders, patients and families with CHI were intrinsically involved in the development of HYPO-CHEAT. Discussions with families around the difficulty of spotting patterns in CGM were held before the development of HYPO-CHEAT. Multiple early versions of HYPO-CHEAT, based on both individual and artificial data, were shown to families and feedback around utility sought. This informed the development of the version of HYPO-CHEAT which was used for patients. For example, early versions of the system did not differentiate between one-off and repeat hypos, and repeat yellow and green markers (see Figure 2) were not present. Patients and families commented on the difficulty in spotting patterns when certain ‘hotspots’ could be generated from a single hypo or one repeated each week. As such, the functionality described above was added and received positive feedback from users.
There are many other aspects of HYPO-CHEAT based upon the BCTs we selected that can be appreciated by interrogating an example of HYPO-CHEAT output in Appendix A. These functionalities, and the evidence behind them, are also discussed further in a separate paper. 14
Evaluation of HYPO-CHEAT
Early versions of HYPO-CHEAT were discussed with patients and families to gather feedback about aspects of the system that were helpful and those that were not. Following this, we performed a pilot study to assess the feasibility of implementing HYPO-CHEAT as well as an initial evaluation of its persuasive potential. Here, we discuss the methods of evaluation as well as present the results of this pilot study and user feedback from interviews. This work received full ethical approval from the Health Research Authority (HRA) (Integrated Research Application System (IRAS) project ID 268245) and Research Ethics Committee (REC) (reference 07/H1010/88).
Methods of evaluation
A detailed description of recruitment to the study and flow through it is described in detail in a separate publication, 14 summarised in Figure 3 and not repeated here so as to retain focus on behavioural change and PT rather than algorithmics. In summary, patients were provided with a Dexcom G6 CGM device which was blinded (no real-time data) for the first 4 weeks. This period acted as an assessment of baseline hypoglycaemia and divided the patients into two groups dependent on the presence or absence of initial hypoglycaemia (TBR >1%). Devices were then unblinded for four weeks and patients had access to real-time data. This allowed the families to self-identify any hypoglycaemic patterns, and these patterns could then be compared with those patterns identified by HYPO-CHEAT.
Finally, at Week 8, families of patients who demonstrated an initial TBR >1% were provided with a personalised HYPO-CHEAT output. Those with no initial hypoglycaemia had no use for HYPO-CHEAT as there were no patterns to be found. Once patients had had a chance to process their HYPO-CHEAT output, the analysis and suggestions were discussed with families, and opinions were sought on whether they thought HYPO-CHEAT was helpful. The CGM device was subsequently blinded for the final 4 weeks of monitoring. Patients took their HYPO-CHEAT output home with them to aid further reflection in their own time and were asked to focus fingerprick tests on their personalised targets. Patients then came back for one final visit and used HYPO-CHEAT for a second time to understand how their profiles had changed during the 4 weeks and to receive feedback on behaviour and outcomes.
Analysis was undertaken to compare the three distinct time periods, with interpretations based on relation to the use of HYPO-CHEAT. The analysis primarily focused on proximal outcomes of whether patients had changed their behaviour and followed suggestions for targeted fingerpricks, as well as the more distal outcome of change in hypoglycaemia within targeted areas and overall. The analysis of TBR between blinded and unblinded periods (periods 1 and 2) was performed for all patients as these periods were the same for all. The analysis of the difference between the two blinded periods was performed only for those five who had used HYPO-CHEAT. Our primary outcome was the number of patients to achieve the predefined MCID in TBR between the two comparative blinded periods (before and after using HYPO-CHEAT).
To better understand user perceptions of HYPO-CHEAT and see if and how it had changed their behaviour, we sought some feedback. This work was performed by a member of the research team who had no association with the primary study and who could thus act independently. This also increased the likelihood that users would offer honest feedback and not feel pressured to be positive to a member of the research team which whom they had built a relationship. Full details are provided in a separate publication, 13 but a summary of methods is provided here. An interview guide was developed through researcher, clinician and psychologist consensus, followed by semi-structured interviews with all families and finally a thematic analysis as described by Braun and Clarke 68 Here, we have provided a subset of the interview data that pertained to HYPO-CHEAT and behaviour change.
Results
Recruitment is described elsewhere 14 with a summary of demographics and clinical details in Table 4.
Demographics and clinical details of participants.
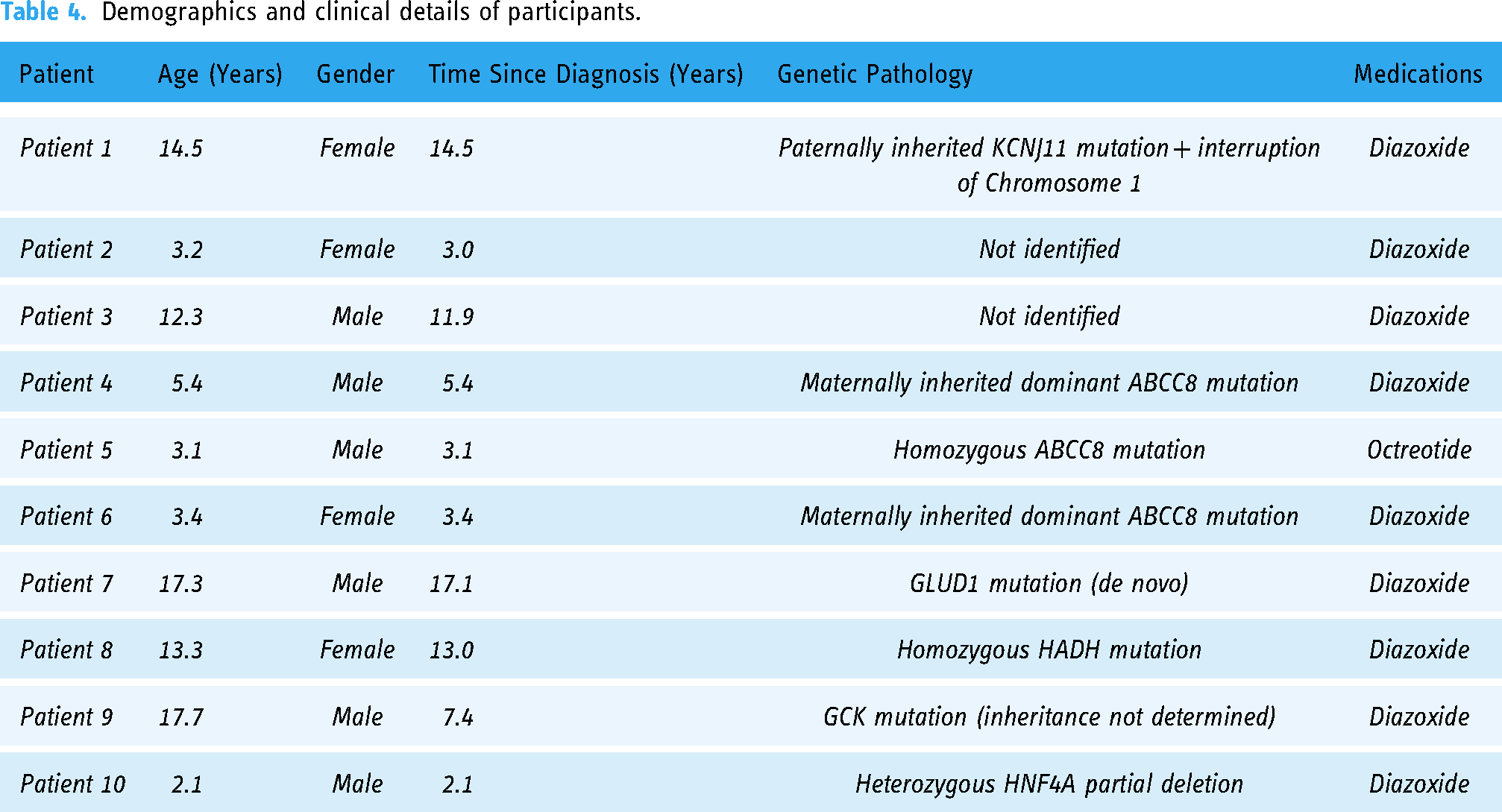
Of the nine patients who completed the study, four had insignificant hypoglycaemia during the initial blinded (baseline) period with a mean TBR of just 0.2% and thus little use for HYPO-CHEAT. These patients provide a useful comparison group (albeit neither matched nor randomised) to see how TBR changed when HYPO-CHEAT was not used.
Results of proximal behaviour change measures. Fingerprick tests were used as a proximal measure of whether HYPO-CHEAT had the potential to change behaviour relating to hypoglycaemia. The five patients with initial hypoglycaemia (TBR > 1%) were shown their hypoglycaemia patterns in HYPO-CHEAT and given up to three targets upon which to focus their fingerpricks. This resulted in 13 targets (52 over a 4-week period) representing 156 hours (each target covers a 3-hour period). Fingerpricks were performed in 31 of the 52 targets (60%) resulting in a fingerprick rate of 0.20 checks per hour. This was 67% higher than the background (non-targeted) fingerprick rate of 0.12 checks per hour (chi-square = 7.1, P = 0.008), demonstrating that provision of targets is likely to have changed users’ behaviours to increase checking of glucose during times of identified hypoglycaemia risk.
Results of distal outcomes (change in TBR). A comparison of blinded and unblinded periods (periods 1 and 2) for all 10 patients showed a change in TBR from a mean of 3.9% to a mean of 4.0%, indicating no average reduction in hypoglycaemia from device unblinding. For the five patients with initial hypoglycaemia, we have shown that HYPO-CHEAT affected a change in fingerprick behaviour within these targets, but we also assessed for the ultimate outcome of a change in TBR. We have presented the results of this outcome in two ways. The first is via an aggregated mean change in TBR followed by a report of individual changes in TBR and whether this met the MCID for each patient.
As documented above, patients were given up to three targets within which they were asked to reflect on the causes of hypoglycaemia. Following the use of HYPO-CHEAT, all patients reduced the TBR in their provided targets (Figure 4, Table 5). The mean (range) reduction in hypoglycaemia within targets was 67% (43–100%) despite users going from a period of unblinded CGM (access to real-time data) to blinded CGM (no access to real-time data). As expected, when devices were reblinded, the aggregated mean total TBR did increase slightly from 4.5% to 5.4%. However, the aggregated total TBR of 5.4% remained 25% lower than the other comparable blinded period (7.1%) before patients used HYPO-CHEAT (Figure 3). Analysed at an individual level, four out of the five patients (80%) achieved the MCID in TBR in blinded periods after using HYPO-CHEAT (Table 5). One patient showed an increase in TBR between these periods, although this patient was admitted to the hospital for 6 days with an intercurrent illness during the final blinded period which significantly increased TBR. Overall the reduction in hypoglycaemia following HYPO-CHEAT was a mean of 25% and failed to reach significance (P = 0.1 on paired t-test). However, if Pt2 is excluded from analysis due to concurrent illness, the reduction is a mean of 32% with a significant P value on a paired t-test of 0.01. The failure of unblinding to reduce TBR for all patients and the reductions in TBR between comparable blinded periods for those using HYPO-CHEAT suggest that the behaviour change resulting from HYPO-CHEAT (rather than live data provision) actually reduced both targeted and total TBR from baseline.

Visualisation of the relative change in TBR between periods for the group with initial hypoglycaemia. When devices were unblinded, TBR improved by 37%. At the end of this period, patients used HYPO-CHEAT and received targets for improvement. Unsurprisingly, when devices were reblinded and live data was taken away, TBR increased slightly by 20%. However, targeted TBR improved by a mean of 67% suggesting that patients were following suggestions made by HYPO-CHEAT. Most importantly, the final blinded period contained 25% less aggregated hypoglycaemia than the initial blinded period, suggesting that HYPO-CHEAT can reduce TBR when patients have no access to live CGM data.
TBR (%) as well as absolute and relative change by the patient. Green shaded boxes indicate those patients who aceived the MCID, with red indicating those who did not. *Pt2 was admitted to the hospital with an intercurrent illness and severe hypoglycaemia during the final blinded period, and thus the TBR in this period is like an overestimate. **Details of Patient 6 (Pt6) are included for the sake of completion but are not included in the calculation of means as the patient did not complete the study. Minimum clinically important difference (MCID) was set at an absolute TBR reduction of 1.5% and was achieved by four of five patients (80%).

For the patient group who showed no discernible pattern of hypoglycaemia in the first part of the trial, HYPO-CHEAT had nothing to report, and so no recommendations were made. Thus, HYPO-CHEAT was not of use to these patients at the beginning of the study. However, throughout the remainder of the study, the TBR for these patients steadily increased from a baseline level of 0.2% in the first blinded period to 3.2% in the final period (Table 5). This is to be expected, as the effect of being observed (Hawthorn effect) is likely to have increased tight glycaemic monitoring, 69 but this effect lessens over time 70 and thus offers further support for the hypothesis that it is the use of HYPO-CHEAT that reduced TBR in the other group and not simply a reduction over time.
User feedback on HYPO-CHEAT
We have presented results that are suggestive of a reduction in TBR, mediated by an intended behaviour change, following the use of HYPO-CHEAT in a test group of patients with CHI. However, because of the lack of a randomised comparator group, it is impossible to say for certain that HYPO-CHEAT was responsible for the behaviour change and subsequent reduction in the TBR. We therefore sought feedback from users to investigate if they felt that HYPO-CHEAT had been useful in changing their behaviour and reducing hypos.
A full report of the semi-structured interview and thematic analysis (primarily focused on CGM in CHI) is provided in a separate paper 13 ; here, we will briefly discuss the elements pertaining to HYPO-CHEAT and behaviour change that emerged during the interviews. All nine families mentioned the way in which knowledge of CGM data and summaries via HYPO-CHEAT had influenced behaviour change.
Patients obtained new insights into their glucose profiles from using HYPO-CHEAT: “it showed some certain times I was getting a lower, like, say on a Friday morning, because I start late, I don’t get out of bed until later on so I start -my blood was dropping” “it's made us more aware that it's happening during the night”
“it made us realise from the graph before what areas to concentrate on for his low sugars”
“there were some Saturday mornings that they were really dipping quite low, but we still have to drag him out of bed to make sure” “we started doing, like, two extra [fingerpricks] a day in between them [meals]” “it's made us more aware that he definitely needs something before he goes to bed after sport even if he doesn’t want it” “[I] make sure that he had something a bit more sugary in the evening or have a late dinner, just to make up for those late hours in the morning where he's getting those low sugars”
“Previously we would […] wait until something was to happen to actually look for, you know, the blood sugars. But with this one, it kind of made us […] pay more attention to her and, you know, tell her ‘have you eaten’, ‘make sure you eat your breakfast’ ”
Discussion
In this paper, we have outlined the devastating impact that hypoglycaemia can have on children with CHI. We have discussed the current methods for the prediction and prevention of hypoglycaemia in children and how these primarily focus on either using ML and continuous CGM to predict future values or on the provision of CGM with a review of data. Unfortunately, the ML approach has yet to demonstrate any real-world effect, and the CGM approach is insufficient with most methods of data review lacking proper data aggregation, and failing to provide helpful visualisations upon which patients can act to change future glucose profiles.
The primary problem with existing approaches is that they fail to account for the human in the loop and the importance of not simply providing information but of designing the delivery of that information such that it has the capacity to trigger reflection and subsequent behaviour change. The fields of behaviour change and PT thus have much to offer and provide an evidence-based background upon which we can develop new approaches to genuinely prevent hypoglycaemia in free-living conditions rather than detect and react to it.
HYPO-CHEAT has been designed as a BCSS, based upon a dual approach incorporating the behavioural theory-informed BCTs of behaviour change and the delivery-orientated PSD model of PT. This method of design has ensured that HYPO-CHEAT is rooted in established behavioural theory while not ignoring the importance of mode and methods of delivery which themselves contribute to the persuasiveness of a system.
Unlike the vast majority of technological approaches to prevent hypoglycaemia, we have conducted an in vivo analysis of HYPO-CHEAT in free-living conditions, in 10 patients with the target condition. To better understand not only if but how our system works, we designed our evaluation to measure both proximal and distal outcomes and thus contribute important knowledge to multiple areas of the literature. Our associated paper 14 demonstrates that HYPO-CHEAT is an effective way to reduce TBR for patients with CHI, and here we provide evidence that it likely achieves this through targeted behaviour change. We demonstrated that HYPO-CHEAT achieved its proximal goal of affecting changes in fingerprick behaviour to target checks around periods of high risk. The impact of this was to increase patient awareness of times of hypos as well as to confirm or refute the presence of troublesome and repetitive hypoglycaemia. There is, however, scope for improvement: despite patients understanding the potential risk of hypoglycaemia and being shown times of heightened risk, 21 of 52 targets went unchecked by fingerprick test. Patients had no access to real-time CGM data in this period, and it is likely that many simply forgot to check a fingerprick during their targets. Reminders delivered by email or SMS are likely to increase adherence to suggestions 33 and will certainly be incorporated into future versions of HYPO-CHEAT to ensure more high-risk periods are monitored with fingerpricks.
HYPO-CHEAT also likely achieved its distal goal (and primary outcome) of reducing the TBR for patients with CHI without access to real-time CGM data. There was a clear reduction in the mean TBR on a group level, but this did not achieve statistical significance unless Pt2 (who was acutely unwell with recurrent hypoglycaemia) was excluded from the analysis. In addition to a likely mean reduction in hypoglycaemia, 80% of patients also achieved a reduction in TBR of at least the MCID. While these results are promising, they must be viewed in light of the small sample size available for this study. From the pilot data we have available here, it is not possible to definitely conclude that HYPO-CHEAT is an effective method of hypoglycaemia prevention. However, from thematic analysis of data from semi-structured interviews, we were able to ascertain that this observed reduction in hypoglycaemia was achieved through predictive and proactive measures taken by families to prevent hypoglycaemia and adds weight to the hypothesis that using HYPO-CHEAT does reduce hypoglycaemia. This behaviour change is coordinated using multiple BCTs from a wide range of categories that can target multiple methods for behaviour change. Importantly, the means by which HYPO-CHEAT is delivered used the considered and evidence-based approach of the PSD model to maximise the chances of optimal and effective persuasion. There are very few algorithmic approaches that have been shown to effectively reduce hypoglycaemia without the need for ongoing CGM, thus a pilot of HYPO-CHEAT suggests its unique ability in this area. The relatively simple algorithmics, low cost and short lead-in time of this system allow it to be scaled up very quickly and tested in large patient groups in multiple environments without the need for either large grants or CGM manufacturer collaboration, both frequent stumbling blocks for hypoglycaemia prevention technologies. 23 More importantly, this is the first innovation aiming to reduce hypoglycaemia for patients with CHI and thus providing much-needed support for this often-underserved group.
Limitations and future work
In our associated paper, 14 we outline the limitation of HYPO-CHEAT being unable to predict acute and unexpected hypoglycaemia in the same way that a glucose forecasting algorithm might. While this limitation is undoubtedly true, the data presented in the background and results of this paper may offer a limited solution to that limitation. We have demonstrated that HYPO-CHEAT is capable of stimulating reflection on the causes of hypos and thus unexpected hypoglycaemia is likely to be less common in a group often reflecting on behavioural precipitants of hypoglycaemia. Furthermore, HYPO-CHEAT can be used alongside CGM if desired and thus acute alarms will remain present in addition to the pattern recognition capacity of HYPO-CHEAT. This would require an increase in the price and burden of CGM, beyond that already required for the use of HYPO-CHEAT as described within this study. Finally, CGM is not currently the standard of care for patients with CHI, and thus by providing HYPO-CHEAT to this patient group, no monitoring or treatment is removed.
It is important to acknowledge the limitation of CGM with regard to point accuracy. This is poor for patients with CHI 12 and is a significant limitation of this technology's utility. However, HYPO-CHEAT somewhat negates this limitation through its focus on repeating patterns. Inaccuracy of CGM devices is consistent across the week and thus repeating patterns of hypoglycaemia are likely to accurately reflect true patterns for patients.
Another, important, limitation of our work is the short time frame and small patient numbers involved in the pilot study. Furthermore, this pilot was not randomised and thus results have to be interpreted with the caution appropriate for pilot studies until larger-scale, randomised trials can be undertaken.
Even within the relatively short 4-week period of using HYPO-CHEAT, patients by no means undertook all suggestions for fingerpricks. It is possible, and even likely, that engagement with HYPO-CHEAT would fade over time as patients improve their TBR and gather less benefit from the use of HYPO-CHEAT. It is important to plan for this so-called ‘gateway’ effect when the persuasive system will no longer be required, 71 and the system can even be expressly designed with this effect in mind, 72 and to plan for user abandonment. As patients reduce their TBR, future iterations of HYPO-CHEAT will suggest less frequent interactions, with the intention to reduce the chance of patients becoming bored with the system. The addition of prompts and nudges to perform fingerprick checks at high-risk times, or to think about pre-bed routines for preventing predicted overnight hypos would theoretically increase engagement with HYPO-CHEAT as well as possibly improve its efficacy. 33 Finally, gamification will be added to future versions of HYPO-CHEAT to try and improve engagement and adherence. 73 Once the initial CGM phenotyping is complete and patients are provided with their individualised heatmap, they will be challenged to try and clear the heatmap by performing targeted fingerprick tests demonstrating normoglycaemia (normal blood glucose) at times of historic hypos and thus slowly remove hotspots from their heatmap. A system of rolling fingerprick checks will slowly check the rest of the week and ensure new and/or old hypos have not (re)emerged.
Future work will concentrate on determining the minimal time period required to establish a reliable and stable digital phenotype of an individual's glucose profile upon which HYPO-CHEAT can then work with fingerprick data. Future studies must evaluate the impact of prompts and gamification on the efficacy of HYPO-CHEAT and a large, randomised controlled trial is required to confirm or refute pilot findings and investigate long-term behaviour change. This larger-scale study would also allow for the collection of group-level data to investigate the correlation of certain classes of behaviour (e.g. exercise, meals) with the risk of hypoglycaemia. Finally, a micro-randomised trial approach would allow for fine-tuning of the system 74 with multiple decision points each day being assessed for efficacy on an individual basis. Once HYPO-CHEAT has been fully established as effective, it will be important to define how this is integrated into existing clinical pathways.
Conclusions
In this paper, we have provided a detailed description of the behaviour change basis of HYPO-CHEAT, a novel approach to aggregate and present CGM data to patients with CHI with the intention of changing proximal behaviours to reduce hypoglycaemia in free-living conditions. We have demonstrated the utility of HYPO-CHEAT in a small pilot study within the target patient population and provided a thorough analysis of how this utility has been achieved. Preliminary results indicate that HYPO-CHEAT may offer an effective and practical approach to reduce hypoglycaemia for patients in free-living conditions in the immediate future. This system does not require expensive algorithmics, monitoring equipment or industry collaboration. HYPO-CHEAT's mechanism of action is through targeted provision of actionable information designed to change behaviour and empowers patients to engage with self-care and improve their future health outcomes. Further, large studies are required to validate these findings.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231192011 - Supplemental material for The behaviour change behind a successful pilot of hypoglycaemia reduction with HYPO-CHEAT
Supplemental material, sj-docx-1-dhj-10.1177_20552076231192011 for The behaviour change behind a successful pilot of hypoglycaemia reduction with HYPO-CHEAT by Chris Worth, Paul W Nutter, Maria Salomon-Estebanez, Sameera Auckburally, Mark J Dunne, Indraneel Banerjee and Simon Harper in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors would like to thank Sumera Ahmed, Elaine O'Shea and Sarah Worthington for their assistance in the practical aspects of running the clinical trial of HYPO-CHEAT.
Contributorship
CW designed and built the analytics required for the project. CW designed and ran the clinical trial of HYPO-CHEAT. CW wrote the manuscript. SA performed the interviews and the thematic analysis. All authors provided expert input into the design and running of the study, reviewed and edited the manuscript and approved the final version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
This work received full ethical approval from the HRA (IRAS project ID 268245) and REC (reference 07/H1010/88).
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article. This work was supported by the Health Innovation Manchester (grant number Momentum Grant).
Guarantor
CW
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
